Abstract
Purpose:
To evaluate the thermomechanical and in vitro biological response of poly(lactic-co-glycolic acid) (PLGA) plates for craniofacial reconstructive surgery.
Methods:
PLGA 85/15 craniofacial plates were produced by injection molding by testing two different temperatures (i.e., 240°C, PLGA_lowT, and 280°C, PLGA_highT). The mechanical properties of the produced plates were characterized by three-point bending tests, dynamic mechanical analysis, and residual stress. Crystallinity and thermal transitions were investigated by differential scanning calorimetry. Finally, in vitro cell interaction was evaluated by using SAOS-2 as cell model. Indirect cytotoxicity tests (ISO 10-993) were performed to prove the absence of cytotoxic release. Cells were then directly seeded on the plates and their viability, morphology, and functionality (ALP) checked up to 21 days of culture.
Results:
A similar performance of PLGA_lowT and PLGA_highT plates was verified in the three-point bending test and dynamic mechanical analyses. Also, the two processing temperatures did not influence the in vitro cell interaction. Cytotoxicity and ALP activity were similar for the PLGA plates and control. Cell results demonstrated that the PLGA plates supported cell attachment and proliferation. Furthermore, energy-dispersive X-ray spectroscopy revealed the presence of sub-micron particles, which were identified as inorganic mineral deposits resulting from osteoblast activity.
Conclusion:
The present work demonstrated that the selected processing temperatures did not affect the material performance. PLGA plates showed good mechanical properties for application in craniofacial reconstructive surgery and adequate biological properties.
Introduction
Craniofacial plates and screws are important biomedical devices for the treatment of craniofacial bone fractures, which require specific mechanical properties for bone regeneration.1–5 Titanium-based implants are usually used in fracture plate systems due to their good mechanical properties and biocompatibility.2,6 However, once the bone healing process is completed, metallic implants lose their functionality and problems such as possible in vivo corrosion and bone resorption, due to stress shielding, could occur. Because of these problems, metallic plate and screws could require a second surgery for implant removal and implantation of new ones. 7
Biodegradable synthetic polymers and copolymers have been proposed as alternative materials for craniofacial fracture repair.3–5,8,9 Bioresorbable implants, mainly based on poly-α-hydroxy ester (e.g., polylactic acid, PLA, polyglycolic acid, PGA), act as an initial support for osteosynthesis and progressively degrade as the bone healing process advances. Due to its lower elastic modulus compared to metal implants, and gradual degradation, the choice of these materials protects against bone weakening due to stress shielding and avoids the need for subsequent surgery.7,10–13 Moreover, the application of bioresorbable implants is particularly advantageous for pediatric patients, since intracranial translocation of plates and screws as well as problems associated with metallic implants, such as tenderness, palpability, visibility, and thermal sensitivity, can be prevented.1,10
PLA/PGA copolymers (poly(lactic-co-glycolic acid), PLGA) are under constant development and investigation as materials for craniofacial plate production. Major problems related to the PLGA synthesis, such as the copolymer composition and ratio of the L and DL isomeric forms in PLA, have been investigated in several studies.3,12,14 Moreover, the polymer needs further processing steps to obtain the final devices (i.e., craniofacial plates), and the mechanical and physicochemical properties may be affected by the manufacturing process and device geometry. 15 Due to the complex geometry of craniofacial plates and screws, injection molding is an optimal technique to produce these implants. Nevertheless, only few studies have investigated the influence of manufacturing process parameters on the properties of biodegradable synthetic polymers.16,17 In fact, several parameters can influence the properties of craniofacial plates, including the injection flow rate, holding pressure, and injection molding temperature, 18 thus potentially affecting their performance. In particular, the injection molding temperature affects the material melting and represents a critical parameter, leading to changes in polymer chain orientation and to an increase in viscosity, altering material crystallinity. Consequently, crystallinity has an impact on several properties of biodegradable PLA/PGA materials, such as stiffness, heat distortion temperature, and impact properties. 15 Since these properties are intimately connected to the in vivo device performance, it is essential to understand the influence of processing conditions on the properties of the final products.
Thus, in this study, PLGA plates for use as craniofacial implants were prepared by injection molding. Among processing technologies, injection molding was selected as other technologies (e.g., CNC machining process) can induce local heating, causing degradation of the polymer. In addition, in the specific case of plates for craniofacial fracture repair, the appropriate mold can guarantee a better shape fidelity compared to other processing technologies. 17 The investigated plate geometry was based on commercially available devices. 10 The influence of the processing temperature on the material properties was investigated; in particular, two processing temperatures, 240°C (PLGA_lowT) and 280°C (PLGA_highT) were evaluated. PLGA_lowT was chosen as the minimum processability temperature while PLGA_highT was defined as the upper working limit temperature without possible polymer degradation. The injection molded PLGA plates were investigated for mechanical and thermal properties as well for in vitro cell response.
Materials and methods
Materials
Poly(
Design and manufacturing of the plates
The craniofacial plate was designed by 3D CAD SolidWorks 2014 software (Concord, MA). The plate design and dimensions (Supplementary Figure 1) were selected based on previous studies; 17 thickness, width, and length of the device were 2 mm, 5.8 mm, and 38.7 mm, respectively.
