Abstract
Magnesium alloys have been investigated as biodegradable metallic materials because of their good biocompatibility with natural bone, while zinc has basic safety properties for biomedical applications and is one of the nutritionally essential elements for the human body. The microstructures and properties of some novel designs of bio-Mg-Zn alloys are studied to provide implantations which have an improvement in mechanical properties and corrosion resistance.
Introduction
Magnesium and magnesium alloys have been attractive for biodegradable implants due to their good biocompatibility. 1 The magnesium alloys have low elastic modulus, which is closer to that of natural bone, and osteoinductive coating on the implant that promotes the interaction with bone and implant fixation. When titanium alloys are used, no stress shielding effects are reported. 2 The stress shielding effects are caused by the mismatch of elastic modulus between the bone and metallic implants, which can lead to critical clinical problems such as implant loosening, skeletal thickening, and chronic inflammation. 3 However, new metallic device matrices that can better compare the elastic modulus with natural bone need to be developed, and so designing biodegradable magnesium implants has become a research focus in recent years.4, 5 Many kinds of magnesium alloys, such as Mg-Al, Mg-Zn, Mg-Mn, Mg-Ca, and Mg-RE systems, are investigated for in vitro and in vivo tests. 6 However, some elements in magnesium are poisonous for the human body; such as in the Mg-Al alloy, which is widely applied, where the Al shows toxicity on the neurons and induces dementia. The rare earth (RE) elements lead to hepatotoxicity and genotoxicity. The Mn has neurotoxicity and therefore excessive intake of Mn may cause Parkinson’s disease. So, the only elements which have no toxicity for humans when alloyed with biodegradable magnesium biomaterials are zinc and calcium. 6
Zinc is one of the most abundant nutritionally-essential elements in the human body and plays an important role in many physiological functions, 7 and the Zn-containing magnesium alloys have been paid more attention lately. 8 Zinc can effectively strengthen the magnesium matrix through a solid solution hardening mechanism and improve the corrosion resistance synchronously (pure Mg: 0.20 in vitro (mm year-1); Mg-6%Zn alloy: 0.16 in vitro (mm year-1).9, 10 Some alloying elements, such as Zr, Sr, or Mn, are added in Mg-Zn binary alloys to enhance the mechanical properties or create a small grain dimension, 10 but investigations seldom focus on different compositions of Mg-Zn binary series alloys to evaluate their potential biomedical characteristics as for implants. As is well known, the corrosion resistance of Mg-Zn alloy cannot meet the requirements of bone replacement or repair, because the corrosion rates of many Mg alloys are too rapid to allow sufficient time for healing (at least 12 weeks). 11 The release of hydrogen gas due to the biodegradation of the Mg matrix in body fluid is the main disadvantage of Mg-Zn alloys. 12 Surface treatment is one of the methods to decrease the corrosion rate, so that the material is stable for more time in vivo, but another effective solution is the composition modification of Mg-Zn alloys. 13 Therefore, according to the Mg-Zn binary phase diagram, 14 the microstructures and properties of series typical Mg-Zn alloys are studied in this investigation to provide the impetus to design some novel Mg-Zn alloys with improvements including mechanical properties and corrosion resistance. The composition and characteristics of the newly designed Mg-Zn binary alloy can be treated as the matrix to further develop metallic biomaterials with good biocompatibility.
Materials and methods
Material production
The designed chemical compositions (at%) of the experimental Mg-Zn binary alloys are Mg99Zn1, Mg97.5Zn2.5, Mg75Zn25, Mg50Zn50, Mg25Zn75, and Mg1Zn99. The sintering process is a promising method to prepare metal-based alloys and composites. After sufficient mixing, the individual components can be homogeneously mixed to reduce spatial heterogeneity. The high purity magnesium powder (>99.99%, provided by Wenxi Yinguang magnesium industry, Shanxi) with the average size of 107 μm and zinc powder (>99.999%, provided by North Mine Zinc Industry, Beijing) with the average size of 24 μm were mixed in a three-dimensional blender for about two hours (the powder dimension is the one declared by the producer), then hot pressed at 500°C under a pressure from 20 to 100 MPa with the protection of Ar gas in a vacuum furnace. The diameter of the sintered billet was 64 mm and the thickness was 18 mm. Three samples of each of the alloys were tested in mechanical and metallurgical testing, density testing and in vitro corrosion testing, and the average values are presented as the results.
