Abstract
A neurodegenerative disorder, glaucoma is a leading cause of blindness in the world. The conventional treatment strategies do not allow the significant penetration of the drug in the cornea. Therefore, we prepare a brinzolamide (Brz) loaded core-shell nanoparticles (NPs) to enhance the coronial penetration of the drug and thus treating the glaucoma. The shell of the NPs was composed of phosphatidylserine (PS; 1,2-diacyl-sn-glycero-3-phospho-L-serine), whereas the core of the NPs contains the Brz encapsulated in brinzolamide–phosphatidylserine–polymer poly-(DL-lactic acid-co-glycolic acid)–phosphatidylserine (Brz-PS-PLGA). The synthesis of Brz-PS-PLGA was achieved by using a coaxial electrospray process (CEP), which allows the preparation of the particles in a single step. The size of Brz-PS-PLGA with PS shell and brinzolamide–poly (lactic-co-glycolic) acid (Brz-PLGA) without shell was 571 ± 27.02 nm and 456 ± 19.17 nm, respectively. The charges on the surface of Brz-PS-PLGA and Brz-PLGA were (-) 27.45 ± 2.98 mV and (-) 19.47 ± 2.83 mV. The transmission electron microscopy images clearly reveal the PS shell as a light black layer over the dark black PLGA core. The CEP allows the high encapsulation of Brz in Brz-PS-PLGA where percentage of entrapment efficiency for Brz-PS-PLGA was 88.13 ± 6.43%. The release study conducted in a simulated tear fluid revealed the sustained release patterns of Brz from Brz-PS-PLGA and these were nontoxic to the cells as revealed by the cytotoxicity studies. Further, the Brz-PS-PLGA enhanced the coronial penetration of Brz and was capable of significantly reducing the intraocular pressure (IOP) after administration to the rabbit eye in comparison to the Brz-PLGA and free Brz. The results clearly suggest that the PS coating significantly enhances the capability of the particles in reducing IOP.
Keywords
Introduction
Glaucoma is a multifactorial and leading cause of blindness around the globe and is characterized by neurodegenerative disorders 1 . The primary characterization of glaucoma is scarcity of retinal ganglion cells in the optic nerves, microglia activation, and atrophic changes in the layers of the photoreceptor 2 . Up to date, intraocular pressure (IOP) is a key factor in clinics that can modify glaucoma development. The current therapeutic studies modulate IOP by reducing the aqueous production 3 . Meanwhile the reduction of IOP via various pharmaceutical drugs or surgical methods are in practice used in order to reduce IOP for treatment of glaucoma.
Brinzolamide (Brz) is a well-known carbonate anhydrase inhibitor, which is a second- generation therapeutic drug used for IOP and open-angle glaucoma treatment 4 . Solubility of Brz is a critical factor which limits its use. However, it can be overcome by encapsulation of Brz in nanoparticles (NPs)5,6. A recent investigation has highlighted that 10–1000 nm ranged NPs are ideal for passage of poorly soluble and large molecules through the ocular barrier system 7 . These superficial barriers mostly hinder the systemic and direct drug delivery to a specific target site. The NPs loaded with Brz have biological properties which enhance eye drop residence time, minimize the toxicity, and maximize the Brz penetration ability in the ocular structure 8 . Beside that the aqueous nature reduces the precorneal drug loss due to tears. NPs have been reported with excellent outcomes for delivery of ophthalmic drugs.
Polymer poly-(DL-lactic acid-co-glycolic acid)–phosphatidylserine (PLGA) is widely used for microencapsulation of antigens and therapeutics9,10. The PLGA-NPs have also been reported for the delivery of pranoprofen, carprofen, and Brz to the ocular system for glaucoma treatment 11 . PLGA have excellent biodegradability and biocompatibility properties which make them ideal for successful drug delivery. Phosphatidylserine (PS), a cell membrane phospholipid, plays an important role in apoptosis and cell cycle signaling12–14. The arrangement of lipid–polymer NPs has also been a subject for reasearch due to their special properties, which can alleviate some drawbacks allied with polymeric NPs and liposomes15–17.
