Abstract
Objective:
The aim was to compare restorative marginal integrity of ceramic crowns luted with bioactive and resin cements using micro-computed tomography (micro-CT) microleakage evaluations and bond strength assessment.
Methods:
Thirty molar teeth were prepared by sectioning and polishing for dentin exposure for resin cement build-ups. Teeth were randomly divided among three groups of cements: (1) bioactive (ACTIVA); (2) glass ionomer cement (GIC; Ketac Cem); and (3) resin luting agent (Nexus 3). Bonding regime and build-ups (4 mm × 2 mm) were performed using the recommended protocol. For microleakage assessment, 30 premolar teeth were prepared for dentin-bonded crowns using lithium disilicate ceramic and the computer-aided design and computer-aided manufacturing technique. Crowns were cemented with standard load, cement amount, and duration using three cements (Group A: bioactive; Group B: GIC; Group C: resin) and photopolymerized. Cemented crowns were evaluated for volumetric infiltration using micro-CT (Skyscan, Bruker 1173- at 86 kV, 93 µA, 620 ms) after immersion in 50% solution of silver nitrate (AgNO3) (24 hours). Shear bond strength (SBS) was assessed by fracture of cement build-ups at a cross-head speed of 0.5 mm/min in a universal testing machine.
Results:
Mean SBS among bioactive (21.54 ± 3.834 MPa) specimens was significantly higher than that for GIC (14.08 ± 3.25 MPa) specimens (
Conclusion:
Bioactive cement exhibited less microleakage and comparable SBS to resin luting agents in in vitro conditions.
Introduction
Ceramic restoration in the form of indirect crowns and fixed partial dentures form the mainstay of oral rehabilitations. Contemporary all-ceramic materials exhibit excellent esthetic attributes and dentin bonding (dentin-bonded crowns) augments mechanical properties, improving the potential for successful oral prognosis.1,2 However, the cementation of indirect restorations produces a vulnerable interface between the restoration and dentin surface, which is prone to a myriad of complications. These complications at the interfacial restorative margin can range from minor staining to formation of micro/nano size gaps resulting in plaque accumulation, sensitivity, and secondary caries. 3 It is a prerequisite that restorative luting agents should demonstrate formation of a seal at the margins to prevent leakage of contaminants. 4
Ideal luting agent properties include low chemical solubility, wear resistance, dimensional stability, esthetic durability, low viscosity, bioactivity, and bacterial resistance. 5 Conventional acid–base cements (zinc phosphate and glass ionomer cement (GIC)) show high solubility and low mechanical resistance in the presence of bioactivity and fluoride release. 6 Commonly used resin luting agents (polymerization reaction) show excellent translucency, controlled setting, low cement film thickness, resistance to solubility post-polymerization, and mechanical strength. 7 However, resin cements are sensitive to moisture, undergo dimensional changes (polymerization and thermal), show minimum bacterial resistance, and lack dentin remineralization potential.5 –7 Therefore, the use of acid–base cements is discouraged for ceramic cementations. By contrast, resin cements are employed for cementation, although fail to create a natural chemical bond to dentin and lack bioactivity to prevent microleakage. 7 It is proposed that contemporary bioactive esthetic materials, which associate with oral fluids and show recharge and renewal of restorative material constituents, have the potential to reduce bacterial microleakage and enhance marginal integrity when used as luting agents.8 –10
A contemporary bioactive material (ACTIVA, Pulpdent) has been introduced in recent years for restorative and luting purpose of indirect ceramic restorations. The material integrates bioactivity by showing an exchange of ions, based on oral pH, between oral fluid and the ionic resin matrix. 9 This bioactive material is based on silica glass particles and an ionic-based resin matrix with calcium, phosphate, and fluoride ions. 9 It is claimed by the manufacturer that bioactivity facilitates the benefits of improved durability, antimicrobial resistance, the chemical bond with dentin, and minimizes leakage due to oral contaminants. 9 Owens et al., 11 in a study on microleakage assessment of bioactive restorative materials using methylene blue dye and class V cavities, revealed no difference in microleakage for bioactive and resin restorative materials. However, in a similar study, resins showed better resistance to microleakage than bioactive restorative materials using dye penetration methods. 12 Therefore, there appears to be a controversy with regards to the comparative microleakage potential of bioactive materials. In addition, these studies employed dye penetration and sectioning methods (two-dimensional) for microleakage evaluations. Microleakage occurs in three dimensions and its assessment in restorative material using micro-computed tomography (micro-CT) has been reported.13,14 It is hypothesized that restorative interfacial bond integrity (microleakage and bond strength) for bioactive luting agents will be comparable to resin-based cements when assessed using the micro-CT evaluation method. Therefore, the aim of the study was to compare restorative marginal integrity (microleakage and adhesive bond) of ceramic crowns luted with bioactive and resin cements using micro-CT evaluations and bond strength assessment.
