Abstract
Background:
High purity cobalt has many important applications, such as magnetic recording media, magnetic recording heads, optoelectronic devices, magnetic sensors, and integrated circuits, etc. To produce 5N or higher purity cobalt in an electro-refining process, one of the challenges is to effectively reduce the Fe content of aqueous cobalt salt solution before electrolysis. This paper describes thermodynamic and kinetic investigations of the Fe adsorption process of a new sulfonated monophosphonic resin with the trade mark Monophos.
Methods:
Five cobalt sulfate solutions of different Co concentrations were prepared. Fe ions were removed from the solutions by ion exchange method using Monophos resin. Chemical analysis was carried out using a Perkin Elmer ICP-OES.
Results:
The initial Fe concentrations of about 0.9–2.0 mg/L can be reduced to about 0.3–0.8 mg/L, which is equivalent to an Fe removal rate of 60–67%. The Langmuir isothermal adsorption model applies well to the Fe removal process. A second-order type based on McKay equation fits better with experimental data than other kinetic models. The kinetic curve can be divided into two sections. For t < 30 min, particle diffusion may act as the controlling step, whereas chemical reaction may control the Fe adsorption process in the section t > 30 min.
Conclusions:
Monophos resin is effective for the removal of trace Fe from cobalt sulfate solution. This ion exchange process obeys the Langmuir isothermal adsorption model and the McKay equation of second-order kinetics.
Keywords
Introduction
High purity cobalt has excellent semiconductor and magnetic properties as well as good electrical conductivity. It has become an important material for many applications, such as magnetic recording media, magnetic recording heads, optoelectronic devices, magnetic sensors, and integrated circuits, etc. In particular, 5N or higher purity cobalt is needed for contact layer applications in integrated circuits.1–7
To produce 5N or higher purity cobalt in an electro-refining process, one of the challenges is to effectively reduce the Fe content of aqueous cobalt salt solution before electrolysis. Chemical precipitation and solvent extraction are methods often employed to remove iron from cobalt salt solutions. However, chemical precipitation requires high temperature and high pressure conditions and produces iron precipitates, which are difficult to separate from solutions. With solvent extraction, a general problem is emulsification, which causes loss of extractant and contamination of electrolyte. In addition, both chemical precipitation and solvent extraction methods are not able to reduce iron to a level sufficiently low to meet the requirement of 5N or higher purity cobalt production.
A more in-depth literature study reveals that ion exchange with specially designed resins is a viable method for removing trace iron from cobalt salt solutions. Zhong and Wang were successful in preparing 5N cobalt by electrolysis using cobalt sulfate solution purified with Diphonix and M-4195 resins. 8 The former resin is for iron removal, whereas the latter is for Ni removal. According to McKevitt and Dreisinger,9,10 sulfonated monophosphonic resins are better than sulfonated diphosphonic and aminophosphonic resins for iron removal. The authors conducted an experimental work using a new sulfonated monophosphonic resin with the trade mark of Monophos to treat an aqueous cobalt sulfate solution. Monophos is a resin with monophosphonic and sulfonic acid as functional groups covalently bonded to a porous polymeric support. It has a high selectivity for Fe3+ over divalent cations in acid. When in contact with aqueous cobalt sulfate solution, the resin exchanges H+ ions with Fe3+ and other metallic ions, thus absorbing them and purifying the solution. Results obtained have shown that the Fe content can be reduced from about 2 mg/L to about 0.4 mg/L under an optimal condition. 11 Monophos is thus proved to be very effective in absorbing trace Fe ions from aqueous cobalt sulfate solution. This is very important for producing high purity cobalt, because Fe is one of the major impurities which are not easy to be reduced during electro-refining process. In order to better understand the adsorption process, thermodynamic and kinetic investigations of trace iron removal using Monophos are carried out in this study.
Experimental details
Preparing CoSO4 solutions of different concentrations
A high purity battery grade cobalt sulfate (CoSO4·7H2O, Co content ≥ 20.5%) provided by Kunshan HP Materials Technology Co. LTD and UP (ultrapure) water were used to prepare aqueous cobalt sulfate solutions. By adjusting amounts of both cobalt sulfate and water, five solutions of different cobalt concentrations were prepared: [Co] = 30, 40, 50, 60, and 70 g/L. All five solutions have a pH value of about 4.3. The initial Fe concentrations in the five solutions are [Fe] = 0.880, 1.168, 1.472, 1.770, and 2.044 mg/L, respectively. It is apparent that the initial Fe concentration increases proportionally to the Co concentration, indicating that Fe is mainly from the cobalt sulfate used in this study. Due to the acidity of the solutions, Fe in this study is believed to be mainly in the form of Fe3+.
