Abstract
BACKGROUND:
Electrospun nanofibers could simulate the natural extracellular matrix (ECM) of the host bone, while minocycline (MINO) is a broad-spectrum tetracycline antibiotic which has been found to have multiple non-antibiotics biological effects that promotes osteogenesis in vitro and in vivo.
OBJECTIVE:
The present study aims at constructing a polylactic acid (PLA) electrospun nanofiber membrane loaded with MINO to enhance Bone marrow mesenchymal stem cells (BMSCs) adhesion and proliferation for early clinical treatment.
METHODS:
The MINO-PLA membrane were characterized by scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR) and in vitro drug release study. The antibacterial ability was also investigated. In addition, in vitro cellular proliferation experiment was performed to verify whether the PLA electrospun nanofibers membrane loaded with MINO enhance BMSCs adhesion and proliferation.
RESULTS:
Analyzing the drug release and cell growth results, it was found that only the effective concentration of MINO-PLA could help the growth of BMSCs in the short term. This is related to the drug release rate of MINO-PLA and the initial concentration of MINO.
CONCLUSION:
This study shows that by controlling the concentration and release rate of MINO with electrospinning PLA, BMSCs could proliferate on it, and a new bone repair material had been made in this study.
Introduction
More than one million orthopaedic implants worldwide are implemented per year to treat osteoarthritis or other impaired bone tissues [1]. However, there are many reasons for poor treatment effect while insufficient osseointegration between the interface of implant and bone tissue is the main causes of treatment failure [2]. Osseointegration is a sophisticated process, in which the most critical steps are enhancing angiogenesis and promoting osteogenesis [3,4]. The development of a suitable implant that enables a systematical manipulation of osteo/angiogenesis and osteoimmunomodulation is highly desirable. Nowadays, the improvement of implants usually focuses on three key goals: extending implant lifespan, improving functional outcomes, and reducing complications.
Titanium-based implants have been widely used in clinics. However, original bioinert implants without initial bioactivity failed to promote osseointegration [5]. The ideal implant surface is to maximally mimic the natural extracellular matrix (ECM) of the host bone, which is mainly composed of nanoscale collagen fibers ranging from 50 to 500 nm in diameter [6]. The nanofibers are then aligned to form a higher-order microscale structure that controls cellular behavior and its structure, and ultimately induces a de novo bone formation [7]. The diameter of electrospun nanofibers would meet these criteria, which has been used to ameliorate and accelerate osseointegration in many studies. So we compared the growth of BMSCs on the electrospun membrane and medical titanium, to verify that the electrospun membrane can help BMSCs adhere and proliferate, so that BMSCs can better grow and differentiate into bone cells to help bone repair.
Mesenchymal stem cells can replicate as undifferentiated cells and may differentiate into lineages of different mesodermal sources, such as cartilage, bone, fat and bone marrow matrix [10]. The cell differentiation will be affected by the growth environment and chemical biological signals [11]. We hope that the nanofiber membrane can help BMSCs split and reproduce in early culture, to produce more BMSCs to help bone repair.
Minocycline (MINO) is a second-generation, semi-synthetic tetracycline that has been in therapeutic use for over 30 years [12] because of its antibiotic properties against both gram-positive and gram-negative bacteria. It has recently been found to have multiple non-antibiotic biological effects that are beneficial in experimental models of various diseases with an inflammatory basis, including dermatitis, periodontitis, atherosclerosis and autoimmune disorders such as rheumatoid arthritis and inflammatory bowel disease [13]. Meanwhile, it has been shown to inhibit bone resorption and promote osteogenesis [14] and bone resorption [13]. Pedro and colleagues demonstrated that MINO significantly increased the proliferation of human bone marrow osteoblasts at 1 mg/ml, the standard therapeutic concentration in plasma, without affecting their phenotype or function. However, high concentration of MINO can produce dose-dependent toxic effects on cells, inhibiting the proliferation and differentiation of osteocyte [14]. Therefore it is very important to screen the optimal safety range of MINO concentration promoting osteogenesis [15]. In addition, the presence of MINO can help patients resist potential risks such as bacterial infection after surgery, which is conducive to better recovery of patients.