Methods
The variables tested in the study were shear bond strength (SBS) and microleakage in dentin-bonded all-ceramic crowns cemented with bioactive cement in comparison with GIC and conventional resin-based cements. The testing was performed according to the “checklist for performing in vitro studies (CRIS guidelines).”
Specimen preparation
For bond strength testing, 30 extracted human teeth (molars) were prepared and stored in a 0.1% solution of thymol (Fisher Scientific, New York, USA). The teeth were washed and mounted vertically using a surveyor in orthodontic acrylic resin (Caulk Orthodontic Resin, Dentsply Caulk, MN, USA) in cross-sections of polyvinyl pipes. To expose fresh dentin, the buccal surface of each tooth was cut 3 mm deep, using a grinding machine (Buehler, MN, USA). A dentin area of around 4 mm diameter was exposed for application of the bonding procedures. The dentin surface was polished on a rotary polishing machine (Aropol 2V, Arotec) at 250 rpm with a 400–500 silicone carbide disc. All teeth were placed in distilled water in an ultrasonic bath (5 min) for dentin surface cleaning. All teeth were randomly allocated to three groups (
Group 1: bioactive (ACTIVA bioactive cement, Pulpdent, MA, USA);
Group 2: GIC (Ketac Cem- Maxicap, 3M ESPE, MN, USA);
Group 3: resin (Nexus 3 Third Generation, Kerr, CA, USA).
All cements were applied to the dentin surface according to manufacturers’ instructions. A putty mold was prepared, customized to the tooth–resin mounting with build-up dimensions of 4 mm × 2 mm. For bioactive cement build-up, dentin was dried with cotton pellets to remove external moisture without desiccation. Cement was dispensed with an automix syringe into the mold secured on the tooth. The excess was removed with a plastic instrument and photo-polymerization was performed (Bluephase, Ivoclar, Vivadent) for 20 s (twice) at a distance of 5 mm, before removal of the mold. For resin cement build-up, dentin was etched with 34% phosphoric acid (EZ Etch, Dentsplysirona, MI, USA) for 15 s and washed and dried for 20 s. A bonding agent was applied (Prime & Bond NT, Dentsply, NY, USA) for 20 s and light cured (Bluephase, Ivoclar, Vivadent) for 10 s. Nexus cement (Dualcure) was then applied to dentin using the mold and photo-polymerized for 40 s. GIC build-up on dentin was made following dentin treatment similar to specimens in the bioactive group. All bonded specimens were exposed to thermocycling for 10,000 cycles between 5ºC and 55ºC water temperature, with a dwell time of 30 s (Thermocycler, GMBH, Miebacher Strabe, Germany).
For microleakage assessment, 30 human premolar teeth were mounted in orthodontic resin (Caulk Orthodontic Resin, Dentsply Caulk, MN, USA) vertically in cross-sections of polyvinyl pipes, 2 mm below the Cemento-enamel junction (CEJ). All teeth were prepared for dentin-bonded all-ceramic crowns with 1.7 mm occlusal, 1.5 mm axial reductions, 1 mm heavy chamfer (0.5 mm above CEJ), 4.5 mm height, and 16 degree total convergence angle (single operator-MT). Tooth preparations were standardized using multiple putty indices. For crown fabrication, impressions of individual tooth preparations were recorded using impression trays and a combination of light body and regular body polyvinyl siloxane (PVS) impression materials (Express Impression Material, 3M ESPE, MN, USA). Stone (SheraPure Diestone, Auckland, New Zealand) dies were prepared by trimming and spray of contrast (IPS Labside, contrast spray, Ivoclar Vivadent, NY, USA). Dies were scanned for computer-aided design and computer-aided manufacturing (CAD-CAM) all-ceramic lithium disilicate (EMax Cad, Ivocalr Vivadent, NY, USA) crown fabrication. CAD-CAM fabrication included die scanning (Ceramill Map 400, Amann Girrbach, NC, USA) and designing on STL files using EvoCad software (design software, Amann Girrbach, NC, USA). Crown design comprised thicknesses of 2 mm at the cusp tip and 1.5 mm at the occlusal fissure, 1 mm axial surface, and 0.05 mm of cement space, 2 mm above the margin. Milling of ceramic crowns was performed by Ceramill Motion 2 (Amann Girrbach, NC, USA) by an experienced laboratory technician. The crown surfaces were etched with 9.5% hydrofluoric acid (HF acid, Ceram-Etch Gel, Gresco products, TX, USA) for 30 s and the application of silane (Monobond S, Ivoclar Vivadent, NY, USA) after washing with acid.
