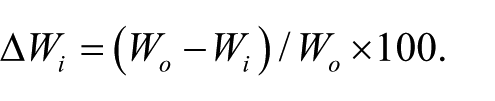Abstract
Introduction:
Injectable scaffolds are emerging as a promising strategy in the field of myocardial tissue engineering. Among injectable scaffolds, microparticles have been poorly investigated. The goal of this study was the development of novel gelatin/gellan microparticles that could be used as an injectable scaffold to repair the infarcted myocardium. In particular, the effect of particle size on cardiac progenitor cell response was investigated.
Methods:
Particles were produced by a water-in-oil emulsion method. Phosphatidylcholine was used as a surfactant. Particles with different diameter ranges (125–300 µm and 350–450 µm) were fabricated using two different surfactant concentrations. Morphological, physicochemical, and functional characterizations were carried out. Cardiac progenitor cell adhesion and growth on microparticles were tested both in static and dynamic suspension culture conditions.
Results:
Morphological analysis of the produced particles showed a spherical shape and porous surface. The hydrophilicity of particle matrix and the presence of intermolecular interactions between gellan and gelatin were pointed out by the physicochemical characterization. A weight loss of 75 ± 5 % after 90 days of hydrolytic degradation was observed. Injectability through a narrow needle (26 G) and persistence of the microparticles at the injection site were preliminarily verified by ex vivo test. In vitro cell culture tests showed a preservation of rat cardiac progenitor biologic properties and indicated a preferential cell adherence to microparticles with a smaller size.
Conclusion:
Overall, the obtained results indicate that the produced gelatin/gellan microparticles could be potentially employed as injectable scaffolds for myocardial regeneration.
Introduction
In the last two decades, cardiac tissue engineering (CTE) has emerged as a potential therapy for the treatment of the infarcted myocardium. 1 The use of three-dimensional pre-formed scaffolds in the form of hydrogels, sponges, films, fibers, and microfabricated meshes has been largely investigated. 2 More recently, injectable scaffolds have raised great interest for myocardial healing in vivo, because they allow implantation by minimally invasive procedures. 3 Thermoreversible polymers, self-assembling peptides, and in situ cross-linked polymers have been investigated as injectable systems. Injectable scaffolds can also be obtained in the form of microparticles, as already proposed in the orthopedic 4 and adipose 5 tissue engineering fields. Microparticles can offer several advantages over other injectable systems, including a high surface to volume ratio and the possibility of cell seeding before transplantation.
Most microparticles used for cell culture are spherical, with a diameter generally ranging from 50 μm to 700 μm.4–7 Reports from literature suggest the size of particles used as a culture substrate and cell transplantation vehicle has a significant effect on cell response. Qu et al. prepared silk fibroin microspheres with diameter in the range of 200 µm to 700 µm, and demonstrated that fibroblast adhesion and growth were promoted on smaller microparticles (200–300 µm). 6 In another study, synthetic microspheres with a size ranging from 180 µm to 425 µm were developed and it was reported that there was a preferential adhesion, proliferation and spreading of human intervertebral disc cells on microparticles with diameter below 250 µm. 7
The use of injectable microparticles has been poorly investigated in the CTE field.8–10 In particular, gelatin (Gel) microspheres were tested as angiogenic factor carriers in the treatment of ischemic myocardium.8,9 Additionally, the effect on myocardial repair of mesenchymal stem cells encapsulated in alginate microspheres was assessed. 10
The present study was addressed to develop novel injectable microparticles based on a blend of Gel and gellan (GE) for the application as a cell carrier in CTE.
GE is a biocompatible and biodegradable polysaccharide, largely investigated in the pharmaceutical and tissue engineering sectors.11,12 However, its high hydrophilicity represents a limitation for the attachment of anchorage-dependent cells. Therefore, GE modification with peptides or proteins has been tested in literature to improve cell adhesion. 13 In this study, the strategy proposed to promote the cell and microparticle interaction was blending of GE with a protein component (Gel). To prepare Gel/GE microparticles, we selected a water-in-oil emulsion method. Phosphatidylcholine (PC) was used as a surfactant.
PC is a phospholipid and is the major component of biological membranes. Its use as a surfactant during the production of microparticles for biomedical applications has been reported in literature 14 and seems to be particularly indicated, thanks to its biocompatibility.
The procedure to obtain Gel/GE microparticles was defined in this work, testing different preparation parameters.
In particular, the specific focus of this work was to assess the effect of microparticle diameter on cardiac progenitor cell behavior. Two microparticle populations of a different size (within the dimensional range of injectable microparticles commonly investigated in literature, as previously discussed) were produced by varying the surfactant concentration. Morphological, physicochemical, and functional characterizations were carried out. Cardiac progenitor cell adhesion and growth on microparticles were tested in both static and dynamic suspension culture conditions.
Materials and methods
Materials
Gel (type B from bovine skin), GE, and PC were supplied by Sigma Aldrich (St. Louis, MO, USA).
Dichloromethane, isopropanol and calcium chloride were purchased by Carlo Erba Reagenti (Italy).
All the other reagents were analytical grade, commercially available, and used as received.
Microparticle preparation
Microparticles based on a Gel/GE blend were prepared by a water-in oil emulsion procedure. PC was used as a surfactant. A solution of Gel (2% w/v) in bi-distilled water was mixed in appropriate volume with a solution of GE (2% w/v) in bi-distilled water to obtain a Gel/GE blend, with 30/70 weight ratio. The Gel/GE blend was used as the water phase; PC, dissolved in dichloromethane (1% w/v concentration), was used as the oil phase.
The water phase was added drop by drop to the oil phase, under constant stirring at 40°C. The emulsion was obtained as follows: 30 mins at 40°C under stirring at 800 rpm; 1 hr at 25°C under stirring at 800 rpm; and 2 hr at 0°C under stirring at 500 rpm. The mixture was then decanted in a funnel. The obtained particles were separated from the oil phase and added for 15 mins to a 2% w/v CaCl2 solution in bi-distilled water for GE cross-linking. Repeated washing with isopropanol was then performed to remove residual PC. Finally, the particles collected on filter paper were dried in a fume hood overnight.
An alternative preparation procedure was tested to reduce particle size, by increasing the surfactant concentration up to 1.5% w/v.
