Abstract
Introduction
Periodontitis, or “gum disease”, describes a set of inflammatory conditions affecting the soft and hard tissues that support teeth. In susceptible individuals, bacteria present on tooth surfaces lead to the inflammation and eventual destruction of connective tissue and alveolar bone, resulting in the formation of periodontal pockets, tooth mobility, and, eventually, tooth loss. Nearly 50% of adults are affected by periodontitis in the United States. 1
Although periodontitis involves a multifactorial disease process, its bacterial etiology is well established. Thus, treatment involves the elimination of bacterial plaque from tooth surfaces via patient education as well as non-surgical and surgical removal. With refractory or aggressive forms of periodontitis, the use of systemic antibiotics is supported. 2 However, targeted antibiotic delivery in localized residual pocketing is appealing because it provides a high antibiotic concentration to the site, decreases the risk of systemic side effects, and eliminates the need for patient compliance. The use of adjunctive, sustained antibiotics release in deep, localized residual pockets is supported by the American Academy of Periodontology. 3
The success of local antibiotic delivery in treating periodontitis relies on the sustained release of antimicrobials; irrigation alone does not seem to have an effect. 4 Several products are available that meet this requirement, including PerioChip® (Dexcel® Pharma Technologies Ltd., Jerusalem), Atridox® (DenMat, Lompoc, CA), and Arestin® (OraPharma, Bridgewater, NJ). These products provide sustained release of antibiotics for at least 7 days and have been shown to have an adjunctive effect when compared to scaling and root planing (SRP) alone. 4 Minocycline microspheres (Arestin®) maintain drug release over 14 days, demonstrating the most significant effect on pocket reduction and clinical attachment loss when used as an adjunct to SRP. 4 Minocycline is effective against periodontal pathogens at low concentrations (1 µg/mL), 5 results in little systemic absorption or development of resistant bacteria, 6 and reduces inflammatory tissue destruction by inhibiting matrix metalloproteinases. 7
Just as minocycline has both antibacterial and anti-collagenase properties, it would be advantageous if the carrier deployed for local antibiotics delivery had beneficial properties in treating periodontitis. Polyphosphates may present one option in this regard; they have been reported to have antibacterial8, 9 and positive bone remodeling10, 11 effects. Furthermore, evidence is available demonstrating the anti-inflammatory effects of polyphosphates in treating periodontitis.12–14
Polyphosphate, in particular calcium polyphosphate (CPP) glass, is being investigated for use in local drug delivery.15–17 CPP is made up of polyphosphate chains that are cross-linked via calcium ions. In the presence of water, a coacervate (gel) forms; on removal of water, a CPP glass (solid) is obtained. This transition between phases can be used to incorporate and release drugs. Furthermore, as CPP hydrolyzes, polyphosphate chains are released.
15
Utilizing this phase transition, drug loading and sustained antibiotic release has been observed.15–17 This CPP system can also be used as an
Materials and methods
Synthesis of drug-loaded coacervate matrix component
Sodium polyphosphate (NaPP) was obtained and characterized using a protocol described by Momeni et al. 18 In short, NaH2PO4·H2O (Sigma-Aldrich) was melted at 700°C and rapidly quenched, resulting in NaPP with an average degree of polymerization (Dp) of ~200. It is subsequently fractioned using serial addition of acetone to obtain an NaPP with Dp of 45, which was used throughout this study. All other materials were American Chemical Society (ACS) grade from Sigma.
