Abstract
Fast degradation rate and inhomogeneous corrosion are obstacles for magnesium alloy bio-corrosion properties. In this paper, a quaternary Mg-Zn-Ca-Mn alloy was designed by an orthogonal method and prepared by vacuum induction melting to investigate its bio-corrosion. Microstructure, corrosion morphology, and bio-corrosion properties of as-cast alloys 1 to 5 with good corrosion resistance were characterized by scanning electron microscopy, energy dispersive X-ray spectroscopy, and X-ray diffraction with immersion and electrochemical tests in simulated body fluid (SBF), respectively. Both the orthogonal method and in vitro degradation experiments demonstrated that alloy 3 exhibited the lowest degradation rate among the tested quaternary Mg-Zn-Ca-Mn alloys. Then, as-cast alloy 3 was treated by solid-solution and solid-solution aging. In vitro experimental results indicated that as-cast alloy 3 showed better corrosion resistance than heat-treated specimens and the average corrosion rate was approximately 0.15 mm/y. Heat-treated alloy 3 exhibited more uniform corrosion than as-cast alloy specimens. These results suggest that alloy 3 has the potential to become a biodegradable candidate material.
Keywords
Introduction
The development of magnesium alloys for biodegradable implant materials is currently under a scientific spotlight. 1 Magnesium is an essential mineral element in the human diet, and magnesium alloys have advantages over traditional metallic biomaterials, such as good biocompatibility, low density, and elastic modulus, with values which are closer to those of human bone when compared with the most commonly used metal implant materials, such as stainless steel, cobalt-chromium alloy, titanium, and titanium alloy.1,2 However, the inferior corrosion resistance, fast degradation rates, and inhomogeneous corrosion of magnesium in the human bio-environment are obstacles for their biomedical application. Therefore, developing magnesium alloys with good corrosion resistance is the main aim of research into biomedical applications of magnesium. 3
Element alloying is one of the most fascinating research areas aimed at improving the corrosion resistance as well as other properties of magnesium. However, few elements can be used in medical applications as implant materials due to biosafety. 4 Magnesium alloys containing Al and RE (rare-earth) showed relatively high strength and good corrosion resistance against NaCl solution, such as AZ91D and WE43 alloy. 5 Nevertheless, it is well known that Al has not only been linked to brain disease and Alzheimer’s disease, but inhibits the phosphorylation process and ATP synthesis, and subsequently reduces the intracellular energy reserve. 6 It has also been reported that the addition of neodymium (Nd) and yttria (Y) into WE43 may cause them to be distributed at the implantation site after the degradation of the magnesium implant.5,7 Therefore, the utilization of elements that exist in the human body and that can retard the biodegradation of magnesium alloys are promising candidates for our approach. 7 In recent years, it has been confirmed that the addition of zinc (Zn), calcium (Ca), and manganese (Mn) in Mg have beneficial effects on an in vitro tolerance of the alloy. 8 Zn is a necessary trace element in the human body and has a moderate degradation rate that is attributed to its higher electrode potential when compared to that of Mg. Therefore, it is expected that the addition of Zn can delay the corrosion of the magnesium alloy. Zhang et al. found that Zn could elevate the electrode potential of Mg in SBF and improve the corrosion resistance and the degradation rate of the Mg-6Zn alloy in vitro, which was slower than that of high-purity magnesium in SBF. 9 It is reported that the corrosion rates of Mg-Zn alloys obviously reduce when the addition of Zn is from 1 to 5 wt.%. 10 In previous studies, Ca has been proved to be beneficial for the human body to promote bone healing and improve the corrosion resistance of Mg alloys. Li et al. 11 developed binary Mg-Ca alloys for use as biodegradable materials within bone and reported that the mechanical properties and bio-corrosion behaviors of Mg-Ca alloys could be adjusted by controlling the Ca content. Moreover, Ca can not only reduce the degradation rates of Mg alloys, but can also improve the mechanical properties of Mg alloys. 12 Furthermore, it has been reported that Mn can refine the grain of Mg alloys, which could elevate both their corrosion resistance and mechanical properties. 13 Gu et al. 14 developed binary Mg-1 wt.% X alloys by adding Al, Ag, In, Mn, Si, Sn, Y, Zn, Zr to magnesium to improve its strength and corrosion resistance. They found that the addition of Al, In, Mn, Zn, or Zr reduced the corrosion rate of as-cast Mg-X alloys in both SBF and Hank’s solutions.
On the other hand, heat treatment is a typical approach that can decrease the corrosion rate of Mg and other properties. 15 Recent research has indicated that aging treatment on the extruded alloy can enhance the corrosion resistance of JDBM alloy (Mg-Nd-Zn-Zr alloy, Jiao Da bio-magnesium series designed by Shanghai Jiao Tong University). 16 Ran et al. 17 have reported that over-aging treatments had an effect on the corrosion resistance of the Al-Zn-Mg-Cu alloy. Song et al. 18 have discovered that T4 treatment weakened the barrier for the fast and inhomogeneous degradation rate of magnesium owing to the reduction of β phase. Previous studies have demonstrated that the corrosion resistance can be improved by increasing the tin concentration of the matrix where the lowest corrosion rate was observed for a Mg-7Sn alloy solution treated at 540°C. 19 Besides that, the enhancement of the aging hardening response of Mg alloys is attributed to the Ca2Mg6Zn3 ternary phase. 20
In this paper, the orthogonal design, an experimental design method that uses an orthogonal table to deal with the multi-factor and multi-level experiments and conducts statistical methods to analyze experimental results, was used to develop Mg-Zn-Ca-Mn alloys with attractive degradation rate, which can be further improved by controlling the addition of Zn, Ca, and Mn. Moreover, we investigated systematically the effect of heat treatment on the corrosion resistance of Mg-Zn-Ca-Mn alloys. Analyses of the open-circuit corrosion potential (OCP), polarization curve, and Electrochemical impedance spectroscopy (EIS) of different magnesium alloys in Hank’s simulated body fluids were also explored in this study. Ultimately, the purpose of this work is to explore the bio-corrosion behavior of Mg-Zn-Ca-Mn alloys and identify a promising alloy for new biological implants.
Methods
Orthogonal composition design and preparation of materials
In order to evaluate the degradation rate of the Mg alloys, the orthogonal method was used to design a quaternary Mg-Zn-Ca-Mn alloy in this experiment. The mean corrosion rate was carried out by immersion tests. In order to determine better alloy compositions toward the corrosion properties of Mg alloys, optimization of each component proportion for Zn, Ca, and Mn was investigated in the range of 1.0–2.0 wt.%, 0.5–1.0 wt.%, and 1.3–2.5 wt.%, respectively.6,7,21,22 The contents of Zn, Ca, and Mn were regarded as being effecting factors and marked as A, B, and C, respectively. Each factor contained five levels and all data of the orthogonal method are summarized in Table 1. In this experiment, the content of Zn was ranked in the first column because Zn was deemed to be the main factor affecting the degradation rate of the Mg-Zn-Ca-Mn alloy. The remaining two columns were placed in turn as Ca (wt.%) and Mn (wt.%). Commercially pure Mg (99.99 wt.%), Zn (99.99 wt.%), Mg-10 wt.% Ca intermediate alloy powders, and MnCl2 were melted in a vacuum induction melting furnace (Institute of Vacuum Technology, Shenyang, China) under a high-purity argon atmosphere at 750°C for 15 minutes. Then the melt was cast into a steel mold at approximately 730°C with Φ(symbol of diameter) 28 mm × 130 mm ingots. The corresponding corrosion rate of each alloy was set as a detecting index (Table 1).
Orthogonal pilot programs and experimental result.

