Abstract
Hard palate, developed by embryo neural crest stem cells, is a tissue with strong regenerative abilities. It is considered an abundant source of progenitor cells, forming various mesenchymal tissues. Rabbits are more suitable models than murine animals for regenerative preclinical study of the head and neck, owing to their larger size. However, there are no reports of the existence or characteristics of neural crest stem cells in the hard palate of rabbits. In this study, we demonstrate for the first time the presence of nestin-, Sox2-, and p75-positive neural crest stem cells obtained from the hard palate of rabbits and the properties of these cells. Flow cytometry analysis revealed that CD29, CD44, and CD81 were positive; and CD11b, CD34, and CD90 were negative on the
Keywords
1. Introduction
In regenerative translational research studies using rats or rabbits, mesenchymal progenitor cells (MPCs) obtained from bone marrow are popularly used as the cell source for tissue-engineered construction. However, the procedure necessary to obtain bone marrow from humans, bone marrow aspiration, has disadvantages such as pain, donor site morbidity, and poor cell yields. As a result of research efforts to find donor sites that provide abundant MPCs without these disadvantages, studies have reported the existence of MPCs in various areas of the adult body, including the adipose tissue, 1 synovial membrane, 2 muscle, 3 and skin. 4
New efforts to easily obtain hematopoietic stem and progenitor cells that are directly involved in the histogenesis of defective areas around the head and neck have led to research reports confirming MPCs in various tissues of the head and neck located near the defective area, such as the palatine tonsil, 5 periodontal tissue, 6 and sinonasal mucosa. 7 Among these, neural crest stem cells (NCSs), which are dispersed from the neural crest that uniquely exists in vertebrate embryos, can develop not only into nerve cells, such as neurons and glial cells, but also into various cell types, including osteocytes, chondrocytes, and adipocytes. In addition, it has been discovered that MPCs exist within adult organs or tissues that are created from the neural crest progenitor cell of the embryo.8–11
Clinical observation has revealed that hard palate in the mouth is a tissue with extremely strong regenerative abilities. 12 It has been considered an abundant source of stem and progenitor cells. In the palate of human and adult rats, neural crest progenitor cells can differentiate into functional neurons and glia cells. 10 NCSs have pluripotency including osteogenesis, chondrogenesis, and adiopogenesis. 13 In clinical practice, it is easy to access and to obtain the tissue from the hard palate under local anesthesia, and the donor site heals rapidly with little discomfort. Taking into consideration such advantages, the hard palate will be used as an invaluable progenitor cell source in various research fields in the future.
Owing to their physiological similarity with humans and appropriate size for laboratory procedures, rabbits are more suitable than rodents as a model for conducting regenerative studies. Moreover, rabbit mesenchymal stem cells have stability and properties similar to those of human mesenchymal stem cells. 14 Rabbits have been gaining popularity as animal models for preclinical studies of the head and neck area, particularly for studies of the bone, cartilage, and cornea. The rabbit has also become a popular animal model in stem cell translational research.15–17 Using this animal provides added advantages in terms of stability and economy. However, there are no reports available on the existence or characteristics of progenitor cells in the hard palate of rabbits.
The aim of this research is to confirm the presence of MPCs in the hard palate of rabbit and to examine their properties. The rabbit is the most suitable animal candidate for stem-cell driven regenerative translational research with scaffolds for bone and cartilage defects of the head and neck area. We also investigated the potential differentiation in three different lineages,
2. Materials and methods
2.1. Rabbit hard palate stem cell isolation and primary cell culture
This study was reviewed and approved by the Institutional Animal Care and Use Committee (20150324002) of the Samsung Biomedical Research Institute (SBRI). SBRI is an Association for Assessment and Accreditation of Laboratory Animal Care International and abides by the Institute of Laboratory Animal Resources guide. Hard palatal tissue was harvested from 1-year-old New Zealand white rabbits (Orient Bio, Seongnam, Korea). The anesthesia injection was administered by an intravenous air bolus injection through the ear vein. Harvested palatal tissues were placed in phosphate-buffered saline (Gibco; Thermo Fisher Scientific, Waltham, MA, USA). Thereafter, the tissues were diced in alpha-minimal essential media (MEM) (Gibco; Thermo Fisher Scientific) composed of 3 mg/ml collagenase I (Sigma-Aldrich, St. Louis, MO, USA) and 4 mg/ml dispase (Gibco; Thermo Fisher Scientific) and passed through a culture process under temperature conditions of 37ºC for 1 hour. After filtering out the single cells with a 70 µm-cell strainer (Corning, NY, USA), they were washed twice in 10% fetal bovine serum (FBS) alpha-MEM. Further, T75 tissue culture flasks (Corning, NY, USA), which contained 2 mM L-glutamine (Gibco; Thermo Fisher Scientific), 10% FBS (Gibco; Thermo Fisher Scientific), 100 U/ml penicillin (Gibco; Thermo Fisher Scientific), and 100 μg/ml streptomycin (Gibco; Thermo Fisher Scientific) in the alpha-MEM culture medium were seeded with rabbit hard palate stem cells (rbHP-PCs) at a density of 1 × 105 cells per cm2. Cell culture was performed under conditions of 37ºC and 5% CO2. After 24 hours, we replaced the culture medium with a new one, cultured it for 3–5 days, and split it once its density level reached 90%.