CPP precipitation was carried out according to the protocol described by Momeni et al. 18 At room temperature, 1.5 g of NaPP was added to 250 mL of deionized water and stirred continuously until fully dissolved. Then 7 mL of 1 M CaCl2 was added to the solution to achieve a 0.5 Ca/P mole ratio. The precipitation reaction proceeded for 5 minutes with continuous stirring. CPP precipitate was collected, washed three times using deionized water, and then freeze-dried using a FreeZone® 1 Liter Benchtop Freeze Dry System (Labconco Corp., USA) at −43°C and pressures below 13.3 Pa for 2 h. Subsequently, 20 mg of minocycline was dissolved in 1.5 mL of deionized water and added to the freeze-dried CPP. This mixture was stored at ~8°C overnight in a scintillation vial. Following refrigeration, the CPP/minocycline was mixed using a hand spatula to allow for homogenous distribution of minocycline throughout the coacervate. Samples of minocycline-loaded CPP coacervate were collected to assess initial drug-loading, and the remaining coacervate was used to produce microspheres.
Microsphere synthesis
Using an overhead stirrer (IKA® Eurostar 40) and a four-blade propeller, 25 g of polycaprolactone (Mn: 80,000) serving as a thickening agent was dissolved in 250 mL of chloroform, followed by addition of 1 mL of an emulsifier, Span80. Minocycline-loaded CPP coacervate (~ 1 mL in volume) was then poured directly into the solution and stirred for 90 minutes at 2000 rpm. Stirring speed was reduced to 400 rpm and 500 mL of acetone was added; stirring continued for 3 hr. Subsequently, the solution was divided equally by volume into four 50 mL Falcon tubes prior to centrifuging at 4400 rpm for 1 minute. As large microspheres settle to the base of the beaker once stirring is halted, the first three Falcon tubes (aliquots) contained essentially small, suspended microspheres, with the final Falcon tube (aliquot) containing primarily large microspheres. The resulting “small” microspheres from the three aliquots were subsequently combined to form one population for further evaluation. Supernatant was removed from all tubes, and the microspheres were washed twice with chloroform and twice with acetone. Acetone was added to the tubes containing microspheres prior to storing at ~8°C.
Microsphere characterization
“Small” and “large” microsphere suspensions were placed drop-wise on scanning electron microscope (SEM) stubs. Following drying, a 10 nm gold/palladium coating was applied using a sputter coater (Leica EM ACE200, Leica Microsystems) prior to imaging using a Hitachi S-4700 FEG SEM at 5 kV. In addition, particle size analysis of the “large” and “small” microsphere samples was performed by laser diffractometry (Malvern Mastersizer 3000) using ethylene glycol as the dispersant.
The chemical and structural characteristics of the polyphosphates throughout the process cycle were also evaluated. To assess polyphosphate Dp, four replicates of small and large microsphere batches were dissolved in 200 mM Ethylenediaminetetraacetic acid (EDTA) (pH 10) and immediately analyzed using a Bruker AV300 MHz nuclear magnetic resonance (NMR) spectrometer (101.26 MHz, 15° pulse, 65,536 (64 k) data points giving a 0.8 s acquisition time, a 7.0 s repetition rate and greater than 100 scans); Na-EDTA was used here as a calcium chelating agent to disrupt CPP bonds and enable subsequent dissolution of the now NaPP chains. Spectra were evaluated by Bruker TopSpin 1.3 software and reported using the d scale, with positive values downfield, and referenced to an 85% solution of H3PO4 in H2O. Peaks at approximately 0, 5 to 10, and 20 to 22 ppm represent the Q0 (orthophosphates), Q1 (terminal phosphorus atoms) and Q2-middle (middle of chain phosphorus atoms in polyphosphates), respectively. Based on the relative peak areas, the Dp was calculated according to Momeni and Filiaggi 19 using equation (1):

The Ca/P mole ratio of the coacervate and resulting microspheres was determined using inductively coupled plasma optical emission spectroscopy (ICP-OES) (OptimaTM 7300 V, Perkin Elmer, USA). Following drug loading, samples of coacervate and of large and small microspheres were freeze-dried for 12 h, weighed, and then dissolved in 200 mM Na-EDTA prior to diluting in 2% nitric acid and analyzing using ICP-OES. Standard curves for Ca and P analyses were developed using Perkin Elmer standard solutions.