Heat treatment
Differential scanning calorimetry (DSC, Netzsch–400, Germany) was performed at constant heating and cooling rates of 10 K/min between 20 and 600°C, and used to determine the potential phase transitions and processing temperatures. The results of endothermic or exothermic peak analysis were used for determining the temperature for heat treatment. The cast ingots were subjected to solid-solution heat treatment at 415°C for 10 hours, quenched in water of 80°C, and then aged at 250°C for 8 hours. 23 All treated samples were ultrasonically cleaned in distilled water and dried in air before further characterization.
Immersion test
Samples for immersion tests were prepared and cleaned according to the standard ASTM G1-1999, and the specimens for corrosion testing were processed into disks with a diameter of 20 mm and a height of 3 mm. Their surfaces were cleaned with isopropanol before testing. The immersion tests were carried out at a steady temperature of 37±0.5°C using Hank’s solution (pH = 7.4). 24 Experimental specimens were removed after 10 days from Hank’s solution, and the corrosion products were removed in a chromic acid solution of 200 g/L CrO3 and 10 g/L AgNO3 (Sinopharm Chemical Reagent Co. Ltd, China) at room temperature for 10 minutes.23,25 There were three parallel samples set for each group. The change of pH value as an indicator of the corrosion resistance was recorded as the immersion time passed and the weight loss was used for calculating the average corrosion rate of the experimental samples. Corrosion morphologies of Mg alloys after immersion in Hank’s solution for 10 days were characterized by scanning electron microscopy (SEM).
Electrochemical measurements
The electrochemical measurements were carried out in Hank’s solution (pH = 7.4) at room temperature according to the standard protocol DIN50918. 26 The size of the cubic corrosion test samples were 10 × 10 × 5 mm3 and the bare area of samples was 1 cm2. A copper conductor was attached onto the bottom side of the specimens, which was packed by an electro-conductive epoxy resin with a 100 mm2 monitoring area of the magnesium surface. The experimental setup consisted of a conventional three-electrode system, comprising the working electrode (samples), a saturated calomel electrode (SCE), and a platinum mesh as the auxiliary electrode. The electrochemical measurements were conducted by monitoring the current density as a function of the free open-circuit potential using the potentiostat of an electro-chemical workstation (CHI600B/D, CH Instruments, Inc., China). After a stabilization period of 3 minutes in solution, the potentiodynamic polarization was carried out at a scan rate of 2 mV/s. The polarization resistance (Rp) and corrosion current density (Icorr) were calculated through curve fitting analysis. Electrochemical impedance tests were performed with a frequency range of 105 Hz to 10-2 Hz and a disturbance voltage of 5 mV.
Results
Analyses of the orthogonal method
Through the analyses of Table 1, the conclusions are as follows: (a) Zn content is the most important factor, Ca and Mn are the second; (b) the best test conditions by analysis and calculations are listed in Table 2, and the best corrosion resistance is registered for alloy 3 (A2B1C3, Mg-1Zn-0.3Ca-1.7Mn).
Chemical composition of Mg100 – x – y – z-Znx-Cay-Mnz alloys (wt.%)