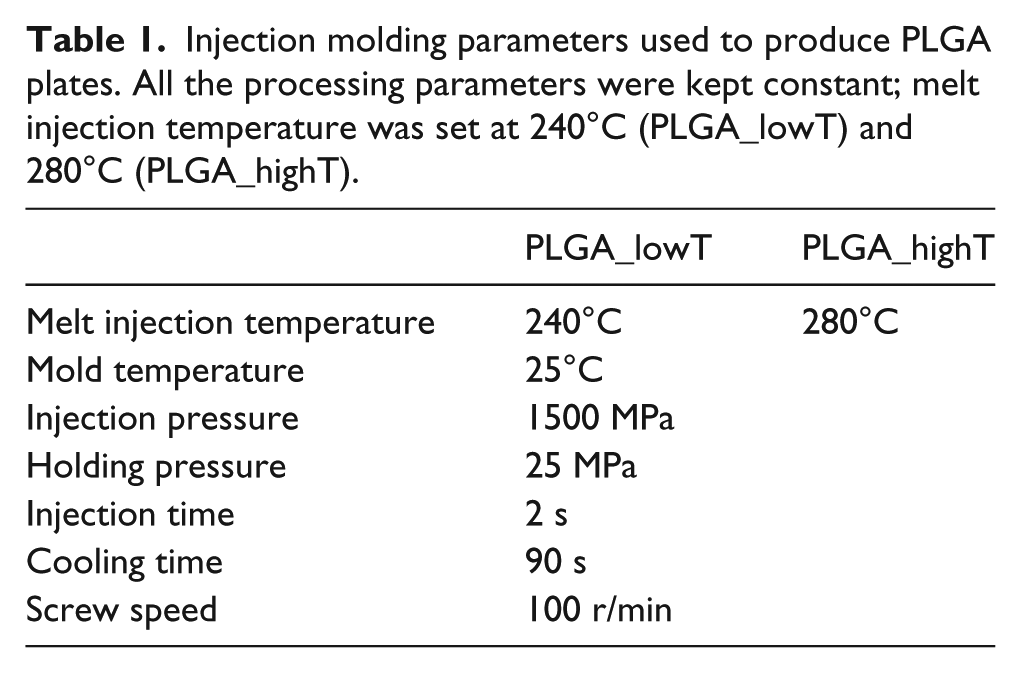
The plates were manufactured using PLGA pellets and processed using an injection molding machine (Arburg 270S, 250-70 model). To investigate the influence of processing parameters on the properties of the PLGA plates, two different melt injection temperatures were tested: a low processing temperature (T = 240°C, PLGA_lowT) and a high processing temperature (T = 280°C, PLGA_highT); the other processing parameters were kept constant, as summarized in Table 1.
Injection molding parameters used to produce PLGA plates. All the processing parameters were kept constant; melt injection temperature was set at 240°C (PLGA_lowT) and 280°C (PLGA_highT).
Mechanical characterization
Three-point bending tests
PLGA_lowT and PLGA_highT specimens (n = 5, gauge length = 10 mm) were tested using a dynamic mechanical analyzer (DMA Q800, TA Instruments), in the three-point flexural mode (ASTM D790), at 37°C, with a test speed of 2 N min−1. The mechanical parameters evaluated were the bending structural stiffness, Ele, bending stiffness, K, and maximum bending load, Y. Parameters were calculated for each specimen and averaged.
Dynamic mechanical analysis
Dynamic mechanical analysis (DMA) was performed. Samples (n = 6, gauge length = 16 mm) were tested at 1 Hz frequency and 0.1% strain, in the temperature range 30–80°C, at 3°C min−1 temperature rate. The storage modulus (E′), loss modulus (E′′), and tan δ (E′′/E′) were obtained and represented as a function of the testing temperature.
Residual stress
The residual stress of the manufactured plate specimens (n = 10) was evaluated by the polarized light technique with a polariscope (Photolastic Inc., Malvern, PA, USA) with polarizing and quarter-wave 250 mm diameter lenses, following the ASTM D 4093 standard. This method enables the qualitative observation of the optical anisotropy or birefringence along the plate geometry that could occur during the injection molding process.
Differential scanning calorimetry (DSC)
The crystallinity and thermal transitions (i.e., Tm, Tg, cold crystallization enthalpy, ΔHc, and melting enthalpy, ΔHm) were obtained using a differential scanning calorimeter (Shimadzu DSC-6000) in a nitrogen atmosphere, using aluminum oxide as the standard. The applied heating rate was 10°C min−1, in the temperature range 10–250°C, using specimens (7 mg) taken from the central region of the molded samples (n = 3). Crystallization temperature (Tcc) was obtained from the second heating run. The first endothermic enthalpy (ΔHg) in the DSC thermogram was associated to the residual stress.
In vitro cell interaction
Indirect cytotoxicity tests and direct cytocompatibility tests were performed to evaluate the in vitro biological response of the injection molded PLGA plates. Human primary osteogenic sarcoma cell line SAOS-2 (ECACC 89050205) was selected as cell model.
