Abstract
Bone defects remain a significant health issue and a major cause of morbidity in elderly patients. Composites based on collagen/calcium phosphate have been widely used for bone repair in clinical applications, owing to their comparability to bone extracellular matrix. This study aimed to evaluate the effects of a scaffold of collagen/calcium phosphate (COL/β-TCP) on bone formation to assess its potential use as a bone substitute to repair bone defects. Bilateral full-thickness critically sized calvarial defects (8 mm in diameter) were created in New Zealand white rabbits and treated with COL/β-TCP or COL scaffolds. One defect was also left unfilled as a control. Bone regeneration was assessed through histological evaluation using hematoxylin and eosin and Masson’s trichrome staining after 4 and 8 weeks. Alizarin Red staining was also utilized to observe the mineralization process. Our findings indicated that COL/β-TCP implantation could better enhance bone regeneration than COL and exhibited both new bone growth and scaffold material degradation.
Introduction
Complications associated with bone disorders induced by trauma, osteomyelitis, and osteosarcoma remain a clinically significant issue. Autologous bone grafts obtained from the rib, iliac crest, or tibia are the gold standard for bone defect repair.1–3 However, the procedure requires additional surgery, which can cause postoperative problems and result in an insufficient amount of provided bone to restore the defects, 4 and is also associated with complications, including limited donor tissue and donor site morbidity, pain, and infection. Allogenic bone for osteoconductive graft implantation has also been developed. However, this is accompanied by increased rates of nonunion, rejection, and possible fracture. 5 As an alternative strategy, bone tissue engineering has gained increasing attention, owing to the disadvantages of traditional treatments, such as immune rejection, chronic inflammation, or the need for complicated surgical procedures. 6 This technology utilizes a combination of various osteogenic cells, biocompatible scaffolds, and growth factors.
Materials used to fabricate bone grafts must be osteoinductive, osteoconductive, and biocompatible. The structure and mechanical properties of these materials should closely resemble those of natural bones. 7 Type I collagen (COL), a major component of bone extracellular matrix, is a good candidate for bone tissue engineering, since it provides an osteoconductive environment in which cell migration, proliferation, and differentiation facilitate new bone growth. This biomaterial has also exhibited therapeutic advantages in preclinical and clinical studies.8–13 However, pure collagen cannot be employed as a bone substitute, owing to its poor mechanical properties. Thus, increasing attention has been focused on preparing composites made of collagen and bioceramics. 14 Calcium phosphate ceramics, which feature good osteoinductive ability, could enable better bone regeneration in vivo, even without the addition of cells or growth factors. It is also worth mentioning that the defect anatomy and macrostructure of the biomaterial also affect new bone formation.15–17 For clinical bone repair, the use of calcium phosphate has proven equally effective in autologous bone grafts. 18 Substitutes based on these bioceramics have been used for more than two decades as fillers or scaffolds in dentistry and orthopedics.19, 20 To fill bone defects, β-tricalcium phosphate (β-TCP), which has excellent resorbability, biocompatibility, and osteoconduction, has also been a focus of research. During the bone remodeling process, β-TCP is slowly degraded and replaced by newly mature bone in animal experiments.21, 22 It has been suggested that this ceramic could promote osteogenesis by increasing adenosine signaling in phosphate metabolism and providing osteoinductive growth factors.23–25 Additionally, this bioceramic has been proven to facilitate osteoblast differentiation, mineralization of extracellular matrix, and subsequent bone formation. 26 Calcium phosphate scaffolds aid in osteogenesis and osseointegration; this facility could be related to their topography, surface charge, and chemical properties. These scaffolds contain minerals similar to those in natural bones; thus, they provide sufficient Ca2+ and PO43− during the bone regeneration process. 27
Importantly, it is necessary to keep the composition of the bone scaffold similar to that of natural bone to provide a desirable environment for cell attachment and proliferation. 28 Both COL and β-TCP could mimic the basic composition of bone and have been used as bone graft substitutes, owing to their osteoconductive and biocompatible properties.27, 29, 30 Several products based on COL and calcium phosphate have been introduced in the market. These two materials, together or in combination with other materials, have shown potential for use as an alternative approach for bone regeneration.14, 31 For example, the Integra Mozaik™ (Integra LifeSciences, USA) consists of 80% β-TCP and 20% type I bovine COL. The efficacy of the osteoconductive scaffold is equivalent to that of autografting. In a clinical study, this graft was applied to patients with posterolateral lumbar fusion, and 100% fusion resulted in all single- and two-level procedures, with an overall fusion rate of 90%. This product was developed to mimic the composition and pore structure of natural human bone. 32
Successful bone grafts should be replaced by new functional bone tissue that is biocompatible with the host tissue. 4 The ability of calcium phosphate ceramics to enhance bone formation depends highly on crystallinity, crystalline phase, and calcium/phosphorus ratio, which leads to the release of calcium and phosphate ions for bone mineralization.33, 34 More recently, we fabricated COL/β-TCP scaffolds with a β-TCP/COL weight ratio of four using a freeze-drying method. Comparison of the alkaline phosphatase activity showed that scaffolds containing β-TCP could significantly improve the differentiation of bone marrow-derived mesenchymal stem cells into osteoblasts compared with collagen, without the addition of growth factors. In fact, the introduction of β-TCP powder into the collagen matrix improved vascularization as well as the biological and mechanical properties of the collagen scaffold; this was consistent with previous studies.35, 36 Here we further evaluated the effects of a complex COL/β-TCP against those of COL scaffold on bone formation in rabbit calvarial bone defects to assess the potential use of COL/β-TCP as a bone substitute to repair bone defects.
