Abstract
Background:
Encapsulation is one of the methods used to trap active ingredients in the wall material of microparticles.
Aim:
The aim of this study was to evaluate the encapsulation of red ginger oleoresin using an emulsion crosslinking method with chitosan as the wall material.
Methods:
Emulsions were formed of red ginger oleoresin with chitosan in concentrations of 1%, 2%, 3%, and 4% (w/v), respectively. The emulsions were then mixed with corn oil and stirred for one hour to obtain a second set of emulsions, and glutaraldehyde saturated toluene (GST) was added dropwise in quantities of 20, 10, 6.7, and 5 ml, respectively. This was followed by the addition of 2 ml of 25% glutaraldehyde and the emulsions were stirred for two hours. The resulting microcapsules were washed with petroleum ether followed by hexane and then dried in an oven at 70oC.
Results:
The emulsion crosslinking method used to trap the red ginger oleoresin in chitosan produced microcapsules of good spherical geometry with the mean diameter ranging from 75.61 ± 11.8 µm to 178.65 ± 40.7 µm. The highest yield was 98.93% and encapsulation efficiency was 83.1%. Thermogravimetric and differential thermal analysis showed that the melting point was at a temperature between 120 and 130oC.
Conclusion:
Chitosan concentration has little effect on encapsulation yield, whereas the amount of GST tends to strengthen the crosslinking bonds of chitosan and reduces the mean diameter of microspheres.
Introduction
Ginger contains essential oils and oleoresins, in which its quality depends on the geographical location and climate in which the plants are grown.
1
Ginger contains approximately 4% to 7.5% oleoresin.2,3 Red ginger (
The weakness of oleoresin is that it is susceptible to the effects of heat and light. Changes in the components of fatty oil and monoterpinic hydrocarbons in the polymer can cause degradation of oleoresins. 8 Encapsulation of oleoresin has proven to be one method that can prevent its deterioration. 9 The wall materials used for encapsulation are subject to certain criteria: (a) they should not react with the core material, (b) they should have low viscosity even at high concentration, (c) they should have good emulsion stability, and (d) they should have properly controlled release. 10 A wall material that can potentially be used for encapsulation of red ginger oleoresin is chitosan, because it is biocompatible, biodegradable, and non-toxic. 11 Encapsulation generally consists of physical and chemical methods. Encapsulation by physical methods includes: spray drying, freeze drying, extrusion, fluidized bed, etc. 12 Chemical methods for encapsulation are: coacervation, interfacial polymerization, co-crystallization, and molecular inclusion. 10
The encapsulation used in this study was achieved by a modified emulsion crosslinking. The modifications adopted in this study were the use of double emulsions and vegetable oil as a continuous phase.
The emulsion crosslinking method utilizes the reactivity between the amine group in chitosan as a coating agent and the aldehyde group as a crosslinking agent. This method can be used to encapsulate either soluble or insoluble materials, liquid or solid materials. Moreover, it can also produce microcapsules and nanocapsules.12,13 The concentration of chitosan and the amount of glutaraldehyde saturated toluene (GST) influence, such as encapsulation yield, encapsulation efficiency, and characterization of microcapsule properties. Higher concentration of chitosan polymer increases encapsulation efficiency14,15,16 and decreases the porosity of the microcapsules. 17 The purpose of this study was to evaluate the emulsion crosslinking method for red ginger oleoresin encapsulation based on changes in chitosan concentration and GST volume. The evaluation parameters are: yield, encapsulation efficiency, mean diameter, and characterization of the microcapsules.
Materials and methods
Materials
Red ginger oleoresin was obtained from Lansida Group, glutaraldehyde 25% was obtained from Merck, and technical toluene 99.6% from CV. Tri Jaya Dinamika. Chitosan (degree of deacetylation (DD) = 87.2%) was obtained from PT. Biotech Surindo. The corn oil was produced by CV. Surya Agung. Glacial acetic acid 100% was obtained from Merck, and petroleum ether, n-hexane, and methanol were technical grade product obtained from CV. Labora.
Preparation of GST
The GST solution was prepared by mixing 25% glutaraldehyde solution and toluene at volume ratio of 1:1 under 500 rpm stirring for three hours. The solution was kept overnight for stabilization. The upper layer of the solution was separated and applied as glutaraldehyde saturated toluene (GST).
