Abstract
Glass ceramics are widely used to enhance the functionality of inert metallic materials typically used for hard-tissue engineering. Biofunctionality of glass ceramics can in turn be significantly boosted with the addition of trace element dopants. Herein, we synthesized a nanostructured glass ceramic and used magnesium (Mg), which is known to promote osteoblast adhesion and proliferation, for further functionalization. The nanostructured akermanite glass ceramic (Ca2MgSi2O7) was used to coat Ti6Al4V substrates by the sol-gel method. Scanning and transmission electron microscopy as well as X-ray diffraction were used to assess the structural morphology and phase composition of the coating, respectively. The micrographs showed a uniform and crack-free coating structure. Atomic force microscopy observation revealed a disordered surface roughness for coated samples. In vitro cytocompatibility tests revealed that Saos-2 cells cultured on bare samples adopted a rounded morphology, whereas cells cultured on the coated samples represented a more spread out configuration and also increased proliferation. The characterizing tests confirmed the efficiency of the synthesis method and the in vitro biocompatibility of the synthesized coating, indicating its suitability to be used for bone implants.
Introduction
Titanium and its alloys are considered the gold standard for hard-tissue implant technology thanks to their biocompatibility and stable mechanical properties.1–3 Host tissue response to inert titanium, however, is not always desirable and can often be prone to fibrous tissue encapsulation. 4 Surface treatments introducing bioceramic coatings are commonly used to functionalize the surface of metallic implants to increase their biocompatibility, 5 corrosion resistance, 6 or fixation characteristics. 4
The most common bioceramic coatings used for orthopedic implants are calcium phosphate (CaP)7–9 and Si-based glass ceramics.10–13 Hydroxyapatite (HA)-coated Ti6Al4V, for example, has been accepted as 1 of the most suitable alternative materials for damaged bone tissue. 6 Previous studies have confirmed the bioactivity of Si-based glass ceramics and their positive role in formation of an HA layer on an implant surface after soaking in simulated body fluid (SBF).14,15 Nevertheless, HA coating has been reported to exhibit poor bonding characteristics because of its notably different thermal expansion coefficient (CTE) with titanium alloys. 16 The CTE of some glass ceramics such as sphene (CaTiSiO5), 17 monticellite (CaMgSiO4), 18 and akermanite (Ca2MgSi2O7) 11 were recently reported to be closer to that of Ti6Al4V alloy. Akermanite, which is crystallized in a tetragonal lattice system and consists of magnesium oxide (MgO) tetrahedral and Si2O7 double-pyramid sheets linked together by CaO bonds, 19 is reported to exhibit not only adequate mechanical strength, but also high chemical stability and biocompatibility. 20 Ionic release of akermanite was shown to considerably enhance osteoblast cells’ adhesion and proliferation. 11 These ions are key factors that influence the process of nucleation and growth of HA, and are reported to affect the mineralization process and the bone-bonding mechanism. 21 There are also indications that Ca and silicon (Si) released from bioactive glass dissolution can promote apatite formation, and stimulate osteoblast proliferation and gene expression. 22
Considering the efficiency of ceramic coatings on biomedical implants, various methods including plasma spray, sol-gel, and electrophoretic deposition (EPD) have been suggested for their deposition. 17 Herein, we used the sol-gel method to coat Ti6Al4V substrates with a nanostructured akermanite (Ca2MgSi2O7). The sol-gel method was used specifically for its advantages over other commonly used methods, including lower process temperature, providing the possibility of producing nanoscale particles and lower costs in addition to the flexibility of use for both coating and powder production. 23 Mg ions were incorporated into the synthesized coating, as it has been suggested that they can significantly enhance osteoblast adhesion and stimulate osteoblast proliferation.24–26 Lack of Mg is also reported to contribute to reduced bone formation and cause osteoporosis. 27 Furthermore, Mg is known to facilitate more than 300 enzymic reactions in the human body. 28 Scanning electron microscopy (SEM), transmission electron microscopy (TEM), and atomic force microscopy (AFM) methods have been used to study the morphology and structure of the obtained coating. X-ray diffraction (XRD) and energy-dispersive X-ray spectrum (EDX) were applied to examine phase composition and elements on the coating, respectively. In vitro tests were also performed to evaluate adhered cell morphology and proliferation on the coated samples.