Materials and methods
Materials
All materials and reagents were purchased from Sigma-Aldrich (Germany) unless otherwise specified.
Scaffold synthesis
The COL/β-TCP composite was prepared, as previously explained. 37 Briefly, collagen type I (NZA, Iran) was dispersed in diluted hydrochloric acid (pH = 2) resulting in a 1% (w/v) solution. The β-TCP powder was then added to the collagen solution while stirring. The ratio of β-TCP/COL weight was determined to be 4/1. The homogeneous suspension was poured into a plastic mold and frozen at −20 °C and −80 °C for 4 h and overnight, respectively. Then, the frozen sample was lyophilized to create a porous COL/β-TCP composite scaffold (freeze drier, ALPH1-2LD, UK). To prepare COL scaffolds, a similar protocol was repeated without β-TCP. To crosslink the obtained scaffolds, glutaraldehyde (0.5%) solution was used for 24 h. Samples were then soaked in deionized water for 4 days, with daily water refreshing to remove remnants of the glutaraldehyde, followed by lyophilization again for experimentation.
Experimental animals and surgical procedures
Animal studies were conducted according to the NIH Guidelines approved by the Ethics Committee of Tarbiat Modares University, Iran (IR.TMU.REC.1395.388). In this study, 12 New Zealand white rabbits (2.5–3 kg) were randomly divided into three experimental groups (n = 4/group) and kept in individual cages. Rabbits were controlled in a standard environment (humidity and temperature) with a 12/12 hours light/dark cycle and standard access to food and water. Animals received an intramuscular injection of xylazine (10 mg/kg) (2%, Alfasan, The Netherlands) and ketamine (90 mg/kg) (10%, Alfasan, The Netherlands). Their calvaria were then shaved, washed with 1.5% aqueous chlorhexidine digluconate, and draped, utilizing povidone iodide to sterilize the area. Calvaria were exposed via a skin incision; the muscle and periosteum of the calvarium were ablated, and three standardized full-thickness defects (8 mm in diameter) were created in the proximal-medial bone area of the calvaria using surgical trephine drills at low speed under continuous irrigation with saline buffer. Care was taken to avoid injury to the dura or midsagittal sinus. One defect was filled with COL/β-TCP scaffold (Group A). The second defect was filled with the COL scaffold (Group B), and the third was left unfilled as a negative control (Group C). The periosteum, muscle, and skin were then repositioned and sutured; subsequently, tetracycline was sprayed on the skin of the calvaria. Amoxicillin (0.1 mL/kg intramuscularly) (15%, Tolid Darou, Iran) was administered after surgery to prevent infections. Animals were protected and kept warm in individual cages until they had completely recovered. They were then sent to the holding room and had free access to water and food. To control postoperative pain, animals were administered ketorolac tromethamine (Tarasyn, Korea) for 3 days.
Clinical and histological analysis
Rabbits were killed with an overdose of anesthetics at 4 and 8 weeks after surgery. The cranial vault and attached pericranium were excised carefully to observe both the cranial and dural sides for gross inflammation and were photographed. The treated calvarium defects were subjected to microscopic studies and histological and radiological analyses.
Specimens were fixed in 10% formalin and decalcified by immersion in 10% nitric acid for 14 days; this solution was renewed every 24 h. After that, samples were dehydrated in a graded series of ethanol solution (80–100%) and cleared with toluene. Each sample was embedded in paraffin via the usual method; specimens were sectioned (5 µm) (Leica Microsystems SP 1600, Germany) and stained with hematoxylin and eosin and Masson’s trichrome. Alizarin Red staining was also utilized to observe the mineralization process. Histological evaluation consisted of examination of at least three sections of each implant under light microscopy (Leica Microsystems AG, Germany).