Preparation of red ginger oleoresin-loaded chitosan microspheres
The emulsion crosslinking method for preparation of red ginger oleoresin-loaded chitosan microcapsules was a modification of the research reported by Jameela and Jayakrishnan 18 and Campos et al. 19 About 4 g of red ginger oleoresin was added to 40 ml of chitosan solution with concentrations of 1%, 2%, 3%, and 4% (w/v) respectively. The mixture was stirred using a homogenizer (IKA-Werk Ultra-Turrax) for 30 minutes to form an oil in water (O/W) emulsion. The emulsion was added to 150 ml of corn oil as the oil medium, and then was agitated for one hour to form an oil in water in oil (O/W/O) emulsion. GST was added dropwise to the emulsion (O/W/O) every 15 minutes. When the addition of GST was complete, the mixture was stirred for 15 minutes, and then 2 ml of 25% glutaraldehyde was added and stirred for two hours. Microcapsules were then separated by 10 minutes centrifugation. The microcapsule product was washed in petroleum ether and n-hexane solution and was dried at 70oC.
Encapsulation yield
Encapsulation yield is the ratio between the weight of red ginger oleoresin microcapsules that have been dried and the total weight of the core material (red ginger oleoresin) and polymers (chitosan). The encapsulation yield is determined by the equation (1)

Encapsulation efficiency
Tan et al. 20 and Tonon et al. 21 calculated the encapsulation efficiency as in Equations (2) to (4). Dried microcapsules (1 g) were added with 20 ml of methanol and then stirred for one minute to dissolve the surface oil. The suspension was filtered and the residue was washed twice with 20 ml of methanol. The powder residue was dried for 30 minutes and weighed.

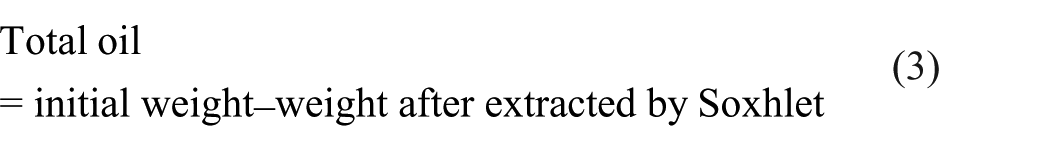

Total oil was determined using a Soxhlet extraction unit and 1 g of microcapsules was extracted using 200 ml of methanol in six hours of operation. The powder was dried and weighed.
Gas chromatography mass spectrometry (GCMS)
The components of red ginger oleoresin were analyzed using a GCMS-QP2010S Shimadzu (AgilentJ% W HP-5 column, length 30 meters, and ID 0.25 mm) with helium as the carrier gas. The column oven temperature was 60 oC and injection temperature was 300 oC. The mode of injection was split; pressure was 13.0 kPa; and the total and column flow were 80.7 ml per minute and 0.51 ml per minute, respectively.
Scanning electron microscopy
Analysis of the morphology of the chitosan microspheres containing red ginger oleoresin was performed using a JEOL JSM-6510LV type scanning electron microscope (SEM). The microcapsules were coated with platinum. Resolution high vacuum (HV) mode was 3.0 nm (30 kV), low vacuum (LV) mode was 4.0 nm (30 kV), and accelerating voltage ranged from 0.5 kV to 30 kV.
Thermogravimetric analysis
Thermal stability analysis was conducted by thermogravimetric analysis (TGA) using a DTG-60 detector. The sample was heated from 30oC to 300oC at a rate of 10oC per minute with a constant nitrogen rate of 30 ml per minute.