Immersion test of as-cast alloys
Five alloys with best test conditions by analysis and calculations are listed in Table 2. These alloys were immersed in Hank’s solution for 10 days to evaluate their corrosion rate (as shown in Figure 1). The fluctuation of average corrosion rate was detected according to the corrosion weight loss rate. 23 It was observed that the highest average corrosion rate was 0.85 mm/y and the lowest average corrosion rate was 0.15 mm/y. The corrosion rate of tested alloys had clearly risen by the seventh day and then declined during the last 3 days. It was worth noting that the corrosion rate of alloy 3 accounts for only 66% of that of alloy 1 at the beginning of immersion. Moreover, the average corrosion rate of alloy 3 was minimal during the test period. It suggested that the corrosion resistance of alloy 3 significantly improved due to the increase of Mn content as the corrosion resistance of alloy 3 was the best among the five alloys.
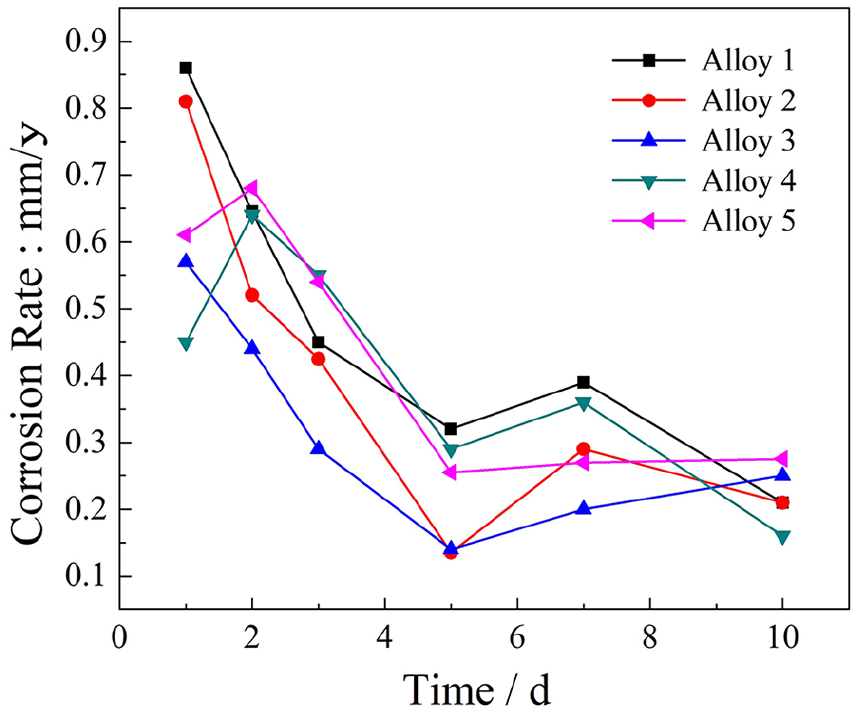
Immersion test results of as-cast Mg-Zn-Ca-Mn alloys: average corrosion rate
The corrosion morphologies of the above as-cast alloys are shown in Figure 2. It can be seen that a reticulate corrosion structure formed on the corrosion surface morphologies of five alloys after immersion in Hank’s solution for 5 days. Simultaneously, a number of corrosion pits were observed on the corrosion surface of each alloy. The deeper corrosion pits appearing in alloys 4 and 5 indicated that more severe localized corrosion had formed, as is shown in Figures 2(e) and 2(f). Among the five alloys, the corrosion pit areas on the corrosion regions of alloys 2 and 3 were smaller and exhibited relatively uniform distribution. Furthermore, corrugated-like morphology appeared in alloy 3 during the corrosion process, which could be attributed to the continuous generation and mutual intersection of new corrosion pits.

Corrosion morphology of as-cast Mg-Zn-Ca-Mn alloys: (a) alloy 1; (b) alloy 2; (c) alloy 3; (d) ×1000 of alloy 3; (e) alloy 4; (f) alloy 5
Electrochemical measurements of as-cast alloys
The OCP curves of five as-cast alloys in the Hank’s solution are shown in Figure 3(a). It was observed that all alloys gradually performed more positive and stabilized OCP values. In addition, the OCP values of alloy 3 increased from −1.78 VSCE to −1.63 VSCE approximately. Generally, the more positive OCP values meant better corrosion resistance of alloys. When there were more positive OCP values, magnesium atoms would have to overcome the higher barrier in the process of leaving the lattice to form magnesium ions; that is, more energy was required for this process. Alloy 3 presented more positive OCP values, which indicated that its corrosion resistance was better than for the other alloys.

Electrochemical corrosion test results of as-cast Mg-Zn-Ca-Mn alloys: (a) open circuit corrosion potential (OCP) curves and (b) potentiodynamic polarization curves
The potentiodynamic polarization curves of alloys are reproducible (Figure 3(b)). The relationship between the current response of the alloy and the potential change can be obtained by the determination of the dynamic potential polarization curve, and thus the OCP, the self-corrosion current density and the linear polarization resistance of the alloys were obtained. The contents of Zn and Mn elements had a significant influence on the cathode reaction of the alloy corrosion process. The self-corrosion current (Icorr) of alloy 1 was lower than that of the other alloys by ~1.5–3 A. From observation of SEM (Figure 5), it is possible to deduce that many of the precipitates, in non-equilibrium phases, would act as cathodes to construct galvanic corrosion within the matrix. That is consistent with what would be expected if significant quantities of Zn were redistributed from precipitate particles, and is also why there are distinct differences among the corrosion currents in the cathode branches across all samples. The results were also consistent with what would be expected if significant quantities of Zn and Ca were redistributed from precipitate particles. Anodic reaction dominates the overall corrosion process. Once the curve went across the free-corrosion potential spot and before the passive state, the corrosion current of alloy 1 was higher than that of other alloys, meaning it had the fastest corrosion rate. We observed that alloy 3 has the highest linear polarization resistance, which is almost twice as high as that of alloy 1.
The Nyquist plots of the alloys immersed in Hank’s solution for 24 hours and 120 hours are shown in Figure 4. As can be seen from the figure, all of the tested alloys had similar degradation behaviors during the immersion in Hank’s solution after 24 hours and 120 hours. The alloys contained one high-frequency capacitance loop and one low-frequency inductive loop. For Nyquist plots after 24 hours, compared with the high-frequency arc, the radius of the inductive loop for the low-frequency arc tended to infinity when the transition from high-frequency arc to low-frequency arc took place. This could be attributed to the loss of oxide film on the alloy surface, which accelerated the corrosion process of the matrix. As for Nyquist plots after 120 hours, the low-frequency arc reflected the corrosion degree of the tested alloys. Therefore, the greater the reduction of the degree of low-frequency arc, the lower the corrosion rate of the tested alloy. The low-frequency arcs of alloys 3 and 2 almost disappeared, and the decrease for alloys 1 and 5 was small. Therefore, the corrosion resistance of alloy 3 was greater than that of the others. Figure 4 illustrates that alloy 3 had the largest capacitance arc, which exhibits relatively low corrosion rate, corresponding to its polarization curves.