Crown-tooth specimens were randomly divided into three groups based on cement type: Group A: bioactive cement; Group B: GIC, and Group C: resin cement (

(a) Cemented dentin-bonded crown. (b) Specimen placed in photo-developing solution. (c) Mounted specimen for micro-computed tomography scans.
Bond strength testing
All bonded specimens were tested for SBS under standard loads using a universal testing machine (Instron 8500 Plus, Canton). Loads were applied by securing the specimen in the load cell through a chisel probe placed parallel to the bonding interface. Loads were applied at a cross-head speed of 0.5 mm/min until fracture of build-up. The SBS values were obtained in megapascals (MPa). The interface of the debonded specimens was assessed for type of failure mode using a microscope (SZX7, Olympus, Hamburg, Germany) at 40× magnification. The failure modes were classified as adhesive, cohesive, and mixed failures.
Microleakage assessment
To assess microleakage, root surfaces of the cemented ceramic crown samples were covered with two layers of nail varnish, following which specimens were immersed in 50% silver nitrate (AgNO3) in the dark for 12 h. Following rinsing in water the samples were further immersed in photo-developing solution under a fluorescent light for 12 h (Figure 1).
Each sample was mounted and positioned inside the specimen chamber by which scanning was acquired using micro-CT (Bruker SkyScan 1173 (high-power), Bruker SkyScan, Kontich, Belgium). The scanner configuration used was 86 kV voltage, 93 µA anode current, 620 ms exposure time, isotropic resolution of 16 µm image pixel size, brass filter, 0.25 rotation step for 360° angle, frame averaging of 4 for improved signal to noise ratio, and random movement of 8 to minimize ring artifacts. A flat-field correction was performed before the scanning procedure in order to correct variations in the camera pixel sensitivity.
After the scanning, a reconstruction of the projected images was performed using N-Recon® program version 1.6.1.3 (Bruker Skyscan, Kontich, Belgium) to produce reconstructed cross-section images. Numerical parameters needed to establish the best image results were checked and adjusted. A ring artifact reduction of 5 for non-uniformity of the background image taken by the X-ray camera; 30% beam hardening compensation to prevent the specimen from appearing artificially denser at or near its surface, and less dense at its central parts; and a smoothing of 2 using the Gaussian kernel were applied. A 16 bit TIF file format was the choice selected for saving the images because of the variety of densities comprising the specimen.
Reconstructed images were three-dimensional (3D) registered and loaded in the Dataviewer® program version 1.5.6.2 (Bruker Skyscan, Kontich, Belgium) software to determine image quality, reorient, and resize and for accurate positioning and visual inspection. Every slice pertaining to the region of interest was scrutinized for silver nitrate penetration, as would be evidenced by an intense grayish shade along the interface. The volume of silver nitrate penetration was quantified with the CTAn® program version 1.17.7.2 (Bruker Skyscan, Kontich, Belgium) by selecting a binarized threshold value corresponding to the amount of silver nitrate exhibited in cubic millimeters (mm3).