19
The culture medium was prepared using McCoy’s 5a culture medium, with added 15% v/v fetal bovine serum,
Cytotoxicity test
The in vitro indirect cytotoxicity test was performed according to the UNI EN ISO10993-5 standard. Injection molded samples were prepared in cylindrical form (0.50±0.01 g per sample), disinfected in 70% v/v ethanol solution overnight, and dried for 24 h. Eluates were extracted by immersing samples (n = 3 per each processing condition and time point) in the culture medium (0.4 g mL−1) and by storing them in the incubator (37°C, 5% CO2) for 1, 3, 7, and 14 days. For each time point, as control, three wells were additionally incubated with culture medium without samples. SAOS-2 cells were seeded (density = 1 × 104 cells/well) in 96-multiwell tissue culture plastic (TCPS, Corning) and cultured with 150 μL complete fresh medium for 24 h. After 24 h, the medium was replaced with 150 μL of the extracts obtained from the PLGA samples and cells were cultured for other 24 h in contact with the extracts. After 24 h, cell viability was measured by Alamar BlueTM assay. Briefly, at each time point, the culture medium was replaced with the same volume of 10 %v/v Alamar Blue solution in fresh medium and incubated for 4 h. After 4 h, 100 µL of solution was transferred to a 96-multiwell TCPS and fluorescence (λexcitation = 540 nm, λemission = 595 nm) was measured with a spectrophotometer (Tecan, Genius Plus plate reader). Cell viability was calculated as follows:
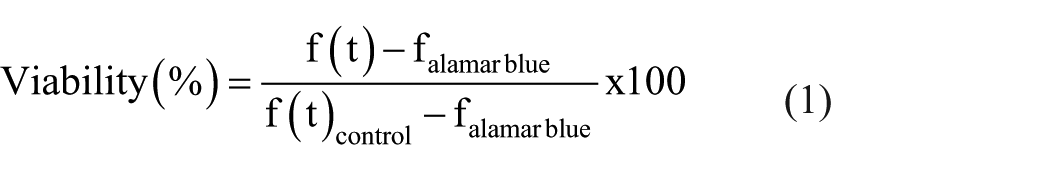
where f(t) is the fluorescence of the solution in contact with the cells cultured in eluates (extracted after contact with the material at the time points t = 1, 3, 7, 14 days), falamar blue is the mean fluorescence of Alamar Blue solution, and f(t)control is the average fluorescence of solutions incubated in contact with cells cultured with the control medium.
In vitro cytocompatibility
Injection-molded PLGA plate samples (n = 3 for each injection temperature, length = 12.5 mm) were obtained cutting the PLGA plates in halves, disinfected in 70% v/v ethanol solution for 4 h. The disinfected samples were put in 12-multiwell TCPS and SAOS-2 cells were drop-seeded (density = 1.2 × 104 cells/sample, drop volume = 8 µL) onto the center of the sample, between the two holes of the plate (Supplementary Figure 2(a)). The same cell density was seeded as control, on TCPS wells. Cells were cultured in the incubator (i.e., 37°C, 5% CO2) up to 21 days, and the medium was changed every three days. At each time point (t = 1, 3, 7, 14, and 21 days), cell viability was measured by Alamar Blue assay, as previously described. Samples were washed twice with PBS, fresh medium was added, and cells were kept in culture until the following time point.
Scanning electron microscopy (SEM) was used to observe the surface morphology of the PLGA plates to investigate a possible degradation during the in vitro cultures, and to analyze SAOS-2 cells morphology on the plates. At each time point, culture medium was removed and samples were immersed in 1.5% v/v glutaraldehyde solution in PBS for 2 h; after that, samples were washed for 2 h in PBS and dehydrated following an ethanol grade scale (10 min in 20, 30, 40, 50, 60, 70, 80, 90, and 100% v/v ethanol in distilled water). Dehydrated samples were gold/palladium sputter-coated (Edwards Sputter Coater 5150B) and observed by SEM (Cambridge Instrument Stereoscan 360 SEM) at 7 and 50× magnifications, with 10 kV accelerating voltage. Energy-dispersive X-ray spectroscopy (EDX, SEM Zeiss EVO 50 EP Plus EDS Oxford INCA Energy 200 with detector PENTAFET LZ4) was used to map C, Ca, and P on the surface of the seeded PLGA plates and controls (i.e., not cells seeded plates).
Alkaline phosphatase (ALP) activity of SAOS-2 cells cultured on injection molded PLGA plates and TCPS, as control, was quantified at each time point. The test was conducted using a commercial kit (BioVision Alkaline Phosphatase Assay Kit), based on the hydrolysis of p-nitrophenol (p-NPP), according to manufacturer’s instructions. At each time point, samples (n = 3) were washed twice with PBS, immersed in 500 µL of Triton X-100 solution and stored at −80°C. The day before the test, all the samples were removed from freezer and placed at room temperature, scratched to promote cell lysis and ALP test was performed by reading the O.D. at 405 nm. The ALP activity was related to the measured O.D. by using standards of 1 mM pNPP solution and ALP enzyme. The ALP activity was normalized to the total protein content of the samples, measured by the bicinchoninic acid assay (BCA). The BCA test was performed using a commercial kit (PierceTM BCA Protein Assay Kit, ThermoFischer Scientific) following the manufacturer’s guidelines; protein content was related to the measured absorbance by a calibration curve built using bovine serum albumin (BSA) standards.