Results and discussion
Composition of red ginger oleoresin
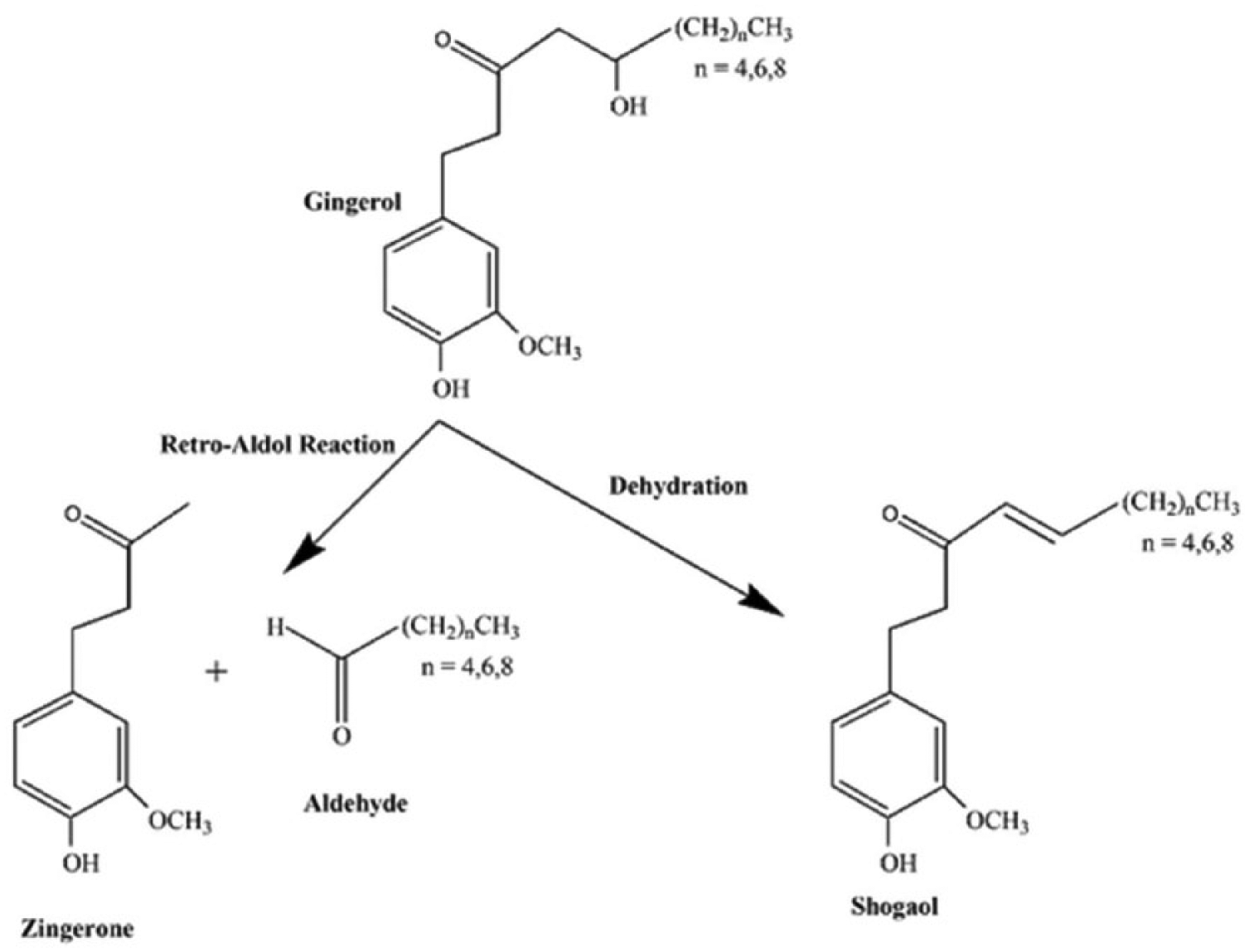
The main components of the red ginger oleoresin were shogaol, zingerone, and zingiberene, as shown in Figure 1. GCMS analysis of the red ginger oleoresin used in this study did not detect the presence of gingerol. The absence of gingerol may be due to it having been degraded during the extraction process to obtain red ginger oleoresin. 22 Gingerol can also be degraded due to the effects of storage and the drying process used to isolate other active components such as shogaol and Zingerone. 23 Figure 2 illustrates the degradation of gingerol to shogaol and zingerone.
