Abstract
This study was to develop a feasible and safe animal model for minimally invasive injectable lumbar interbody fusion using a novel biomaterial, mineralized collagen-polymethylmethacrylate bone cement (MC-PMMA), with unilateral pedicle screw fixation in an in vivo goat model. Eight goats (Capra aegagrus hircus) were divided into three groups: MC-PMMA, unmodified commercial-polymethylmethacrylate bone cement (UC-PMMA), and a control group (titanium cage filled with autogenous bone, TC-AB). Each group of goats was treated with minimally invasive lumbar interbody fusion at the L3/L4 and L5/L6 disc spaces (injected for MC-PMMA and UC-PMMA, implanted for TC-AB). The pedicle screws were inserted at the L3, L4, L5, and L6 vertebrae, respectively, and fixed on the left side. The characteristics of osteogenesis and bone growth were assessed at the third and the sixth month, respectively. The methods of evaluation included the survival of each animal, X-ray imaging, and 256-layer spiral computed tomography (256-CT) scanning, imaged with three-dimensional microfocus computed tomography (micro-CT), and histological analysis. The results showed that PMMA bone cement can be extruded smoothly after doping MC, the MC-PMMA integrates better with bone than the UC-PMMA, and all goats recovered after surgery without nerve damage. After 3 and 6 months, the implants were stable. New trabecular bone was observed in the TC-AB group. In the UC-PMMA group a thick fibrous capsule had formed around the implants. The MC-PMMA was observed to have perfect osteogenesis and bone ingrowth to adjacent bone surface. Minimally invasive injectable lumbar interbody fusion using MC-PMMA bone cement was shown to have profound clinical value, and the MC-PMMA showed potential application prospects.
Introduction
Interbody fusion is generally preferred over other fusion techniques to treat symptomatic lumbar degenerative disease. Common intervertebral bone devices including polyetheretherketone (PEEK) have disadvantages of rejection, cross-infection, poor osteogenic effects and biocompatibility, damage to the nerve and dural sac, epidural scar formation, destruction of epidural small blood vessels, and with no degradation or rapid degradation.1-5 The use of autologous iliac crest bone is the gold standard, but some complications cannot be ignored.6,7
However, polymethylmethacrylate (PMMA) bone cement has advantages such as injectability, immediate cure, and good mechanical properties and operating performance.8,9 PMMA has become one of the most common bone substitutes in clinical applications.10-12 However, it still has some disadvantages, such as being non-bioactive, non-unifiable with the host’s bone, and non-osteoconductive. Therefore, it is unable to stimulate bone formation. To improve upon PMMA-based cement–bone interaction to increase its biocompatibility and osseointegration capacity, many attempts have been made to modify the bone cement by various strategies, such as adding hydroxyapatite or chitosan to PMMA, changing monomer components, etc.13-17 However, there has been no ideal modification method that meets the requirements of clinical applications.13-17
This study is based on the scientific research of a modification to PMMA bone cement using a biomimetic mineralized collagen (MC) artificial bone graft in terms of both biomechanical properties and biocompatibility.18,19 The nano-hydroxyapatite (HA)-coated bone collagen (mineralized collagen, patent number: ZL01129699.2, MC) is a combination material of recombinant human collagen and nano-hydroxyapatite that is made in vitro. 20 In previous studies, MC in different proportions was added to the PMMA bone cement to detect the compressive strength, compression modulus, coagulation properties, and biosafety; results confirmed that 15.0% (wt) impregnated MC-PMMA had the best material properties and MC showed strong biological and osteogenic activities in bone defect repair.19-21 The MC artificial bone graft was designed and prepared based on a biomineralization mechanism of the natural bone tissue and in vitro biomimetic mineralization technology.22,23 MC has been commercialized and applied in many clinical applications, such as orthopedic, dental, neurosurgery, and other fields.24-27 In the field of spine surgery, MC has been used to promote bone regeneration, as well as to modify PMMA bone cement in percutaneous vertebroplasty (PVP) and percutaneous kyphoplasty (PKP). In previous research, the appropriate blending ratio of the MC to PMMA bone cement powder, as well as the particle size of the MC and optimal blending method, were systematically studied.28-30 Via the modification, bone formation at the interface between the bone cement and host bone could be observed, and secondary fracture was significantly decreased as shown by long-term follow-ups.31-33 Although MC-modified PMMA bone cement has many advantages compared with traditional bone cement, it has not been used for interbody fusion. Moreover, an appropriate biomaterial and applicable operative method need to be developed. This study used an in vivo goat (Capra aegagrus hircus) model to develop a feasible and safe method for minimally invasive injectable lumbar interbody fusion by using MC-modified PMMA bone cement (MC-PMMA) with unilateral pedicle screw fixation.