Results and discussion
Owing to their high biocompatibility and bioactivity, bioceramics based on β-TCP have been widely used for bone repair in clinical applications.38–42 Some researchers have also considered β-TCP to be osteoinductive. 43 In this study, bone regeneration was assessed by implanting COL/β-TCP and COL disks into critically sized rabbit calvarial defects, which were then studied after 8 weeks. The anatomical and physiological characteristics of the rabbit make it especially appropriate for the study of certain human diseases. The New Zealand rabbit has been considered a suitable experimental model for analyzing the osteogenic capability of biomaterials.44–46 By creating bilateral defects, we were able to compare the efficacy of the two scaffolds within the same animal. The scaffolds were placed in 8 mm full-thickness rabbit calvarial bone defects; one defect was left unfilled in each rabbit, as a control (Figure 1).
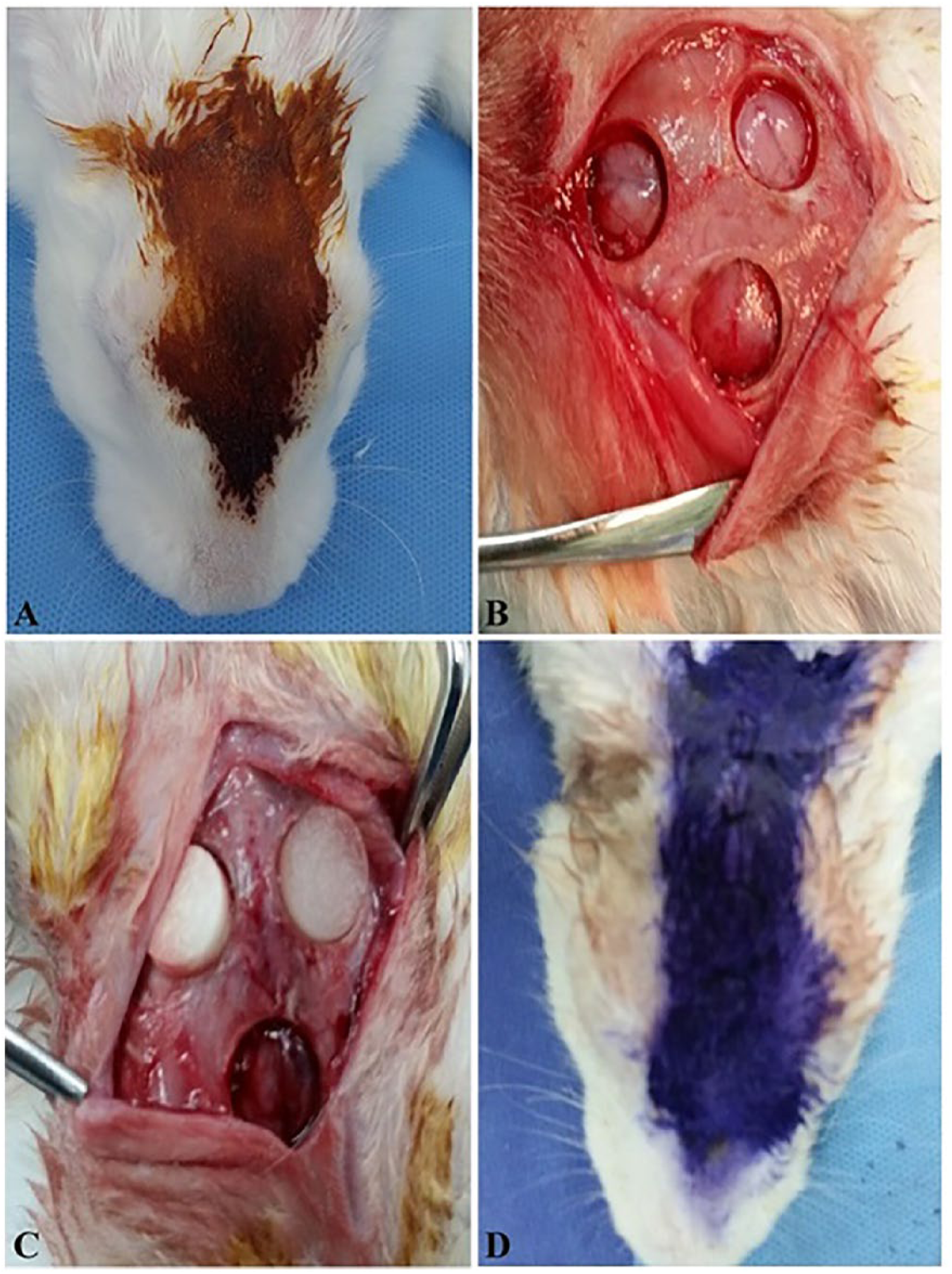
Bone graft in rabbit calvarial bone defects. A. Prepared rabbit calvarial skin. B. A 3 cm incision was made along the midline of the scalp and bilateral 8 mm full-thickness defects were made in both parietal bones. C. Scaffolds were placed directly onto the dura, leaving one defect unfilled. D. The skin was closed and sutured.