Materials and Methods
Substrate Preparation
Disc-shaped Ti6Al4V samples with a diameter of 2 cm and thickness of 0.1 cm were sequentially polished by silicon carbide papers ranging from 100 to 600 grit sizes. The samples were subsequently ultrasonically washed in acetone for 15 minutes.
Coating Preparation and Characterization
Akermanite powder was synthesized by the sol-gel method using (C2H5O)4Si (TEOS), Mg(NO3)2.6H2O, and Ca(NO3)2.4H2O as precursors of Si, Mg, and Ca, respectively. Briefly, tetraethyl orthosilicate (TEOS) was mixed with water and nitric acid (2N HNO3, as a precipitant) and stirred for 30 minutes. Afterward, Mg(NO3)2.6H2O and Ca(NO3)2.4H2O was added to the solutions with a molar ratio of TEOS/Mg(NO3)2.6H2O/Ca(NO3)2.4H2O = 2:1:2. The mixture was then stirred at room temperature for 5 hours. For microstructure observation, dry gel of the prepared akermanite was characterized by TEM (EM208S). The distribution of akermanite particles was analyzed by EDX-mapping analysis.
The Ti6Al4V discs were fixed on a sol-gel coating (dip-coating) machine and were placed into the akermanite solution for 10 seconds. Samples were entered vertically in the solution at an approximate speed of 0.5 m per second and then with the same speed out of the solution. After being heat-treated at 900°C for 2 hours, the samples were characterized by a Philips PW3710 XRD instrument. The particle size was estimated using the Scherrer equation described in Equation 1: 29
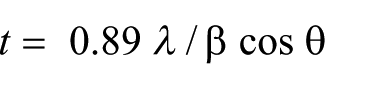
Where
A Philips XL30 SEM was used for coated surface observation as well as coating thickness evaluation. The surface morphologies of the coated and uncoated samples were studied using FlexAFM (Switzerland).
Cell Culture
Coated and uncoated samples were sterilized in an autoclave at 120°C for 30 minutes. Then, the samples were incubated in Dulbecco’s modified Eagle’s medium (DMEM) and supplemented with 10% fetal bovine serum before cell seeding. The DMEM concentration was prepared according to previously published research.
30
SaSo-2 osteoblast cells were seeded on the samples and the culture medium was changed every 2 days. The culture was kept at 37°C in a humidified atmosphere of 95% air and 5% CO2. A total of 5 × 104 cells passaged at these experiments. The culture media was changed every 2 days during the proliferation assay. After 3 days, the adhered cells were fixed with 2.5% glutaraldehyde buffer for 30 minutes at 4ºC. A dehydration process was performed using a graded ethanol solution of 30%, 50%, 70%, 95%, and 100%. Adhered cells’ morphology was observed by SEM (VEGA TESCAN) and optical microscopy (OM). 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay was used to assess the cells’ viability on coated, uncoated, and control samples after 1, 3, and 7 days in culture. Ultra-high–molecular weight polyethylene (culture plate) was used as negative control. The discs were kept under the same culture conditions with 720 μL of fresh culture medium and 80 μL of MTT solution. Subsequently, dimethyl sulfoxide was added to completely dissolve the purple formazan. The cell viability percentage was estimated by spectrophotometry (enzyme-linked immunosorbent assay reader) at a wavelength of 570 nm. A
Results and Discussion
The XRD pattern of the coating (Figure 1) confirms the presence of Ca2MgSi2O7 peaks (according to Joint Committee on Powder Diffraction Standards 00-076-0841). Akermanite is known to crystallize at temperatures above 700°C and to develop the crystalline phase by heat treatment at 800°C. 31 On the other hand, the Ti6Al4V surface is seriously damaged above 900°C. 32 Therefore, a heat treatment was applied at 900°C with the aim of increasing the crystallinity, reducing the residual stresses, and enhancing ductility without creating tension between the coating and the underlying titanium substrate and not damaging the coating and the substrate. 33 Peaks observed in Figure 1 can be considered as an indication of the high crystallinity of the coating. Few impurities of merwinite (Ca3MgSi2O8) are present in the XRD pattern. The rutile peaks detected by XRD are also due to the limited thickness of the coating layer. The akermanite grain size was measured to be about 32 ± 2.5 nm according to the Scherrer equation.