Methods
Experimental animal model and preoperative preparation
This study received institutional animal ethical board approval from the Orthopedic Hospital of Xingtai (No. ZCKT-0016).
Eight goats were provided by the animal experiment center of Hebei medical university. The goats were seven males and one female, aged 16–20 months, with a weight of 26–33 kg. The cattle and goat blood was 7.6–8.35% of live weight proportion. 34 Based on this reference, blood was drawn from each goat 1 week before operation for intraoperative autologous blood transfusion.
Grouping of experimental animals
The goats were divided randomly into three groups. Group 1 was injected with MC-PMMA bone cement (MC, Bongold® MC bone graft material, Beijing Allgens Medical Science and Technology Co., Ltd. China), group 2 was injected with unmodified commercial PMMA bone cement (UC-PMMA, OSTEOPAL® V, Heraeus Medical GmbH, Germany), while those in the control group (group 3) were implanted with a titanium cage filled with autograft (TC-AB, titanium cage, ZTRH01, ¢10×3 mm, Jiangsu Ideal Medical Science and Technology Co., Ltd. China). The operative intervention was applied to the animals grouping described below (Table 1). Every goat was operated at two lumbar vertebral spaces: L3–L4 and L5–L6, respectively. The material-embedded goat bone tissues was harvested after sacrifice at the third month (Nos. 1–4) and the sixth month (Nos. 5–8), respectively.
Grouping of experimental animals.

MC-PMMA: Mineralized collagen-polymethylmethacrylate bone cement; UC-PMMA: Unmodified commercial-polymethylmethacrylate bone cement; TC-AB: Titanium cage filled with autogenous bone. All eight goats were inspected via X-ray photography and thin spiral CT examination postoperation immediately and before the specimens were harvested. For goat Nos. 1–4, the material-embedded intervertebral space was harvested after sacrifice at the third month. The specimens were imaged with three-dimensional micro-CT and histological analysis. For goat Nos. 5–8, (1) at the third month after surgery, they were inspected via X-ray photography and thin spiral CT examination. (2) The material-embedded intervertebral space was harvested after sacrifice at the 6th month. The specimens were imaged with three-dimensional micro-CT and histological analysis.
Anesthetic protocol
All goats were premedicated with atropine half an hour prior to surgery (0.05 mg/kg intravenous injection) and xylazine hydrochloride injection (0.01 g/kg, diluted with 0.9% sodium chloride injection to 10% concentration). Local anesthesia was used prior to skin incision to improve surgical safety; this consisted of 0.2 g of lidocaine hydrochloride injection mixed with 178.8 mg of ropivacaine mesylate, diluted with 20 ml of 0.9% sodium. During anesthesia, endotracheal intubation was performed. Anesthetic monitoring was connected to the goat, including cardiac electric activity, respiratory rate, partial CO2 concentration in expired air, partial blood O2 concentration, and noninvasive arterial blood pressure.