Mg-Zn-Ca-Mn alloys immersed in Hank’s solution at different times of Nyquist plots: (a) alloy 1; (b) alloy 2; (c) alloy 3; (d) alloy 4; (e) alloy 5
Microstructures of alloy 3
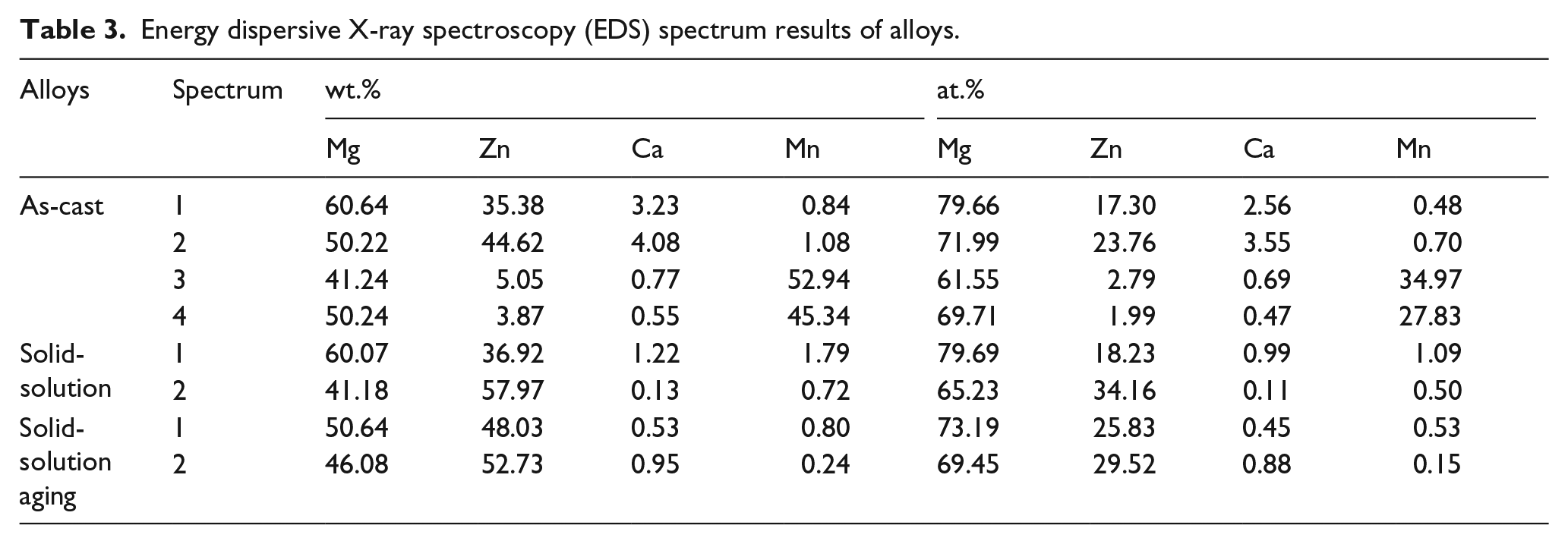
Through the analyses of Table 1 and Table 2, alloy 3 (A2B1C3, Mg-1Zn-0.3Ca-1.7Mn) exhibited a lower corrosion rate than the other tested alloys. We treated alloy 3 by heat treatment and investigated the effect of the solid-solution and aging treatment on its corrosion resistance. Figure 5 illustrates the distribution and the morphology of the second phases in all the stages of alloy 3. The second phases of as-cast alloy, distributing at the grain boundary, were mainly strip-like while some granular phases were also found in the inner grain (Figure 5(a)). After heat treatment, the strip-like second phases disappeared and dispersed into the grain and some fine granular second phases also appeared at the grain boundaries after solution and aging treatment (Figures 5(b) and 5(c)). The chemical composition of the second phase was identified by energy dispersive X-ray spectroscopy (EDS) and the results are given in Table 3 combined with the results of the X-ray diffraction (XRD) patterns of as-cast and heat-treated alloy 3 (Figure 6). It was observed that the as-cast alloy was mainly composed of α-Mg, MgZn, MgZn2, Mg2Ca, and α-Mn particles. There were also some nonequilibrium phases such as Mg7Zn3 and Ca2Mg6Zn3, appearing as precipitates. Ca tended to combine with O to form CaO at first, and then the rest of the Ca reacted with Mg to form Mg2Ca. The addition of Mn was adopted due to its excellent biocompatibility and potential, which could influence the corrosion properties. However, no eutectic reaction could take place between Mg and Mn when the content of Mn was lower than 2.0%, based on a proposed binary alloy Mg-Mn phase diagram. There was also no dissolution when the temperature was under 200°C. Hence, the granular phases of the as-cast alloy were mainly α-Mn particles and the strip-like phases were mainly composed of Mg, Zn, and Ca elements (as shown in Table 3). Compared with the as-cast alloy, the peaks of the nonequilibrium phases—especially the Ca2Mg6Zn3 phases—and α-Mn were significantly decreased after solid-solution and aging treatments. The peaks of Mg2Ca and Mg7Zn3 phases disappeared due to dissolution in the magnesium matrix, which suggested heat-treated alloy 3 may contain particles dispersed fairly homogeneously throughout.