Corresponding minocycline levels were monitored by UV/VIS spectrophotometry (Synergy HT, BioTek Instruments). For these drug-loading studies, a 400 nm absorbance wavelength characteristic of as-prepared minocycline and demonstrating low background absorbance was used. Drug loading and encapsulation efficiency were calculated using equations (2) and (3), respectively.
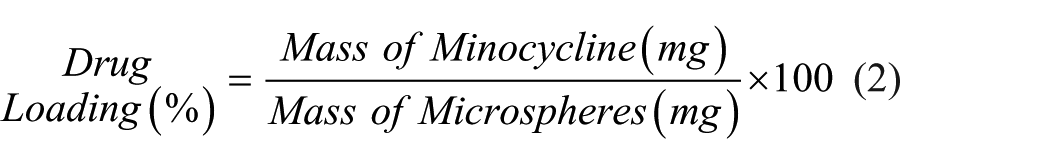
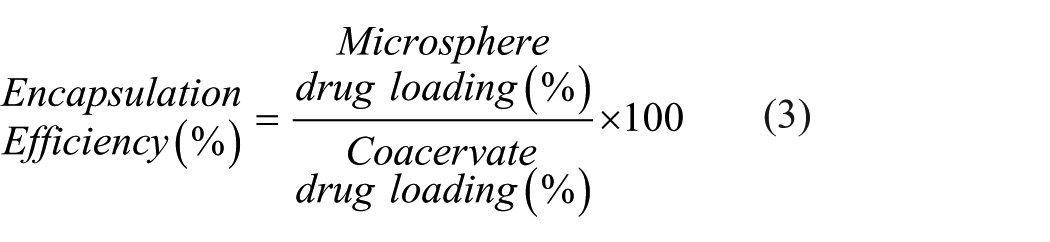
Elution protocol
Small and large minocycline-loaded microspheres (

Ca and P release over the 7-day elution study was also evaluated using the same samples collected for minocycline analysis. Here, samples were diluted in 2% nitric acid containing Na-EDTA prior to analyzing using ICP-OES.
Statistical analysis
A Kolmogorov-Smirnov test and Probability-Probability (P-P) plot were used to test for normal distribution of data. Student’s
Results
Physicochemical characteristics of the microspheres