2.2. Flow cytometric analysis
After trypsinizing and harvesting 80–90% cultured rbHP-PCs, cells were washed with flow cytometric (FC) buffer. We used a 100 μl volume suspension of 1×106 cells with a fluorochrome-conjugated antibody: CD11b 1:100, CD90 1:100, CD81 1:100 (Thermo Fisher Scientific) in FC buffer and then cultured it at 4ºC for 30 min. A 100 μl volume suspension of a non-conjugated antibody, primary antibody CD34 1:100, CD29 1:100 (Thermo Fisher Scientific), CD44 1:100 (SeroTec, USA), was also made in FC buffer and cultured it at 4°C for 30 min. Then, a 100 μl volume incubation for a phycoerythrin (PE)-conjugated secondary antibody (1:100) (Thermo Fisher Scientific) was done in FC buffer at 4ºC for 30 min. After washing, 500 μl volume of 2% paraformaldehyde was added to fix the cell and a FC analysis was performed.
Cultured rbHP-PCs were harvested and seeded onto a 96-well plate (1x103 cells per well). After 24 hours, 10 ul CCK-8 reagents (Dojindo Laboratory, Kumamoto, Japan) were added and incubated for 2 hours. Then, the supernatant was transferred to a new plate, and the optical density was measured at a 450 nm wavelength.
2.3. Reverse transcription-polymerase chain reaction
Total RNA of the rabbit hard palate tissue was isolated using a Qiagen RNeasy mini kit (Qiagen, Hilden, Germany). cDNA was synthesized into 1 μg RNA by using a Maxime RT premix kit oligo (dT) primer (Intron Biotechnology, Seongnam, Korea). Cycling conditions were as follows: initial denaturation at 98°C for 3 min and 30 seconds, 30 seconds at the annealing temperature, 30 seconds at 98°C, and 5 minutes of final elongation at 72°C. Details of the sequence of gene primers and the annealing temperatures are described in supplementary data.
2.4. Osteogenic differentiation
To induce osteogenic differentiation, 6×104 cells per well of rbHP-PCs on a 24-well plate were seeded in MPC growth medium (Cambrex Biosciences Inc., Verviers, Belgium), which is composed of 2 mM L-glutamine, 10% FBS, 100 U/ml penicillin, and 100 μg/ml streptomycin. After culturing the cells until the density reached 100%, culture medium was replaced with a MPC osteogenic differentiation medium (Cambrex Biosciences Inc.), composed of 10% FBS, 50 μg/ml ascorbate, 10 mM β-glycerophosphate, 100 I.U./mL penicillin-100 μg/mL streptomycin, and 10 nM dexamethasone. 18 The culture medium was changed every 3 days for 21 days.
After removing the osteogenic differentiation medium from osteogenic differentiation-induced cells, the cells were washed with Dulbecco’s modified phosphate-buffered saline (DPBS) without Ca++/Mg++ (Thermo Fisher Scientific) and fixed for 30 minutes with 4% formaldehyde solution (Sigma-Aldrich, St. Louis, MO, USA). After washing twice with DPBS (without Ca++/Mg++), the cells were stained with alizarin red S working solution (Merck Millipore, Billerica, MA, USA) for 5 minutes. Thereafter the cells were washed two to three times more with DPBS (without Ca++/Mg++).
2.5. Chondrogenic differentiation
To induce chondrogenic differentiation, 3×105 rbHP-PCs in a 15 ml polypropylene conical tube (Thermo Fisher Scientific) were seeded and a 160 g centrifuge for 7 minutes was performed. The cells were cultured in a chondrogenic differentiation medium (Cambrex Biosciences Inc.) composed of 10% FBS, 10 ng/ml TGF-β3, 100 nM dexamethasone, 25 μg/ml ascorbate, and 100 μg/ml sodium pyruvate. 18 In 24 to 48 hours, the cells gathered to form a 3D aggregate shape. Culture medium was changed every 3 days for 21 days.
The chondrocytes that differentiated into a 3D aggregate were fixed in 4% paraformaldehyde and embedded in paraffin. After sectioning into 4 um, staining was done using hematoxylin and eosin. The slides were stained with Alcian blue 8GX solution (Sigma-Aldrich) for 30 minutes and then washed two or three more times with distilled water and then stained further with hematoxylin.
The slides were stained with anti-collagen II primary antibody (1:200, Abcam) overnight at 4°C. The secondary antibody (1:1000, Abcam) was incubated for 30 minutes at room temperature. Finally, the slides were stained using 3,3-diaminobenzidine solution (Dako, Ft. Collins, CO).