Interestingly, various methods are in practice for the fabrication of PLGA-NPs, but the most successful one is determined by many drug-related factors (partition, solubility, and coefficient) and based on the nature of polymers (molecular weight, composition, and chemical functional groups)18, 19 . Hence, single and double emulsion, solvent evaporation, porous glass membrane emulsification, and spray-drying are commonly applied techniques for synthesis of biodegradable PLGA-NPs20–22. However, each of these methods have some limitations including particle-size polydispersity, low drug-loading, and encapsulation efficiency and hydrophilic drugs incorporation, etc. 23 .
Electrospray (ES) techniques utilize the electrostatic forces to synthesize various sizes and shapes of fibers or particles 24 . The ES method is preferred over conventional encapsulating techniques due to its narrow particle-size distribution, higher loading efficiency, and ease of particle synthesis25, 26 . Moreover, the ES technique would also eradicate the need for a surfactant. The size, shape, composition, and morphology of NPs can be optimized by modulating the material choice and process parameters. The advantages of the ES technique also include the production of multilayered structures using a coaxial process and individually monitored delivery of multiple drugs. We especially used a coaxial electrospray process (CEP) which could produce the lipid-coated PLGA micropaticles in a single step process 27 . The whole setup is briefly shown in Figure 1. The advantages offered by CEP over other nanoparticle fabrication methods include the production of uniform particles with encapsulation of the drug up to 100%. It also protects the protein cargo from denaturation as no heat is required in the process28–30. Further, the use of a surfactant could be avoided in the CEP. However, in the process of emulsification, although the process setup is easy in comparison to CEP, the particles produced were not uniform and have wide size distribution 31 . The comparison of advantages and disadvantages of CEP over other methods is shown in Figure 2.

Detailed instrumental setup for fabrication of brinzolamide–phosphorothioates–polymer poly-(DL-lactic acid-co-glycolic acid) using the coaxial electrospray process.

The advantages and disadvantages of the coaxial electrospray process in comparison to other methods used for fabrication of nanoparticles.
In the present study, we have developed Brz-loaded core-shell microparticles using the CEP for the delivery in glaucoma. The CEP was optimized and the developed particles were well characterized. The potency of these particles in reducing the IOP and the ability of corneal penetration were evaluated.
Materials and methods
Materials
The PLGA was obtained from LACTEL Absorbable Polymers, Shandong, People’s Republic of China. PS (1,2-diacyl-sn-glycero-3-phospho-L-serine), poly (vinyl alcohol) (PVA, 87–90% hydrolyzed, Mw = 30–70 kDa) and phosphate buffer saline solution (PBS, pH 7.4) were purchased from Sigma-Aldrich.
Preparation of Brz-loaded PS-PLGA (Brz-PS-PLGA) NPs
The Brz-loaded PLGA-NPs having a PS shell (Brz-PS-PLGA) were fabricated via CEP in a single step process. The CEP system consists of a dual flow pump, a coaxial needle which consists of an inner needle with a diameter of 70 μm inserted and fixed in an outer needle having a diameter of 150 μm, a positive electrode, which was fixed at the top of the coaxial needle, and a negative electrode, which was fixed at the collector. The collector comprised 1% PVA solution in a glass Petri dish which was kept on the aluminum plate. The distance between two electrodes was set as 12 cm. The electric field was generated via voltage difference in a nozzle chip and collector which resulted in ES. The inner solvent contained 4% of PLGA and 1% of Brz dissolved in acetonitrile while the outer solvent consisted of 1% solution of PS in acetonitrile. The flow rate of the pump was adjusted at a speed of 10 µL/minute. Both electrodes at different voltage helped with particle acceleration toward the collecting solution. The whole setup was placed inside a Taflon box with controlled flow of air which adjusts the temperature and humidity. When the solution emerged from the coaxial needle in the presence of an electric field, a Taylor cone was formed at the tip of the needles, further resulting in formation of a jet. The jet consists of particles in a solution, which were collected in the collector. The process was well optimized for the effect of voltage and flow rate on the particle size, morphology, and drug entrapment. To ensure homogenous particles the solution was continuously stirred. The suspension of particles was centrifuged at a speed of 30,000 revolutions per minute (rpm) for 30 minutes in an Eppendorf 5804R centrifuge. The supernatant was removed and replaced with fresh medium. The method was repeated three times to remove unbound drug and excess PVA. The pure clean Brz-PS-PLGA were stored at -20°C following lyophilization. The Brz-PLGA were also prepared without PS coating by using a single axial needle and they were collected as described above.