A clinical presentation of the examined groups at 4 and 8 weeks is given in Figure 2. A fibrous scar covered the entire defect area. The COL/β-TCP-filled defect appeared to be more intact than its COL-filled counterpart. No inflammation was detected in either experimental group macroscopically. Eight weeks after implantation, the scaffold-treated defects were thinner than the surrounding bone. However, those that contained COL/β-TCP were contiguous, being scarcely distinguishable from the surrounding bone tissue, and with no mobility. The COL-treated defect was thinner than the COL/β-TCP defect, macroscopically, confirming the results of a previous study. 47
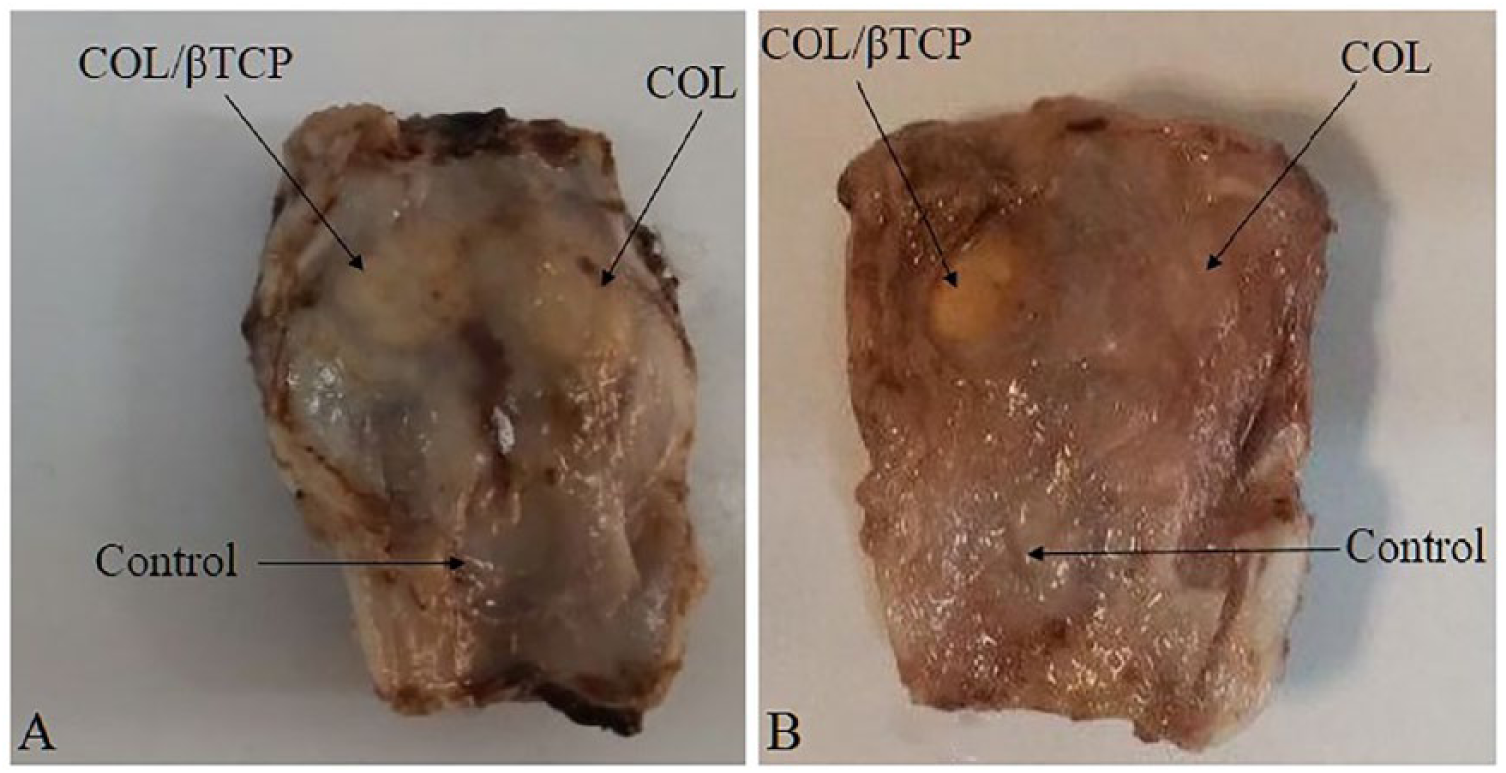
Cranial gross appearance of the defects, showing COL and COL/βTCP scaffolds and control (unfilled). A. After 4 weeks. B. After 8 weeks.