2.6. Adipogenic differentiation
To induce adipogenic differentiation, 6x104 cells per well of rbHP-PCs on a 24-well plate were seeded in a MPC growth medium (Cambrex Biosciences Inc.) composed of 2mM L-glutamine, 10% FBS, 100 U/ml penicillin, and 100 μg/ml streptomycin. Once the density of the cells reached around 80–90%, adipogenic differentiation was induced. The culture medium was changed to an adipogenic differentiation medium (Cambrex Biosciences Inc.) composed of 10% FBS, 100 I.U./mL penicillin-100 μg/mL streptomycin, 1 µM dexamethasone, 0.2 mM indomethacin, 10 µg/ml insulin, 0.5 mM 3-isobutyl-1-methilxantin.
18
After confirming the cell density reached 100%, the medium was changed to a differentiation medium composed of FBS, penicillin-streptomycin, glutamine, and insulin. After 24 hours, the medium was changed to an adipogenic differentiation medium. The adipogenic differentiation medium was changed with differentiation medium and
After removing adipogenic differentiation medium from adipogenic differentiation-induced cells, the cells were washed with DPBS, without Ca++/Mg++ (Thermo Fisher Scientific) and fixed for 30 minutes with a 4% formaldehyde solution (Sigma-Aldrich). After washing with DPBS two times, the cells were stained for 30 minutes with a filtrated Oil Red O solution (Sigma-Aldrich). Once the staining was complete, the cells were washed in DPBS two more times.
2.7. Western blot
The differentiated cells were lysed by using 1X Lysis buffer (Merck Millipore, Billerica, MA, USA) that contained phosphatase/protease inhibitor (Sigma-Aldrich), and centrifuged for 15 minutes at 4°C and 13,000 rpm. Later, the amount of protein was measured with the BCA protein assay reagent (Pierce Biotechnology Inc., Rockford, IL, USA) and sodium dodecyl sulphate polyacrylamide gel electrophoresis was performed. Next, the product was transferred to polyvinylidene difluoride membranes (Thermo Fisher Scientific). After blocking the membrane with 5% skim milk in phosphate-buffered saline with 0.1% Tween 20 Detergent (PBST), primary antibodies, namely, anti-osteocalcin (OCN, 1:500; Abcam, Cambridge, MA, USA), anti-alkaline phosphate (ALP, 1:1000; Abcam), anti-PPAR-γ (1:500, Abcam), anti-FABP4 (1:200, Abcam), anti-Aggrecan (1:100, Abcam), and anti-GAPDH (1:5000) antibodies were diluted in 3% skim milk in PBST and incubated overnight at 4°C. Thereafter, peroxidase-conjugated secondary antibodies, namely anti-mouse (1:5000, Sigma-Aldrich) and anti-rabbit (1:5000, Sigma-Aldrich), were diluted in 3% skim milk in PBST and cultured in room temperature for 1 hour. The reacted protein was detected over film using an ECL Prime Western Blotting Detection Syprogenitor (GE Healthcare).
2.8. Immunofluorescent staining
For fixing, 30-minute incubation was carried out in 4% paraformaldehyde. For permeabilization, incubation was carried out with 0.4% Triton X-100 (Sigma-Aldrich) in PBS for 20 minutes. Blocking was done by incubating 1% bovine serum albumin (BSA) for 1 hour at room temperature, and the primary antibody β-actin (mouse anti-actin, 1:1000, Sigma-Aldrich), OCN (mouse anti-OCN, 1:100, Abcam), FAPB4 (rabbit anti-FABP4, 1:100, Abcam), and collagen II (mouse anti-collagen II, 1:100, Abcam) were incubated overnight at 4°C. Finally, the FITC-conjugated secondary antibody (donkey anti-rabbit, goat anti-mouse, Abcam) was incubated for 3 hours in room temperature. The stained samples are observed using confocal laser-scanning microscopy (CLSM) (M780; Zeiss).
2.9. Fabrication of PCL and PCL/TCP scaffold
Polycaprolactone (PCL) is a clinically approved material in many countries and is easy to use for 3D bioprinting. 19 In particular, tricalcium phosphate (TCP) helps bone formation as an implant material in bone reconstruction. PCL was fabricated for cartilage tissue engineering and PCL/TCP for bone tissue engineering. For PCL/TCP fabrication, PCL (99023710, Evonik Industry, Pharma Polymers, Germany) and β-TCP (average diameter 100 nm, Berkeley Advanced Biomaterials Inc, Berkeley, CA, USA) were blended together by melting to fabricate PCL/TCP scaffold. Chip-typed PCL (4 g) was placed on a glass container and melted at 120°C for 15 minutes. The blended PCL/TCP mixture was prepared by adding powdered β-TCP (1 g) to the molten state of PCL. Β-TCP and molten PCL were manually stirred for 10 minutes.
The chip-typed PCL and blended PCL/TCP mixture were placed in a 10 ml steel syringe equipped with extrusion-based 3D printer. A 3DX-printer (T&R Biofab Co., Ltd, Korea) was used in the fabrication of 3D-printed substrates. A moving path of the head and working base were followed according to pre-generated G code, created using computer-aided manufacturing software that was developed in-house. Details are provided in the supplementary data.