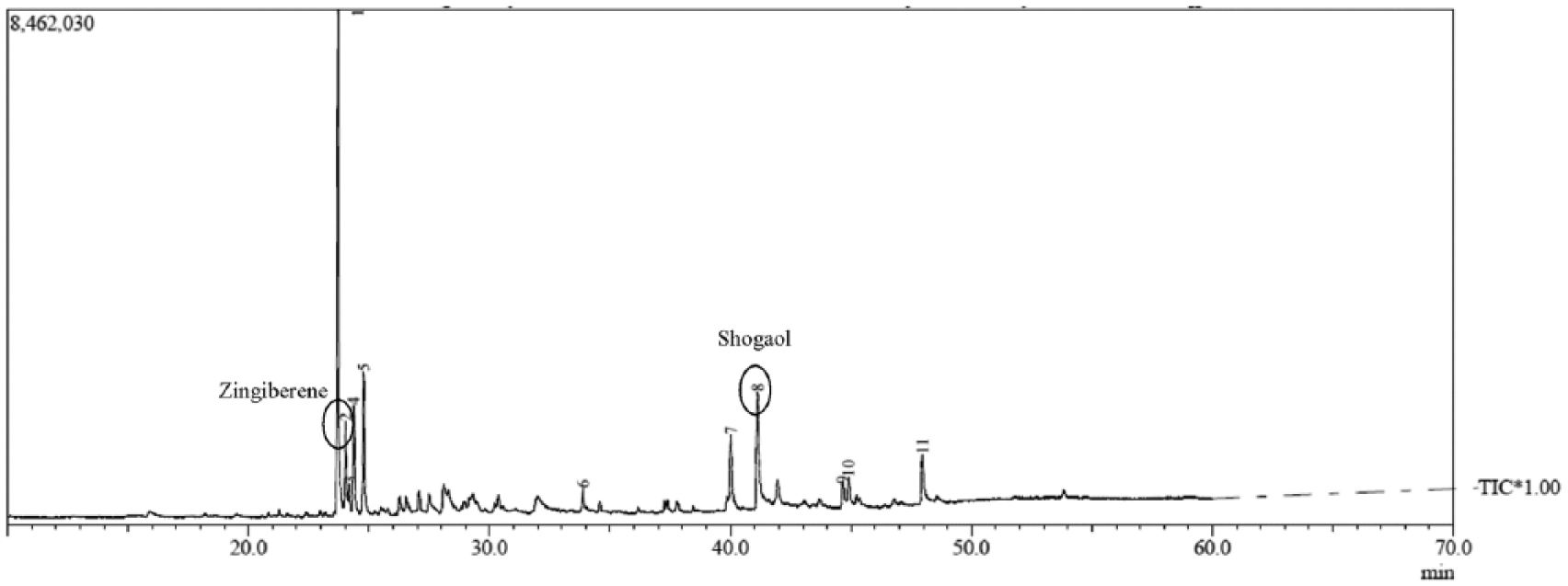
GCMS analysis of red ginger oleoresin.
Encapsulation yield
In this research, the method of emulsion crosslinking used for red ginger oleoresin encapsulation was successfully performed. The yield of microcapsules ranged from 81.35% to 98.93%, as shown in Table 1. Changes in chitosan concentration and GST volume resulted in the changes in yield of microcapsules. Table 1 shows that decreasing GST volume also decreased the yield of microcapsules at the same chitosan concentration. The decrease in GST volume would reduce the glutaraldehyde content in GST, thereby reducing the process of solidification of the emulsion into microcapsules. Higher amounts of glutaraldehyde would increase crosslinking reactions. 24 In contrast to studies conducted by Ofokansi, 25 the yield was irregular. The largest yield of 99.19% was obtained using 25 ml of crosslinking agent.
Physical properties of red ginger oleoresin microcapsules.