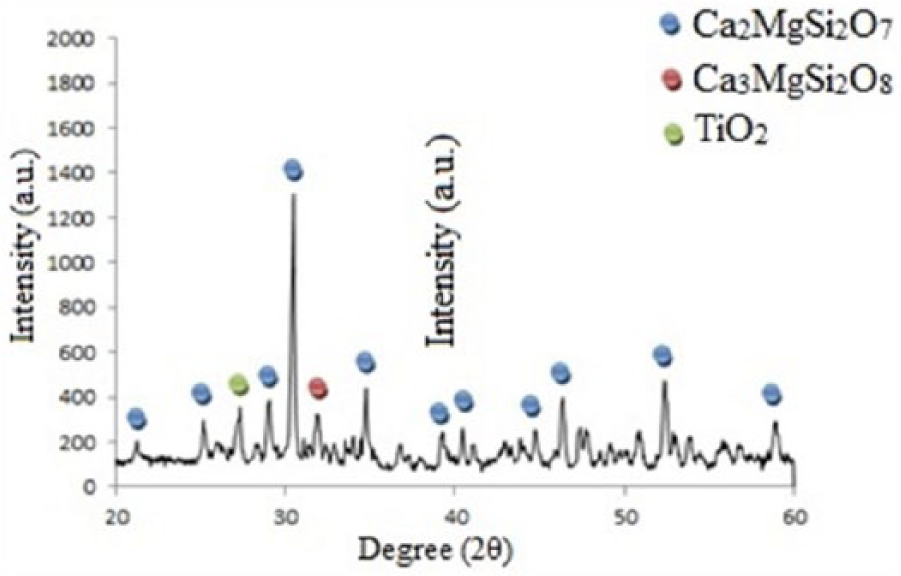
XRD Pattern of the Synthesized Coating at 900°C
TEM observation of the synthesized powder (Figure 2A) demonstrated nanosize powder particles with an almost oval shape and a smooth surface morphology. Figure 2B shows an SEM a top-surface micrograph of the synthesized coating. The micrograph indicates that the obtained coating has a disordered surface morphology with the presence of some pores. The cross-section observation in Figure 2B displays a uniform and crack-free structure that is deposited uniformly all over the substrate. The coating thickness is quite regular and is measured to be about 5-10 μm with a defect-free microstructure. According to SEM observations, the created coating shows a disordered topography that is also confirmed by AFM analysis. EDX mapping analysis confirmed the presence of Ca, Si, Mg, Ti, and Al elements on the surface of the coated sample (Figure 3). The EDX cartography of the akermanite coating showed that all akermanite elements have been preserved in deposited nanostructures and are uniformly spread all over the sample’s surface. The presence of the elements on the surface is expected to induce coating bioactivity, which can significantly promote the formation of HA. 27

A, Transmission Electron Microscopy Micrograph of the Synthesized Powder; B, Scanning Electron Microscopy (SEM) Micrographs of the Coating Top Surface; and C, SEM Cross-Section Observation of the Coating.

Energy-Dispersive X-Ray Spectrum (Mapping Results of Chemical Elements’ Distribution, A, Calcium (Ca); B, Silicon (Si); C, MAGNESIUM (Mg); D, Titanium (Ti); and E, Aluminum (Al) on Surface Samples.