SEM images of minocycline-loaded CPP microspheres are shown in Figure 1. Microspheres in both “small” and “large” groups were found to have a similar morphology, chiefly a smooth outer surface with a regular spherical shape. Particle size analysis revealed volume mean diameters (D[4, 3]) of 193 ± 4.52 µm and 22.2 ± 1.03 µm for the “large” and “small” microspheres, respectively (Table 1, Figure 2).

Minocycline loaded calcium polyphosphate microspheres. Small microspheres (left) and large microspheres (right) are shown with (from top to bottom) 500, 100, and 50 μm scales.
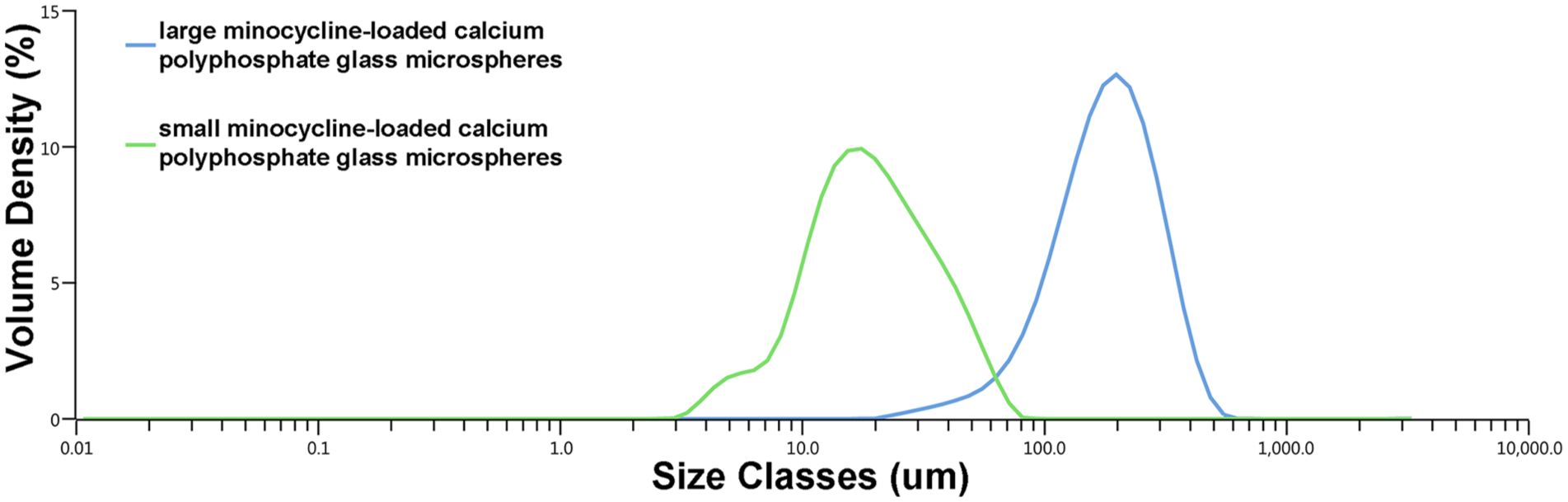
Particles size analysis using laser diffraction of “small” and “large” minocycline-loaded calcium
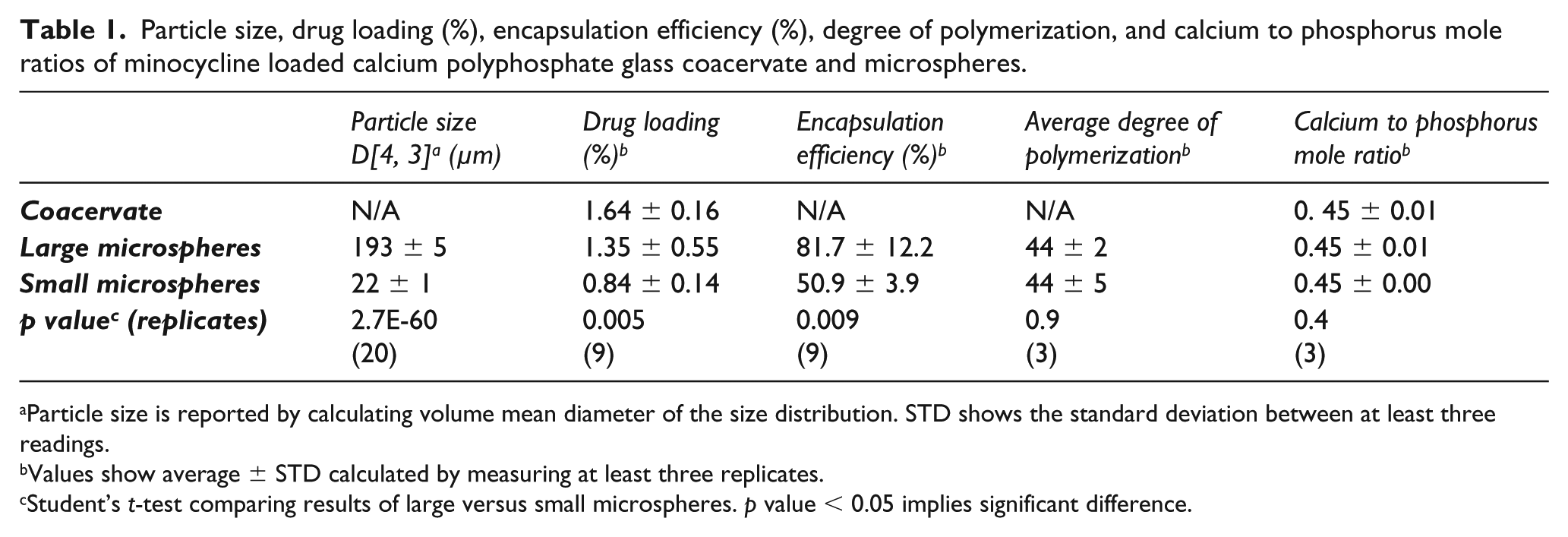
Particle size, drug loading (%), encapsulation efficiency (%), degree of polymerization, and calcium to phosphorus mole ratios of minocycline loaded calcium polyphosphate glass coacervate and microspheres.