The electrospinning technique has been identified as a cutting-edge approach to fabricating polymer nanofibers [16], which have large surface area with high porosity. Electrospun nanofibers offer several advantages to develop as wound dressings. It has good drainage and strong breathability, and can be easily loaded with therapeutic drugs [17]. In general, nanofibers loaded with drugs are prepared by using a mixture of polymer and drugs for electrospinning, both of which must be dissolved or finely dispersed in the same solvent. It has been observed that during the electrospinning procedure, drug molecules in the suspension are capable of migrating towards the nanofiber surface during the process of rapid jet-stretching and evaporation of the solvent solution [18]. Thus, a significant burst release in the initial stage of the release is inevitable. Also, the release kinetics depend on how well the drug molecules are distributed inside the nanofibers.
Therefore, it is feasible to construct MINO loaded electrospun nanofibers with a control released rate of MINO to help the process of bone repairing. In the article, we chose polylactic acid (PLA) without inflammatory response and biotoxicity [19] as the electrospinning carrier containing various concentration of MINO. The characterization of SEM, FTIR, drug release, antibacterial study and in vitro cellular experiment were conducted to determine whether it could promote osteogenesis.
Material and methods
Materials
Polylactic acid (PLA) was purchased from NatureWorks LLC (United States), Organic solvents chloroform and dimethylformamide (DMF) were supplied by Sinopharm Chemical Reagent Co., Ltd. Minocycline (MINO), Escherichia coli(ATCC25923) and Staphylococcus aureus(ATCC25922) was purchased from Solarbio (Beijing, China). Titanium was obtained from Baoji, China. SD rats were provided by the Second Affiliated Hospital of Anhui Medical University. DMEM medium and α-MEM basic medium were purchased from Solarbio (Beijing, China).
Synthesis of PLA membranes loaded with MINO
The polymer solution was prepared by dissolving 1 g of PLA and MINO (0.5%, 1% and 2wt% relative to the PLA weight) in 10 ml of solution made of 3 ml of dimethylformamide and 7 ml of chloroform. The solution was stirred with a magnetic stirrer bar until the solution became completely clear. The resulting solution was then placed into a 10 ml syringe with a metal needle. A voltage of 15 kV was applied to the syringe needle and the collector was placed 20 cm from the needle tip. To control the same cell growth area of electrospun membrane and medical titanium. The round titanium sheets with a diameter of 0.5 mm were placed on the receiving device for cellular experiments. The solution flow rate was set at 5 μL/h. The electrospinning was carried out at room temperature (22 ± 2 °C) and relative humidity of 50 ± 5%. The collected nanofiber membranes were kept overnight in a desiccator to ensure complete evaporation of the solvent.
Characterization of MINO-loaded PLA membrane
Scanning electron microscopy (SEM)
SEM (SU8220, Hitachi, Japan) was used to observe the morphology of PLA electrospun nanofiber membrane containing MINO (0%, 0.5%, 1%, 2%) after sputter-coating with gold for 90 s. The fiber membranes were carefully fixed on stubs, and then gold coated by a sputtering device before SEM observation. The obtained fiber images were analyzed by ImageJ software (50 fibers were randomly selected for each sample).
Fourier transforms infrared spectroscopy (FTIR)
The chemical compositions of PLA and 0.5%, 1%, 2% MINO-PLA electrospun membrane were determined by FTIR (MA). The samples were observed using the attenuated total reflection (ATR) method. The wavelength range of infrared absorption was 500∼4000 cm−1. The FTIR spectrum of samples were compared with standard FTIR curve of MINO.
In vitro drug release
The release of MINO from electrospun membranes was determined by cutting the membranes into slices of equal weight and incubating in 5 mL of phosphate buffered saline (PBS). The optical density (OD) values of MINO released from membranes were measured at absorbance wavelength at 375 nm. The absorbance of standard curve at 375 nm was also measured by using PBS solutions with different MINO concentration.