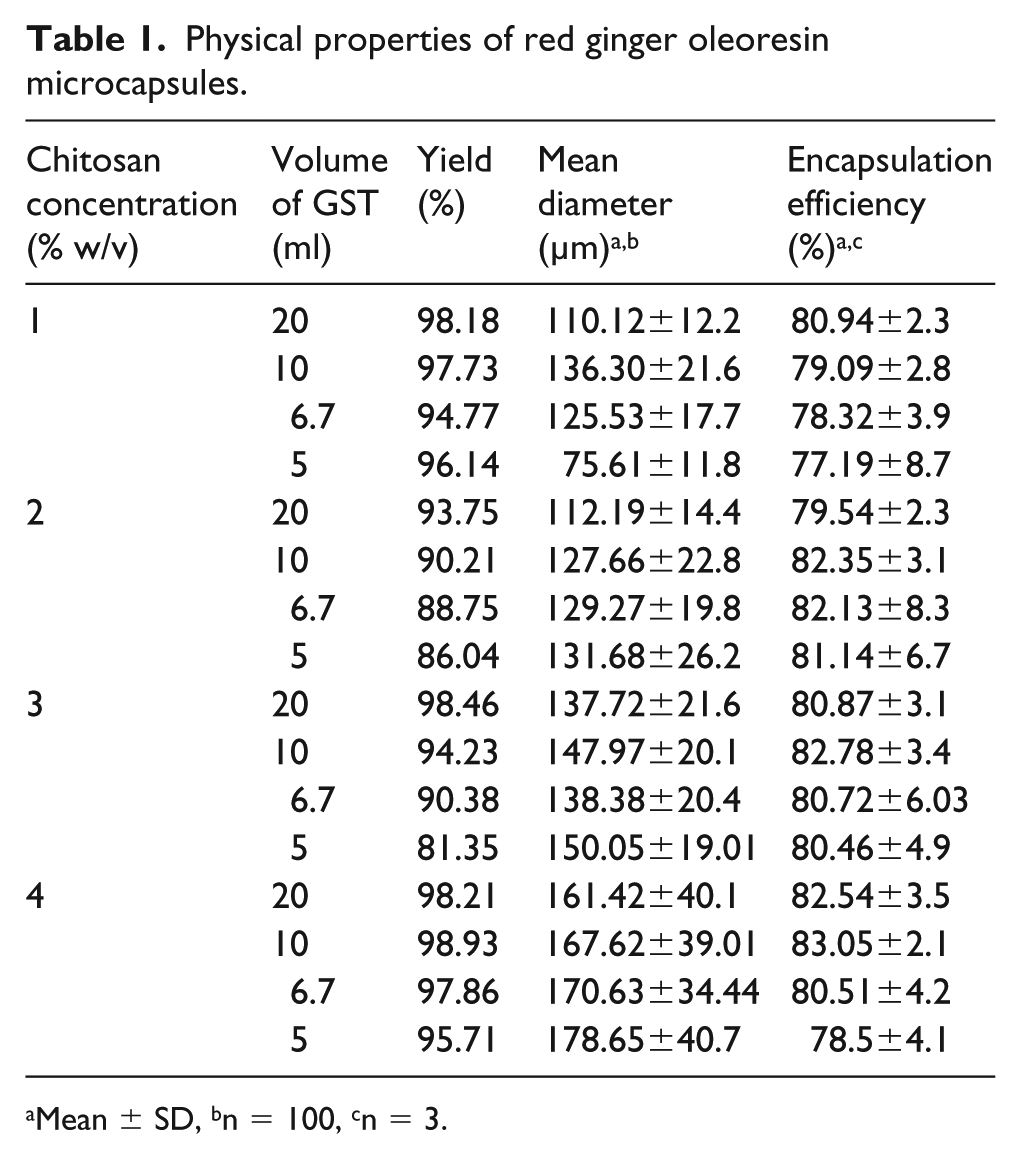
Mean ± SD, bn = 100, cn = 3.
The yield of microcapsules due to the effect of changes in chitosan concentration in the same GST volume was irregular. The highest yield was obtained at 4% chitosan concentration with 10 ml GST volume, while the lowest yield was obtained at 3% chitosan concentration with 5 ml GST volume. Table 1 shows that different chitosan concentrations had no significant effect on yield compared with the effect of GST volume.
Mean diameter analysis
The emulsion crosslinking method used for red ginger oleoresin encapsulation was successful in making spherical microcapsules. The particle size of the microcapsules was analyzed and calculated by digital microscopy. The results of the analysis of microcapsule size are shown in Table 1. Increasing chitosan concentration was observed to result in microcapsules of greater diameter. Microcapsule diameter increased due to the higher degree of chitosan molecular crosslinking under GST solution. The effect of the amount of GST used as the crosslinking agent upon the particle size may be related to the thickness of chitosan as the wall material of the encapsulation. The mechanism of red ginger oleoresin encapsulation by chitosan microsphere under the emulsion crosslinking method is illustrated in Figure 3.