Surgery
After preoperative disinfection, the pedicles of the vertebral arch of the operated level were marked on the skin with a C-arm machine. A midline incision was performed at the waist, and through the skin and subcutaneous tissue. The left side of the paravertebral muscle was stripped, the L3 to L6 facet joint and laminar space were exposed, and pedicle screws were inserted at L3, L4, L5, and L6 and fixed on the left side.35-37 Intraoperative fluoroscopy confirmed the position of pedicle screws were proper. A partial laminectomy was performed between L3 and L4, and L5 and L6, and a partial facetectomy was performed at the inferior facet joint of L3 and L5, respectively. Generally speaking, in order to not pull the dural sac, removal of most or the entire left side facet joint was required. Then the pedicle screws were used to moderately open the intervertebral space to expose the adjacent upper segmental nerve roots between L3 and L4, and L5 and L6 prior to disc excision. This was a key point when removing the intervertebral disc in order to gently open the nerve root under direct vision. This allowed for exposure of the rear side of annulus fibrosus so that a window could be created to remove as much disc tissue as possible. A special intervertebral processor was then used for the upper and lower vertebral endplates to reduce interference with the nerve root. The purpose of the above operative steps was to remove the cartilage endplates and clean the intervertebral space. There was intraoperative neurophysiological testing and monitoring with electrocardiography (ECG) during surgery.
Implantation of biomaterials
The MC powder was prepared as reported previously. 22 For the UC-PMMA and MC-PMMA bone cement preparation, briefly, MC was added to unmodified commercial PMMA bone cement (UC-PMMA) during the early period of the dough phase, and then mixed thoroughly for the MC-PMMA group. The UC-PMMA and MC-PMMA bone cements were prepared at 23°C with relative humidity of 50%. The sterile cement was then injected into the intervertebral space by the bone cement-filler system device. We found the mean time of injection was less than 1 min, and the injected volume was approximately 0.6 ml (Figure 1). The titanium cage filled with trivial osseous blocks was implanted according to the height of the intervertebral space. In order to make the bone cement or titanium cage closely integrated with the endplates, we properly pressurized after interbody fusion. For group 1 and group 2, the intervertebral space was expanded before injecting the bone cement. The distribution of bone cement could then be observed in perspective. Then we installed the titanium rods embedded in pedicle screws, and the intervertebral space pressurized.

(a) The preparation of MC-PMMA and PMMA bone cement before injection, (b) MC-PMMA or PMMA bone cement was injected into the intervertebral space.
Postoperative care
The X-ray image and 256-layers spiral computed tomography (256-CT) scans were performed immediately after surgery. The animals were treated with antibiotics (1 g/ day, subcutaneous) for 3 days. The animals received an intravenous drip (0.9% sodium chloride, 5% dextrose, and compound amino acid injection 17AA-I 250 ml, respectively) once a day for 2 days. After the third month (Nos. 1–4) and the sixth month (Nos. 5–8) of implantation time, the animals were sacrificed by xylazine hydrochloride intravenous injection. Before harvesting the vertebral specimens, each goat was inspected with X-ray and 256-layers spiral CT scan to observe the position of the pedicles screws, rod, cage, and bone cement. Samples were then taken, and material–bone integration was assessed by micro-CT and histologic studies.
Micro-computed tomography
The material-embedded goat bone tissues were harvested after sacrifice with 0.01 g/kg xylazine hydrochloride injection at the third and the sixth month and imaged with three-dimensional microfocus computed tomography (micro-CT, SHIMADZU inspeXio SMX-90CT Plus, Japan), at a voltage of 90 kV and an electric current of 110 μA. The number of views was set at 1200, and the image matrix was 1024×1024 pixels. The fusion status between the embedded materials and surrounding bone tissues was observed via the images of the cross-sectional slices of the samples.
Histological analysis
When all the samples were imaged with micro-CT, they were analyzed with hard tissue slicing. Samples were fixed in 4% paraformaldehyde for 3 days, washed with running water for 24 h, dehydrated in serial concentrations of ethanol (70, 80, 90, and 95% vol/vol) for 5 days at each concentration, and dehydrated in 100% ethanol three times with each time lasting 5 days. After soaking, the samples were placed under vacuum for 30 min, and sealed in a refrigerator at 5°C for 3 days. After the steps were repeated two times, the sample was taken out, placed in a water bath at 27°C for curing, and placed in an oven at 60°C for further curing for 5 h, before curing, slicing, and staining.