The sample was collected at different time point and the OD values of MINO were converted to concentrations using a standard curve. The results were expressed as the cumulative amount of released MINO at each time point, according to the formula:
Antimicrobial susceptibility test
The antimicrobial activity of the electrospun membranes coated titanium sheet was assessed by the agar diffusion test. The strains used were Staphylococcus aureus strain ATCC25923 and Escherichia coli strain ATCC25922.
First, the prepared Staphylococcus aureus and Escherichia coli were inoculated on a petri dish using an inoculating loop device and cultured in a 37 °C incubator for 24 h. Then, the bacteria on the petri dish were scraped, and fully dispersed in physiological saline to prepare a bacterial solution with a concentration of 1 × 105 CFU/ml by McFarland turbidimetry. Finally, a quantitative amount of the bacterial solution was placed in the center of the petri dish, spread evenly with a glass rod and left to dry out. The sterilized titanium sheet was then placed on the petri dish with a sterile tweezer. The zone of inhibition was observed after culturing for 24 h in a 37 °C incubator.
Cell culture
Extraction and culture of rat BMSC
SD male rats were killed by intraperitoneal injection of excessive pentobarbital sodium (8 mg/100 g.b.w). In a sterile environment, the bilateral femur and tibia of the rats were removed. The surface muscle and fascia were then removed, and the metaphyses on both sides were cut off using a bone rongeur. 10 ml of α-MEM basic medium was sucked into a 1ml syringe and used to repeatedly wash the bone marrow cavity of the rats until it turned white. The washed primary bone marrow cells were planted in a 10cm culture dish, beaten evenly, and cultured in a thermostatic cell incubator (37 °C, 5% CO2). The adherent cells were observed to grow in fibrous colonies under the microscope when they were placed in the cell to be aggregated and adhered to the wall, and the culture medium was changed every 2 days. Primary bone marrow mesenchymal stem cells (BMSCs) were cultured in DMEM medium supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin in 5% CO2 at 37 °C. The medium was replaced every 2–3 days.
CCK-8 (cell proliferation)
The viability of BMSCs in different materials were evaluated by CCK-8 (Sigma-Aldrich, St. Louis, MO, USA) method. After BMSCs were seeded in 96-well plates at 5 × 103/well for 1 day, 3 days and 7 days, respectively, 10 μL of CCK-8 was added to each well and cultured for 2 h. The absorbance of samples was measured at 492 nm with Rayto RT-6100 microplate reader immediately.
Scanning electron microscopy (SEM) (cell attachment)
After BMSCs were seeded at 5 × 103/well in 96-well plates for planned days, the samples were washed three times with PBS and then fixed in electron microscopy fixative for 12 h, and dehydrated in gradient ethanol (50%, 75%, 90% and 95%). The cell morphology was observed by SEM.
Results
Characterization of MINO-loaded PLA membrane
The morphology of PLA fibers containing 0%, 1%, 2% and 3% MINO observed by SEM was shown in Fig. 1. These fibers are typical continuous ribbon without beads or broken wires. It was shown that the morphology of PLA fibers was not affected after loading with different concentration of MINO. Analyzed by ImageJ software, the mean diameters of PLA fibers containing 0%, 0.5%, 1% and 2% MINO were 329±156, 330±100, 315±42 and 331±53 nm, respectively. The 50 fibers selected were showed in Fig. 1. The diameter of PLA fiber was, therefore, not affected by the concentration of MINO because there was no statistical significantly difference (P > 0.05).

SEM of fibers (A) PLA, (B) 0.5%MINO-PLA, (C) 1%MINO-PLA, (D) 2%MINO-PLA.
The FTIR of PLA fibers containing different concentration of MINO was shown in Fig. 2. The spectrum showed the bands of the -CH, -CH2, -CH3 stretching vibrations (2950 cm−1), the carbonyl C=O stretching vibrations (1747 cm−1) and the C–O stretching vibrations(1085 cm−1). The FTIR spectrum did not significantly change with an increased concentration of MINO which proves no new molecular structure was created.