Operation temperature was maintained at 110°C. Disk-type PCL and PCL/TCP scaffolds (diameter 10 mm, height 1 mm) were fabricated. The line width, pore size, and line height were 300, 300, and 100 µm, respectively. Fabricated scaffolds had a triangular pore architecture, the calculated porosity was about 50%, and the pores were fully interconnected. Unlike the conventional method of making a scaffold, the stacking technique of 3D bioprinting can digitally control the pore size and matrix structure, as can be seen in the scanning electron microscope (SEM) results. 20
The morphology of the scaffolds was observed using a high-resolution field emission SEM (Nova NanoSEM 200, FEI Co, USA) at 10 kV. The scaffolds were coated with carbon using a sputter-coater for 3 minutes. The surface morphology of the PCL/TCP scaffold was compared with that of the PCL scaffold.
2.10. Cell culture in scaffolds
PCL or PCL/TCP scaffolds in a 24-well plate were pre-wet for 30 minutes in serum-free medium. The harvested rbHP-PCs 5×104 cells were dropped over the PCL of PCL/TCP scaffold. After 3 hours of incubation, the culture medium was added. The alpha-MEM culture medium contained 2 mM L-glutamine (Gibco; Thermo Fisher Scientific), 10% FBS (Gibco; Thermo Fisher Scientific), 100 U/ml penicillin (Gibco; Thermo Fisher Scientific), 100 μg/ml streptomycin (Gibco; Thermo Fisher Scientific). After 1 week of culturing, the medium was changed to a differentiation medium and cultured for 14 more days. For osteogenic differentiation, MPC osteogenic differentiation medium (Cambrex Biosciences Inc.) was used. Chondrogenic differentiation medium (Cambrex Biosciences Inc.) was used for chondrogenic differentiation and adipogenic differentiation medium (Cambrex Biosciences Inc.) was used for adipogenic differentiation.
3. Results
3.1. The existence of NCSs on rabbit hard-palatal tissue
In the hematoxylin and eosin-stained section (Figure 1(a)), the cells were distributed around the hard palate tissue of the rabbit. Nestin, an NCS marker, was detected (Figure 1(b)–(d)) by performing immunofluorescence staining. As NCS markers, nestin, Sox2, and p75 (Figure 1(e)) were detected by Reverse transcription-polymerase chain reaction (RT-PCR) analysis. The existence of NCS in the hard palate was proved.