Particle size is reported by calculating volume mean diameter of the size distribution. STD shows the standard deviation between at least three readings.
Values show average ± STD calculated by measuring at least three replicates.
Student’s
31P NMR spectra of the NaPP used for microsphere preparation and of the polyphosphate after microsphere dissolution are shown in Figure 3. Here, the large peak around −21 ppm represents phosphate groups in the middle of the chains, the doublet peak around −5 ppm represents terminal phosphate groups, and the small peak at around 2 ppm indicates the presence of orthophosphates. Corresponding Dp values for the large and small microspheres were found to be 44 ± 2 and 44 ± 5, respectively, essentially equivalent to the initial Dp of the NaPP (45 ± 3) used to make the microspheres. Similarly, a nominal Ca/P mole ratio of 0.45 was observed in all cases (Table 1), though notably lower than the theoretical value of 0.5 used for coacervate preparation.
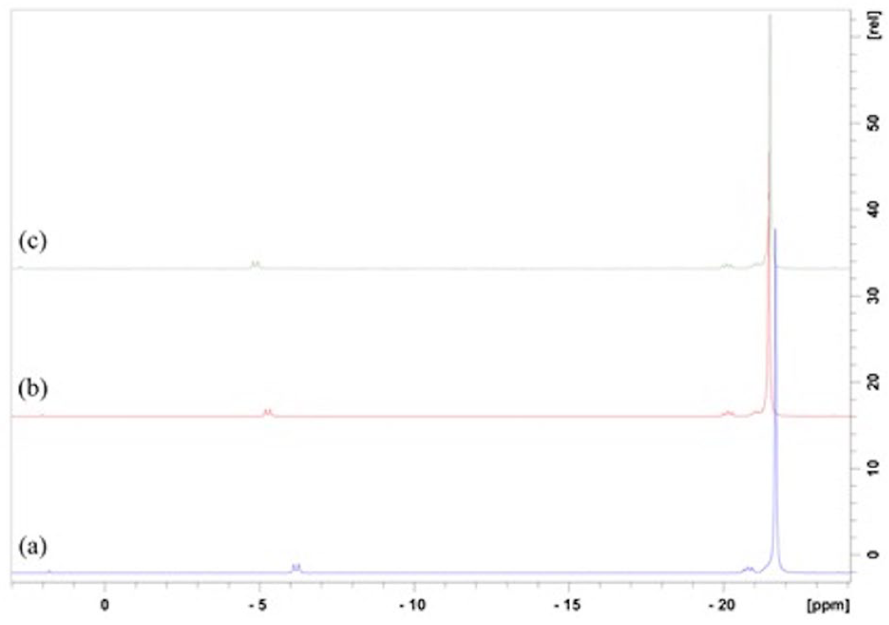
Phosphorus-31 Nuclear Magnetic Resonance of (a) sodium polyphosphate (NaPP) used for microsphere preparation, (b) polyphosphate from large microspheres, and (c) polyphosphate from small microspheres. The observed slight shifts in peak positions between the samples is caused by pH differences between the samples after their dissolution.
Drug loading and release
Average drug loading (%) for the CPP coacervate and the resulting large and small microspheres was 1.64 ± 0.16 %, 1.35 ± 0.55 %, and 0.84 ± 0.14 %, respectively (Table 1). Large microspheres exhibited a higher encapsulation efficiency (81.7 ± 12.2 %) compared to small microspheres (50.9 ± 3.9 %).