Mechanical and microstructure testing
The samples for mechanical tests and microstructure analysis were cut from the sintered billets. The in vivo compression is the main force of implants for bone defect repair, and the compressive strength is an important reason for mechanical instability of implants. 15 Therefore, the compression test is more suitable than the bending and tensile tests due to the measurement of the samples’ mechanical properties in this experiment. The compression testing was carried out using the MTS 810 material test system. Specimens machined from sintered billets for mechanical testing were a shape with diameter of 10 mm and a thickness of 20 mm, and the compression speed was 1.0 mm/min, based on ASTM E9-89a. The densities of the experimental alloys with diameter of 10 mm and thickness of 20 mm were tested through the standard Archimedes method and an electronic balance with pure ethanol. The surface hardness of Mg-Zn binary series alloys was measured by a Brinell Hardness Tester HBE-300. The diameter of the steel ball (D) is 5 mm, the load force (F) is 250 N and the loading time is 30 s. Taking the average of three points as the Brinell hardness, based on ASTM E10-2014, we measured the diameter of a round indentation (d) and the Brinell hardness of the samples was calculated.
The microstructures of the experimental alloys were observed with a Jeol JSM-5600Lv scanning electron microscope (SEM) with an energy-dispersive X-ray spectroscope (EDX).
In vitro corrosion testing
The corrosion resistance of experimental Mg-Zn alloys was measured in Ringer’s solution at 37°C. The specimens for the corrosion test were ground with 1000-grid silicon carbide (SiC) sandpaper and cleaned with ethanol. Immersion corrosion testing under static conditions was performed in Ringer’s solution at 37 ± 0.2°C for 448 h. The Ringer’s solution was a solution of recently boiled distilled water containing 8.6 g of sodium chloride, 0.3 g of potassium chloride, and 0.33 g of calcium chloride per liter. 16 The Ringer’s solution was not stirred during the experiments, was saturated with atmospheric oxygen, and the solution temperature was controlled with an HTW-10B water bath. The amount of hydrogen bubbles generated from the soaked specimens were measured from the different heights of measuring cylinders.
Cytocompatibility assessments
Mouse fibroblast L929 cells were incubated with extracted medium (10%, 50%, and 100%), culture medium as negative control, and 0.1% phenol solution as positive control in three days. The cytotoxicity tests were performed according to the MTT method, and the relative growth rate (RGR) was calculated according to the optical density (OD) values measured by Thermo Election Corporation at 490 nm wave length based on ISO 10993-2009 standard. As the average OD values of the negative control was defined as 100%, the RGR of the rest groups was calculated according to the following equation: RGR (%) = (average OD values / negative control average OD values) ×100%.
Results and discussion
The microstructures and phase identification of six experimental alloys are shown in Figure 1. Different alloys exhibit obviously different morphologies at sintered state. The sintered magnesium matrix grains with an average size about 100 μm exist in Mg99Zn1 alloy and only small-sized second phases in white color distribute along the grain boundary in Figure 1(a). Such white color intermetallic becomes more distinct in the grain boundaries of Mg97.5Zn2.5 alloy in Figure 1(b). The morphology of Mg97.5Zn2.5 alloy shows a cellular dendrite structure and the composition of intermetallic phases are detected by EDX analysis in Figure 1(c). The ratio of magnesium atom to zinc atom is about 7:3. According to the Mg-Zn phase diagram, the intermetallic phase as the ratio of Mg atom to Zn atom should be Mg7Zn3 compound, 8 and this intermetallic Mg7Zn3 phase obtains obvious strength, increasing its effect on the magnesium matrix. The Mg7Zn3 phase could hinder the dislocation movement and contribute to the dispersion strengthening. 17

The morphologies and phase identifications of sintered experimental Mg-Zn alloys: (a) morphology of Mg99Zn1 alloy; (b) morphology of Mg97.5Zn2.5 alloy; (c) phase identifications in Mg97.5Zn2.5 alloy; (d) morphology of Mg75Zn25 alloy; (e) morphology of Mg50Zn50 alloy; (f, g, h, i) phase identifications in Mg50Zn50 alloy; (j) morphology of Mg25Zn75 alloy; (k) phase identification in Mg25Zn75 alloy; (l) morphology of Mg1Zn99 alloy.
With the increasing addition of zinc in magnesium to produce Mg75Zn25 and Mg50Zn50 alloys, the morphologies of microstructures exhibit typical highly-developed dendritic structures (Figure 1d and e). The dark contrast is mainly magnesium matrix and the white contrast is the intermetallic compounds. With an identification of these different phases in Figure 1(f), it can be confirmed that three phases possess different compositions and show different contrasts. The atom percentage of the compound at point A in Figure 1(g) shows it is also the Mg7Zn3 phase; and at point B, the composition shows it should be MgZn2 phase because the atom proportion of Mg and Zn is close to 1:2 (Figure 1h). 8 At point C in the boundary of the dendrite, the enrichment of zinc illustrates that zinc might be melted at the sinter temperature of 500~600 °C due to its melting point at only 419.5 °C (Figure 1i). The sintering in this study was the hot-press sintering. Under pressure, the melting of an alloy is a non-equilibrium condition, and parts of zinc were melted at 500°C. Such melted zinc might lead to some shrinkage and porosity in the sintered specimens and be harmful to the mechanical properties.