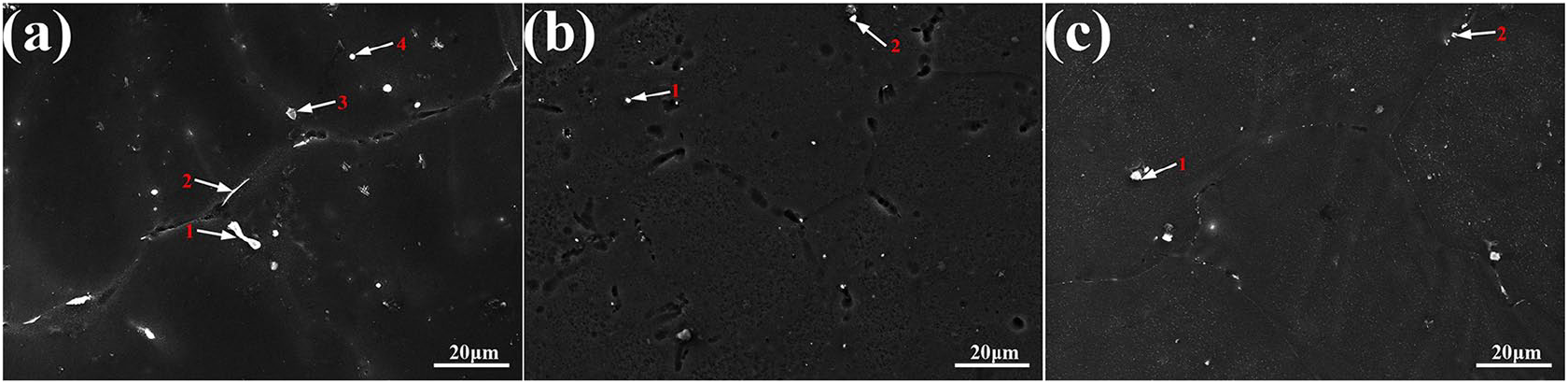
Scanning electron microscopy (SEM) microstructure of as-cast and heat-treated alloy 3
Energy dispersive X-ray spectroscopy (EDS) spectrum results of alloys.
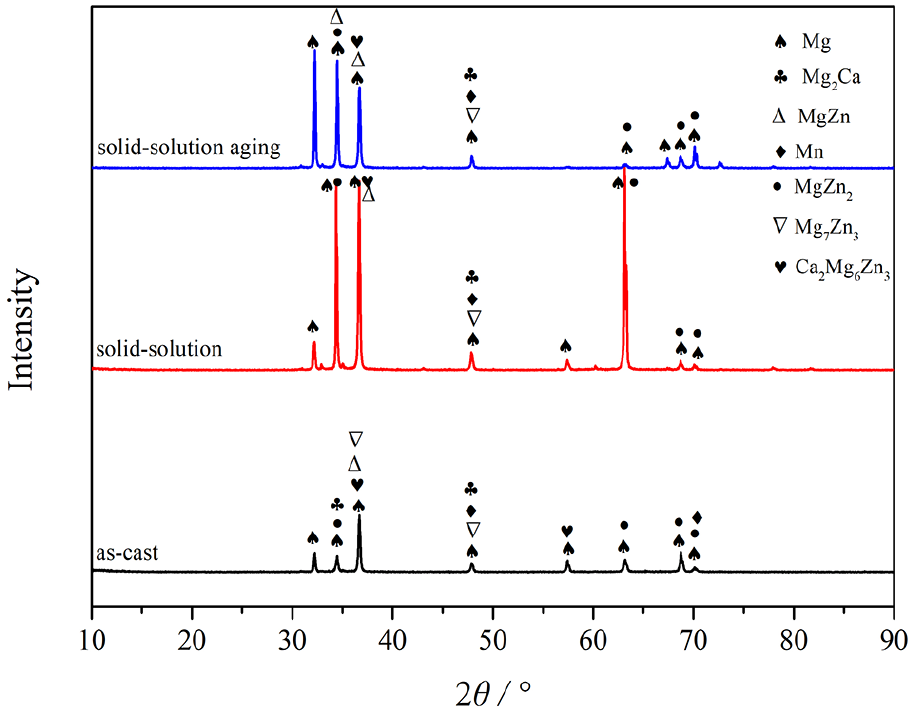
X-ray diffraction (XRD) patterns of Mg-1.0Zn-0.3Ca-1.7Mn alloy in three states: (a) cast; (b) solid-solution; (c) solid-solution and aging
Effect of the solid-solution and aging treatment on alloy 3 corrosion resistance
The OCP of as-cast, solid-solution, and solid-solution aging alloys in Hank’s solution are shown in Figure 7(a). At about 600 seconds, it can be seen that the OCP curve of the solid-solution aging alloy rapidly increased from −1.74 VSCE to −1.54 VSCE. Then, the OCP gradually increased to some potential and the OCP curves of the as-cast and solid-solution alloys became gradually stabilized with the immersion time. Comparing the curves, the Ecorr of the as-cast alloy was lowest when it reached a stable value, at a value of −1.602 VSCE, and the Ecorr in the solid-solution aging alloy was highest, at −1.496 VSCE.
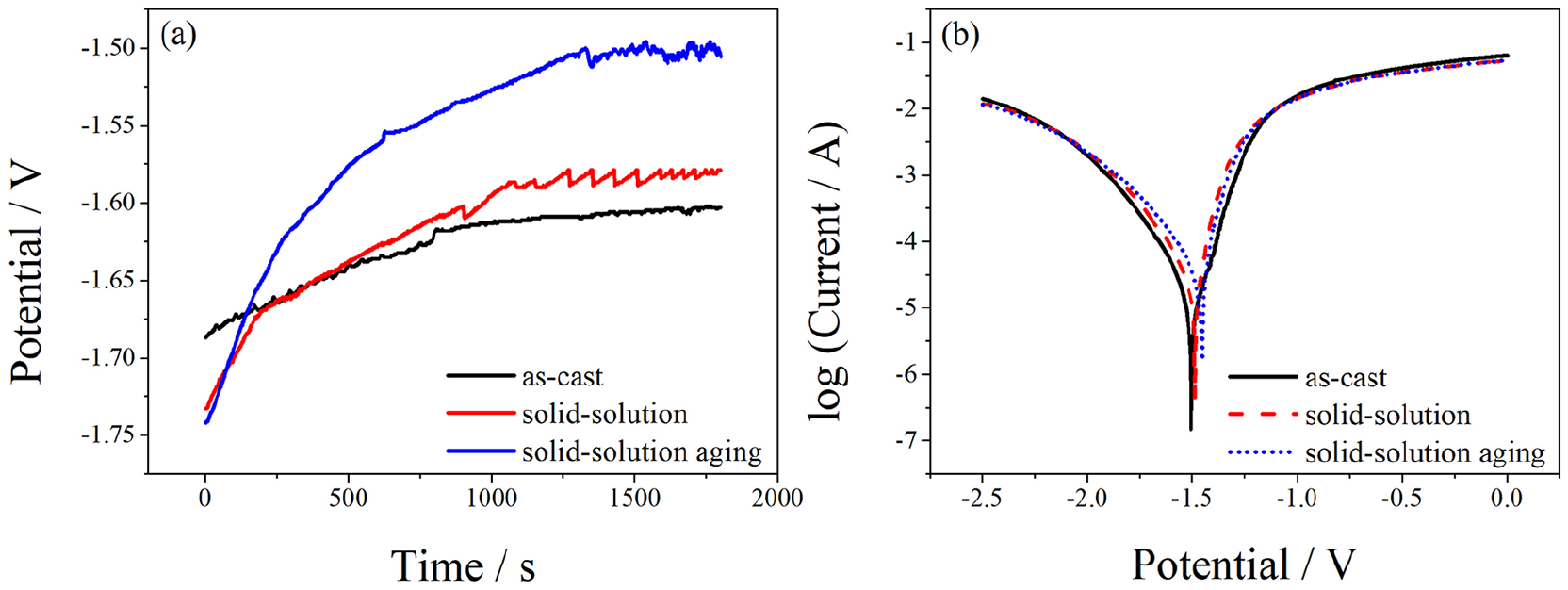
Electrochemical corrosion test results of Mg-Zn-Ca-Mn alloys after different treatments: (a) curves of the open-circuit corrosion potential (OCP) and (b) curves of potentiodynamic polarization
Figure 7(b) shows the polarization curves of the tested alloy 3 in Hank’s simulated body fluids. It indicates that the self-corrosion potential (Ecorr) of alloy 3 shifted positively and the self-corrosion current (Icorr) reduced. It is obvious that the electrochemical polarization behavior of as-cast, solid-solution, and solid-solution aging alloys were significantly different. The Ecorr of specimens after heat treatment was more positive than the as-cast alloy while the Icorr became higher. Furthermore, the Ecorr of the solid-solution aging alloy was higher than for the other two alloys, and the Icorr obviously increased. Combined with SEM and XRD analyses, the volume fraction of the Mg2Ca phase in the matrix of the alloy was greatly reduced after the solid-solution and aging treatments, which resulted in a decrease of the effective area of the harmful cathode phase, and thus the probability of galvanic corrosion decreased.