Morphologic analysis
The morphology of the obtained particles was investigated using a scanning electron microscope (SEM JSM 5600, Jeol Ltd). Before analysis, particles were deposited on metal stubs and sputtered with gold.
Particles were also observed under an optical microscope (integrated within the Spotlight 400 FTIR Imaging System, Perkin Elmer), to evaluate their average dry diameter, size distribution, and swelling percentage. Samples of at least 100 dry particles from both preparations (high and low surfactant concentration) were used.15,16 Diameters were measured starting from optical images and results were reported as average value ± standard deviation. The resulting distributions of microparticle sizes were plotted as histograms. After the determination of the average dry diameters, a swelling test was carried out. Particles were immersed in phosphate buffer solution (pH = 7.4) for 15 mins. Then they were observed again under the optical microscope and the average diameter of swollen particles was determined. The percentage of swelling was calculated as follows: (Ds-Dd)/Dd×100, where Ds is the average diameter of swollen particles and Dd is the average diameter of dry particles.
Physicochemical characterization
Infrared spectra of the samples were obtained with a Fourier transformed infrared spectrometer (Spectrum One, Perkin Elmer). The analysis was performed in attenuated total reflectance (ATR) mode, with a resolution of 4 cm-1.
Thermogravimetric analysis (TGA) was performed using a thermal analyzer (Perkin Elmer TGA-6). Samples of circa 10 mg, in six replicates, were heated from 30 to 800°C at a rate of 10°C per min, under nitrogen flow.
Degradation tests
Degradation tests were performed in agreement with the ISO norm 10993-13 “Biological evaluation of medical devices. Part 13: Identification and quantification of degradation products from polymeric medical devices”. 17
Tests were performed on six replicates. The in vitro hydrolytic degradation properties were evaluated in phosphate buffered saline (PBS, pH = 7.4). Microparticle weight loss during degradation was determined by measuring changes in dry weight after incubation for fixed time periods.
Samples were dried at room temperature to a constant mass. The starting dry weight, W0, was determined for each sample using a precision balance. Samples were then fully immersed in the degradation solution inside a closed container and maintained in a bath at 37±1°C under stirring. At appointed times, ti, samples were removed from the degradation solution, rinsed in bi-distilled water, dried to a constant mass, and weighed. The dry weight at time ti of degradation, Wi, was determined for each sample. Percentage weight loss was evaluated according to the following equation:
In addition to mass loss determination, degraded samples were characterized by SEM and infrared analysis.
Injectability evaluation
To evaluate microparticle injectability, their viscous behavior as a function of temperature was estimated using a rotational rheometer (Kinexus series, Malvern Instruments Ltd, Worcestershire, UK), in the following experimental conditions: fixed shear stress value equal to 5·10-1 Pa, 1 Hz of constant frequency, heating rate equal to 3°C per min, in the range 15–45°C. Values of viscosity at room temperature (25°C) and physiological temperature (37°C) were recorded. Six replicates were tested and results are reported as the average value ± standard deviation.
Ex-vivo injectability tests were also performed to evaluate both the injectability and the adherence of microparticles to the myocardial tissue. A known amount of microparticles, stained with methylene blue (Carlo Erba Reagenti, Italy), was loaded into an insulin syringe with a 26 G needle and PBS was added. After 15 min, once microparticle hydration was reached, the solution was injected into the myocardium of a porcine heart, collected from a local slaughterhouse. The heart was then dissected and the microparticle distribution at the implantation site was evaluated by infrared analysis.
In vitro studies
Rat cardiac progenitor cell isolation and culture
Cardiac progenitor cells (CPCs) were isolated from green fluorescence positive (GFPpos) rats 18 and cultured as previously described.19,20
CPCs at passage 3 and 4 (P3-P4) were employed for the experiments in this study.
In vitro studies in static and dynamic suspension conditions were performed employing Gel/GE microparticles with different diameters, obtained by high or low surfactant concentration (as detailed in the previous paragraph).
Gel/GE microparticles were sterilized by ultraviolet light irradiation (254 nm wavelength) for 30 mins and then suspended in culture medium for 15 min to obtain microparticle swelling, before starting cell culture tests.
Cell culture tests in static conditions
CPCs isolated from GFPpos rat hearts were resuspended in culture medium together with Gel/GE microparticles and seeded at the density of 1.5 x 105 cells per ml.
Cell and microparticle adhesion in static conditions were evaluated after 2 days under an inverted fluorescence microscope (Leica DMI6000B).
Cell culture tests in dynamic suspension conditions
In vitro cell culture tests in dynamic suspension conditions were performed within an impeller-free bioreactor. The bioreactor, part of a peristaltic pump-based closed-loop circuit, guarantees cell and microparticle low-shear dynamic suspension within the culture vessel at a laminar mixing flow regime, without using rotational components. A more detailed description of the bioreactor has been given previously. 21
CPCs were seeded on microparticles at a density of 105 cells per 100 µl in a 96-well plate to promote cell and microparticle adhesion in static conditions. To assess the suspension efficiency, analysis by fluorescence microscopy was carried out immediately after cell and microparticle mixing.
Bioreactor components were sterilized in autoclave at 121°C for 60 min and assembled under biological hood.
Cell-seeded microparticles (4.0–6.0 x 105) were injected in the bioreactor vessel and the culture was carried out for 1 day with complete growth medium in dynamic suspension.
After 1 day, cells were harvested from the bioreactor, re-suspended in fresh medium, seeded in a 96-well plate, and analyzed by optical and fluorescence microscopy. The analysis was repeated after 5 days of static culture to verify cell adhesion and survival (Figure S1).
Controls were represented by cells cultured in dynamic suspension conditions without microparticles (GFPpos CPCs CTRL). All experiments were performed in triplicate.
Quantitative analysis of cell-microparticle adhesion
Photomicrographs of GFPpos CPCs collected from the culture vessel or cultured in static conditions were analyzed to quantify cell adhesion and distribution on Gel/GE microparticles. The fractional area occupied by GFPpos CPCs adhering to Gel/GE microparticles was evaluated by computing green fluorescent signals and their intensity as an integrated optical density, using Image Pro Plus 4.0 (Media Cybernetics, Inc., USA), a dedicated software for image analysis. 22
Data management and statistics
SPSS statistical software was employed (Chicago, IL, USA). Statistical variables were SEM, paired Student t-test, one-way analysis of variance (post-hoc analyses: Holm-Sidak or Tukey test, when appropriate). Statistical significance was set at p<0.05, p<0.01, and p<0.001.