Characterization of Brz-PS-PLGA
The Brz-PS-PLGA morphology was analyzed by scanning electron microscopy (SEM; Hitachi SU-70, Fei). The SEM analysis was performed at +10 kV. Before SEM imaging, the Brz-PS-PLGA were coated with gold and placed on the aluminum tubs for imaging. Size distribution was determined by a Malwern Zetasizer. The lipid coating on the Brz-PS-PLGA surface was determined by transmission electron microscopy (TEM; TECNAI-10, Phxc ilips, Eindhoven, the Netherlands). The particles were stained with 0.1% phosphotungstic acid for 15 minutes, thoroughly washed, and dried before TEM analysis.
Determination of entrapment efficiency (EE) for Brz-loaded PLGA
To determine the percentage of EE of Brz in the Brz-PS-PLGA, the particles were centrifuged at a speed of 30,000 rpm for 30 minutes and the supernatant was collected. The Brz was extracted using acetonitrile and the amount of encapsulated Brz was quantified by high-performance liquid chromatography (HPLC, Waters Alliance 2695) using acetonitrile and PBS of pH 3 in a ratio of 40:60 as a mobile phase in an isocratic mode. 20 µL of the sample was injected and a reverse phase C18 column (25 cm × 0.46 cm, 5 μ) at a flow rate of 1 mL/minute resulted. The sample was detected by the ultraviolet detector at the wavelength of 254 nm 32 .
The equation for percentage of EE calculation was as follows:
The analyses were performed three times and values were expressed as mean ± standard deviation.
In vitro Brz release from Brz-PS-PLGA
The dynamic dialysis technique was used to investigate the drug release of Brz-PS-PLGA investigated in simulated tear fluid (STF) comprising sodium chloride 6.8 g, sodium carbonate 2.2 g, calcium chloride dehydrate 0.08 g, potassium chloride 1.4 g, and ultra-pure water to 100 mL. In brief, the equivalent amounts of free Brz, Brz-PLGA, and Brz-PS-PLGA were placed in dialysis bags (MW: 10K Da) and were shaken at the speed of 100 rpm at 37°C. 0.5 mL of sample were collected at different time intervals from the release medium and replaced by fresh medium. The samples were analyzed by the HPLC method to calculate the content of Brz. The analyses were carried out in triplicate.
In vitro cytotoxicity studies
A cytotoxicity assay was performed in the L929 cell line, which is a mouse fibroblast cell line and ARPE-19, which is a human retinal pigment epithelial cell line obtained from the China Center for Type Culture Collection using a MTT, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay 33 . The safety of Brz-PS-PLGA was evaluated on the basis of cell cytotoxicity caused by it. The cells were cultured using Roswell Park Memorial Institute medium and were seeded in 96 well plates. 100 µL medium containing 5 × 103 cells was added per well and incubated in an incubator for 24 hours. Following the incubation, the cells were treated with different concentrations of blank-PS-PLGA or Brz-PLGA and Brz-PS-PLGA for 48 hours. Afterwards, 10 μL of MTT solution in a concentration of 5 mg/mL was added to each test group. Subsequent to the incubation for another 4 hours, dimethyl sulfoxide (100 μL) was added to every well of 96 well plates. The data were recorded by using enzyme-linked immunosorbent assay at 490 nm. In order to calculate the cell viability percentage, we used a viability formula:
In vivo studies
The animal studies were conducted after the prior approval from the institutional animal ethical committee of the First People’s Hospital of Lianyungang, Lianyungang (Protocol Number:
FPHLACUC061134). Male rabbits of age 5 weeks were used for the study. Animals were caged in the animal house and were provided with the recommended diet.