In Mg25Zn75 alloy, the compound distribution on the dendritic boundary is the MgZn2 phase, according to the proportion of atom percentage of Mg and Zn (Figure 1j and k). Such compound is very stiff but brittle and results in a high hardness with a bad ductility for the alloy specimens. The strength and hardness of Mg25Zn75 reaches an obviously higher value than those of other Mg-Zn alloys, but with the disadvantage of bad ductility (ductility of 4.2±1.7). In the Mg1Zn99 alloy, such phase can also be observed, but its size becomes smaller and surrounded with pure zinc matrix (Figure 1l). So, the ductility of Mg1Zn99 alloy (ductility of 6.7±1.0) can be improved compared with the Mg25Zn75 alloy.
The typical physical and mechanical properties of the six experimental alloys are listed in Table 1. In these experimental alloys, only Mg75Zn25 and Mg50Zn50 obtain some obvious shrinkage or porosity after sintering, due to the melting of zinc during the solidification process. In the sintering process, different Mg-Zn alloys produced different volume content and components of the liquid phase. When the liquid phase transited to the solid phase, different volume shrinkages occurred, and some of them were intense, which caused the shrinkage or porosity. The varying diffusion rates between Mg and Zn during the sintering process differed widely; and the closer the atomic weight of magnesium and zinc, the more volume shrinkage was induced by the Kirkendall effect. 18 Such defects of constriction cause bad effects on the properties of sintered Mg-Zn alloy, such as strength or corrosion characteristics. The compression strength of Mg99Zn1 alloy exhibits the lowest among the six experimental specimens as it is almost entirely composed of Mg matrix, so it seems to have no potential for biomedical implant use due to the low strength. The situation is similar for the Mg1Zn99 alloy, which contains mostly zinc. Mg99Zn1 and Mg1Zn99 alloys can be used as implants only required for low strength or non-bearing applications. Although Mg25Zn75 obtains a significant high strength and density, it is too brittle to be machined to the implant standard needed, so it is hard to be applied to implant biomaterial. The most potential alloy for implant in all the experimental alloys might be the Mg97.5Zn2.5 alloy, which exhibits good comprehensive physical and mechanical properties.
Physical and mechanical properties of different experimental Mg-Zn alloys. 11

The release of hydrogen gas is an important parameter to evaluate the corrosion properties of biodegradable Mg-Zn alloys in the body fluid. From Figure 2, it can be seen that the release of hydrogen gas of Mg75Zn25 and Mg50Zn50 alloys increases quickly in the body fluid due to their poor corrosion resistance; but the corrosion properties of other experimental alloys are in the tolerable range. The change of weight of biodegradable Mg-Zn alloys in the body fluid shows similar results as the release of hydrogen gas in 120 h, as shown in Figure 3. Mg25Zn75 and Mg1Zn99 had weight remaining of 100%, because the quality of the corrosion products adhered to the alloy surface was greater than that dissolved in the corrosion medium, which was declared the best corrosion resistance. Mg75Zn25 and Mg50Zn50 alloys exhibit most change of weight (9% and 17%) among a series of Mg-Zn alloys in 100 h of immersion. Due to micro-galvanic corrosion, the more the magnesium matrix around the intermetallic phases and zinc matrix was attacked, the more corrosion occured. 13 Therefore, Mg75Zn25 and Mg50Zn50 alloys cannot maintain stability for long enough on account of their over-fast corrosion rate. The Mg99Zn1 alloy exhibited the best corrosion resistance, because the lesser Zn content and more homogeneous Mg-Zn phases induced the less-localized corrosion attacks and more heterogeneous corrosion. 19
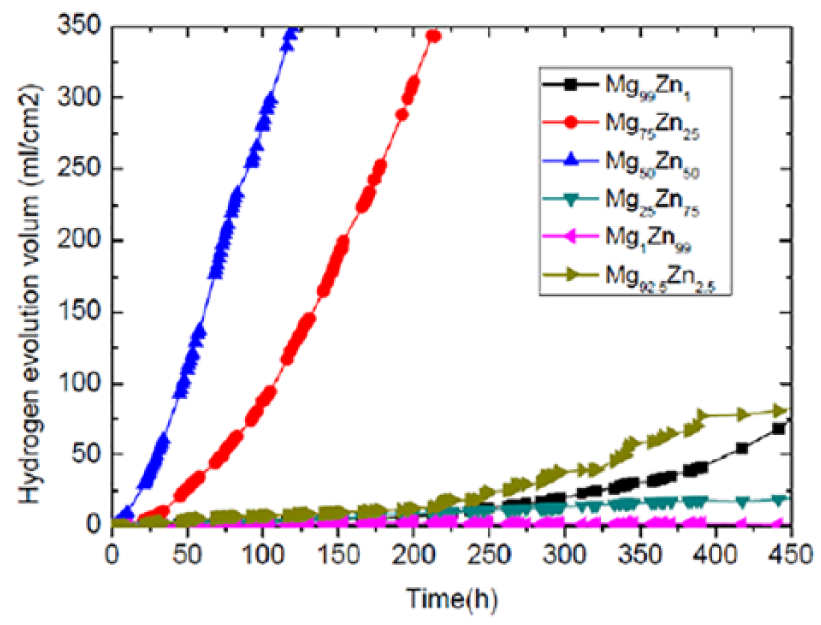
Release of hydrogen gas of Mg-Zn alloys in body fluid.