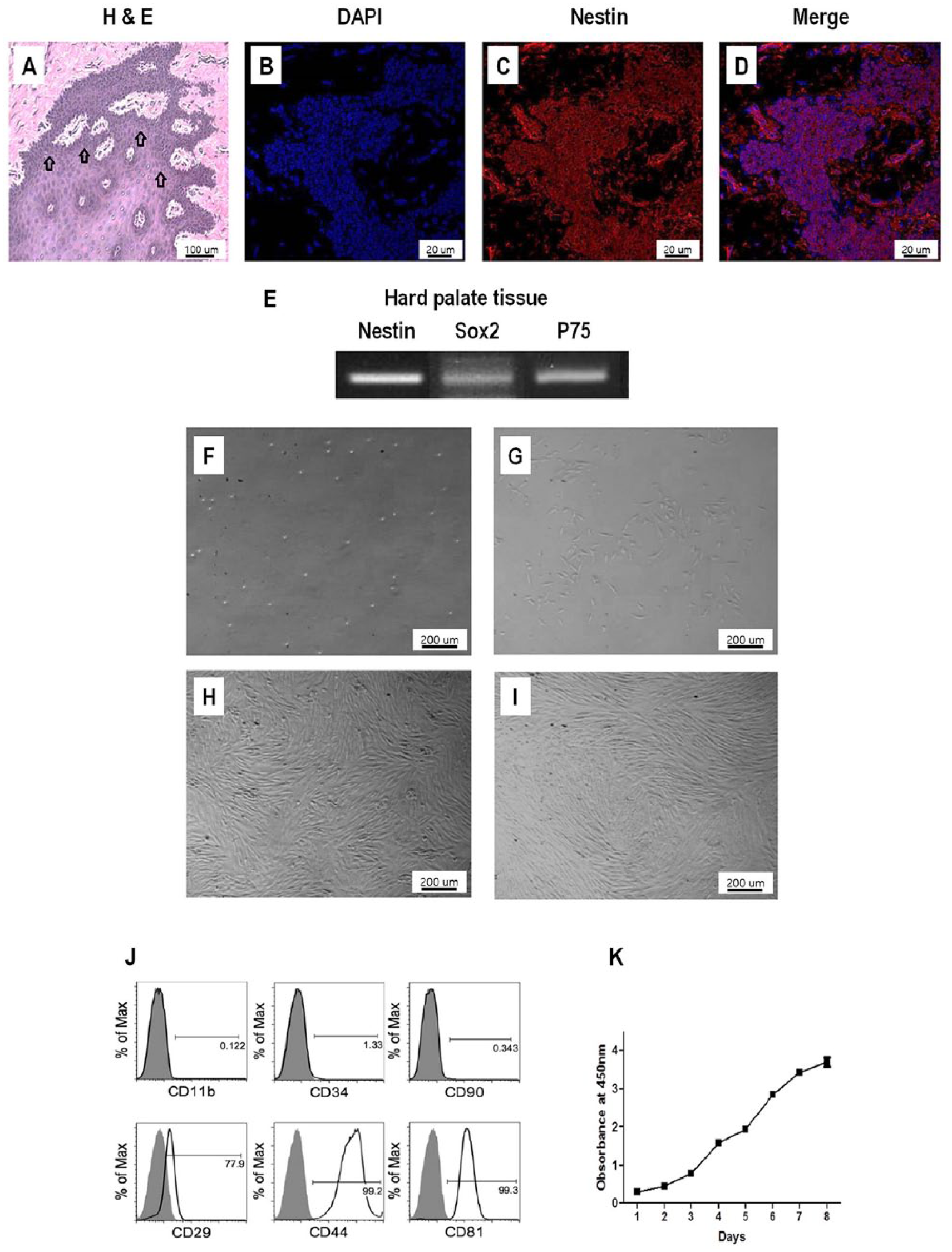
After paraffin embedding sections and (a) hematoxylin and eosin staining on the tissue, cells were distributed around the hard palate tissue (arrow) of the rabbit. (b) Immunofluorescence staining of the tissue: 4’,6-Diamidino-2-Phenylindole (DAPI), (c) nestin, (d) DAPI and nestin were merged by confocal laser scanning microscopy (CLSM), and (E) neural crest progenitor cell markers as a result of Reverse transcription-polymerase chain reaction (RT-PCR) (nestin, Sox2, p75, and GAPDH). Morphology change of rabbit hard palate stem cells (rbHP-PCs) in accordance with the number of cultured days; (f) day 1, (g) day 4, (h) day 7, (i) day 11. Confirmed surface marker on rbHP-PCs using flow cytometry analysis. (j) Positive markers (CD29, CD44, and CD81) and negative markers (CD11b, CD34, and CD90). (k) Cultured rbHP-PCs were harvested and seeded on a 96-well plate (1×103 cells per well). After 24 hours, 10 µl CCK-8 reagent (Dojindo Laboratory, Kumamoto, Japan) was added and incubated for 2 hours. Then the supernatant was transferred to a new plate, and optical density (OD) was measured at 450 nm wavelength.
3.2. Primary culture and characterization of rbHP-PCs
During the first day of incubation of the isolated rbHP-PCs, we observed a clear and bright circular border around the cell. During days 3–5, the cells adhered to the bottom and began to spread in a long line shape. Later, the cells began to aggregate. After day 7, the cells showed compact confluency, exhibiting an overall spiral shape (Figure 1(f)–(i)).
According to rabbit MPC markers, positive markers showed CD29 (77.9%), CD44 (99.2%), and CD81 (99.3%) whereas negative markers showed CD11b (0.4%), and CD34 (3.13%) from the cultured rbHP-PCs (Figure 1(j)).
The proliferation assay was performed for 8 days after culturing rbHP-PCs using a CCK-8 assay kit. The rbHP-PC growth curve after 3 days showed an increasing tendency (Figure 1(k)).
3.3. rbHP-PCs have mesenchymal tri-lineage differentiation potential
As a result of the osteogenic differentiation of rbHP-PCs (Figure 2(b)) for 21 days, red stains with Alizarin red S of the mineral component were observed. In contrast, there were no stained minerals from rbHP-PCs (Figure 2(a)) cultured in MPC growth medium.