Subconjunctival administration and IOP measurements for Brz-PS-PLGA
One of the most considerable factors for ophthalmic preparations is corneal permeability. Hence the corneal permeability of Brz-PS-PLGA was studied using a Franz diffusion chamber. The chamber is maintained at at a fixed temperature and comprises a donor compartment having volume of 0.5 mL and a receiver compartment consisting of 1.0 mL volume. Once the rabbits were sacrificed the cornea were removed with a 1.5 mm sclera ring, and the sample was stored in glutathione-bicarbonate-Ringer (GBR) solution of pH 7.5. The standard composition of GBR was used for the study. The extended cornea was fixed in clamped donor-receptor compartments by sclera. On the epithelial side, 1 mL of the GBR following 0.5 mL of Brz-PLGA or Brz-PS-PLGA was added. Further on, at fixed time intervals (0.5, 1, 1.5, 2, 2.5, 3, 3.5 and 4 hours) 0.3 mL of samples was sampled and fresh GBR solution of the same amount was added. The obtained samples were analyzed by the HPLC method and the drug infusion across the cornea was calculated.
The IOP was calculated by following reported methods in the literature 34 . In our studies, we took fifteen healthy rabbits and randomly divided them into four groups. To induce the increased IOP, we have used α-chymotrypsin, which was injected in the right eye of the rabbit. The rabbit’s right eye was treated with 50 µL of sterile PBS (control), free Brz, Brz-PLGA, and Brz-PS-PLGA. IOP values were monitored using a Schiotz Tonometer (Chongqing Vision Star Optical Co., Ltd.) at specific time intervals (0.5, 1, 2, 4, 8, 12, 16, 20 and 24 hours). In this analysis, the left eye of all rabbits was analyzed along with the treated right eye of the animals and their IOP was monitored throughout the experiment.
At the end of the study, the cornea was isolated and analyzed for any toxicity after hematoxylin and eosin staining using a microscope.
Result and discussion
The preparation of Brz-PS-PLGA particles was conducted using a coaxial electrohydrodynamic process. Brz encapsulating Brz-PS-PLGA and Brz-PLGA were synthesized using a CEP and single axial ES process, respectively. The process allowed the encapsulation of Brz in a PLGA core with a lipid shell of PS in a single step. The Brz-PS-PLGA have a PS coating whereas Brz-PLGA, prepared with a single needle having the flow of inner solution, did not have the PS coating. The simultaneous flow of outer fluid and inner fluid allows the preparation of Brz-PS-PLGA in a single step. The process was well optimized for the effect of voltage and flow rate on the prepared particles. The effect observed on particle size has been summarized in the supplementary online information. Particle sizes of Brz-PS-PLGA and Brz-PLGA were determined using a DLS Zetasizer (Malwern) which reveals the particle sizes of 571 ± 27.02 nm and 456 ± 19.17 nm, respectively. The charges on the surface of Brz-PS-PLGA and Brz-PLGA were (-) 27.45 ± 2.98 mV and (-) 19.47 ± 2.83 mV, respectively. It was observed that the presence of PS on the surface of Brz-PS-PLGA increases the negative charge on the particles. The morphology of these particles was confirmed by using SEM analysis which were uniform and spherical in shape as shown in Figure 3(a). Figure 3(b) represents the TEM micrograph of Brz-PS-PLGA which confirms the PS shell over the PLGA core. The TEM image clearly reveals that the PS shell has a light black layer over the dark black PLGA core. The ES process allows the high encapsulation of Brz in Brz-PS-PLGA. The percentage of EE for Brz-PS-PLGA was 88.13 ± 6.43 whereas for Brz-PLGA it was 84.37±3.76. The process allows the enhanced encapsulation of drug in comparison with other methods of NPs’ fabrication. The results are shown in Table 1.