Statistical analysis
Analysis of variance was performed by GraphPad Prism software considering a 0.05 statistical significance; p-value was investigated for significance of the results obtained for the two melting temperatures. All data are reported as mean ± standard deviation.
Results and discussion
Mechanical properties
Representative stress–strain curves obtained by the bending test performed on PLGA_lowT and PLGA_highT plates are shown in Figure 1(a).
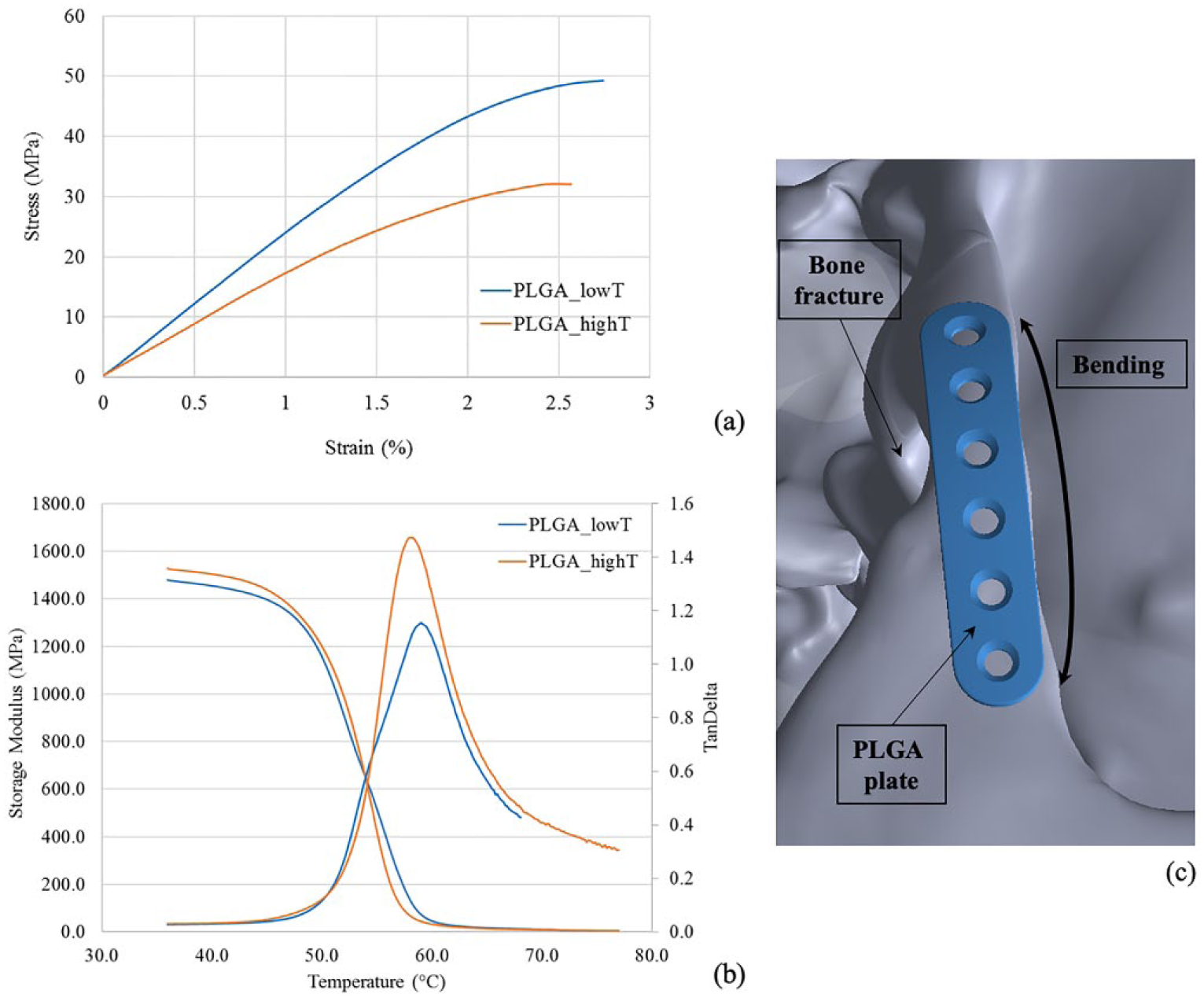
Mechanical properties of injection molded PLGA plates: (a) representative stress–strain curves obtained by bending tests and (b) representative E′ and tan δ curves versus temperature obtained by DMA. (c) Schematic representation of a PLGA plate fixed on the bone fracture and subject to bending.