Results and discussion
Morphological analysis
SEM micrographs of the produced microparticles are shown in Figure 1. Particles obtained at a low surfactant concentration (Figures 1a–1b) appear well separated and show an almost spherical shape, a porous surface, and a diameter in the 350–450 µm range.
SEM micrographs of particles obtained at low (a–b) and high (c) surfactant concentration. Scale bar is reported for each micrograph. (d–e) Diameter histograms for microparticles obtained at low and high surfactant concentration, respectively. For both microparticle populations, the size range was divided in bins of 10 µm each.
The increase in surfactant concentration does not significantly modify particle morphology (Figure 1c) in terms of shape and surface porosity. A reduction of particles size can be observed, with diameters in the 125–300 µm range.
Average dry diameter, size distribution (Figures 1d–1e) and swelling percentage were evaluated by particle observation under an optical microscope, in dry and swollen conditions.
The particles produced at a low surfactant concentration (Figure 1d) were polydispersed, whereas in the sample produced at a high surfactant concentration (Figure 1e), about 40% of the particles showed a diameter between 140–160 µm. A reduction of particle polydispersity with the increase of surfactant concentration was already observed in literature. 23
With GE being a polyelectrolyte, its swelling properties are dependent on pH, therefore the particle swelling degree was evaluated at controlled pH 7.4, simulating the biological environment. Images of swollen particles obtained by optical microscopy showed that, when immersed in aqueous medium, the particles acquire a spherical shape. Particles obtained at a low PC concentration had an average dry diameter of 391 ± 64 µm and showed a swelling degree of 55%. For the particles obtained at a high PC concentration, an average diameter of 186 ± 36 µm and a swelling degree of 58% were measured. The results of the swelling test demonstrated the hydrophilicity of the particle matrix.
Physicochemical characterization
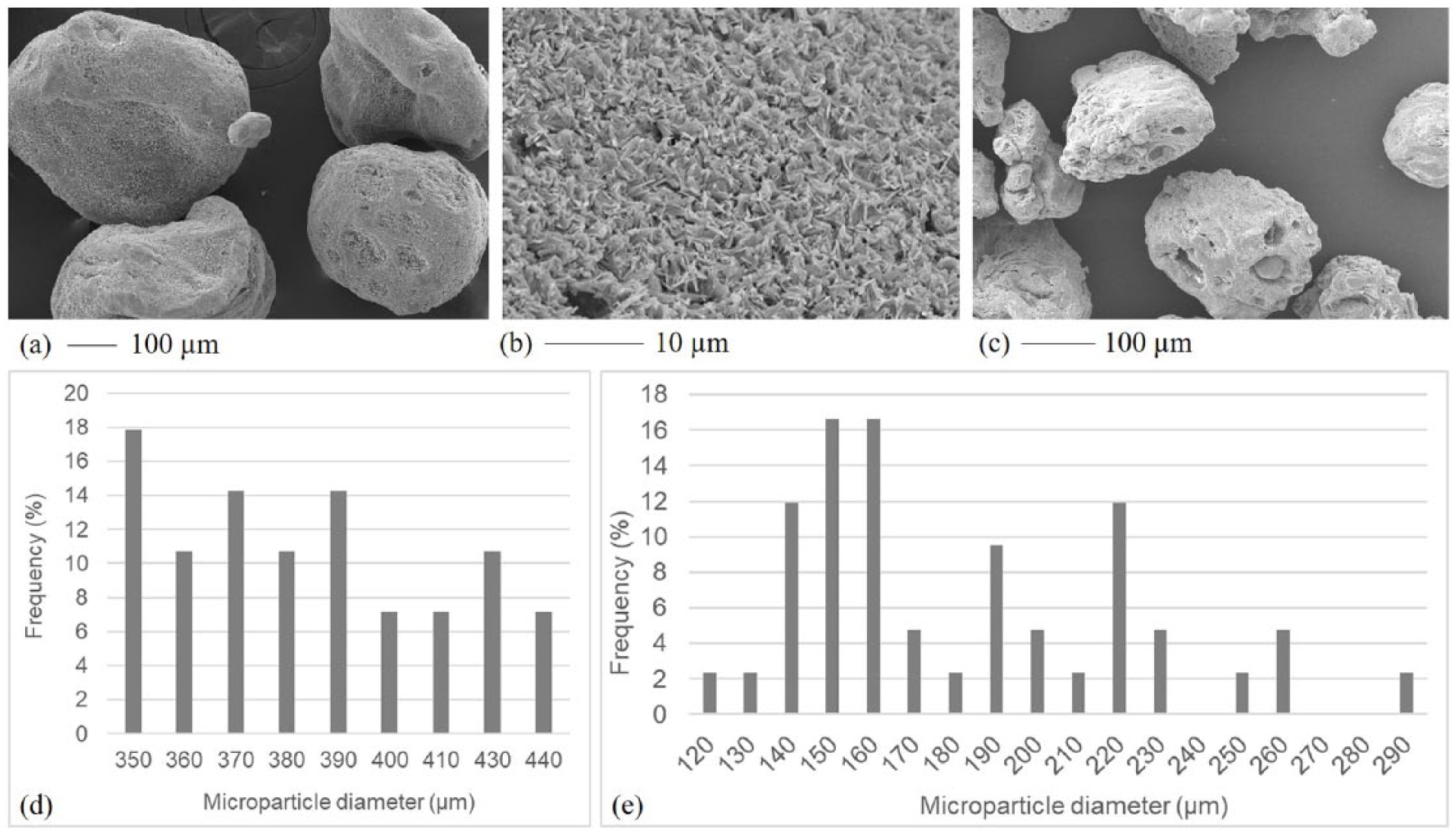
Infrared analysis and TGA were performed on the produced microparticles to investigate chemical composition and thermal degradation. Similar results were obtained for both groups of microparticles, independently of their size range, and they were compared with those related to pure polymers. Modifications in absorption bands and thermodegradation behavior of a polymer blend with respect to pure components can be considered experimental evidence of intermolecular interactions occurring between the functional groups of the two polymers. 24 Infrared spectra were acquired for the produced microparticles and for pure Gel, GE, and PC (Figure 2).