AFM images of the coated and uncoated surfaces, shown in Figure 4, exhibit a significantly higher surface roughness for the coated samples compared to the bare material (radium = 1.5 μm vs 0.1 μm). The coated surface represents random surface topography. This hierarchical surface topography is expected to have positive effects on cellular activity. 34 The positive effects of micrometer-sized roughness of biomaterials on osteoblast behaviors have been well documented. 35 Disordered topography, as is the case for most biological tissue and almost all clinically accepted biomaterials, is reported to highly affect the morphology of osteoblast cells. 36
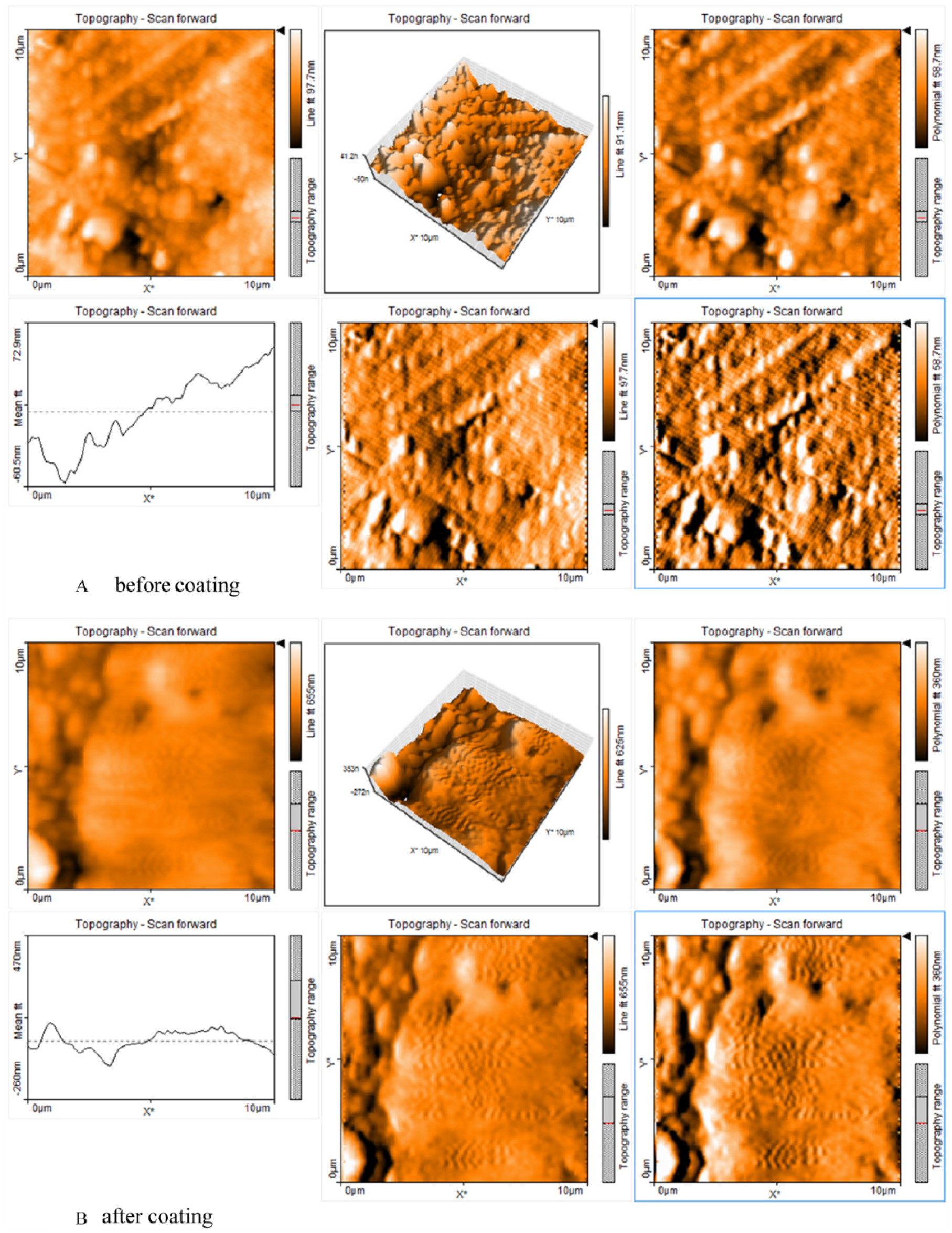
Atomic Force Microscopy Images A, Before Coating, and B, After Coating.