The considered mechanical parameters obtained in bending tests performed on PLGA_lowT and PLGA_highT samples are summarized in Table 2; the bending tests were selected to simulate a possible in vivo application where the plates would be subjected to bending, as schematically illustrated in Figure 1(c). In the test, the PLGA plates showed fracture under both the processing conditions. The bending structural stiffness was improved (p < 0.05) when PLGA was injection molded with temperature of 280°C, compared to PLGA_lowT. This is probably due the presence of a different crystallinity and chain organization while cooling time occurs.
Mechanical proprieties of injection molded PLGA 85/15 plates (mean ± standard deviation; n = 6): bending stiffness, K, bending structural stiffness, Ele, and maximum bending load, Y (*p < 0.05).

DMA representative curves for PLGA_lowT and PLGA_highT are reported in Figure 1(b). Storage modulus showed similar trend for the two injection molding temperatures. The tan δ behaved differently for the two processing conditions. In fact, even if the peak of the two samples is at the same temperature, the higher values of tan δ is related to greater contribution of the viscous component for the PLGA_highT than the PLGA_lowT (i.e., higher E′′ for PLGA_highT).
Mechanical properties of the obtained PLGA plates have lower mechanical properties compared to those of bone (Ebone = 6–20 GPa). 11 Nonetheless, PLA, PGA, and their copolymers are widely investigated for craniofacial bone repair due to their advantages; in fact, some devices are already commercially available (e.g., ProToeTM EndoSorbTM, Merete Medical Inc.; LactoSorb® SE, Biomet Microfixation Inc.) confirming the possible use in terms of biodegradation rate and mechanical properties. 20 Moreover, PLGA plates can retain their mechanical strength long enough to support bone healing, and then gradually and harmlessly degrade in the patient’s body.
Differential scanning calorimetry
The transition temperatures and enthalpy detected for PLGA_lowT, PLGA_highT, and for the PLGA pellets are shown in Figure 2. The glass transition temperature occurs at 56±1°C for PLGA_lowT and at 53±1°C for PLGA_highT. The glass transition temperature of the PLGA pellets was detected at 60±2°C. The difference in the transition temperatures for PLGA_lowT and PLGA_highT, compared to the PLGA pellets, can be due to changes in the molecular mobility caused by the injection molding processing. The endothermic melting peak of PLGA_lowT appeared at 157±1°C, while the PLGA_highT peak occurred twice, at 151±2°C and 158±1°C, detecting the presence of higher crystallinity in the PLGA_highT plates.
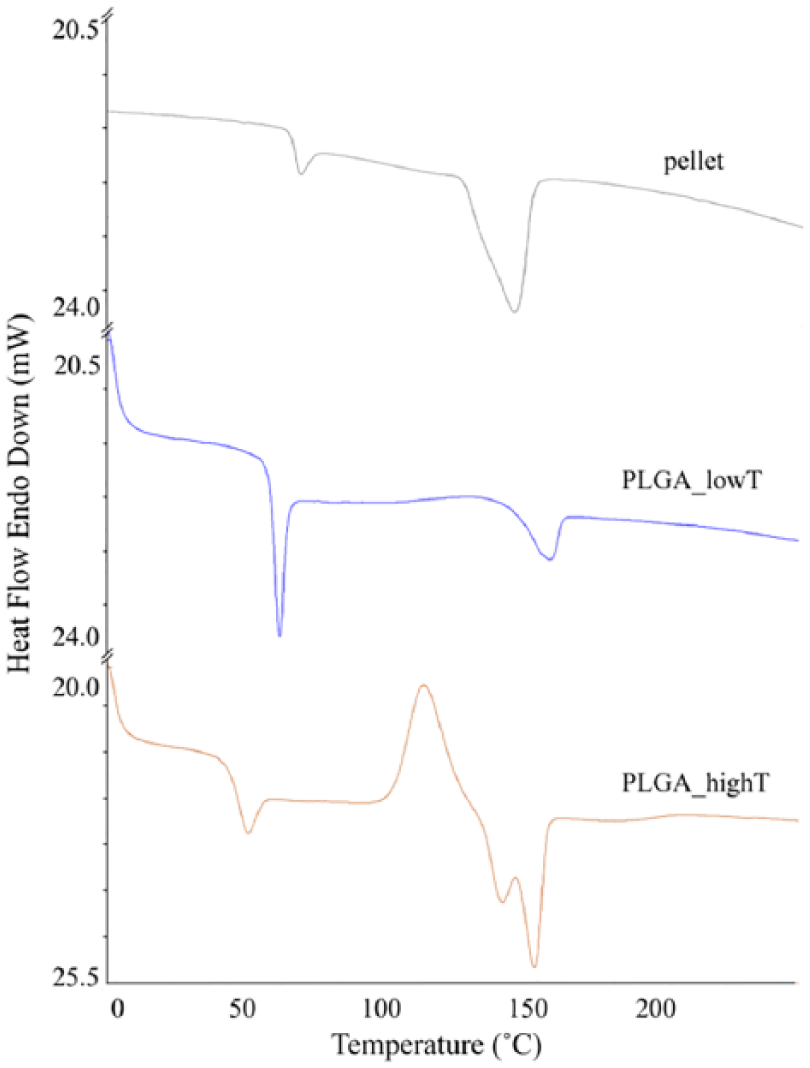
Transition temperatures of injection molded PLGA_lowT and PLGA_highT, and PLGA pellets.