Infrared spectra of gelatin (Gel), gellan (GE), phosphatidylcholine (PC), and microparticles. Typical absorption bands of GE, Gel, and PC are indicated by arrows, both in the spectra of pure components and in the microparticles spectrum. The amide II of Gel, present in the microparticle spectrum as a shoulder, is indicated by a circle.
GE spectrum was characterized by the following absorption peaks: 3382 cm-1 (O–H stretching), 2926 cm-1 (C-H stretching) and 1046 cm-1 (piranosidic ring). The Gel spectrum showed two typical peaks: 1550 cm-1 (N-H bending and C-N stretching, amide II) and 1635 cm-1 (C=O stretching, amide I).
The microparticle spectrum showed the presence of all the typical absorption peaks of both protein and polysaccharide components. In particular, the amide II absorption peak appeared as a shoulder. In addition, a small displacement of the peak due to Amide I (from 1635 in pure Gel to 1629 cm-1) was observed, suggesting the presence of hydrogen bond interactions between GE and Gel. 24 A similar shift of this peak was observed by Pranoto et al. 25
The absence of the typical PC absorption peak (at 1735 cm-1, due to the bond of the choline head) in the microparticle spectrum demonstrated complete surfactant removal by washing with isopropanol. Moreover, no absorption band attributable to the solvents used during the preparation procedure was observed.
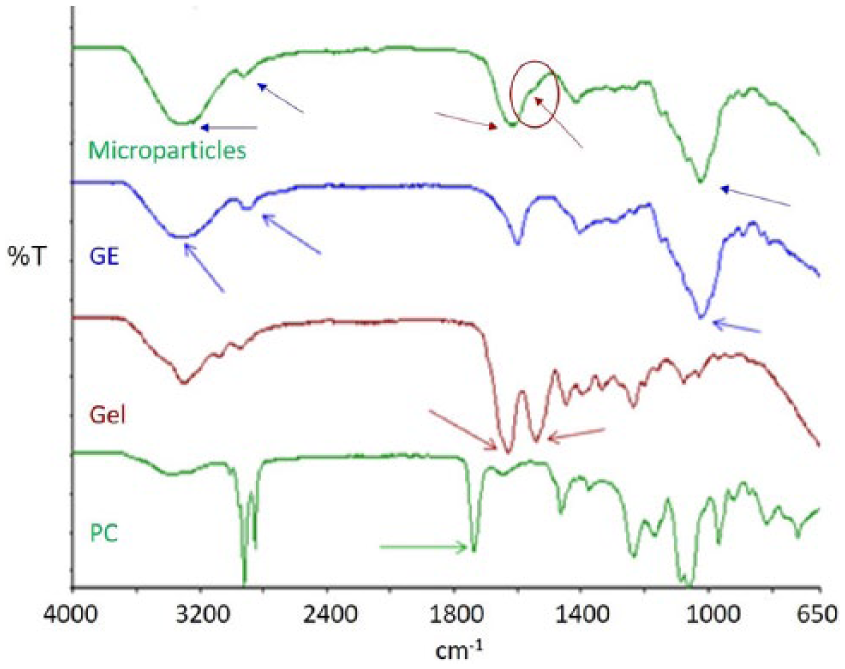
TGA thermograms were acquired for Gel/GE microparticles and for pure Gel and pure GE. The results are collected in Table 1.
Results of thermogravimetric analysis performed on gelatin (Gel), gellan (GE), and Gel/GE microparticles: temperature range and weight loss percentage for free water loss and polymer degradation events. Weight loss percentages are reported as the average value ± SD.
All the thermograms were characterized by two events of weight loss: the first due to the loss of free water (between 30 and 150°C) and the second related to polymer degradation. Differences in the percentage of weight loss due to the loss of water were not significant for the three samples (p>0.05, as calculated by Student’s t-test), demonstrating a similar capability to absorb free water. For polymer degradation, the range of temperatures registered for the microparticles resulted in an intermediate between those of the pure components. Moreover, the percentage of weight loss due to the thermal degradation of Gel/GE microparticles was lower than the theoretical one, which can be calculated for a blend of Gel and GE, with 30/70 weight ratio (25.5%). These results confirmed the presence of interactions occurring between the two natural polymers. 24
Additionally, TGA confirmed the absence of thermal events related to solvent evaporation.
Overall, the presence of interactions between GE and Gel, pointed out by the physicochemical characterization, could enhance the affinity of the resulting material towards anchorage-dependent cells, as they mimic those occurring between proteins and polysaccharides in the extracellular matrix of natural tissues. 26
Degradation test
A degradation test was performed maintaining a weighted amount of microparticles in PBS at 37°C for up to 90 days. The test was performed on the particles with smaller dimensions (125–300 µm) that appeared the most promising on the basis of the biological characterization. At appointed time instants, the percentage of weight loss was measured. The weight loss kinetic is reported in Figure 3a.