For the observations using hematoxylin and eosin staining, the specimens were examined using a light microscope at magnifications of ×40 and ×100. The defects filled with the COL/β-TCP scaffold showed immature bone formation within 4 weeks. New bone formation was observed to start bridging the defects at 8 weeks, with noticeable cell migration. It was difficult to distinguish the border of the defect from surrounding bone tissue. However, no cell permeation or bone formation was observed in COL-treated defect at 4 weeks, and the border between the original bone and the defect was not clearly linked with newly formed bone. Delayed cell penetration and the start of bone formation were observed at 8 weeks for this graft. The unfilled defect did not heal and new bone was only observed at the defect margin. Fibrous tissue covered the rest of the defect (Figure 3).
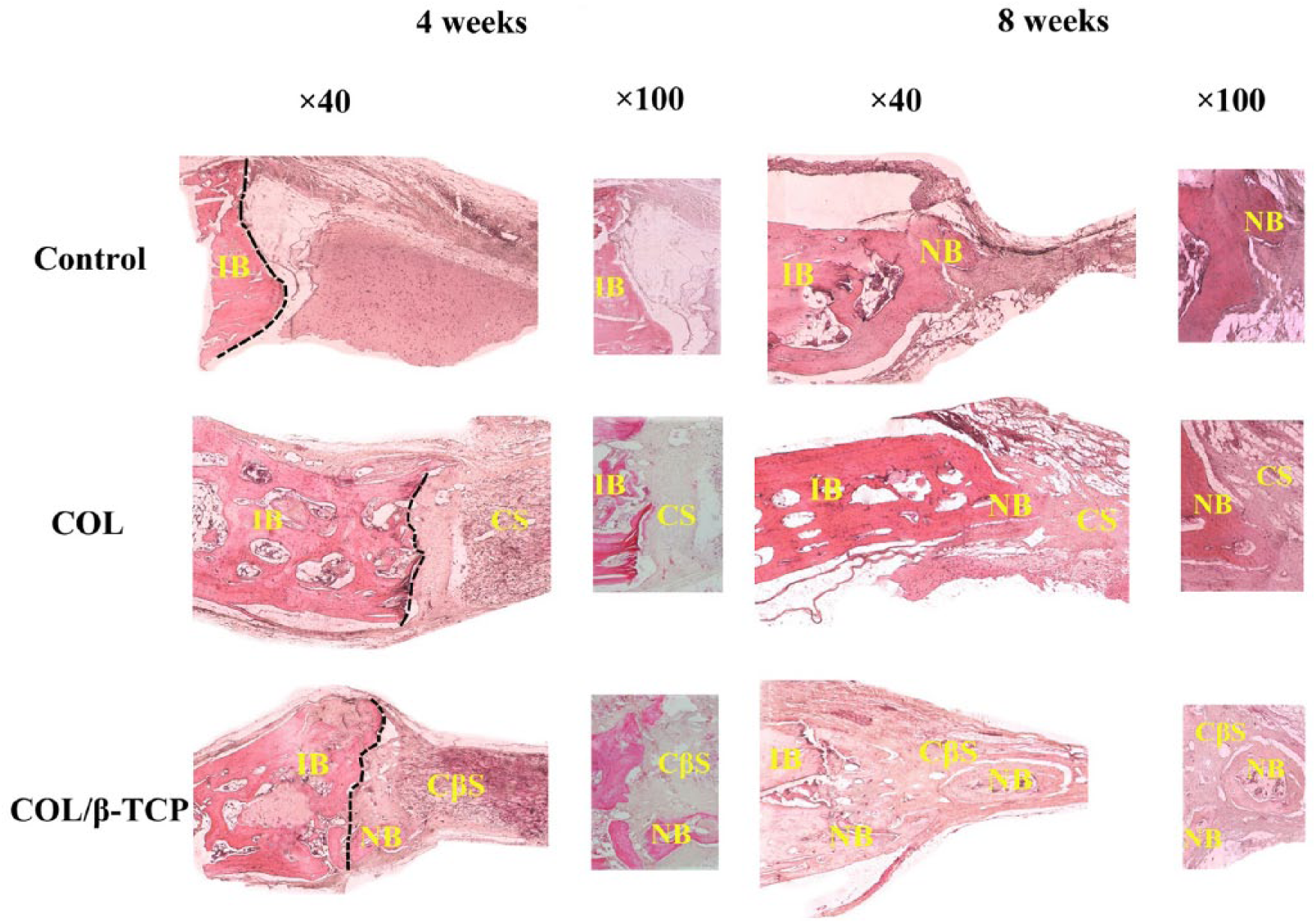
Histological findings at 4 and 8 weeks; hematoxylin and eosin stain (magnification, ×40 and 100). The dotted line indicates the border between intact bone and scaffolds.