Results
Eight goats successfully underwent minimally invasive injectable discoplasty with unilateral pedicle screw fixation. The X-ray photography and spiral thin CT showed that the position of pedicle screws, rod, cage, and bone cement was correct (Figure 2).

X-ray images of lumbar vertebrae at 6 months, (a) L3/L4 was implanted with TC-AB and L5/L6 was injected with MC-PMMA bone cement, (b) L3/L4 was injected with MC-PMMA bone cement and L5/L6 was injected with UC-PMMA bone cement.
Micro-CT scanning and evaluation
Micro-CT was used for measuring the amount and distribution of bone cement or titanium cage filled with autogenous bone for samples. The interface was sharp and clear in the UC-PMMA group, with almost no trabecular ingrowth into the cement at the third and sixth month. This phenomenon also appeared in the TC-AB group, but we could observe some trabecular bone embedded within the titanium cage (Figure 3(e) and Figure 3(f)). However, in the MC-PMMA group, the interface was closely combined between the vertebral endplate and bone cement from the third to the sixth month, forming a better interface with interspersed integration (Figure 3(a) and Figure 3(b)).
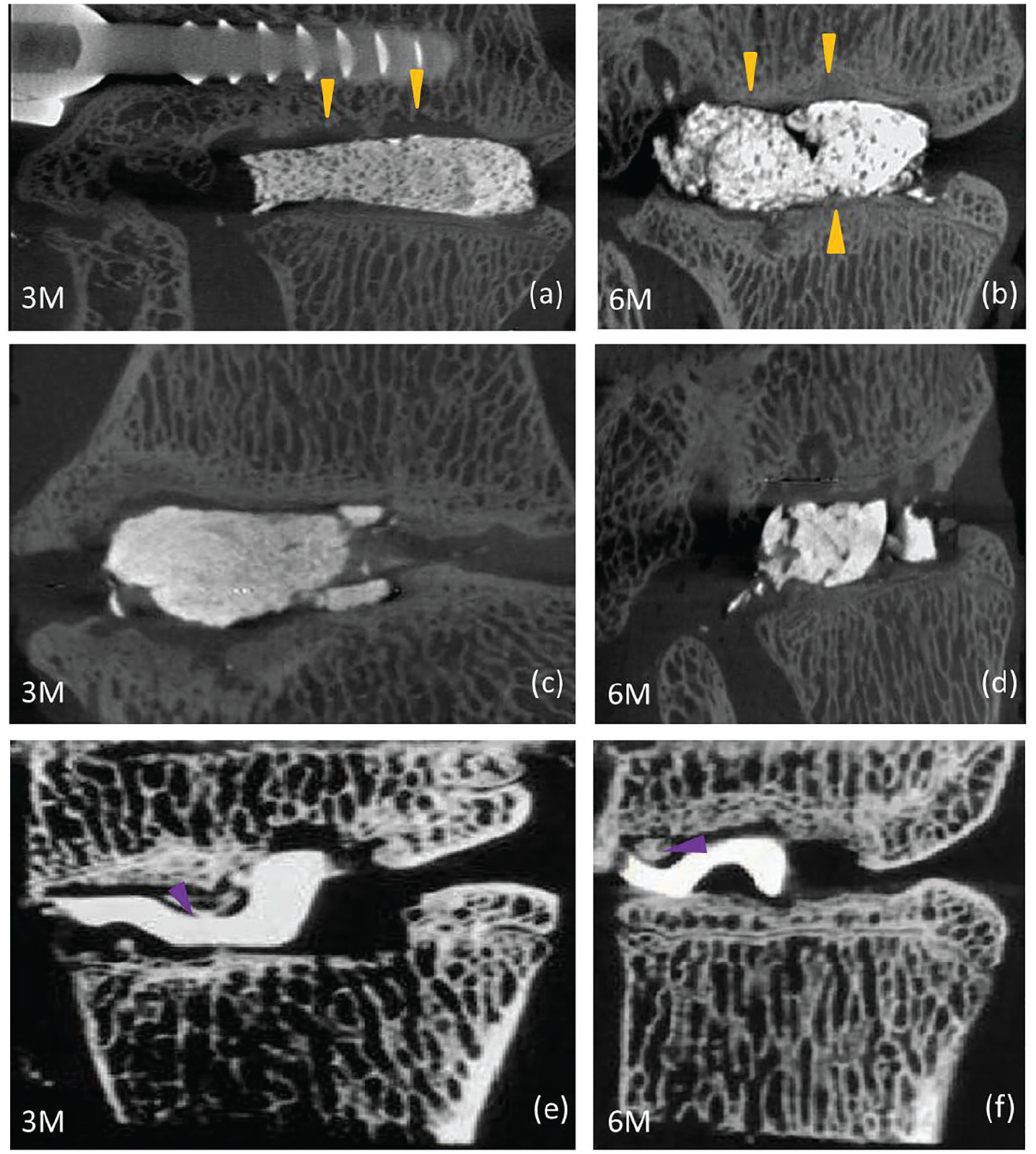
The micro-CT images of MC-PMMA (a, b) UC-PMMA (c, d) and TC-AB (e, f) 3 months (a, c, e) and 6 months after implantation (b, d, f). The yellow arrows (a, b) and purple arrows (e, f) indicate the ingrowth site of trabecular bone.
Histological evaluation
The sections of samples were polished and surface-stained with basic fuchsine and toluidine blue staining, as previously reported. 38 The staining results showed that the MC-PMMA tightly combined with the vertebral endplate. There was obvious bone tissue ingrowth into the surface of the cement, forming a firm anchor in the MC-PMMA group at the third and sixth month, as indicated by the yellow arrows in Figure 4(a) and 4(b). For the UC-PMMA group, the bone cement loosely adhered to the surrounding tissue and a thick fibrous capsule formed around the implants, as indicated by the blue arrows in Figure 4(c) and 4(d). In the TC-AB group, there was only some point contact between the two vertebral endplates and between the titanium cage and bone tissue. Regeneration of new bone tissue at the third and sixth month could be observed, with new trabecular bone indicated by the green arrows in Figure 4(e) and 4(f).

The surface-stained with basic fuchsine and toluidine blue staining of MC-PMMA (a, b), UC-PMMA (c, d) and UC-AB (e, f) after implantation for 3 months (a, c, e) and 6 months (b, d, f). Yellow arrows indicate the ingrowth site of bone tissue (a, b), blue arrows indicate fibrous capsule (c, d); green arrows indicate trabecular bone (e, f).