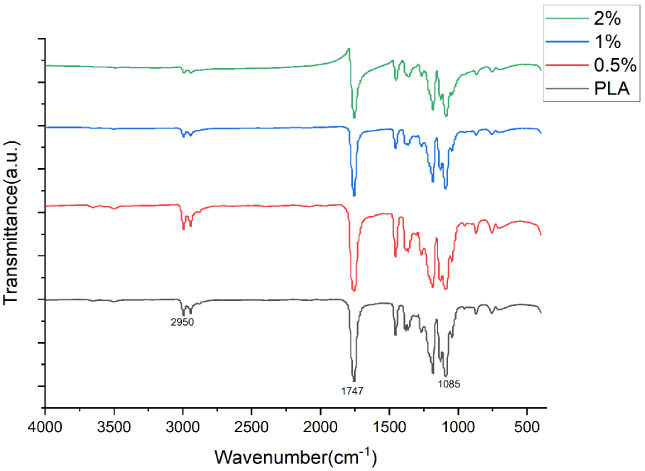
FTIR spctra of PLA, 0.5% MINO-PLA, 1% MINO-PLA, 2% MINO-PLA.
The kinetics of MINO release from PLA fibers and the standard curve of MINO concentration and absorbance was shown in Fig. 3. An initial burst release of MINO was observed on the first day, with 56.67%, 40.11% and 44.94% of MINO released from 0.5%, 1% and 2% MINO-loaded PLA fibers, respectively. The drugs in 0.5% MINO-loaded PLA fibers were completely released on the seventh day. The correlations obtained by linear fitting analysis were 0.5%: R2 = 0.968, 1%: R2 = 0.935, 2%: R2 = 0.911. The slopes were 0.5%: 0.067, 1%: 0.054 and 2%: 0.062, respectively.
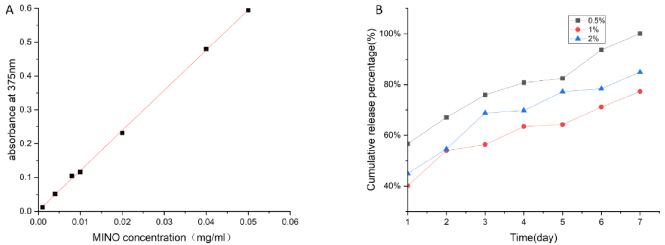
(A) Standard curve of MINO concentration and absorbance at 375 nm, (B) MINO release properties of the 0.5%, 1%, 2%MINO-PLA membranes in a week.
The zone of titanium sheet inhibition was illustrated in Fig. 4. The diameters of the inhibition zone were as follows: (1) Escherichia coli: 0.5%: 1.3cm, 1%: 2.0cm, 2%: 2.5 cm; (2) Staphylococcus aureus 0.5%: 2.0 cm, 1%: 2.5 cm, 2%: 3.0 cm. It can be concluded that with the increase of MINO concentration, the antibacterial effect of titanium sheets coated with MINO-PLA nanofibers was enhanced, and the Staphylococcus aureus had a stronger antibacterial effect. The agar diffusion test showed that with the increase of MINO concentration, the diameter of the inhibition zone increased and the bacteriostatic ability increased. Connecting with the result of FTIR, it was speculated that MINO exerted the bacteriostatic effect due to MINO releasing from the fiber rather than generating new functional groups on the surface of fiber.
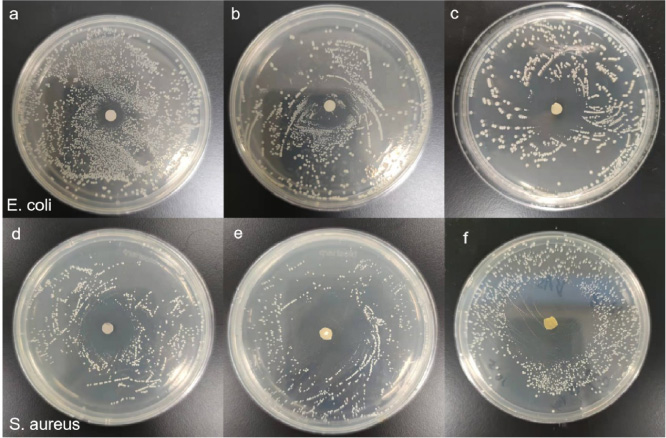
Bactericidal activity of 0.5% (a, d), 1% (b, e), 2% (c, f) MINO-PLA on titanium.