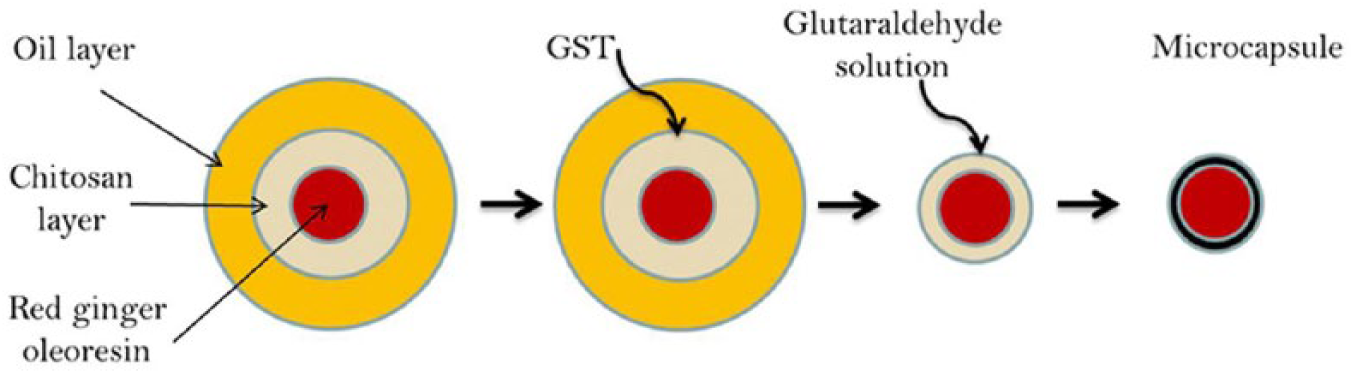
Illustration of the encapsulation process using the emulsion crosslinking method.
Table 1 shows the difference in particle size based on chitosan concentration: chitosan concentration of 4% produced microcapsules of greater diameter than that of chitosan concentrations of 3%, 2%, or 1%. Chitosan concentration will affect the viscosity of the emulsion. Increasing concentration of chitosan increases viscosity. Increasing viscosity of the polymer in the internal phase would result in the formation of a coarser emulsion with large droplets and would lead to the formation of large microparticles. 26 Increasing concentration of chitosan increases the size of microcapsules, as shown in Figure 4. The largest microcapsule (Figure 4(a)) was from chitosan concentration of 4%, while the smallest microcapsule (Figure 4(d)) was from chitosan concentration of 1%.

Photographs of chitosan microcapsules: (a) chitosan 4%; (b) chitosan 3%; (c) chitosan 2%; (d) chitosan 1%.
Higher chitosan concentration could increase microcapsules diameter. Patel and Patel 27 also found that chitosan concentration has an effect on microcapsule diameter. Increase of chitosan concentration may increase the size of microcapsules. This was because high chitosan viscosity facilitates large droplets, which increases the size of the resulting microcapsules. Eroǧlu et al. 28 and Lee and Rosenberg, 29 in contrast, found that the amount of crosslinking agent had no effect on the particle size.
Encapsulation efficiency
The efficiency of encapsulation is a measure of the success of encapsulation as it relates to the amount of active ingredient in the microcapsule. The efficiency of encapsulation depends on the value of the surface oil and the total oil. Table 1 shows that the lowest encapsulation efficiency value is 77.19 ± 8.7% resulting from 1% chitosan concentration and 5 ml GST volume. The highest encapsulation efficiency is 83.05 ± 2.1% from 4% chitosan concentration and 10 ml GST. Encapsulation efficiency presented in Table 1 show that increased chitosan con-centration also increased encapsulation efficiency. The influence of GST is similar to the effect of chitosan concentration. Encapsulation efficiency increased with an increase in GST volume.
There were some irregular values of encapsulation efficiency, such as in the 4% chitosan concentration and 20 ml GST, which had lower encapsulation efficiency than 4% chitosan concentration and 10 ml GST. Likewise, with 1% chitosan concentration and 20 ml GST, the encapsulation efficiency value was higher than that for 2% chitosan concentration and 20 ml GST. This was probably due to the effect of unsuitable total oil value produced from imperfect extraction so that the red ginger oleoresin was still in the microcapsules.