Data obtained both for SBS and microleakage volume was assessed for normality (Kolmogorov–Smirnov test). A statistical software (Graphpad Instat, NY, USA) was employed to identify means and standard deviations among the study groups. Using analysis of variance (ANOVA) and Tukey–Kramer multiple comparisons tests, the statistical difference among study groups was analyzed. A
Results
The maximum and minimum SBS among study groups were observed among Group 3 (resin, 24.73 ± 4.32 MPa) and Group 2 (GIC, 14.08 ± 3.25 MPa) specimens. Overall mean SBSs among the study groups were significantly different (
Comparisons of means and SDs of shear bond strength among study groups.
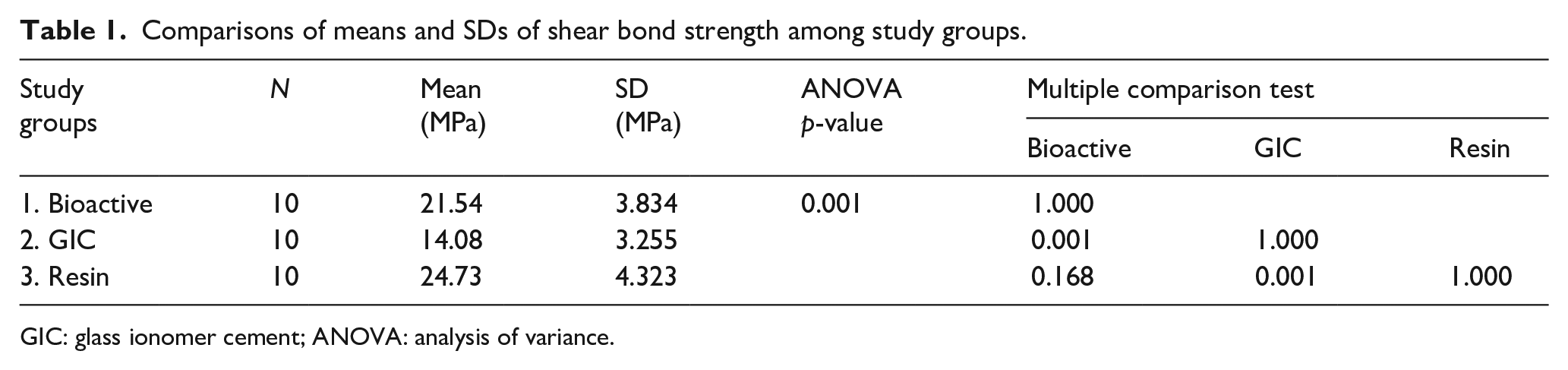
GIC: glass ionomer cement; ANOVA: analysis of variance.
Failure modes among study groups for shear bond strength test.

Regarding microleakage scores, the means and standard deviations in cubic millimeter (mm3) are presented in Table 3 and Figure 2. Highest and lowest microleakage scores were observed in GIC Group B (1.057 ± 0.399 mm3) and bioactive (ACTIVA Group A) (0.381 ± 0.134 mm3) samples (Figures 3 and 4). Overall microleakage scores among the study groups were significantly different (ANOVA
Comparison of means and SDs for microleakage among study groups.

GIC: glass ionomer cement; ANOVA: analysis of variance.

Graphical presentation of microleakage in volume (cubic mm) among different cement study groups. GIC: glass ionomer cement.

Image showing silver nitrate penetration in coronal (a), transverse (b), and sagittal (c) slices of micro-computed tomography images, in a bioactive specimen.