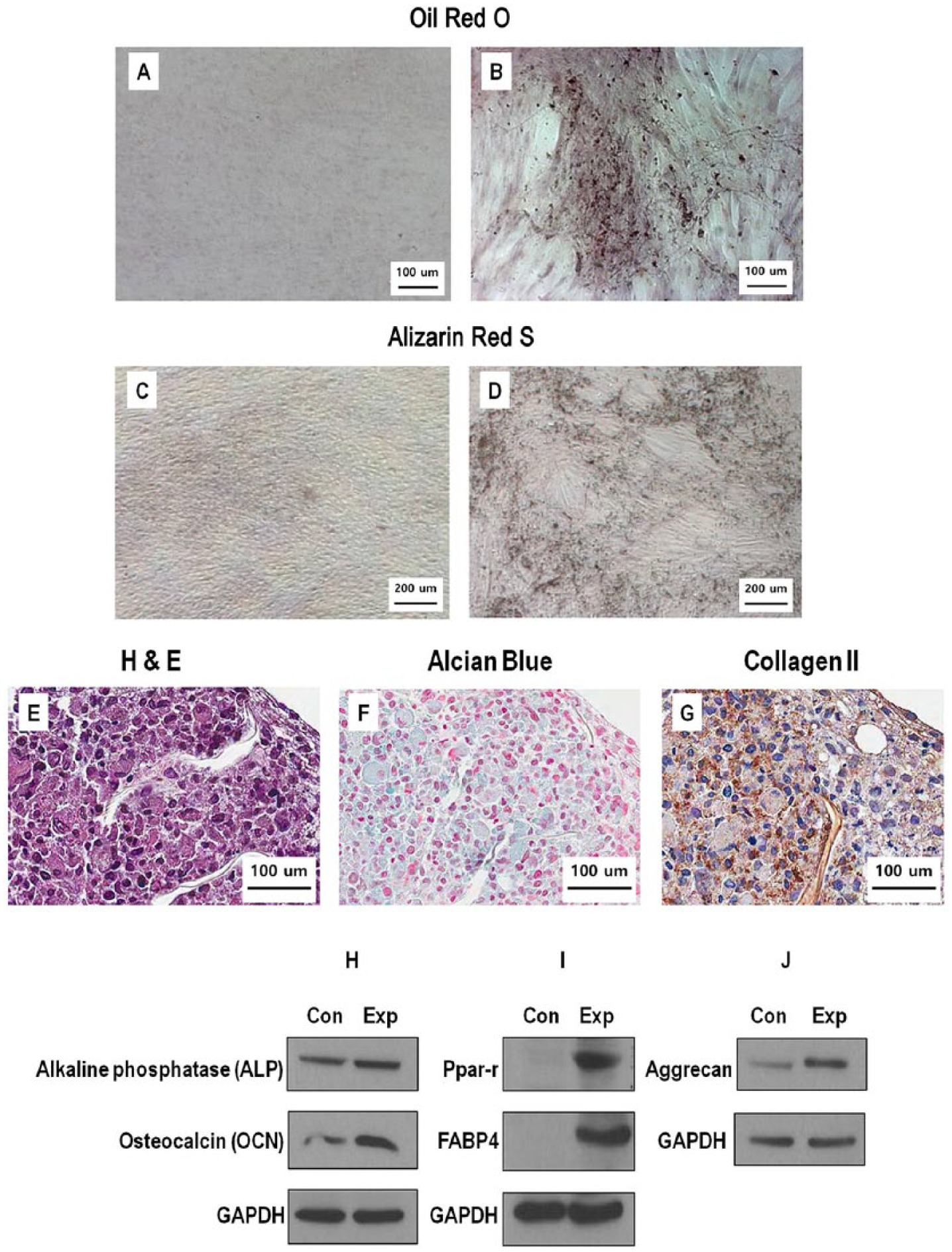
Rabbit hard palate stem cells (rbHP-PCs) were cultured for 21 days with control medium and osteogenic, adipogenic, and chondrogenic medium. (a) and (b) Osteogenic differentiation: Alizarin red S staining. (c) and (d) Adipogenic differentiation: Oil red O staining. (e)–(g) Chondrogenic differentiation: paraffin embedded section: hematoxylin and eosin (HE), Alcian blue, and collagen type II expression for immunohistochemistry (IHC). After the rbHP-PCs were differentiated with osteogenic, adipogenic, and chondrogenic medium for 3 weeks, the relevant markers were confirmed. (h) Osteogenic differentiation markers: alkaline phosphate (ALP), osteocalcin (OCN). (i) Adipogenic differentiation markers: PPAR-γ, FABP4, (j) chondrogenic differentiation marker: Aggrecan.
A 3D aggregated shape was observed 21 days after culturing the pellet in chondrogenic differentiation medium. For histological evaluation, the chondrogenic differentiated pellets were embedded in paraffin and positively stained with Alcian blue, and collagen type II expression was evaluated (Figure 2(e)–(g)). Chondrocytes were stained with hematoxylin and eosin and extracellular matrix (ECM) was positively stained with Alcian blue for glycosaminoglycan content. Staining of adipogenic differentiation cells of rbHP-PCs was done for 21 days in Oil red O. By comparing the stain results of the adipogenic differentiation medium-induced rbHP-PCs that were stained in red (Figure 2(d)) with the control group culture using MPC growth medium (Figure 2(c)), we could confirm the lipid droplet in the rbHP-PCs.
After osteogenic, adipogenic, and chondrogenic differentiation of rbHP-PCs for 21 days, each of the markers was confirmed using immunoblot assay. As a result, ALP and OCN, the osteogenic differentiation markers (Figure 2(h)), PPAR-γ and FABP4, the adipogenic differentiation markers (Figure 2(i)), and aggrecan, the chondrogenic differentiation marker (Figure 2(j)), were higher than those of the non-differentiated control.
3.4. Fabrication of 3D-printed biodegradable PCL and PCL/TCP scaffolds
The PCL and PCL/TCP scaffolds were successfully fabricated using an extrusion-based 3D printer (in-house syprogenitor, Figure 3(a)-(d)). The scaffolds had a diameter of 10 mm. In addition, interconnected pores play an important role in the transportation of oxygen and waste. The line width and pore sizes of scaffolds were 300 and 300 µm, respectively. The surface roughness of PCL and PCL/TCP scaffold was different. A rougher surface was observed on PCL/TCP scaffolds as shown in Figure 3(b) and (d). This is due to the incorporated β-TCP in the PCL.