The cell proliferation was evaluated by the cck-8 method as shown in Fig. 5. On day 1, the growth activity of cells on titanium sheet coated with MINO-PLA nanofiber was greater than that of cells on titanium sheet only. There was statistical significantly difference (P > 0.05) observed in titanium sheet coated with 2% MINO-PLA nanofiber. On day 3 and day 7, no significant difference was observed among all groups (P < 0.05). Therefore, the morphology of cells grown on titanium sheet and titanium sheet coated with 2% MINO-PLA nanofiber on day 1 was observed by SEM. The results were shown in Fig. 6. The cells grown on titanium sheet coated with nanofibers spread with a large number of filopodia and evenly attached to the nanofibers, while the cells on titanium sheet remained circular with smooth edges, and the adhesion area was small. These findings indicated that MINO-PLA nanofibers membranes, especially the 2% MINO-PLA nanofibers membranes promoted BMSCs growth compared with traditional medical titanium sheets.
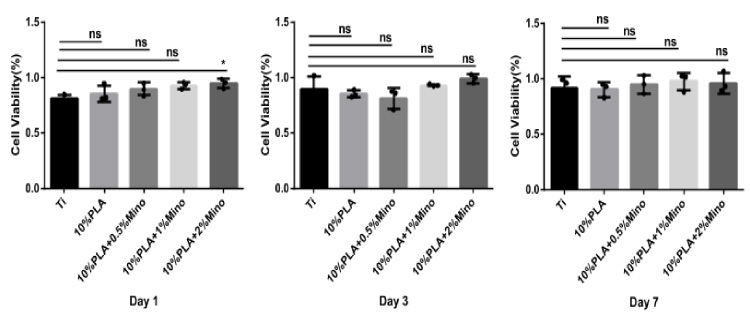
CCK-8 assays of cells cultured on titanium sheets coated with 0%, 0.5%, 1%, 2% MINO-PLA membranes and Ti.

SEM of cells grown on (A) Ti, (B) PLA, (C) 2%MINO-PLA at the first day.
Previously published articles have shown that the network architecture of electrospinning fibers can promote cell growth [6,7]. Longs experiments proved that the network porous coating on titanium can help bone integration [9]. Some researchers noticed that MINO can promote the proliferation and differentiation of kinds of bone cells [20–22]. Combining the previously experimental conclusions, in this experiment, the titanium surface coating of PLA electrospinning fiber loaded with MINO was made. It could control the concentration of MINO while providing a network structure to help BMSCs adhere and grow. MINO also produced an antibacterial effect to avoid bacterial infection. Our study found that MINO-PLA, especially containing 2% MINO, promoted the proliferation of BMSCs. It was based on the increased attachment of BMSCs on the MINO-PLA membranes and the enhanced cell number detected by CCK-8 assay.
The fiber morphology is the basis for electrospinning fiber membrane to simulate the extracellular matrix. From the picture of SEM, we produced smooth, continuous, beadless, randomly distributed nanofibers successfully and made sure no new molecular structure was created during electrospinning by FTIR.
As the basic ability for MINO, antibacterial assay was assessed by incubating prepared samples in an agar disc. Bacterial infections are associated with many medical situations, resulting in slower wound recovery, fever, and other complications [23,24]. Therefore, antibacterial activity is necessary for biomaterials. The results indicated that the PLA-loaded MINO displayed good antibacterial activity aimed at both Gram-positive and Gram-negative bacteria.
The drug release rate of 10% PLA with different concentrations of MINO was measured. The results showed that the drug release rate was more related to the porosity of PLA than to the concentration of MINO. Generally, there will be an explosive release on the first day, which may be determined by the surface area of the spinning.