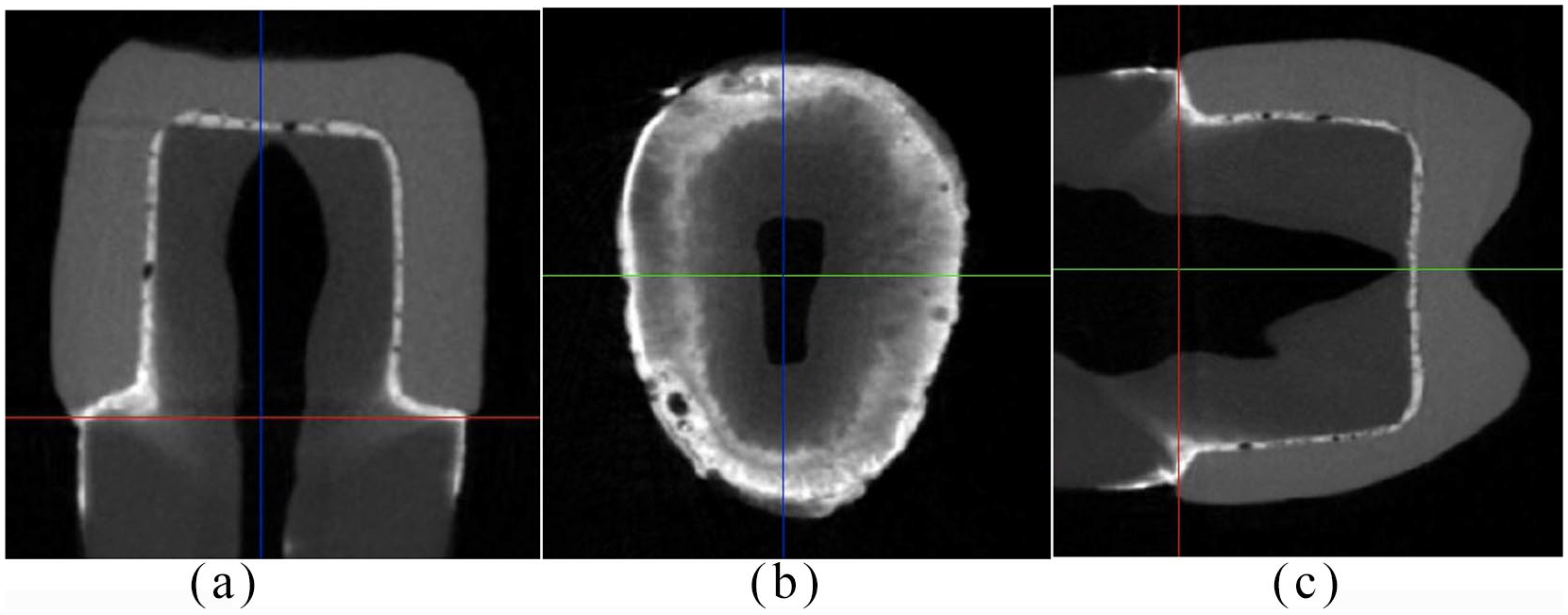
Image showing silver nitrate penetration in coronal (a), transverse (b), and sagittal (c) slices of micro-computed tomography images, in a glass ionomer cement specimen.

Image showing silver nitrate penetration in coronal (a), transverse (b), and sagittal (C) slices of micro-computed tomography images, in a resin specimen.
Discussion
The present study was based on the hypothesis that restorative interfacial bond integrity (microleakage and adhesive bond) for bioactive luting agents is comparable to resin-based cements when assessed using micro-CT and bond strength evaluation methods. The experiments revealed that the bioactive luting agent exhibited lower microleakage than comparative groups, but its bond strength to dentin was statistically comparable to resin luting agents. Therefore, the hypothesis was partly accepted. The explanations for these findings are manifold, including chemical compositions of the materials and their interaction with tooth dentin.
Microleakage is related to the interfacial contamination of bacteria through ingress of oral fluids. The present study is unique as it evaluated volumetric leakage at the cement–ceramic crown (non-sectioned specimen) interface, in contrast to commonly used class V cavity configurations, which are not restored with luting cements clinically. The microleakage interface is assessed with either the use of tracer compounds followed by penetration evaluation or analysis of restorative marginal gap using microscopes. Multiple agents are employed in microleakage studies, including tracers like methylene blue, erythrosine, and eosin; radioactive markers, silver nitrate, and the bacterial penetration technique. 15 Some of these tracer compounds are not stable and react with acidic primers in luting agents and hydroxyl ions to produce colorless molecules, showing false negative outcomes. 16 In addition, tracers can also show solubility and can be washed out if used in combination with sectioning techniques. By contrast, silver nitrate does not show dissolution as it reacts with substances in the developer solution and forms opaque precipitates of metallic silver, and therefore it was used in the present study. Studies have shown variations in penetration of different tracers when compared in a single study, and therefore concluded that comparison among microleakage studies was not possible due to differences in methodological parameters.13,16
The use of dye and tracer penetration methods is commonly combined with sectioning of specimens for evaluation of tracer penetration. This allows for a two-dimensional assessment of the 3D phenomenon of microleakage, which is non-uniform in nature. 17 In addition, the majority of studies employ one or two sections of specimens, limiting points of evaluations, and therefore producing underestimated or overestimated results with little validation.13,18 By contrast, a volumetric visualization of a radio-opaque silver nitrate through micro-CT allows for non-destructive evaluation with the possibility of re-evaluation over a period of time. 19 Therefore, the present study employed a silver nitrate tracer and micro-CT X-ray visualization in a standardized protocol for the evaluation of microleakage at the luting agent–dentin interface of cemented ceramic crowns.