The Nyquist plots of as-cast and heat-treated alloys immersed in Hank’s solution for 24 hours and 120 hours are displayed in Figure 8. The EIS of tested alloys were demonstrated. From the Nyquist plots of specimens immersed in Hank’s solution for 24 hours, it can be expected that the corrosion product layer formed in the early stage of immersion had a protective effect on the matrix for a short time, but it lost this protective effect for the matrix alloy very quickly during the corrosion process. A large reticular boundary structure formed on the corroded metal surface, which was not conducive to the accumulation of corrosion products. When the pores of the matrix metal were seriously corroded, the corrosion products were easily detached from the net grain boundary structure, so that the corrosion zone was exposed in the solution and the pitting corrosion continued to develop along the longitudinal direction.
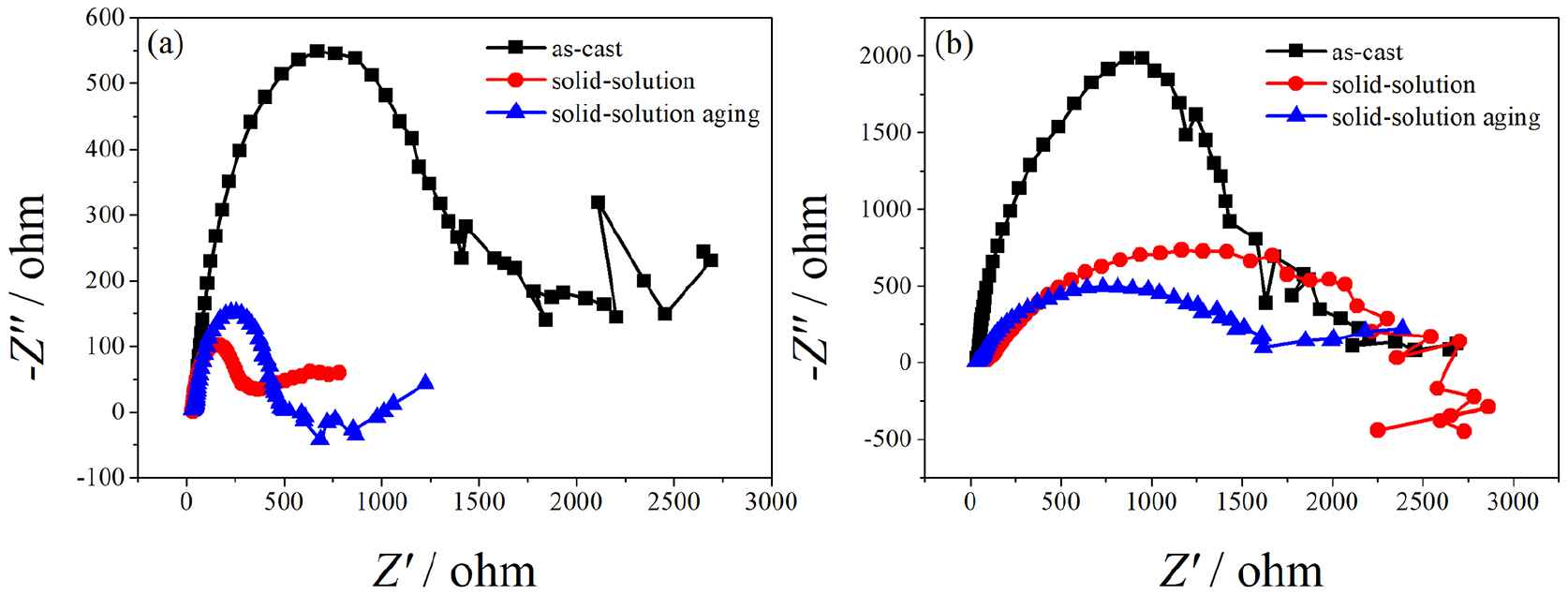
Nyquist plots of Mg-Zn-Ca-Mn alloys after different treatments
From the Nyquist plots of alloys immersed in Hank’s solution for 120 hours, the high-frequency capacitance arc was transformed into a low-frequency inductance arc, which was due to the change of the surface state of the alloy. Compared with as-cast alloys (Figure 2), during the immersion process, corrosion products were generated continuously on the surface of heat-treated alloys (as shown in Figures 9 and 10). Figure 9 shows the corrosion morphology of the solid-solution alloy. A large number of corrosion pits were seen evenly distributed over the surface, and the size of these corrosion pits was relatively uniform. A dense network structure was formed among the pits and the surface was relatively smooth. This indicated that the solid-solution alloy exhibited the characteristics of uniform corrosion. Compared with as-cast alloys and the solid-solution alloy, the thickness of the corrosion product layer on the surface of the solid-solution aging alloy increased rapidly and improved the density of the film. These results indicated that the solid-solution aging alloy showed more uniform corrosion than other alloys during the corrosion process and the diffusion resistance of infiltration and exudate increased, which meant that water molecules and other anions did not find it easy to enter and exit the film (Figure 10). According to oxide-film theory, when the corrosion-resistant alloy reached its critical composition, the metal surface could form a complete compact passivation film, thus increasing the resistance of the alloy and its effect.