Discussion
Interbody fusion facilitated by spacers or cages, several different biomaterials, and transpedicular stabilization is a widely accepted surgical procedure for the treatment of certain forms of lumbar degenerative disc disease. 39 In current orthopedic practice, how to find a kind of new biomaterial that can induce osteogenesis, biocompatibility, and biosafety and use minimally invasive surgery to inject into the intervertebral space to achieve intervertebral bone fusion is a clinical problem. Many studies have tried to modify PMMA, such as with HA, whose chemical composition, Ca10(PO4)6(OH)2, is the most stable state of calcium phosphate and the main inorganic component of bone mineral. Therefore, HA is the most commonly used bioceramic based on calcium phosphate.34,40,41 Li et al. investigated the mechanical properties and bioactivity of MC-PMMA, and the results showed excellent biocompatibility and improved osteointegration. 42 Komang-Agung et al. used a caprine model to explore the optimum ratio of PMMA/HA for PVP, and they found that a mixture of PMMA and HA could increase the quality of callus formation. 34
In recent years, the discoplasty technique has been reported. Varga et al. 43 and Carlos Sola et al. 44 published their experiences with UC-PMMA as a stand-alone intervertebral spacer (percutaneous cement discoplasty in the case of vacuum phenomenon within lumbar intervertebral discs). They proved the feasibility of percutaneous discoplasty again. The results also proved the feasibility of using MC-PMMA in interbody fusion. However, more studies are needed to determine whether the percutaneous discoplasty technique is limited to the elderly, and whether it can be used in a wider population of patients with low back pain. Moreover, pure PMMA bone cement is a bioinert material that cannot osteointegrate with the host bone at the implantation site. In previous spine applications of PVP and PKP, PMMA bone cement was considered as one of the main factors inducing secondary fracture at the adjacent vertebral bodies due to its relatively high compressive modulus. So, it is necessary to improve the mechanical properties and biocompatibility of the PMMA bone cement in order to better meet the requirements of percutaneous cement discoplasty. Based on previous modification studies on PMMA bone cement using the biomimetic MC, the current study introduces a novel minimally invasive technique with MC-PMMA bone cement (Bongold®, Mineralized Collagen-Polymethylmethacrylate) to improve interbody fusion.