Figures 5 and 6 shows the OM and SEM micrographs representing the morphology of Saos-2 cells cultured on coated and uncoated samples after 3 days in culture. The cells cultured on the coated surface represent a rather flat morphology whereas the cultures on uncoated samples represent a more rounded configuration. SEM images under higher magnification confirm the higher tendency of the cells to spread in a more flattened configuration on the coated samples compared to the uncoated ones.
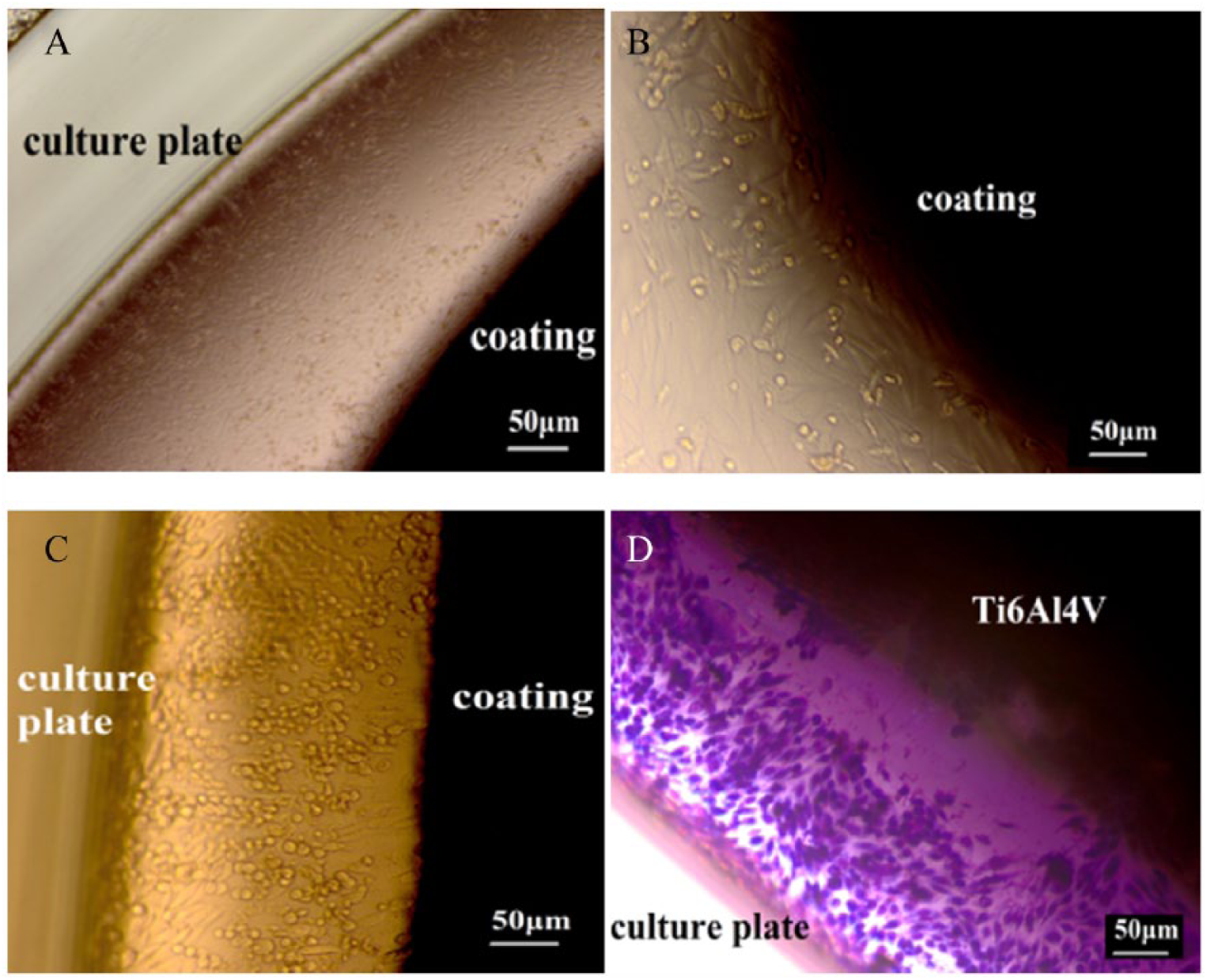
Optical Microscopy Images of Saos-2 Cells Cultured on A, B, Coated and C, D, Uncoated Discs After 3 Days.