In the present study, microleakage scores were significantly lower for bioactive luting agents as compared to resin-based luting cements. Bioactive cement (ACTIVA) is based on the ionic resin matrix responsible for the chemical bond between the material and the dentin. In the presence of water, ionization causes the replacement of hydroxyl groups in phosphate acid of the matrix with the calcium in dentin, resulting in a chemical bond. 20 This ionic exchange results in binding of the bioactive luting agent to the tooth structure, forming a hydroxyapatite complex and a marginal seal. 21 In addition, the absence of bonding agent application for bioactive cement allows one to prevent fluid percolation in and out of the hybridized complex of dentin and bioactive cement. Furthermore, bioactive cements are claimed to extract fluoride, calcium, and phosphate ions from the environment, based on the changing pH. This allows for revitalization of the cement and dentin, resulting in a durable chemical bond and seal at the dentin cement complex. 9 Moreover, the comparatively higher microleakage in the resin cement specimens could be attributed to the absence of hydroxyethylmethacrylate (HEMA) in the bonding agent (Prime & Bond NT). The peptides in the dentin collagen are reported to create peptide bonds with the hydrogen (in the absence of HEMA), making the collagen susceptible to collapse, and hence compromising the seal and bond integrity. 22 Similar outcomes have been demonstrated in previous studies.23,24
Bond strength assessment was performed to evaluate the integrity of dentin bond to luting agents in the present study. Resin-based cement showed higher but statistically comparable bond strength to the bioactive specimens. Previous studies have shown similar outcomes.23,25 The higher SBS values could be attributed to the conditioning of dentin with acid etching followed by the application of bifunctional adhesive. Acid etching produces a high-energy uniformly patterned surface of dentin, allowing deeper penetration with hydrophilic adhesives in the bonding agent. Acid etched dentin surfaces produce a hybrid layer with improved thickness, which results in a strong mechanical dentin–resin complex, and therefore showing higher bond strength outcomes.26,27 The quality of the adhesive bond for bioactive and resin cements was also validated by the majority presence of adhesive failures in both observed groups.
The findings of the study suggest that bioactive cements have the ability to produce an adhesive interface with lower volumetric microleakage, and are comparable SBS to resin luting agents. Therefore, with regards to the interfacial integrity of cemented indirect ceramic restorations, bioactive cement (ACTIVA) can compare to resin luting agents in durability. However, it is pertinent to mention that the methods employed in the assessment of microleakage are not standardized and comparisons of outcomes observed to other studies is not justified. In addition, the in vitro study assessed a clinical in vivo phenomenon, with limitations of not having an intra-oral environment. The conclusion of in vitro microleakage screenings cannot be strictly applied to intra-oral conditions; however, as these findings draw comparisons to resin luting agents, further randomized controlled trials evaluating bioactive cements and their influence on clinical restoration durability are recommended. However, as resin cements show superior mechanical and physical characteristics, further studies comparing the surface roughness, flexural strength, and durability of bioactive cements are recommended.
Conclusions
Within the study limitations, it was observed that bioactive cement showed lower volumetric microleakage than resin cements and GICs using a combination of silver nitrate and the micro-CT assessment technique. Furthermore, the SBS to dentin of bioactive luting agents was comparable to that of resin luting agents (total-etch adhesive) in in vitro conditions.
Footnotes
Contributorship
FA, MT, TA, and AA: data collection, study design, manuscript writing, final manuscript approval. YA and MA: data collection, study design, manuscript drafting, data analysis, and manuscript approval. MAd and AAM: data collection, manuscript approval, and data interpretation. KA, LA, and MA: data collection, writing, revise, and editing and final manuscript approval.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Researchers supporting project at King Saud University for funding through Researchers supporting project No. (RSP-2019-44).
Guarantor
Tariq Abduljabbar.