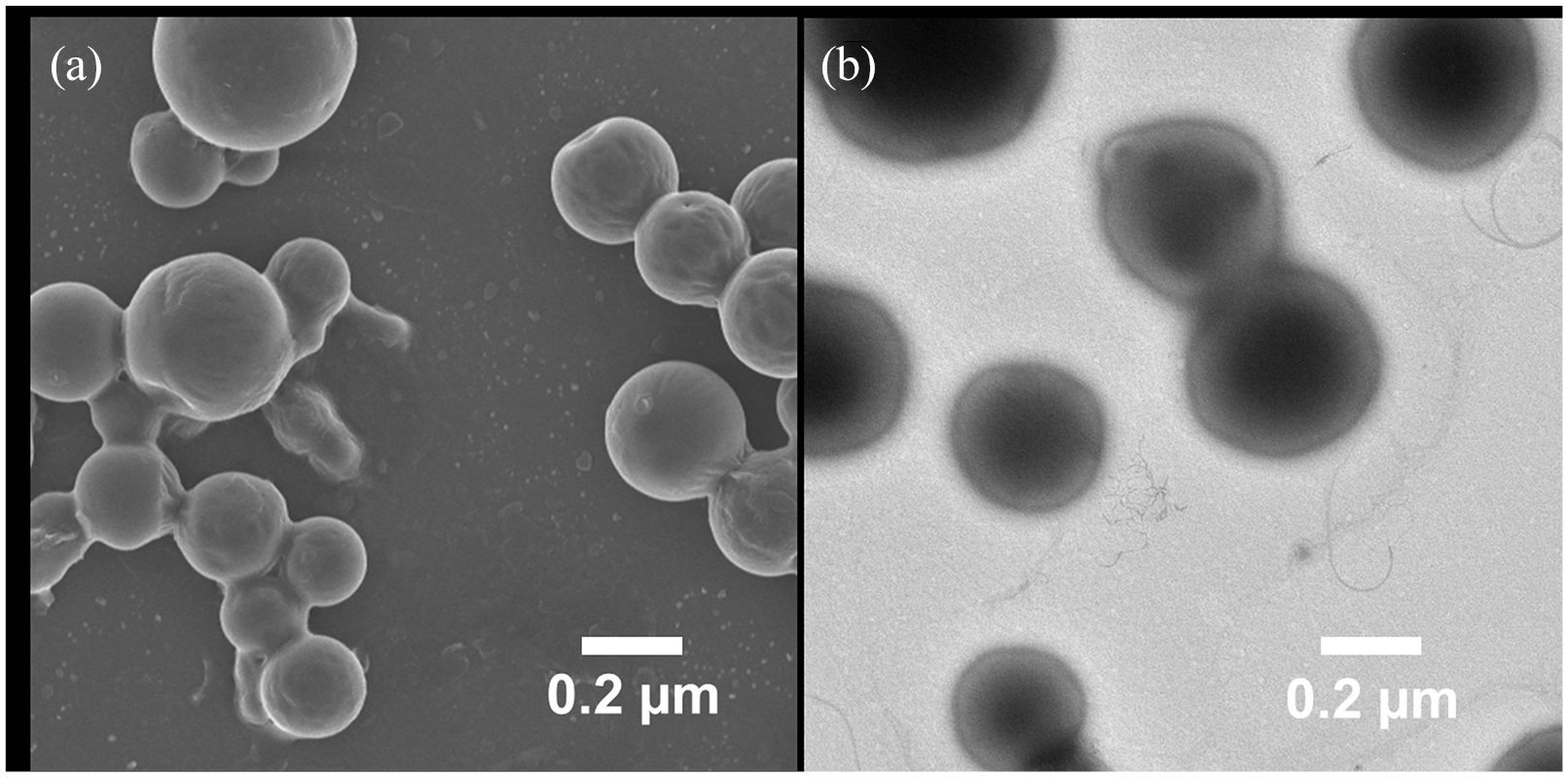
(a) scanning electron microscopy image showing the morphology of brinzolamide–phosphorothioates–polymer poly-(DL-lactic acid-co-glycolic acid) (Brz-PS-PLGA); and (b) transmission electron microscopy images revealing the phosphorothioates coating on the Brz-PS-PLGA as a light black layer over the dark black polymer poly-(DL-lactic acid-co-glycolic acid) core.