Corrosion morphology of Mg-Zn-Ca-Mn alloys with solid-solution treatment

Corrosion morphology of Mg-Zn-Ca-Mn alloys with solid-solution aging treatment
Discussion
It has been reported that Zn, Ca, and Mn are potential alloying elements for Mg alloys due to their good biocompatibility. 11 Zn can elevate the corrosion resistance of Mg alloys because of its higher electrode potential. 9 An appropriate amount of Mn content can refine the microstructure of the as-cast alloy. 27 This is due to the role of Ca increasing the diffusion of Mn in Mg and making it more evenly distributed in the alloy, and therefore hindering the corrosion of the alloy to some extent. However, residual Ca will be formed as the Mg2Ca phase of the cathode phase forms with Mg at the same time, which will lead to accelerated corrosion of magnesium alloys. From the perspective of microstructure evolution, there are some second-phases and inter-metallic compounds (Figures 5 and 6). Their electrode potential is higher than that of the α-Mg matrix, leading to galvanic corrosion and acceleration of the corrosion rate of magnesium alloys, 28 even though the surface of the sample is covered by sedimentary magnesium hydroxide film momentarily. The solid-solution and solid-solution aging processes may make precipitates in the grain boundary but they are mostly merged into the grains to form a uniform dispersed second phase, which is the limit of grain growth. From the XRD patterns (Figure 6), it can be seen that some nonequilibrium phases were eliminated, a number of MgZn phases and MgZn2 phases with higher potential were formed, and the volume fraction of the Mg2Ca phases after solid-solution treatment was reduced. In the process of solid-solution treatment, the eutectic phase Mg7Zn3 was decomposed into α-Mg and MgZn phases, and the nonequilibrium phase Ca2Mg6Zn3 was completely decomposed to form a mixed product of the nonequilibrium phase that was interwoven between the MgZn2 phase and the α-Mg solid solution at 325°C 29 ; therefore, the corrosion tendency was decreased. The solid-solution aging treatment may change the microstructure of the tested alloys so that it is not conducive to the formation of a stable and dense protective film on the alloy substrate caused by corrosion products. Pitting corrosion also appeared in the studied alloy during solid-solution aging treatment (Figure 11). Although the solid-solution aging process could cause the studied alloy’s surface to be covered by a dense corrosion product film (Figure 10), it led to the concentration polarization of the Mg2+ inside and outside the in vitro film. This environment also activated pitting corrosion to a great extent, which was not conducive to the improvement of the corrosion resistance of the studied alloys.