Residual stress was measured by peak area in the glass transition and enthalpy, which was higher for PLGA_lowT (5.1±0.5 J/g) compared to the pellet (1.7±0.4 J/g) and PLGA_highT (3.2±0.2 J/g). PLGA_lowT and PLGA_highT samples were also investigated by photoelastic analysis to verify residual stress caused by the shear stress during injection and/or by the packing pressure during solidification, qualitatively confirming the DSC data. Photoelasticity of PLGA craniofacial plates using polarized light (Supplementary Figure 3) showed a small region with optical anisotropy near the injection point (the gate on the left of the plate pictures), probably due to the higher packing pressure at this area during solidification.
In vitro cell interaction
Indirect cytotoxicity tests
SAOS-2 percentage cell viability (Figure 3) was higher than 90% for PLGA_lowT and PLGA_highT at all evaluated time points (1, 3, 7, and 14 days), indicating that no cytotoxic low molecular weight by-products were released in the culture medium. Moreover, no differences (p > 0.05) were evidenced comparing the viability of cells cultured in contact with elutes of PLGA_lowT and PLGA_highT, demonstrating that the processing temperatures had no influence on possible release of cytotoxic compounds.
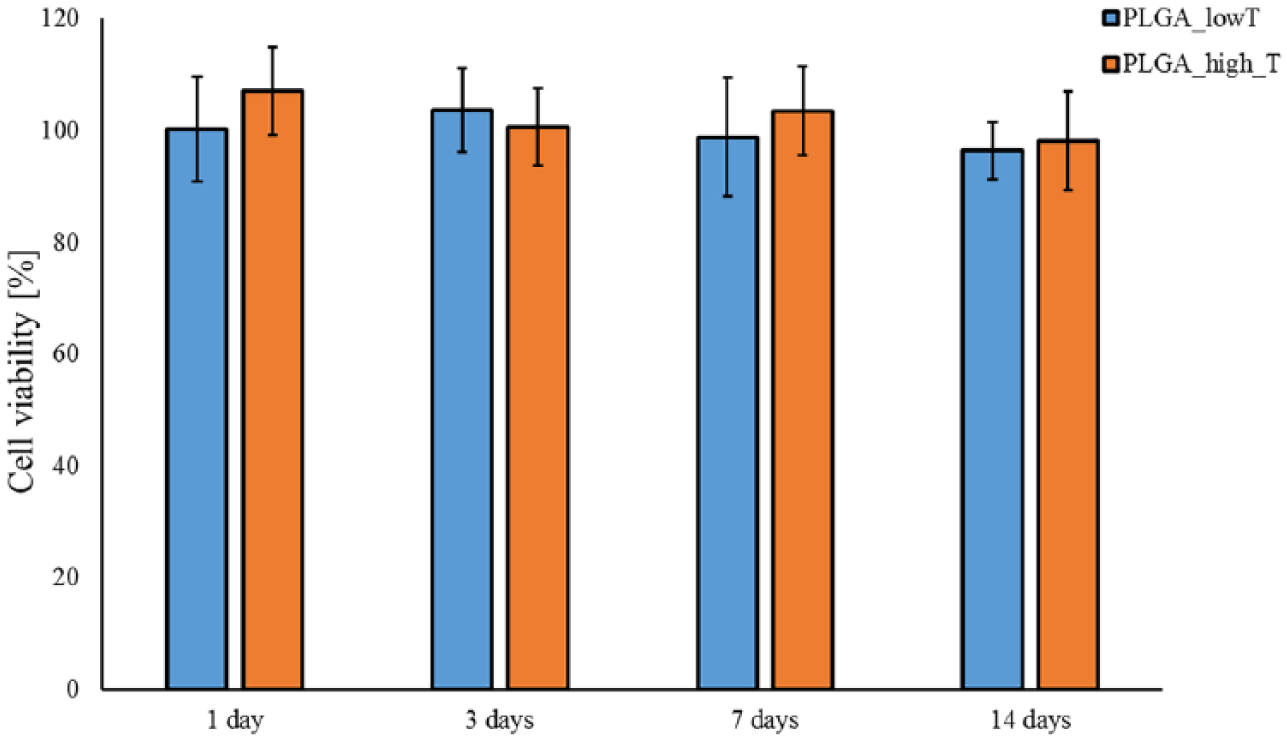
Viability of SAOS-2 cells cultured in eluates of the medium previously kept in contact with injection molded PLGA_lowT and PLGA_highT plates; viability is expressed as the percentage of cell viability of cells cultured with eluates (p > 0.05).