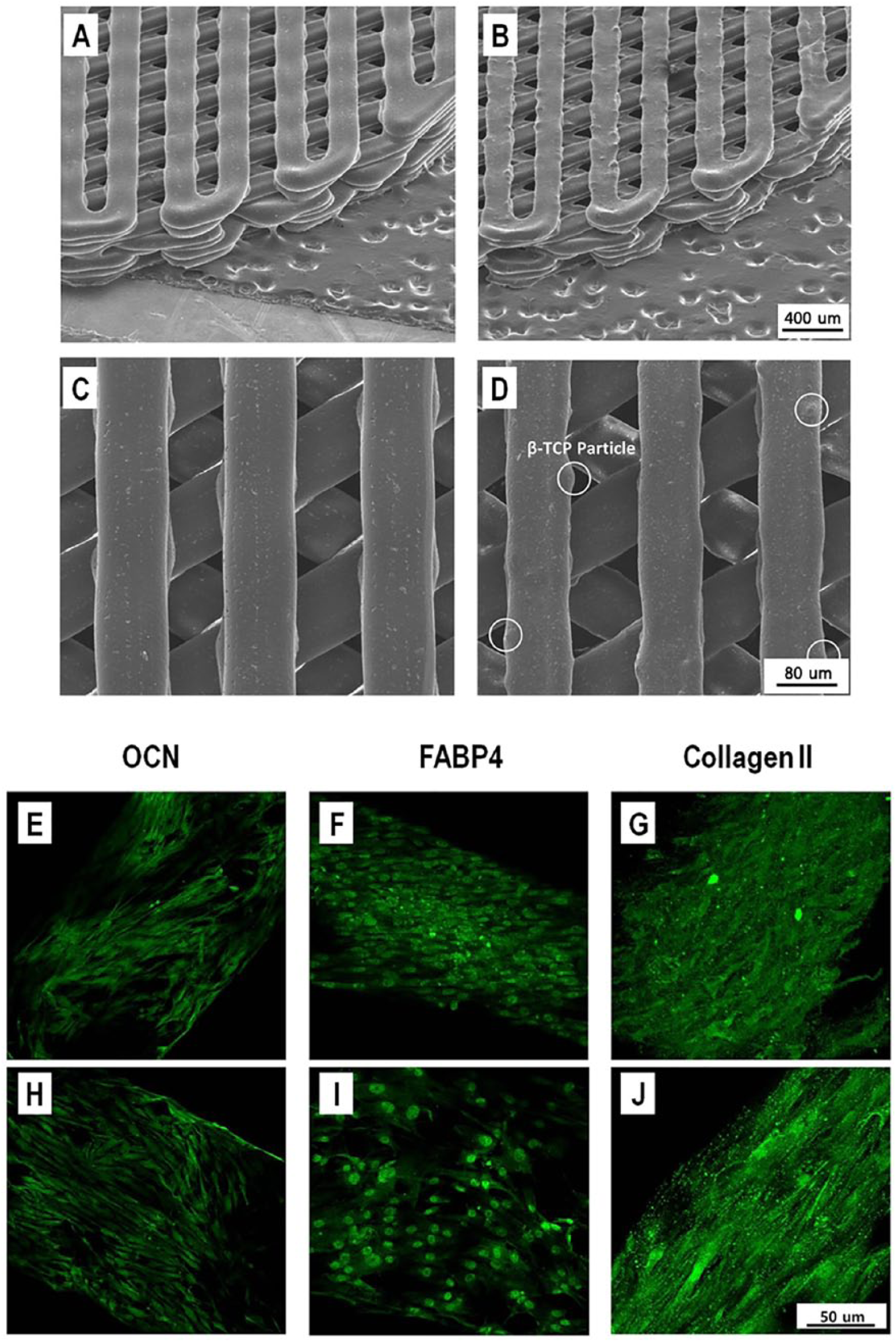
Scanning electron microscope (SEM) images of polycaprolactone (PCL) (a), (c) and polycaprolactone–tricalcium phosphate (PCL/TCP) (b), (d) scaffolds. Fabricated PCL (a) and PCL/TCP (b) scaffolds were confirmed at a magnification of 50×. A line width of 300 µm and pore size of 300 µm were observed at a magnification of 180× (c), (d). In particular, rougher surface was shown in PCL/TCP scaffold (b), (d). Tri-lineage differentiations of rabbit hard palate stem cells (rbHP-PCs) in scaffold was seen by confocal laser-scanning microscopy (CLSM). Osteogenic differentiation in (e) PCL and (h) PCL/TCP, adipogenic differentiation in (f) PCL and (i) PCL/TCP; Chondrogenic differentiation in (g) PCL and (j) PCL/TCP.