Bone formation was further confirmed by Masson’s trichrome staining (Figure 4). The obtained data for the COL/β-TCP scaffold revealed deposition of the mature collagen at the new bone formation zone. At 4 weeks, immature bone generation was detected, followed by new bone formation after 8 weeks. However, immature bone formation was not observed in the COL and control groups after 4 weeks. In the unfilled group, fibrous tissue was observed, with new bone ingrowth around the defect margins. In the scaffold-treated groups, newly formed bone was not only on every side of the margin defect but also surrounded and was between the implanted scaffolds. However, new bone formation was more noticeable in the COL/β-TCP group than in the COL group.
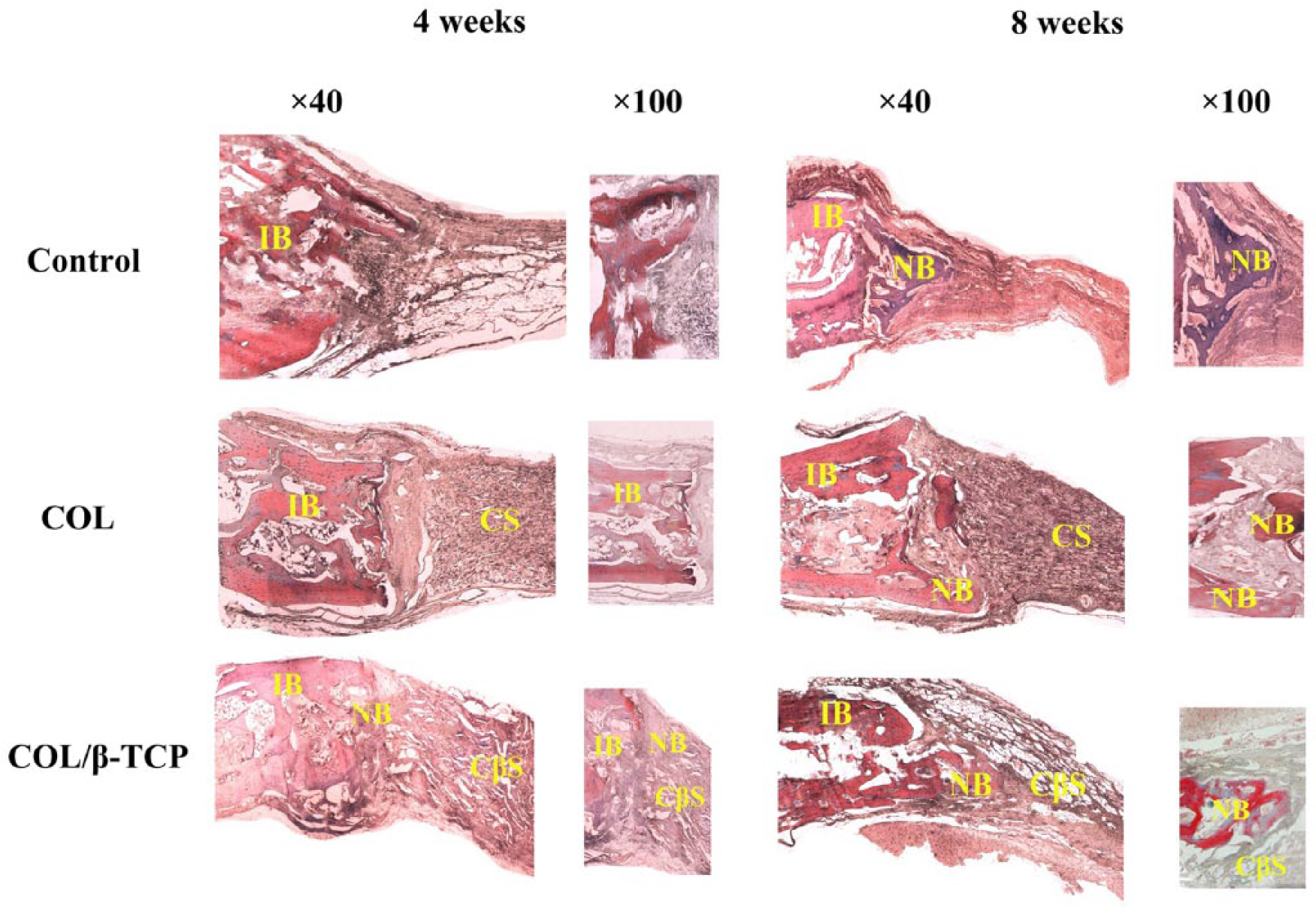
Histological findings at 4 and 8 weeks; Masson’s trichrome stain (magnification, ×40 and 100).