Backscattered photographs and EDS analysis of pitting of Mg-Zn-Ca-Mn alloy after solid-solution aging treatment.
In this work, Mg-1Zn-0.3Ca-1.7Mn alloy (alloy 3) was determined to be the best performing alloy by the orthogonal comparison method and in vitro tests (Table 2). It exhibited a lower average corrosion rate than other as-cast alloys (Figure 1). Through the experiment, it was obvious that the corrosion resistance of the tested alloys was determined by the corrosion properties of each phase in the alloy. Moreover, it was important to study the corrosion reaction of these magnesium alloys. The polarization of magnesium in SBF is usually similar to what is seen in NaCl aqueous solution. The chemical properties of magnesium include high activity in aqueous solutions where the following reactions occur. 30
At the anode

At the cathode

Chemical reaction

Total reaction

In neutral and alkaline media, metastable Mg+ from the anodic dissolution process is extremely unstable. It can be readily converted into Mg2+, and the anode reaction is


In the solution containing Cl-, the surface of Mg(OH)2 can be eroded by Cl- and the following reaction occurs

With the attack of large numbers of active anion ions, such as Cl- and OH- ions in the solution, the area of pitting corrosion changes relative to the level of local corrosion. In particular, passive films are destroyed by Cl- in the backscattered electron image (Figure 12). There were large amounts of Cl- detected in the black area (at point A) where pitting corrosion took place. Cl- penetrated through the local protection film through structural deficiencies such as dislocation, while the oxide film structure could be changed by the concentration of Cl-. Moreover, competitive adsorption is raised when Cl- coexists with other ions that are adsorbed in the membrane surface and, as a consequence, the oxide surface is transformed into a soluble salt layer. In the process of competing adsorption, Cl- can be preferentially adsorbed to damage the passivation layer. Cl-, which was absorbed into the lattice, replaces the water molecules in the membrane and OH- or O2- occupies the positions of the water molecules. Then the electric field effect of the negative charge influences the activation energy of the electrode reaction, accelerating the anode dissolution of the metal. Meanwhile, white areas (at spot B) represent phosphate and carbonate sediments in the corrosion process, so C and P were also detected in the white area.

Backscattered photographs and EDS analysis of pitting of the as-cast Mg-Zn-Ca-Mn alloy.
Because of the complexity of actual body fluids and corrosive environments, sometimes in vitro corrosion tests cannot be used to predict in vivo corrosion rates of magnesium alloys. 31 We cannot treat it simply as a solution containing Cl- and OH-, but also should consider the effects of phosphate, carbonate, and others.
From the view of corrosion electrochemistry, the change of corrosion rate depends on the change of cathode and anode processes. The cathode depolarization process of magnesium corrosion is the cathode reaction that leads to hydrogen evolution. Therefore, the cathode depolarization process may not increase with the changing of the pH values in the process of corrosion. The reason for hydrogen evolution is that the anodic process of magnesium corrosion is accelerated more easily. The corrosion of the studied magnesium may be related to the change of composition as time goes on, and with more Mg2+ dissolving into the solution, the precipitation will be consumed when the anions that inhibit Cl- may react with the Mg2+. Therefore, the corrosion of the solution becomes stronger as time goes on and the anode process of magnesium is accelerated. However, although the corrosion of magnesium alloys has many similarities with pure magnesium, such as anodic hydrogen evolution with anodic dissolution, impurity elements that could negatively impact corrosion, such as Fe, Ni, and Cu, were uniformly distributed in the magnesium alloy matrix due to the heat treatment. 23 Therefore, magnesium alloys studies show different corrosion characteristics from pure magnesium.
Conclusion
The as-cast alloy with the four components Mg-1Zn-0.3Ca-1.7Mn (alloy 3) exhibits the best corrosion resistance with approximately 0.15mm/y among five alloys designed by the orthogonal method.
Localized corrosion of the as-cast alloy plays an important role in the five alloys designed by the orthogonal method. While heat treatment of the alloys studied makes the corrosion morphology of alloy 3 more uniform than that of the as-cast alloy, after the solid-solution aging treatment a compact protective film can form on the surface of the alloy, retarding the corrosion to some extent. However, due to the selective adsorption of Cl-, the tested alloys were found to have corrosive pitting of different degrees.
The corrosion tendency of the alloy is correlated with the decrease of nonequilibrium phases and the potential rising of the alloy matrix after heat treatment. However, although a dense protective film could form after heat treatment, it accelerates the corrosion of the alloy due to the Mg2+ concentration polarization differential inside and outside the film.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NSFC) under Grant (No. 51365029 and No.51664041); the Fundamental Research Funds for the Universities in Gansu Province and the Program for Support Projects of Science and Technology in Gansu Province under Grant (No. 1604GKCA038) and Longyuan Youth Innovation and Entropreneurship Projects.