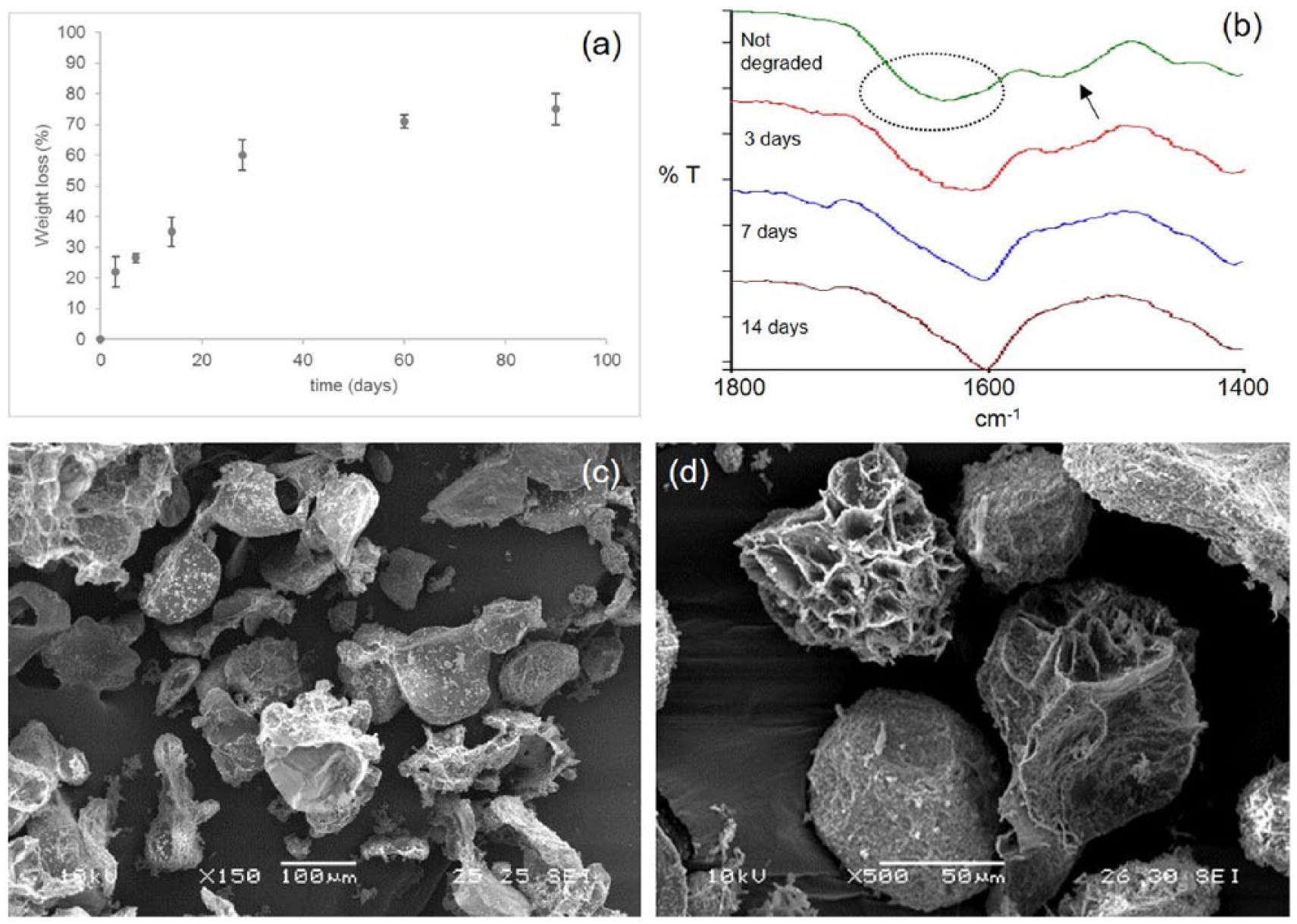
(a) Percentage of weight loss during microparticle (125–300 µm) hydrolytic degradation; (b) infrared spectra of microparticles acquired at different times of hydrolytic degradation (3, 7, and 14 days), compared with the reference spectrum of not-degraded microparticles. The infrared spectrum of not-degraded microparticles was characterized by an enlarged absorption band at 1630 cm-1, due to the overlapping of amide I band of Gel and the band at 1603 cm-1 of GE (indicated by the dotted circle), and an absorption band at 1550 cm-1 due to amide II of Gel (indicated by the arrow). (c–d) SEM micrographs of microparticles after 90 days of hydrolytic degradation, acquired in different regions of the samples, at different magnifications.
The degradation kinetic was characterized by a first quick phase (during the first 3 days), with a percentage of weight loss of 22 ± 4%. Then, the degradation occurred more slowly, reaching 75 ± 5% of weight loss after 90 days.
Our hypothesis to explain the degradation kinetic is that the protein component, which is not cross-linked during the preparation procedure, is quickly released during the first two weeks (35 ± 4% of weight loss at 14 days) then the dissolution of the polysaccharide occurs.
To demonstrate our hypothesis, infrared spectra of degraded microparticles were acquired at different time instants and compared with a reference spectrum of not degraded microparticles (Figure 3b). The infrared spectrum of not-degraded microparticles showed an enlarged absorption band at 1630 cm-1, due to the overlapping of amide I band of Gel and the band at 1603 cm-1 of GE, and an absorption band at 1550 cm-1 due to the amide II of Gel. During the hydrolytic degradation, a progressive reduction of the absorption band at 1550 cm-1 was observed, up to its disappearance after 14 days. Moreover, the enlarged absorption band moved towards 1603 cm-1 (due to GE). These results confirmed the protein was completely released during the first two weeks.
SEM micrographs collected at the end of the degradation test showed marked microparticle disintegration, coupled with an increase of surface porosity (Figures 3c–d).
Overall, the obtained degradation profile should be compatible with the kinetic of new tissue formation, offering sufficient support to cell growth. 27
Injectability evaluation
The first property investigated to evaluate the microparticle injectability was viscosity. On one side, the viscosity value should be maintained in a moderate range, to avoid the need to apply excessive pressure to inject microparticles into the myocardium. In particular, according to the existing literature, 28 microparticle viscosity at room temperature should be lower than 150–200 cP, to ensure safe injection through catheters. On the other side, after injection microparticle viscosity should be sufficiently high to prevent the leakage from the myocardial tissue to the ventricular chamber.
The measured viscosity values of 69 ± 7 cP at 25°C and 239 ± 19 cP at 37°C confirm that microparticles (1) can be injected in the myocardium through a syringe needle at temperature around 25°C, and (2) their increased viscosity at body temperature of 37°C supports the formation of a gel inside the cardiac tissue. 28
Microparticle injectability was further investigated through ex vivo tests, by delivering the microparticles in the myocardial tissue of a porcine heart, using an insulin syringe. After dissection of the porcine heart, injected microparticles appeared well adhered and localized at the injection site (Figure 4a). Chemical imaging analysis in µATR mode, performed on a small tissue sample collected close to the site of injection, confirms this observation. In the optical image, microparticles were well distinguishable from the myocardial tissue (Figure 4a). The chemical map was then acquired at the edge of the injection site (Figure 4b) and the infrared spectra acquired in different areas of the chemical map were compared with a reference spectrum of the myocardial tissue. Collected spectra were markedly comparable with that of the myocardium and did not show the typical absorption peaks of Gel and GE (Figure 4c), suggesting the absence of microparticle leakage phenomena during injection. The correlation map between the chemical map and the reference spectrum of the myocardial tissue showed values close to 1 on all the analyzed areas (Figure 4d), further confirming the absence of microparticles in the region bordering the injection site.