Characterization of brinzolamide–polymer poly-(DL-lactic acid-co-glycolic acid) (Brz-PLGA) and brinzolamide–phosphorothioates–polymer poly-(DL-lactic acid-co-glycolic acid) (Brz-PS-PLGA).

Note: values are expressed as means ± standard deviation.
The CEP method could generate the lipid shell particles in a single step. It also provides the flexibility to generate the particles with different coating thickness by changing the lipid concentration. Also, the concentration of drug and the PLGA could be tuned based on the purpose of fabrication. The thickness of PS and the concentration of PLGA could play an important role in the drug release, and it also influences the drug loading. Furthermore, surface of the prepared particles could also be modified to induce functionalization for targeted delivery.
Cumulative drug release profiles
In vitro release experiments of Brz from Brz-PS-PLGA were evaluated in STF. A fair comparison of drug release was performed between Brz-PLGA and Brz-PS-PLGA along with free Brz. The particles and free Brz were loaded in a dialysis bag at an equivalent concentration of Brz. The comparison with Brz release was conducted until the complete release was obtained as shown in Figure 4. The release profiles were much sustained for the Brz-PS-PLGA which is justified due the presence of lipid shell which might have sustained the release of the Brz from the particles. Release of free Brz was completed within 8 hours. The comparative sustained release of Brz from Brz-PS-PLGA could be possibly due to the PS layer, which is the layer over the polymeric core entrapping the drug. The PS layer provides an additional barrier for diffusion of the drug after it has been diffused from the polymer; thus, it might be possible to tune the release pattern of the drug from the core shell particles by modifying the thickness of the shell.

In vitro dissolution profiles of brinzolamide (Brz) from free Brz, brinzolamide–polymer poly-(DL-lactic acid-co-glycolic acid) and brinzolamide–phosphorothioates–polymer poly-(DL-lactic acid-co-glycolic acid) conducted in phosphate buffer saline solution (pH 7.4) at 37°C.
Cytotoxicity assays
The cytotoxicity assays of free Brz, Brz-PLGA, and Brz-PS-PLGA were carried out using L929 cell lines and ARPE-19 cell lines. The MTT assay revealed that the cytotoxicity caused by free Brz, Brz-PLGA, and Brz-PS-PLGA was < 10% at an equivalent concentration of Brz. The effects of free Brz, Brz-PLGA, and Brz-PS-PLGA on the L929 cell lines and ARPE-19 cell lines were comparable and the results clearly revealed that the developed Brz-PS-PLGA was completely nontoxic to the cells and could be used effectively. The cytotoxicity results are shown in Figure 5(a) and Figure 5(b).

The cytotoxicity assays of free brinzolamide, brinzolamide–polymer poly-(DL-lactic acid-co-glycolic acid) and brinzolamide–phosphorothioates–polymer poly-(DL-lactic acid-co-glycolic acid): (a) L929 cell lines; and (b) ARPE-19 cell lines (n = 3).
Corneal permeability and IOP studies
The Brz-PS-PLGA were developed in order to improve the bioavailability of Brz after topical administration by enhancing its penetration in the cornea. As illustrated in Figure 6, the corneal penetration of Brz from free Brz, Brz-PLGA, and Brz-PS-PLGA was evaluated for up to 5 hours. The results revealed that Brz-PS-PLGA significantly enhances the Brz permeation in comparison with Brz-PLGA and free Brz. The enhanced corneal penetration of Brz-PS-PLGA could be possible due to the presence of PS facilitating its binding with the corneal membrane. Also, the presence of PS shell over the PLGA core with high encapsulation of the Brz, might have enhanced the penetration of Brz-PS-PLGA across the cornea. PS is a phospholipid present in the cell membrane which could be easily recognized by the cells, and this might result in the enhanced penetration of PS-coated NPs.