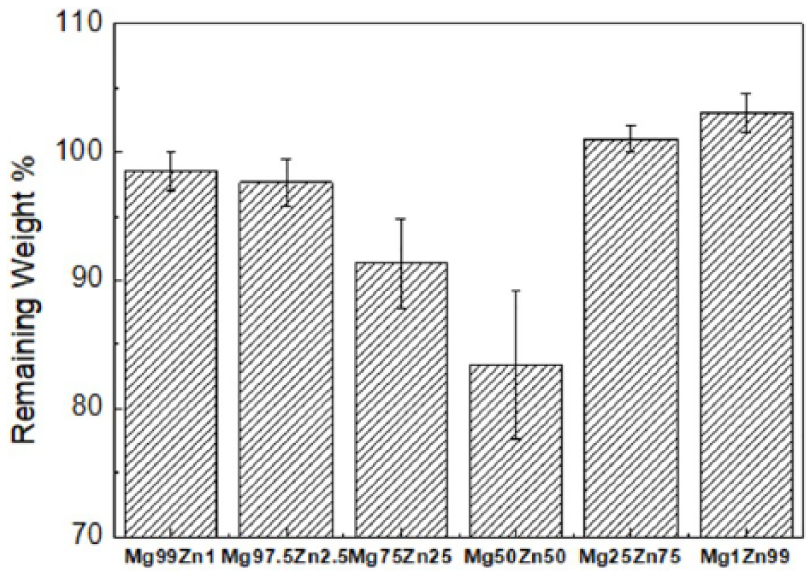
Change of weight of Mg-Zn alloys in 100 h of immersion.
According to the mechanical properties, phase compositions, and the release of hydrogen gas, Mg97.5Zn2.5 alloys seem to be the most potential metallic biomaterials. Mg99Zn1 and Mg1Zn99 can used for load-bearing applications. These three alloys were selected in cytocompatibility tests. The RGRs of the L929 cells for different groups are shown in Figure 4. The results demonstrate no significant differences between the RGR of the cells in the experimental group and those in the negative control. According to the ISO 10993-2009 standard, the cytotoxicity grade of these extracts of Mg99Zn1, Mg97.5Zn2.5, and Mg1Zn99 alloys were 0–1, which means they are not toxic for L929 cells. Finally, it can be seen that the RGR of Mg97.5Zn2.5 alloy is the highest among the three alloys, which illustrates its good cytocompatibility.

RGRs of the L929 cells cultured in different extracts of Mg-Zn alloys.
Conclusions
Compared with the mechanical properties, corrosion resistance, and cytocompatibility test results, obvious shrinkage or porosities in the Mg75Zn25 and Mg50Zn50 alloys decrease their mechanical properties and impede their applications. Although Mg25Zn75 obtains a significant high strength and corrosion resistance, it is too brittle to be machined to the implant standard needed. Mg99Zn1 and Mg1Zn99 showed good corrosion resistance, but can only be used for bearing applications, due to their low mechanical strength. The Mg97.5Zn2.5 alloy should be selected as the best candidate for biodegradable Mg-Zn series alloys.
Supplemental Material
Supplementary_Materials – Supplemental material for Comparison of the microstructure and properties of a series of Mg-Zn metallic devices on biomedical applications
Supplemental material, Supplementary_Materials for Comparison of the microstructure and properties of a series of Mg-Zn metallic devices on biomedical applications by Tao Zhang, Yilong Dai, Yang Yan, Yu Zhang, Kun Yu, Hui Liu, Hongjie Fang, Xier Luo and Li Li in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support of the 2015 Shandong province project of outstanding subject talent group, the project (81472058) supported by the National Natural Science Foundation of China, the Natural Science Foundation of Shandong Province of China (ZR2017MEM005), the project (A2016003) supported by the Health and Family Planning Commission of Hunan Province, the project (2017GK2120) supported by the Key Research and Development Program of Hunan Province and the Fundamental Research Funds of Central South University Project (2015zzts024).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