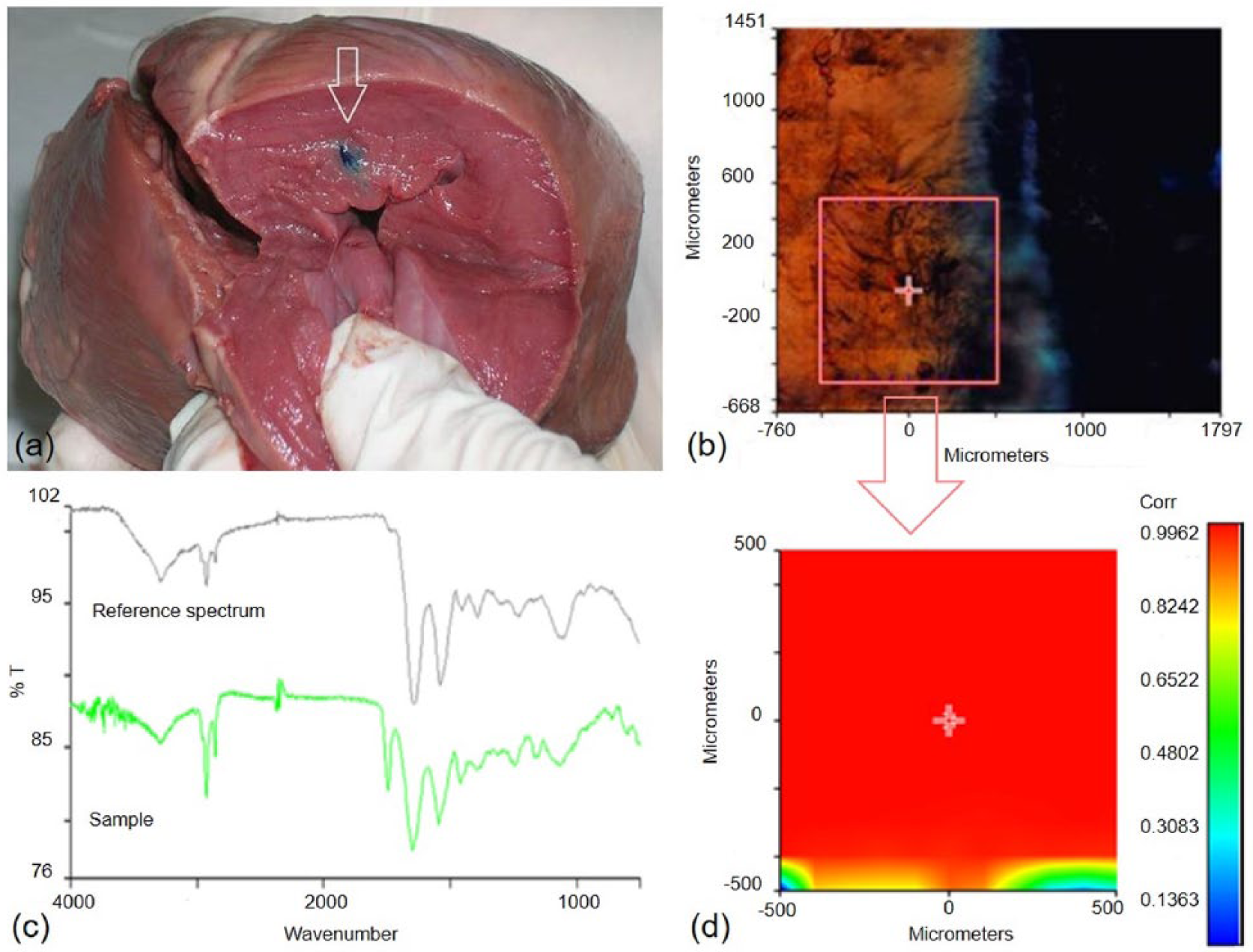
(a) Microparticles colored with methylene blue, injected in a porcine heart; (b) chemical map acquired on a small section of tissue, collected close to the site of injection; (c) comparison between the medium spectrum of the sample and a reference spectrum of porcine myocardial tissue; (d) correlation map between the selected area of the chemical map and the reference spectrum of porcine myocardial tissue.
In vitro studies
Cell culture tests in static conditions
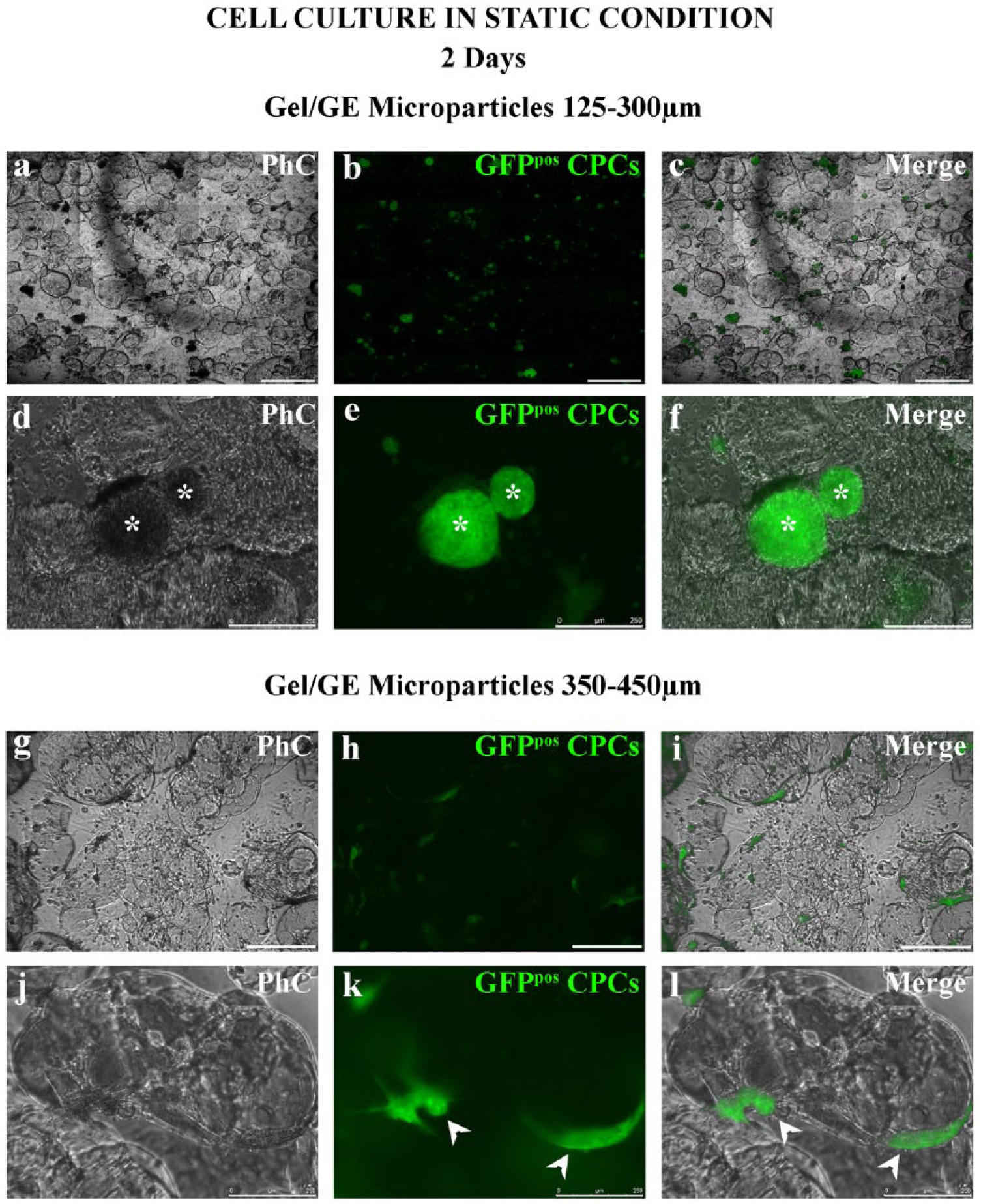
Cell culture tests in static condition were performed using Gel/GE microparticles, averaging 125–300 µm or 350–450 µm in diameter. After 2 days of static culture, GFPpos CPCs adhered to the 125–300 µm microparticles mainly as clusters (Figure 5a–f), and to the 350–450 µm microparticles, mainly as single cells, spreading their cytoplasm on the microparticles (Figure 5g–l).
Cell culture tests in dynamic suspension conditions