Scanning Electron Microscopy Images of Saos-2 Cells Cultured on Ti-6Al-4V A, B with Coating, and Ti-6Al-4V C, D, without Coating After 3 Days in Culture.
Calcified surface of the samples is shown in Figure 6. Calcification is the process by which mineral Ca builds up on biomaterials, and it is proved to have an important role in the bone bonding flow of bioglass and glass-ceramic bone implants. 24 This process depends on various factors including absorption of Ca-binding proteins on implant surface and cellular response of the host tissue to the biomaterials.37,38 Also, the effect of Mg in this process is well documented.38,39 The SEM images confirm good adhesion and anchorage of the cells to the surface coatings that can in turn confirm the cytocompatibility of the synthesized coatings.
Figure 7 shows the cell proliferation results of the MTT assay on coated and uncoated samples as well as control samples up to 1, 3, and 7 days in culture. The cultured Saos-2 osteoblast showed a significantly (
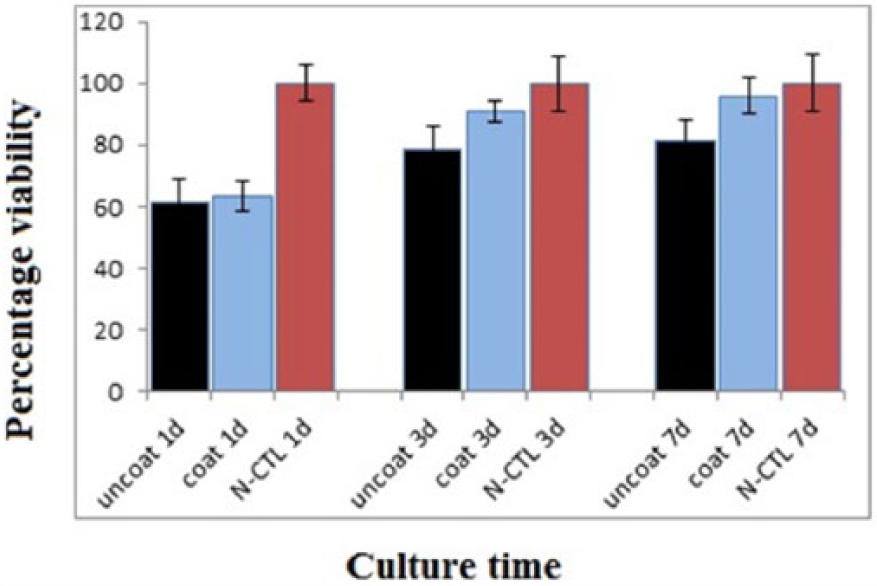
MTT Assay of the Samples After Different Culturing Times
Conclusions
The sol-gel method was used to synthesize a nanocrystallized Ca2MgSi2O7 coating on Ti6Al4V substrates. The obtained results indicate that akermanite phase can be synthesized from a gel precursor by calcining at 900°C. XRD, EDX, and microscopical analysis confirmed that the coating presented a defect-free and homogeneous nanostructure. In vitro assays with microscopical observation and cell-proliferation tests revealed that the obtained samples’ coatings stimulated cell spreading compared to uncoated samples. The coating samples were found to effectively enhance cell-proliferation rate. The results reveal the adeptness of the synthesized coating in promoting cell interaction with bone implants, promising an improved tissue response.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