Alizarin Red staining was used to observe the mineralization stages of the tissue that formed at specific time points. Figure 5 shows the start of immature bone formation in the COL/β-TCP scaffolds at 4 weeks, followed by the detection of new bone formation and bridging at 8 weeks. However, the unfilled defect showed new bone only at the margin, while fibrous tissue covered the rest of the defect. For the COL group, only a little bone formation was observed and most of the defects were empty. Our findings indicated that COL/β-TCP implantation could enhance bone regeneration more effectively than COL in this model. This could be due to the high concentration of calcium and phosphate ions (Ca2+ and PO43−) provided by β-TCP, which creates a desirable environment for increasing the proliferation and attachment of bone marrow-derived stem cells.48–50 The release of Ca2+, PO43−, and HPO42− from the material into the surrounding biological fluid provides nucleation sites for the precipitation of biological carbonated apatite. Moreover, the calcium and phosphate released from the β-TCP dissociation could increase osteoblast alkaline phosphatase activity.51–55 Increased PO43− in the surrounding medium provides an alkaline environment, which has previously been proven to increase the alkaline phosphatase activity within human dental pulp cells 56 and might be more desirable for osteogenesis. For example, Li et al. 57 reported that the addition of β-TCP to COL/hydroxyapatite particles could result in better proliferation of adipose-derived stem cells with osteogenesis-promoting effects. Li et al. 57 also mentioned that its excellent mechanical property could both support bone formation and make the scaffold conducive for use in clinical applications. In our previous experiment, 37 we also showed that the presence of β-TCP in the collagen matrix could provide a much higher compressive modulus for the prepared composite than that of collagen. Moreover, the addition of β-TCP to the collagen assisted vascularization with good integration with the surrounding tissue. In fact, the rapid formation of a functional blood vasculature affects tissue engineering construct success. The good water absorbance and high porosity of this scaffold allowed blood infiltration and material exchange and promoted cell proliferation with the ability for manipulation with normal saline and blood. 57 Calcium phosphate ceramics are able to induce angiogenesis, a requirement for osteoinduction. In fact, a higher content of β-TCP phase has been proven to enhance neovascularization, a prerequisite for osteoinduction. 58
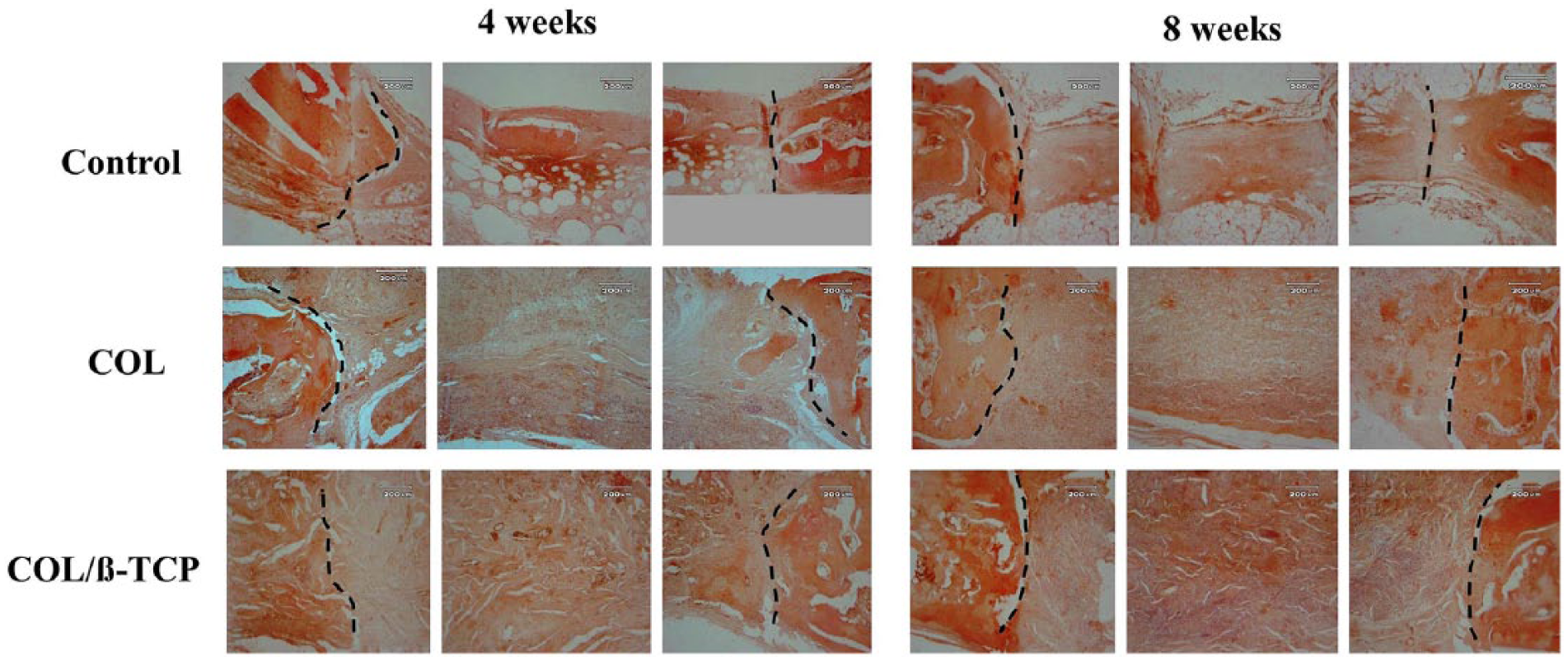
Histological findings after 4 and 8 weeks; Alizarin Red stain (magnification, ×40 and 100).