The effect of increasing chitosan concentration on the encapsulation efficiency was related to the viscosity of chitosan. High chitosan concentration provides higher solution viscosity which lead to greater rigidity of microcapsules due to higher crosslinking bonds between chitosan molecules. Therefore, this could inhibit the release of red ginger oleoresin as a bioactive compound and lower the rate of diffusion in a simulated medium. 30 Higher rigidity could cause solidification of microcapsules leading to reduced porosity. 17
Morphology of red ginger oleoresin microcapsules
Morphology of the red ginger oleoresin microcapsules was shown using SEM at various magnification. The sample of microcapsules were prepared from 3% chitosan concentration with 10 ml of GST. The results of SEM analysis are shown in Figure 5. The microcapsules produced in this study were perfectly spherical and had a smooth and rigid surface as shown in Figure 5(a) and (b). Figure 5(c) and (d) show burst microcapsules; Seems that there are cavities inside the microcapsules.

SEM analysis: (a) magnification 100x; (b) magnification 1500x; (c) magnification 1500x (d) magnification 2500x with burst microcapsule.
Thanoo et al. 31 suggested that a slow and uniform crosslinking of droplets particularly on the surface could produce spheres of good sphericity because instantaneous crosslinking caused the product obtained did not show good spherical geometry and surface smoothness.
TGA and differential thermal analysis (DTA)
TGA analysis was performed on microcapsule samples based on changes in chitosan concentration at 20 ml GST. The mass reduction of red ginger oleoresin microcapsules to temperature changes were divided into two stages. The first stage was evaporation of water at temperatures between 30oC and 110oC. The second stage was melting and decomposition phase of microcapsule which occurred at temperatures between 120oC and 300°C. Decomposition was assigned by several factors, such as dehydration of saccharide rings, depolymerization, and decomposition of the acetylated and deacetylated unit of the polymer32,33 The smallest weight loss of the red ginger oleoresin microcapsules at a chitosan concentration of 1% was 4.3%, while the greatest weight loss at 4% chitosan concentration was 13.4%.