Direct cytocompatibility tests
The metabolic activity of SAOS-2 cells cultured on PLGA_lowT, PLGA_highT plates, and TCPS, as control, is reported in Figure 4(a). Increase in SAOS-2 cell metabolic activity was observed for all the conditions along the time of the experiment, demonstrating the ability of the PLGA plates to support osteoblast-like cell growth and proliferation. A higher metabolic activity (p < 0.05) was observed for cells cultured on TCPS wells after 7, 14, and 21 days of culture, compared to cells cultured on the PLGA_lowT and PLGA_highT plates. Since no cytotoxic effect was observed in the eluate of the PLGA plates (Figure 3), the difference in viability could be associated with the higher surface area of the TCPS well bottom in comparison to the PLGA plates (1.9 cm2 vs. 0.6 cm2). No differences were observed between the PLGA_lowT and PLGA_highT plates for the considered time points.
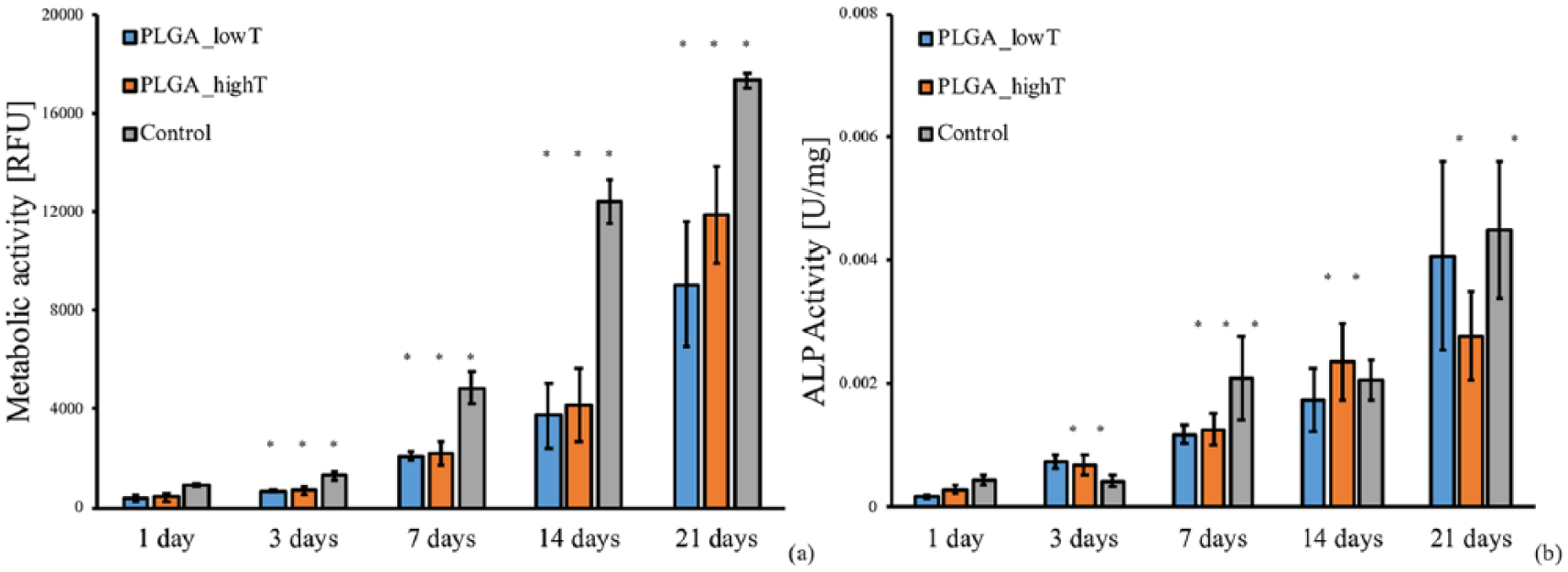
In vitro cytocompatibility tests of SAOS-2 cells cultured on injection molded PLGA plates: (a) metabolic activity measured by the Alamar Blue assay; (b) alkaline phosphatase activity normalized to the total protein content (*p < 0.05 comparing the same sample at subsequent time points: 3 vs. 7, 7 vs. 14, and 14 vs. 21 days).
ALP activity can be used as an early osteogenic marker to evaluate osteoblastic activity. The ALP activity of SAOS-2 cells cultured on PLGA_lowT, PLGA_highT, and TCPS plates is shown in Figure 4(b). The ALP activity was observed to increase after 7 days, in which no significant difference was verified among the PLGA_lowT, PLGA_highT, and TCPS plates. The higher expression of ALP over time indicated that the PLGA plates were able to sustain osteoblast-like cells functionality, preliminarily indicating a correct sustainment of functional bone tissue growth.
Cell attachment and proliferation were further evaluated using SEM-EDX, as illustrated in Figure 5 after 7 days of culture (see Supplementary Figure 2 for images at the different considered time points).

SEM images (scale bar = 50 µm) and EDX chemical mapping of carbon (C, blue), calcium (Ca, red), and phosphorus (P, green) of injection molded PLGA_lowT and PLGA_highT plates seeded with SAOS-2 cells after 7 days of culture and control plates (i.e., craniofacial plates without cells).