In vitro corneal permeability studies of brinzolamide, brinzolamide–polymer poly-(DL-lactic acid-co-glycolic acid) (Brz-PLGA) and brinzolamide–phosphorothioates–polymer poly-(DL-lactic acid-co-glycolic acid) (Brz-PS-PLGA) conducted up to 24 hours using a Franz diffusion chamber (*p < 0.05; Brz-PS-PLGA vs free Brz, #p < 0.05; Brz-PS-PLGA vs Brz-PLGA).
Next, we performed the effect of Brz-PS-PLGA on the IOP reduction following the administration of free Brz, Brz-PLGA, and Brz-PS-PLGA in the rabbit eye and these results were compared with those of the PBS-treated group. The results clearly revealed that free Brz and Brz-PLGA were capable of reducing the IOP after administration to the eye of the rabbit. However, Brz-PS-PLGA were capable of significantly reducing IOP in comparison to the control, free Brz, and also Brz-PLGA. We did not observe any reduction in the IOP of PBS-treated animals. The results are shown in Figure 7. The decrease in IOP by free Brz and other preparations could be attributed to the release pattern of Brz from Brz-PLGA and Brz-PS-PLGA. It was observed that in the initial hours free Brz, Brz-PLGA, and Brz-PS-PLGA reduce the IOP, followed by an increase in the IOP. However, a sustained IOP was observed in Brz-PLGA and Brz-PS-PLGA. The significant (p < 0.05; Brz-PS-PLGA vs free Brz, p < 0.05; Brz-PS-PLGA vs Brz-PLGA and p < 0.05; Brz-PS-PLGA vs control) reduction in the IOP was observed in Brz-PS-PLGA up to the 24 hours study in comparison to the other groups. The results could be ascribed to the existence of PS which might have bound to the corneal membrane and released Brz in a sustained manner. Also, the PS could be recognized by several receptors. The interaction of PS with the proteins of these receptors results in the higher uptake of PS-coated NPs, and results in reduction of IOP. The initial IOP observed for rabbits’ eyes were 14.71 mmHg. However, the left eye of the rabbits after the treatment revealed an average IOP of 11.56 mmHg. Although Brz-PS-PLGA was administered topically, reduction in IOP in the untreated eye was also observed—this might be due to systemic absorption of the Brz-PS-PLGA.

The effect on intraocular pressure administration of free brinzolamid, brinzolamide–polymer poly-(DL-lactic acid-co-glycolic acid) (Brz-PLGA) and brinzolamide–phosphorothioates–polymer poly-(DL-lactic acid-co-glycolic acid) (Brz-PS-PLGA) in comparison with control (*p < 0.05; Brz-PS-PLGA vs free Brz, #p < 0.05; Brz-PS-PLGA vs Brz-PLGA and †p < 0.05; Brz-PS-PLGA vs control).
Conclusions
The Brz-PS-PLGA were developed by the CEP method which enables the preparation of mono-dispersed PS shell over drug-loaded PLGA particles in a single step with high encapsulation efficiency. The CEP results in the rapid production of the particles with shell and core, without affecting the consistency of the produced particles. The particles prepared were uniform in size and the PS coating was clearly observed. The PS coating and the core shell structure of Brz-PS-PLGA sustained the release of Brz. The Brz-PS-PLGA were non-toxic to the fibroblast cell lines and also to the retinal epithelial cell line. Furthermore, PS coating on the particles also allows the significant enhancement in coronial penetration of Brz from Brz-PS-PLGA in comparison to free Brz, and without PS coating Brz-PLGA. The significant reduction in the IOP was observed in Brz-PS-PLGA in comparison to other groups. In summary, the results suggest that Brz-PS-PLGA might provide the future treatment regimen of glaucoma.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The presented work was supported by the National Science Foundation of China.