Drug release was observed over 7 days. Within the first 3 hours of placing the microspheres into the elution medium, the microspheres aggregated and formed a coacervate. This coacervate was yellow, reflecting the presence of the minocycline, with no appreciable change until 24 hours; subsequently, some fading of the yellow colour was observed. This reduction in colour, together with that of the coacervate size, continued gradually throughout the observation period. By 7 days, small microspheres had released 73% and large microspheres had released 69% of their total of minocycline (Figure 4). Release profiles for both small and large microspheres were found to be curvilinear, with an initial linear portion from 3 to 48 hours, followed by a decreased cumulative release between 48 and 168 hours. Approximately 95% and 86% of the expected minocycline amounts loaded within small and large microspheres, respectively, were recovered based on the 7-day cumulative drug release and amounts obtained following dissolution of any remaining coacervate.
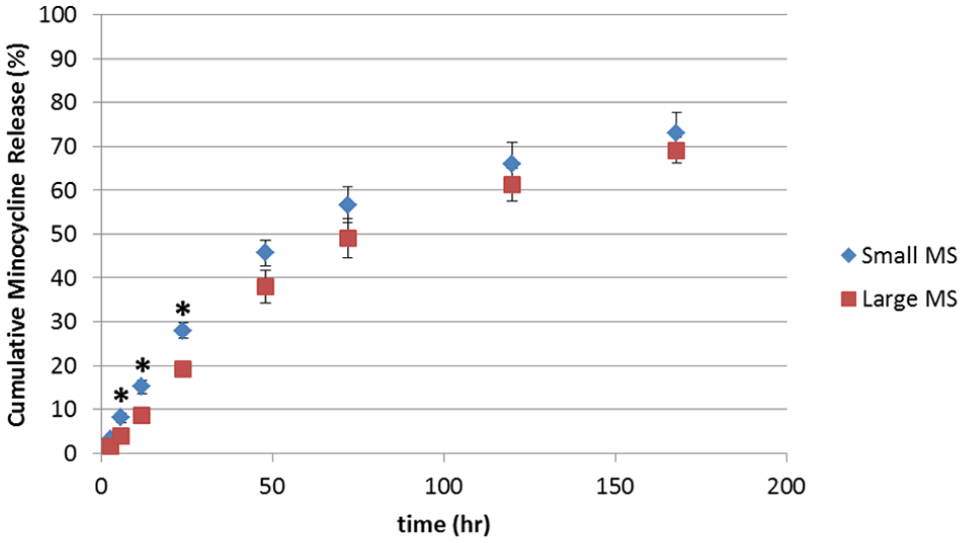
Mean cumulative release of minocycline from small and large calcium polyphosphate microspheres over 7 days. Each point represents average cumulative release of four replicates (
Figure 5(a) shows the cumulative Ca and P released from the microspheres during the elution period. By 7 days ~80% of the P and ~60% of calcium ions are lost, suggesting corresponding rapid mass loss; visual observations corroborated these findings (Figure 5(b)). As was observed with minocycline release, Ca and P release profiles from the small and large microspheres closely approximated each other (Figure 5(a)).