After 1 day, single cells were identified on PLGA plate surfaces (Supplementary Figure 4). At 3 and 7 days, cell proliferation was confirmed by a higher number of cells colonizing the surface of the injection molded plates. After 7 days, a confluent SAOS-2 cell layer was observed covering the surface of PLGA plates. The in vitro tests enabled signs of degradation of the PLGA plates due to hydrolysis. In fact, surface cracks caused by hydrolysis of PLGA due the contact with the culture medium are expected, particularly after 7, 14, and 21 days of culture. Nevertheless, the sample degradation did not interfere with attachments, spreading and proliferation of cells, thus it does not influence the implant colonization. Moreover, from day 7, sub-micrometric white spots, which appeared to be calcium phosphate deposits, were observed on the surface of the samples, thus indicating production of extracellular inorganic matrix by SAOS-2 cells.
To investigate the chemical features of the sub-micrometric particles, EDX was carried out in select areas (Figure 5). A craniofacial plate treated only with cell medium but without cells was used as the negative control. The EDX carbon, (C, blue), calcium (Ca, red), and phosphate (P, green) maps are shown in Figure 5. A qualitative higher concentration of Ca and P on particle surfaces confirmed the inorganic extracellular matrix deposition on the PLGA plates. Moreover, higher concentration of carbon was detected on the negative control plate confirming the active role of cells for inorganic matrix deposition.
The proposed in vitro cell culture procedure was designed to simulate the in vivo colonization and to understand the interaction of osteoblast-like cells with the materials under investigation (Supplementary Figure 2(a)). 21 In craniofacial surgeries, the central region of the plate is positioned perpendicularly to the bone fracture to be repaired.2,22 The plate position on the bone gives greater stability to the plates in relation to the tensile and bending force required in the fixation of the plate-screw system and enables cell colonization. As the healing process starts, bone cells migrate from the surrounding tissue to the central region of the plate (Supplementary Figure 2(b)).
PGA-based copolymers have higher degradation rates than other bioresorbable polymers, which limits their applications. 13 Besides the PGA fraction, several factors can influence the properties of the devices, such as the processing parameters, crystallinity and device geometry. The study of manufacturing condition and its effects is a key aspect during the development of bioresorbable craniofacial plate devices. Therefore, it is important to control the material/implant properties by fine tuning the production process parameters. In particular, in a recent study, we demonstrated that the PLGA plates tested in the present work undergo degradation in PBS after 120 days, proved by the presence of crumbling on the PLGA plates and loss of mechanical properties with increasing immersion time in PBS. 17 The present study contributed in the understanding of the effects of injection molding temperatures on the physic-chemical and mechanical properties of the PLGA plates and on the in vitro osteoblastic cell response. The results found in this work reinforce the potential of PLGA plates as promising implant devices for craniofacial and other orthopedic applications.
Conclusion
PLGA craniofacial plates were manufactured using different injection temperatures. Flexural tests showed values of bending stiffness of 0.8 and 1.2 N mm−1 for plates molded using the lower and higher temperatures, respectively. Dynamic mechanical tests revealed different viscoelastic behaviors for the plates molded using different injection temperatures. DSC analyses also showed different melting behaviors and residual stresses for the plates injected using different temperatures. The photoelastic analyses indicate low levels of birefringence near the gate of the molded plates, suggesting that the residual stress is mainly due to the packing pressure during solidification. The in vitro cellular tests revealed the absence of cell cytotoxicity. Cytocompatibility tests were performed with SAOS-2 osteoblast cells and indicated an increase in cell viability for all PLGA plates. Moreover, the plates stimulated cell proliferation and increased the ALP activity during the cell culture period. Adhered cells colonizing the plate surface were confirmed by SEM. Calcium and phosphate deposition on the PLGA plates was confirmed by EDX analysis.
Supplemental Material
Melo_suppl1 – Supplemental material for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates
Supplemental material, Melo_suppl1 for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates by Liliane Pimenta de Melo, Nicola Contessi Negrini, Silvia Farè, Carlos Rodrigo de Mello Roesler, Izabelle de Mello Gindri and Gean Vitor Salmoria in Journal of Applied Biomaterials & Functional Materials
Supplemental Material
Melo_suppl2 – Supplemental material for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates
Supplemental material, Melo_suppl2 for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates by Liliane Pimenta de Melo, Nicola Contessi Negrini, Silvia Farè, Carlos Rodrigo de Mello Roesler, Izabelle de Mello Gindri and Gean Vitor Salmoria in Journal of Applied Biomaterials & Functional Materials
Supplemental Material
Melo_suppl3 – Supplemental material for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates
Supplemental material, Melo_suppl3 for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates by Liliane Pimenta de Melo, Nicola Contessi Negrini, Silvia Farè, Carlos Rodrigo de Mello Roesler, Izabelle de Mello Gindri and Gean Vitor Salmoria in Journal of Applied Biomaterials & Functional Materials
Supplemental Material
Melo_suppl4 – Supplemental material for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates
Supplemental material, Melo_suppl4 for Thermomechanical and in vitro biological characterization of injection-molded PLGA craniofacial plates by Liliane Pimenta de Melo, Nicola Contessi Negrini, Silvia Farè, Carlos Rodrigo de Mello Roesler, Izabelle de Mello Gindri and Gean Vitor Salmoria in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by PRONEX/FAPESC, CNPQ, CAPES and FINEP.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