Gel/GE microparticles (diameter 125–300 µm)
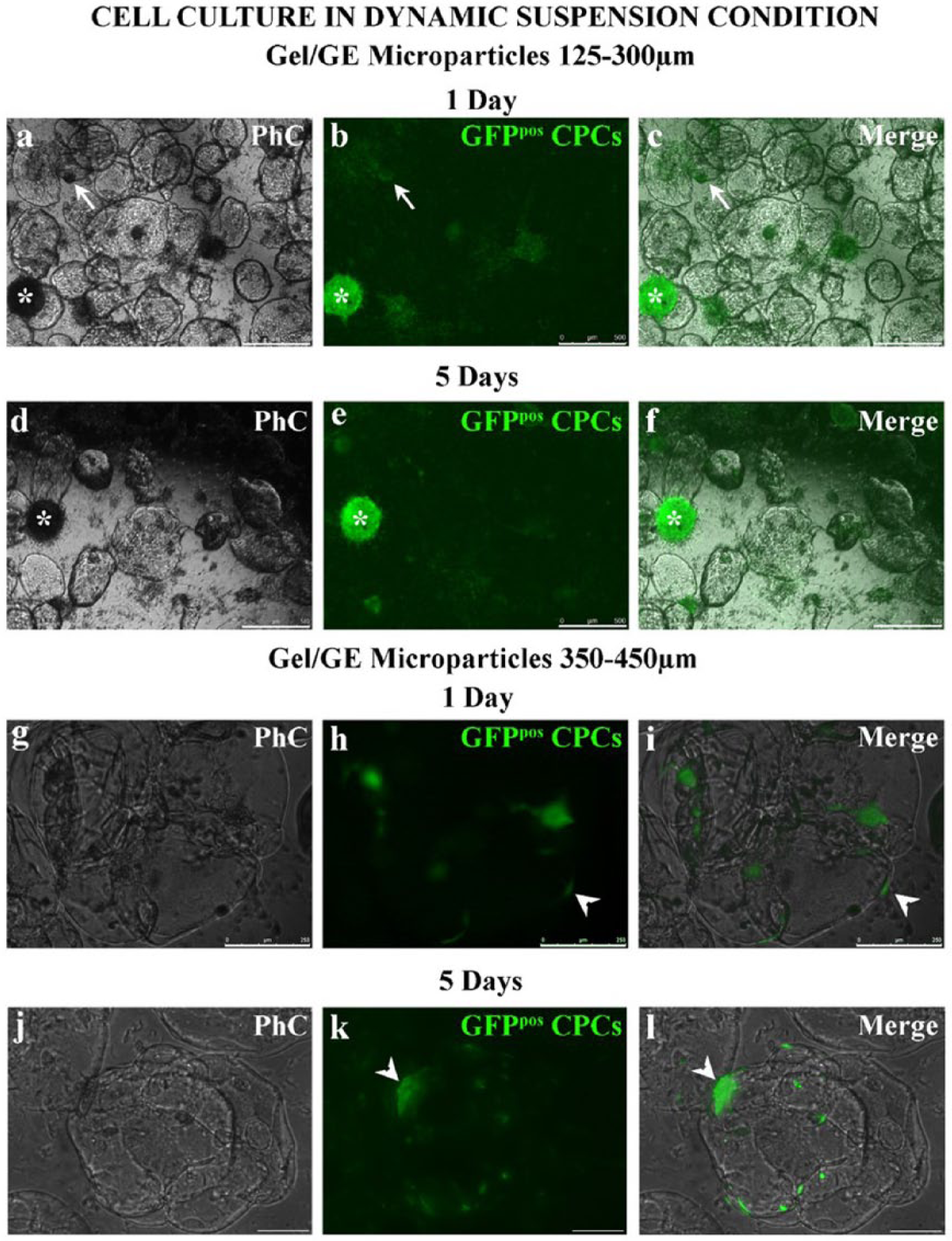
GFPpos CPCs preferentially attached to the 125–300 µm microparticles, entirely covering their surface (Figure 6a–c), while they surrounded the edge of the 350–450 µm microparticles (Figure 6g–i). After 1 day of dynamic suspension culture, GFPpos CPCs not adhering to microparticles formed small circular clusters (Figure 6a–c). This suggest the culture condition guaranteed by the bioreactor could be suitable for the maintenance of stemness. In detail, the laminar vortices developed within the bioreactor, besides allowing microparticle dynamic suspension, guarantee mixing and transport of nutrients and oxygen, ultimately enabling a homogeneous culture environment at very low shear stress culture conditions, as demonstrated previously. 21 This particular dynamic suspension environment could promote the formation of spheroidal cellular aggregates in vitro, a typical feature of stem cells (Figure 6a–c).