Using an in vivo goat model, our study evaluated the use of MC-PMMA bone cement as an injectable biomaterial for minimally invasive injectable lumbar interbody fusion with unilateral pedicle screw fixation. The goat model was chosen since it is a large animal model with vertebrae similar to human vertebrae. The bone remodeling rate is also nearly identical to that of humans. 45 In this procedure, we noticed that intraoperative autologous blood transfusion improved postoperative recovery.
We found the PMMA bone cement can be extruded smoothly after doping MC, and the results of micro-CT scanning and histological evaluation initially confirmed the feasibility and application of minimally invasive injectable lumbar interbody fusion with MC-PMMA. Meanwhile, compared with UC-PMMA, MC-PMMA was shown to have characteristics of osteogenesis and bone induction. However, the concentration distribution of the embedded MC-PMMA was non-uniform, and this may be related to the technique used for mixing the PMMA bone cement and the MC. To establish a standard operating procedure for percutaneous cement discoplasty using MC-PMMA in future clinical application, we will further study and standardize the operation specification to realize a uniform distribution of the materials. In theory, the osteogenesis of TC-AB group should be obvious; our study explains and shows this result to some degree, but the amount of osteogenesis was not complete. We considered that the problem may be related to the treated cartilage endplates and intervertebral space, or due to the bone graft—bone graft was from the vertebra lamina decompression—being unsuitable. This is a noteworthy problem and method of operation.
The goat model in particular has usually been employed to evaluate functional performance. However, large differences exist between the goat and human species in terms of the geometrical characteristics and the biomechanics of their lumbar spine systems, and differences in the normal daily activities between the two species make it rather difficult for the goat model to fully represent a human model. Furthermore, our current study included eight goats. The sample size was limited, but the present study aimed to develop a reproducible and safe model of lumbar interbody fusion and to provide a type of safe and feasible interbody material for spinal fusion. So the next work will enlarge the sample size and prolong the follow-up time to confirm this advanced and innovative technique. Biomechanical testing was not performed because we worried about damaging the integrity of fibrous tissue and bony structures formed between the vertebral endplate and the biomaterials. However, biomechanical testing should be performed in further studies to assess the stiffness and fracture strength in order to verify the bone formation and osteointegration.
Conclusion
The MC-PMMA achieved a better intervertebral fusion effect than the UC-PMMA in this in vivo percutaneous cement discoplasty study. A feasible and safe animal model for minimally invasive injectable lumbar interbody fusion using a novel biomaterial with unilateral pedicle screw fixation in an in vivo goat model was developed. The results confirmed the feasibility of this technique to treat lumbar degenerative disease, and the procedure was suitable for preclinical in vivo studies, mimicking clinical application. Further clinical transformational experimental studies are needed.
Footnotes
Acknowledgements
The authors wish to thank School of Materials Science and Engineering of Tsinghua University and Engineering Research Center in Biomaterials of Sichuan University for providing the micro-CT imaging instrument.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received financial support from The Science and Technology Project of Hebei Province in 2018 (project number 182777172).