3.5. In vitro mesenchymal tri-lineage differentiation of rbHP-PCs on PCL and PCL/TCP scaffolds
RbHP-PCs were cultured for 1 week after seeding on PCL scaffold. To confirm whether the cells were fixed properly onto the scaffold, the cell nucleus and cytoskeleton were stained and verified using CLSM. Once the cells on the scaffold were stabilized, they were differentiated through osteogenic, adipogenic, and chondrogenic differentiation medium. The cells were cultured for 14 days in the respective media, following which each of the markers was confirmed. Immunofluorescent staining was carried out for the entire scaffold, and the expressions of OCN, FABP4, and collagen type II were confirmed (Figure 3(e)-(j)). We demonstrated the
4. Discussion
In this study, the MPCs in rabbit hard palate expressed NCS markers (nestin, Sox2, and p75), similar to that in rats and humans,10, 11 and possess multi-lineage differentiation potential for mesenchymal cells of bone, cartilage, and fat. Widera
We were able to observe the spiral shapes from the proliferated cells of harvested palate tissue. Such a shape signifies the characteristics of a progenitor cell and is based on the traits of MPCs.21, 22 The specific antigen expression of cultured rbHP-PCs was validated through flow cytometry. The expression markers were based on MPC markers originating from human bone marrow. 22 The expressions of CD11b, CD29, CD34, CD44, CD81, and CD90 were observed, and CD11b (0.122%), CD34 (1.33%), CD90 (0.343%) showed negative expression; whereas CD29 (77.9%), CD44 (99.2%) and CD81 (99.3%) showed positive expression. Our results were similar to the negative and positive markers of human mesenchymal stem cells. However, differences in expression were identified for CD90. Although the human mesenchymal stem cells manifested positive expressions, the rbHP-PCs we isolated manifested negative expressions. There have been reports that CD90 for rabbit progenitor cells manifested positive expressions,23,24 and there have also been those that observed negative expressions.23,25 These controversial findings are due to the fact that sufficient research has not been carried out for rabbit progenitor cells, resulting in lack of a solid consensus regarding progenitor cell markers. Because this was the first time hard palate was tested, further research will be required in this field.
We identified differentiation markers ALP and OCN after 21 days of osteogenic differentiation through immunoblot analysis. Although OCN had significantly increased compared to that in non-differentiation, ALP had increased only slightly. The expression of ALP was expected to decrease as an early marker, but it is not fully understood why ALP increased. In fact, previous reports claimed that the expression of ALP decreases as the differentiation progresses. 26 The increase in OCN conforms it is expressed from mature osteoblasts and OCN is secreted 2 weeks after osteogenic differentiation. 26
During the progression of adipogenic differentiation, lipid droplets are formed. These droplets are stained with Oil red O. We measured the adipogenic differentiation markers (PPAR-γ and FABP4) and confirmed their expression increased. 27 To confirm chondrogenic differentiation, we stained the paraffin-embedded section from the pellet with Alcian blue to measure glycosaminoglycan production. 28 Other chondrogenic markers, collagen II and aggrecan, 29 were measured through immunoblot and immunohistochemistry (IHC) methods.
Like the previous studies, 29 we could expand the MPCs and differentiate them into mesenchymal cell lineages (bone, cartilage and fat) from the hard palatum of the rabbit. These data suggest that mesenchymal progenitors exist mostly in the head and neck area in rabbits. These tissues could be a promising new source for progenitor cell-driven tissue engineering research combining scaffolds for head and neck defects. Rabbit is gaining popularity in animal models and has useful properties such as stability, economics, and an appropriate size for procedures. 14
3D-printed PCL- and PCL/TCP-based scaffolds have been used as popular base materials for cartilage- and bone-regeneration research. Recently, printing scaffolds with specific biomaterials or proteins were reported with considerable effects.7, 11, 30 Cho
5. Conclusion
We isolated and characterized MPCs from the hard palate of a rabbit through their morphology, surface marker expression, proliferation, and mesenchymal tri-lineage differentiation potential. Also, the mesenchymal tri-lineage differentiation process was observed for the bio-degradable scaffold PCL and PCL/TCP. With our results, rabbit palatum could be used as a new, easily accessible source of multipotent MPCs for cell-driven tissue engineering research of head and neck mesenchymal tissue.
Supplemental Material
Supplemnt__revision834520 – Supplemental material for Rabbit palatum-derived mesenchymal progenitor cells tri-lineage differentiation on 2D substrates and 3D printed constructs
Supplemental material, Supplemnt__revision834520 for Rabbit palatum-derived mesenchymal progenitor cells tri-lineage differentiation on 2D substrates and 3D printed constructs by Dong Hwan Kim, Bo Young Kim, Dong Hyun Kim, Jin Hur and Chung-Hwan Baek in Journal of Applied Biomaterials & Functional Materials
Footnotes
Author Contributions
Bo Young Kim, Dong Hwan Kim and Dong Hyun Kim: collection and/or assembly of data, data analysis and interpretation, manuscript writing; Jin Hur and Chung-Hwan Baek: conception and design, manuscript writing, data analysis and interpretation, final approval of the manuscript.
Declaration of conflicting interests
The author(s) declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CHB is supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI14C2215). JH is supported by the Basic Science Research Program through NRF (2017R1A2B2012305). DHK is supported by the National Research Foundation of Korea (NRF-2016R1D1A1B03935879).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