Cell culture in dynamic suspension condition.
GFPpos CPCs conjugated to Gel/GE microparticles were harvested from the vessel, cultured for 5 days in static conditions, and analyzed by fluorescence microscopy. This analysis confirmed that GFPpos CPCs mainly adhered to smaller microparticles, covering the entire surface (Figure 6d–f).
Gel/GE microparticles (diameter 350–450 µm)
After 1 day of dynamic suspension culture, GFPpos CPCs were adherent to the microparticles (Figures 6g–i).
After 5 days of static culture, the cells remained attached to the microparticles, with few single cells attached to the dish (Figures 6j–l).
Conversely, after 1 day of dynamic suspension culture of GFPpos CPCs without the Gel/GE microparticles (GFPpos CPCs CTRL), we did not observe cluster formation and cells were individually suspended (Figure S2). This observation highlights the important role of the Gel/GE microparticle addition to dynamic suspension culture to improve cell to cell adhesion and cluster formation.
Quantitative analysis of cell-microparticle adhesion
The quantitative analysis pointed out a more efficient adhesion of GFPpos CPCs to smaller microparticles both after 1 day of dynamic suspension culture and after the subsequent 5 days of static culture. Moreover, during the 5 days of static culture, an increased fraction of GFPpos CPCs was documented, suggesting a positive effect of Gel/GE microparticles on cell adhesion and survival (Figure 7).
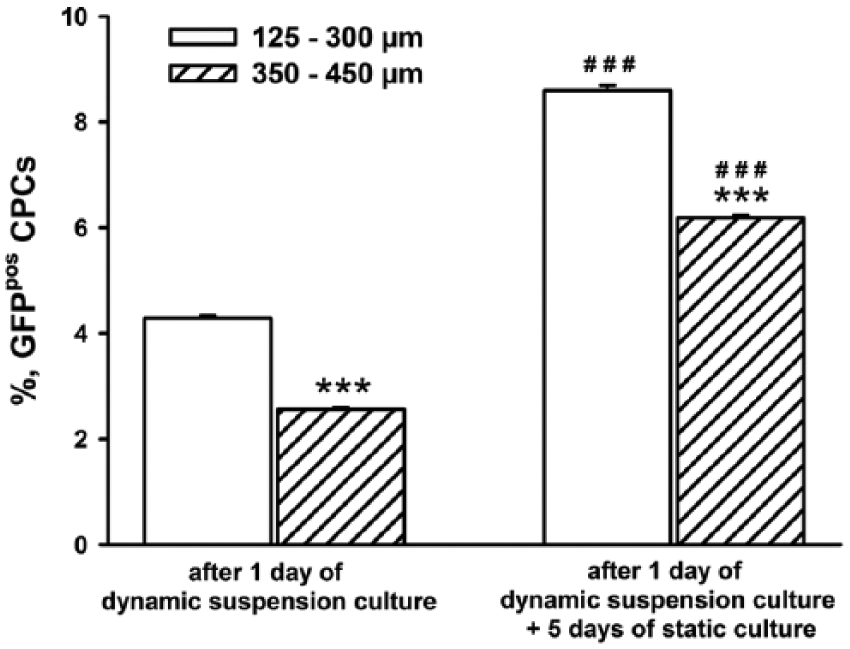
Quantification of the fractional area occupied by green fluorescence positive cardiac progenitor cells after 1 day of dynamic suspension culture and after 5 days of static culture, following dynamic suspension culture. ***p< 0.001 vs 125–300µm; ###p< 0.001 vs 1 day of dynamic suspension culture.
Conclusions
Novel injectable scaffolds, in the form of microparticles, were studied in this work for application in CTE. A blend of GE with a protein component, Gel, was used as scaffold material to favor cell adhesion. Microparticles were produced by a water-in-oil emulsion, using PC as a surfactant, and simply cross-linked by calcium ions, ensuring the biocompatibility of the polymer matrix. Two different size populations were obtained by changing the surfactant concentration. The particles showed a spherical shape and a porous surface. The physicochemical characterization pointed out the hydrophilicity of particle matrix and the presence of intermolecular interactions between Gel and GE. These interactions, mimicking those occurring between protein and polysaccharide components in natural tissues, can result in improved cell compatibility. The degradation kinetic observed during in vitro hydrolytic degradation tests could reasonably fit with the supposed tissue regeneration period. Values of viscosity, measured at room temperature by a rotational rheometer, were adequate for injection through catheters and preliminary ex vivo injectability tests showed the retention of microparticles at the injection site. The analysis of the influence of particle diameter on cardiac progenitor cells documented a preservation of cell integrity and a preferential cell adherence to small microparticles. In addition, following exposure to a dynamic suspension culture in the presence of Gel/GE microparticles, cardiac progenitors tended to form small spherical clusters that are a typical feature of stem cells, and maintained their viability and growth capacity. On the contrary, these spherical clusters were not observed in the absence of the microparticles and the cells were individually suspended.
The obtained results indicate that the produced Gel/GE microparticles could be potentially employed as injectable scaffolds for myocardial regeneration. For this purpose, a more specific characterization, including tests in animal models, is needed, and it will be object of an upcoming paper.
Supplemental Material
Supplement_Material_(Fig_1_and_2) – Supplemental material for Influence of injectable microparticle size on cardiac progenitor cell response
Supplemental material, Supplement_Material_(Fig_1_and_2) for Influence of injectable microparticle size on cardiac progenitor cell response by Elisabetta Rosellini, Niccoletta Barbani, Caterina Frati, Denise Madeddu, Diana Massai, Umberto Morbiducci, Luigi Lazzeri, Angela Falco, Costanza Lagrasta, Alberto Audenino, Maria Grazia Cascone and Federico Quaini in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Commission FP7 Programme, grant 214539.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.