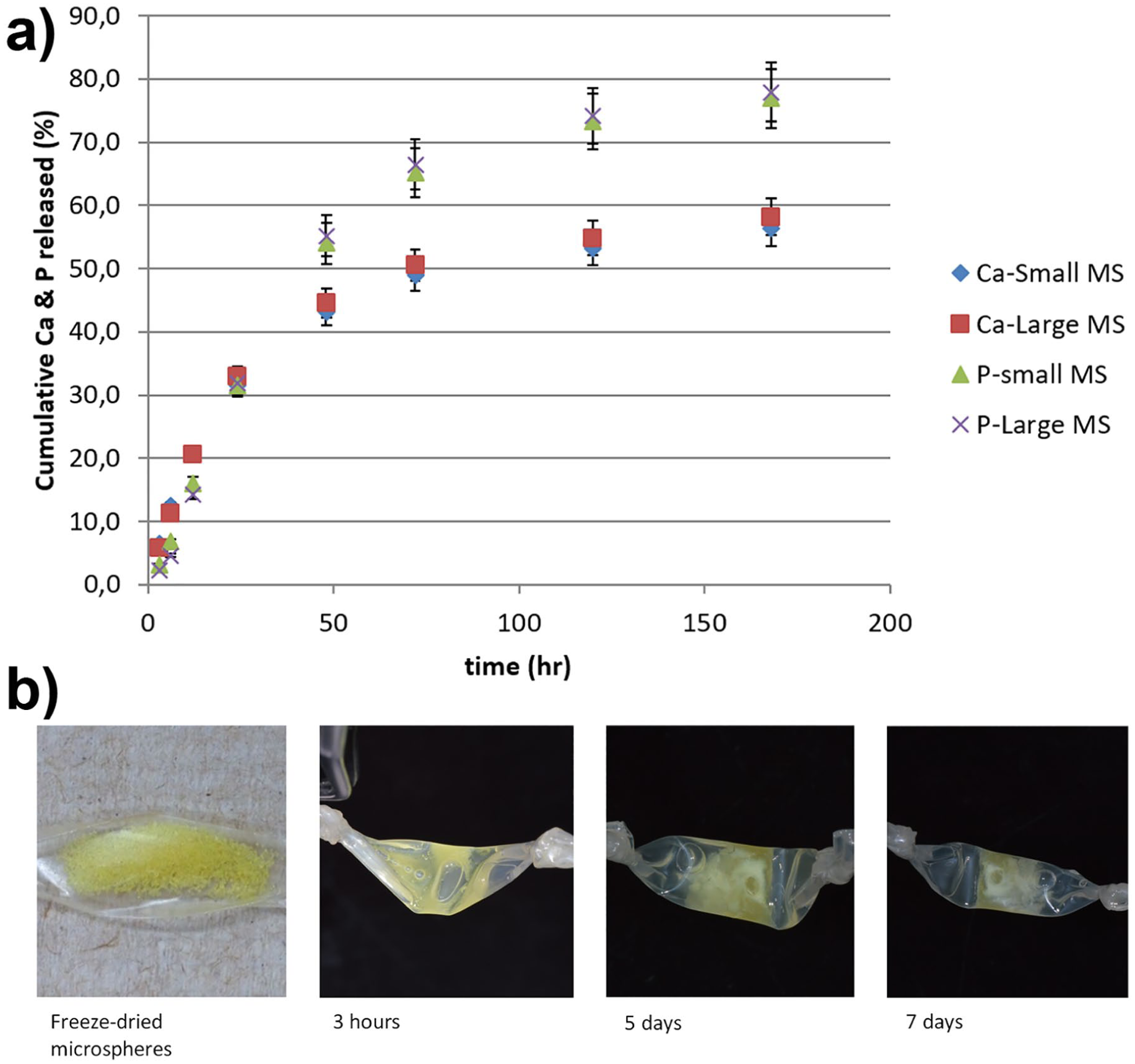
(a) Calcium and phosphorus release as determined by ICP-OES throughout the 7-day elution of minocycline-loaded calcium polyphosphate glass microspheres. Error bars show the standard deviation between three readings. (b) Representative images of the minocycline-loaded calcium polyphosphate glass microspheres over the elution period.
Discussion
Using previously established protocols for CPP coacervation, we were able to produce minocycline-loaded microspheres of varying size that are spherical in shape and have smooth, regular surfaces when observed by SEM. The resulting microspheres can be separated into small (22.2 ± 1 µm) and large (193 ± 5 µm) populations. Furthermore, these microspheres can be sieved to produce a narrower size distribution if desired.