TGA analysis (Figure 6) shows two endothermic peaks on all four graphs. The first endothermic peak occurs at almost the same point on all graphs, that is between 40oC and 80oC; this indicates the evaporation process of water. The second endothermic peak occurs in different temperature ranges on each graph; this shows the melting process of the microcapsules.
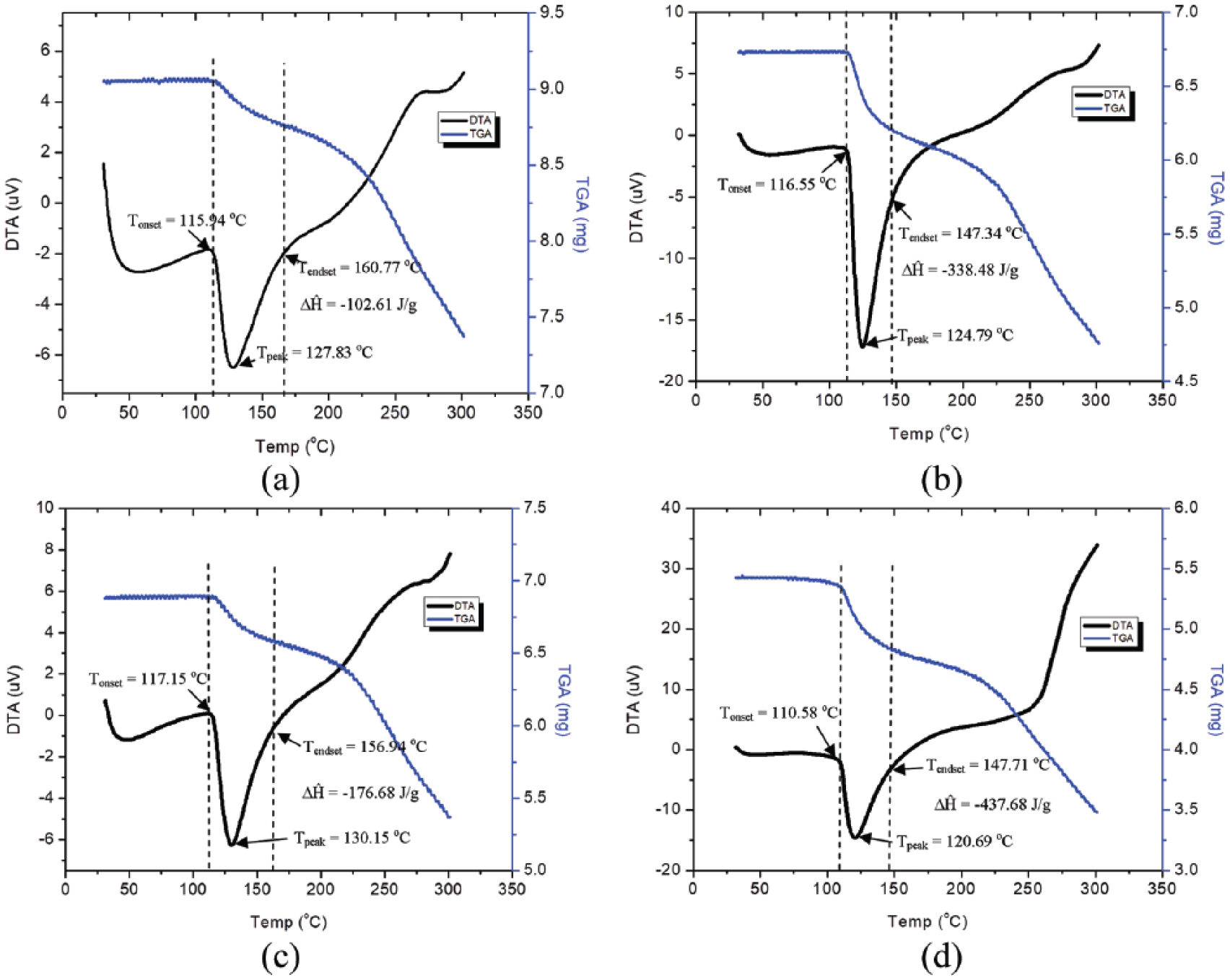
TGA and DTA analysis of 20 ml GST with several chitosan concentrations: (a) 1%; (b) 2%; (c) 3% and (d) 4%.
For Figure 6(a), the second endothermic peak occurs in Tonset = 115.94oC and Tendset = 160.77oC, for melting point at Tpeak = 127.83oC. In Figure 6(b) the melting area occurs between Tonset temperature = 116.55oC and Tendset = 147.34oC and Tpeak = 124.79oC. In Figure 6(c) the change in melting area occurs in Tonset = 117.15oC and Tendset = 156.94oC and Tpeak = 130.15oC. For Figure 6(d) the melting area occurs at Tonset = 110.58oC and Tendset = 147.71oC and Tpeak = 120.69oC. The melting process is associated with the amount of enthalpy to be used; Figure 6 shows that the largest enthalpy occurs in Figure 6(d) (4% chitosan concentration) with an enthalpy of
Conclusion
The preparation of red ginger oleoresin microcapsules by the crosslinking emulsion method on chitosan coating was successful. The resulting microcapsules is spherical and has smooth surface, and the process has a yield of up to 98.93%. The mean diameter size of the microcapsules ranged from 75.61 ± 11.8 µm to 178.65 ± 40.7 µm. The smallest diameter was obtained at 1% chitosan concentration and the largest particle size was obtained at 4% chitosan concentration. The largest encapsulation efficiency of 83.05 ± 2.1% was obtained from microcapsules with 4% chitosan concentration with 10 ml GST volume. For the effect of temperature on the characterization of microcapsules, analysis using TGA and DTA showed that microcapsules were stable at 40oC to 110oC and significant changes occurred at temperatures above 110oC to 160oC, indicated by a melting process. The largest enthalpy for melting process occurred in microcapsules with chitosan concentration 4% and 20 ml GST volume at
Footnotes
Acknowledgements
The authors are indebted to the Ministry of Research, Technology and Higher Education of the Republic of Indonesia and the Chemical Engineering Department, Universitas Gadjah Mada, for all research facilities provided.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