The drug release performance of a drug delivery system affects its efficiency for various biomedical applications. The suffering of patients could be reduced by the control of the drug release over a period since frequent dressing changes can be eliminated. In the in vitro drug release study, it was observed that an initial burst released on the first day followed with a sustained release until the end of the study. This might be because MINO was evenly distributed in the inner and outer layer of fiber during the electrospinning process. After the drug on the surface was released at the initial stage, drugs dispersed in the inner layer were released with the degradation of the matrix material. Moreover, MINO-PLA nanofiber with many pores provided a large contact area with PBS, resulting in burst release on the first day [25,26]. Therefore, we can control the drug release rate by controlling the electrospun structure or changing the spinning materials.
The cell activity was measured by CCK-8 assay. It was found that the cell activity of 2%MINO-PLA on the first day was significantly higher than that of Ti, which indicated that a certain concentration of MINO-PLA really helped the adhesion and growth of bone marrow mesenchymal stem cells (BMSCs) in a short time. The cell electron microscope images also verified this result. Comparing the cell activity of the group of PLA to Ti, the cell activity of PLA was higher than the other, but there was no statistical significance. Observe the cell electron microscope images of the first day of PLA and Ti, we saw that the cells grown on nanofibers membrane spread with a large number of filopodia and evenly attached to the nanofibers, while the cells on titanium sheet remained circular with smooth edges, and the adhesion area was small. So that the electrospun PLA membranes could help cell adhesion and growth. Meanwhile, the cell activity of the first of 2%MINO-PLA was higher than that of other groups, and it was significantly higher than that of Ti. So, the MINO actually helps BMSCs growth although not as effective as it is on bone marrow osteoblastic cells which could be derived from BMSC. However, with the growth of cells, the cell activity among groups was no longer significant on the third and seventh days. According to the drug release results, it was speculated that the concentration of MINO could not reach the working concentration to help bone cells grow after changing the culture medium [14]. Because of metabolism and other reasons, the concentration of MINO after injection fluctuates greatly in the body and cannot play a good role. The clinical solution is to inject MINO weekly for one month to keep the concentration of MINO [8]. This will bring constant and unnecessary pain to the patient. Therefore, it is necessary to develop materials with stable release MINO by electrospun.
Therefore, the release of MINO can be controlled in the future by modifying matrix of nanofiber to promote BMSCs growth. Also, different electrospinning methods will affect the final fiber structure, leading to changes in cell adhesion and the release of loaded drugs. It was better that the releasing of MINO could be stable so we could control the concentration of MINO always been working concentration. Although, this material had been proven to have the effect of promoting bone repair, in vivo study also needs to be explored in future study to further evaluate titanium sheet coated with MINO-PLA membrane on promoting BMSCs proliferation and bone repair. The material we had made in this article was supposed to have the prospect of clinical application.
Conclusions
MINO-PLA electrospun nanofiber membranes were successfully prepared and demonstrated antibacterial properties and the ability to promote BMSCs early proliferation and attachment in vitro. This is probably due to the dual role of nanofiber structure and MINO. This material has a good surface morphology and no new groups are formed during MINO dissolution and electrospinning. The loaded MINO can resist bacteria and promote bone repair at an effective concentration. Despite an initial burst release of MINO was observed on the first day, the followed sustained release was last until the end of study, indicating that it had adequate release rate and duration in vitro. Compared with the traditional medical titanium sheets, MINO-PLA nanofiber membrane promoted cell attachment and proliferation. The release of MINO can then be controlled by using electrospun prepared by different polymer. Therefore, it is a potential therapeutic strategy with loading MINO into PLA electrospun membrane with sustained release. Different electrospinning materials and technologies can be used to control the release of drugs, which is valuable for further research.
Footnotes
Ethical approval
The study protocol was approved by the Experimental Animal Ethics Committee of Anhui Medical University (LLSC20220452).
Conflict of interest
There is no conflict of interest among the authors.
Funding
This work was supported by the Fundamental Research Funds for the Central Universities (No. USTC 20210079), Plasma Applied Technology Joint Laboratory Development Funding (JL06120001H), National Research Program on the Development of Magnetic Confinement Fusion Energy (2022YFE03080000), Anhui Science and Technology Department (202007d07050007) and Hefei Science and Technology Bureau (J2020Y07).