The minocycline-loaded coacervate exhibited a calcium to phosphorus ratio (~0.45) that was less than the expected theoretical value (0.5), suggesting some of the Ca does not incorporate into the coacervate during NaPP precipitation and instead remains inside the supernatant, as reported previously. 18 In contrast, there was little to no difference in the Ca/P mole ratio between the drug-loaded coacervate and either large or small microspheres (Table 1). Similarly, the average degree of polymerization of the polyphosphate present in the minocycline-loaded microspheres was comparable to the initial chain length of the polyphosphates used to make the microspheres (Figure 2). These results suggest that throughout the process of drug loading and microsphere production, there is little to no change in the polyphosphate chain structure. This is significant because altering the chain length can affect the properties of the coacervate, specifically considering the rate at which they break down. 18
Previous studies have used spectrophotometric analysis at various wavelengths when detecting minocycline.20, 21 One study suggests that peak absorbance of minocycline is at 283 nm. 22 The challenge when studying minocycline is that it undergoes an autocatalytic reaction when in aqueous solution. 21 This was observed in preliminary studies, as minocycline solutions would undergo a change in colour from yellow to brown. From these studies it was found that absorbance values at 325 nm remained relatively unchanged over a 7-day period, suggesting that even with possible minocycline degradation, the antibiotic concentration can be reliably estimated using this wavelength. Interestingly, during our elution studies, the minocycline-loaded microspheres (and resulting coacervates) maintained their yellow colour, suggesting a possible protective role of CPP in preventing minocycline breakdown. However, further studies are required to determine if there are any changes to antibacterial activity after storage in and/or during sustained release from CPP microspheres.
In this study drug loading was highest in the CPP coacervate, reduced in the large microspheres, and lowest in the small microspheres (Table 1). This loss of minocycline is likely due to dissolution of the drug into chloroform/acetone during the formation of microspheres. Because small microspheres have a larger surface area, one would expect more drug loss during the emulsion process, leading to reduced encapsulation efficiency overall. Notably, drug loading for CPP microspheres reported in this study is significantly lower than that reported for PLGA microspheres used in Arestin®. 6 However, the use of polyphosphate glass as a carrier for drug release is in its infancy. To our knowledge, this is the first study demonstrating the drug loading of a CPP coacervate and subsequent retention and release of the drug from a microsphere format. Further optimization studies of the drug-loading process, with particular consideration to the initial polyphosphate Dp and minocycline concentration, are required.
Prolonged release of minocycline from the polyphosphate glass microspheres was observed over 7 days. Both large and small microspheres exhibited similar minocycline release profiles except at early time points up to 24 hours, where presumably the higher surface area of the small microspheres contributed to initially higher antibiotic release prior to aggregation of these microspheres and their conversion back to a coacervate in an aqueous environment. The interaction term obtained through a subsequent two-factor ANOVA indicated that the two release curves are not significantly different (
Polyphosphate glass microspheres are an attractive prospect for local drug delivery in the treatment of periodontitis. Potential clinical benefits of this carrier system include: (1) the ability to deploy as a gel or microspheres in response to clinician preference with respect to handling characteristics; (2) inherent bioadhesive qualities that will help to ensure the stability of the carrier/drug within the gingival sulcus without the need for additional retention; (3) anticipated biocompatibility owing to its composition (calcium ions and polyphosphates); and lastly (4) increased bioavailability of polyphosphates that may provide a supplemental role in treating periodontitis. Although previous work has suggested the biocompatibility of these polyphosphate coacervate-based systems and their ability to release functional antibiotics,15, 24 future studies including
Conclusions
Overall this study demonstrated repeatable and reliable drug loading of CPP coacervate at room temperature using minocycline, as well as
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Natural Sciences and Engineering Research Council of Canada (Discovery Grant), Springboard Atlantic (Innovation Mobilization Program) and the Faculty of Dentistry, Dalhousie University.
Guarantor
Mark Filiaggi (MF).
Contributorship
All authors researched the literature and conceived the study. IG and AM were involved in protocol development, data collection and analysis. IG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