Osteoconductivity is also a useful property for synthetic bone graft substitutes. An ideal scaffold with osteoconductivity should have macropores that are 150–500 µm with 60–80% interconnected porosity. 59 We previously confirmed that prepared scaffolds exhibited interconnected macropores that were 150–200 µm in diameter, with an average porosity of 95–98%, which could provide an appropriate environment for inducing osteogenic differentiation. The obtained interconnectivity of the macropores is significant for tissue ingrowth into the material. 60 The collagen matrix also provides a template for depositing mineral crystals. 61 The material’s swelling ability, which helps adsorb nutrients by maintaining the three-dimensional network, also affects cell growth and differentiation, influencing the efficacy of engineered bone constructs. We earlier found that the distribution of β-TCP within the collagen matrix of the composite could increasingly improve the stability of the composite scaffold compared with collagen, which demonstrated structural instability with a much higher swelling ratio. 37
Since complete biodegradation of COL/β-TCP was not confirmed in this study after 8 weeks, prolonged experiments are necessary to achieve a better understanding of the critical parameters for bone repair using this scaffold. One reason for this delay could be apatite formation on the surface of the scaffold after implantation, which hinders the release of TCP in the physiological bone remodeling. 62 It was previously reported that the percentage of newly formed bone in a critically sized bone defect of dog calvaria by octacalcium phosphate/collagen can significantly increase between 3 and 6 months. 63 Therefore, it may be useful to continue our observation for 6 months to evaluate bone formation. Inzana et al. 30 fabricated a synthetic calcium phosphate and collagen bone graft substitute by low-temperature three-dimensional printing. They implanted scaffolds into a critically sized murine femoral defect for 9 weeks and confirmed that the scaffolds were osteoconductive and supported new bone growth but were only partly osteoinductive and unable to completely heal the defect. Inzana et al. 30 stated that supplementing the scaffold material with growth factors or cells to induce new bone formation could be an alternative strategy for completely healing critically sized defects using such synthetic bone graft substitutes.29, 30 Another report stated that β-TCP is an osteoconductive material supporting new bone formation but cannot drive cellular differentiation. Combination with bone marrow aspirate or concentrated bone marrow aspirate might be more effective. 29 In fact, the resorption of β-TCP, which is important for bone formation, may be promoted using such growth factors as fibroblast growth factor-2 64 or bone morphogenetic protein-2 (BMP-2). 65
Furthermore, since bones have different resorption and formation rates, 66 scaffold biodegradation might occur more rapidly in maxillary defects than in calvarial defects. 47 Thus, modifying the scaffold preparation process could be a suitable way to achieve complete scaffold resorption. 47 Walsh et al. 67 assessed the in-vivo performance of two US Federal Drug Administration-approved COL-calcium phosphate bone grafts for repairing critically sized cancellous defects in rabbits. Walsh et al. 67 stated that even in chemically virtually identical bone grafts, a different in-vivo response might be observed. This difference could be due to the collagen source (skin versus tendon) and processing (crosslinking method, washing), as well as calcium phosphate component differences (amount, porosity, or size), which influences the resorption profiles. 67
It is also worth noticing that, apart from the implant’s chemical composition and geometry, microstructural surface parameters, including grain size, roughness, specific surface area, and microporosity, are all considered important factors that affect the osteoinductive abilities of calcium phosphate ceramics. 68 In addition, β-TCP is considered to have a positive effect on BMP-2 gene expression, 18 which was not assessed in this experiment. More information is still needed about the potential usefulness of the synthetic scaffold in orthopedic and dental applications.
Conclusions
Owing to the similarities between calcium phosphate ceramics and the mineral phase of bone, their ability to bond with bone tissue, and their high biocompatibility, these ceramics have been widely used as bone graft substitutes in dentistry, orthopedics, and maxillofacial surgery over the past few decades. 69 The aim of this study was to assess the efficacy of a prepared COL/β-TCP composite for new bone formation in rabbit calvarial bone defects. This study demonstrated that this scaffold might have potential for use as a bone substitute in clinical cases. The prepared composite has proven advantageous properties for use as a bone substitute, including easy handling, biodegradation properties, and replacement by newly formed bone without the use of cells or other external cytokines.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