Resin treatment
Monophos resin was first soaked with deionized water for 24 h, causing the resin to expand, and then soaked with 6 mol/L hydrochloric acid solution for 6 h to elute iron absorbed in previous tests. After soaking in HCl solution, the resin was first rinsed with pure water until water became colorless, and then rinsed with a sulfuric acid solution of pH 2 to convert the resin into a hydrogen type. Finally, the resin was repeatedly washed with deionized water until the supernatant was neutral. The resin treated in this way was kept in pure water for later use.
Thermodynamic tests
Five equal volumes (50 mL each) of Monophos resin were measured after water being filtered out and placed in five beakers. Five equal volumes of the five cobalt sulfate solutions (250 mL each) were taken and added into the five beakers. So each beaker contained 50 mL of resin and 250 mL of cobalt sulfate solution, with a resin to liquid volume ratio of 1:5. But cobalt concentrations in the five beakers were different: [Co] = 30, 40, 50, 60, and 70 g/L. Static adsorption took place for a duration of 300 min at three constant temperatures: 303 K, 313 K, and 323 K. The duration of 300 min is believed to be long enough for the adsorption process to reach an equilibrium state. A 2.5 mL sample was taken for each test to determine its iron concentration at equilibrium. Chemical analysis was carried out using a Perkin Elmer ICP-OES.
Kinetic tests
250 mL of cobalt sulfate solution with [Co] = 60 g/L were placed in a beaker which was then put in a hot water bath to warm up to a temperature of 40℃. 50 mL of Monophos resin was measured after water being filtered out and added into the beaker when the solution temperature was stabilized. A 2.5 mL sample of solution was taken every 10 min till the adsorption time reached 90 min. Afterwards, a 2.5 mL sample of solution was taken every 30 min till the total adsorption time reached 300 min. Analysis using ICP-OES was conducted to measure iron concentration of each sample.
Results and discussions
Thermodynamic investigation
Figure 1 shows the initial Fe concentrations and the equilibrium Fe concentrations at different test temperatures for the five cobalt sulfate solutions. It can be seen in Figure 1 that initial iron concentrations of about 0.9–2.0 mg/L can be reduced to about 0.3–0.8 mg/L (for different cobalt concentrations). This is equivalent to an Fe reduction rate of 60–67%. In addition to Fe adsorption, resin can absorb cobalt at the same time. For example, a cobalt loss as high as about 20% was reached for the sample with an initial cobalt concentration of [Co] = 60 g/L.
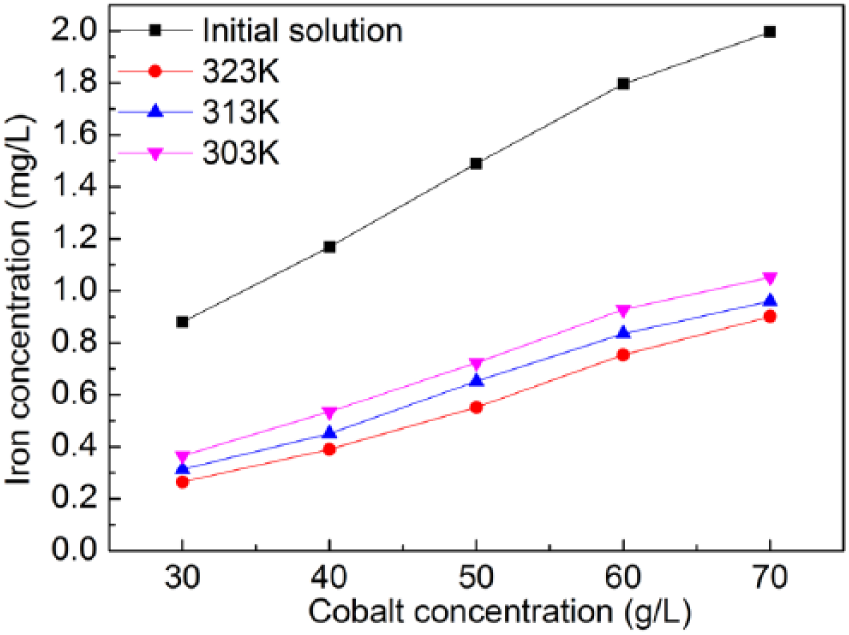
Initial Fe concentration and Fe concentration at equilibrium for different cobalt concentrations at different test temperatures.
Using the data shown in Figure 1, resin adsorption capacity at equilibrium, Qe, plotted as a function of Fe concentration at equilibrium in Figure 2, was calculated according to
where C0 is the initial Fe concentration, Ce is the Fe concentration at equilibrium, V is volume of cobalt sulfate solution (V = 250 mL in this study), and VR is resin volume (VR = 50mL in this study). From equation (1), it can be seen that the adsorption capacity is actually the amount of Fe absorbed by a unit volume of resin under given experimental condition. Table 1 provides a summary of symbols used in this paper.
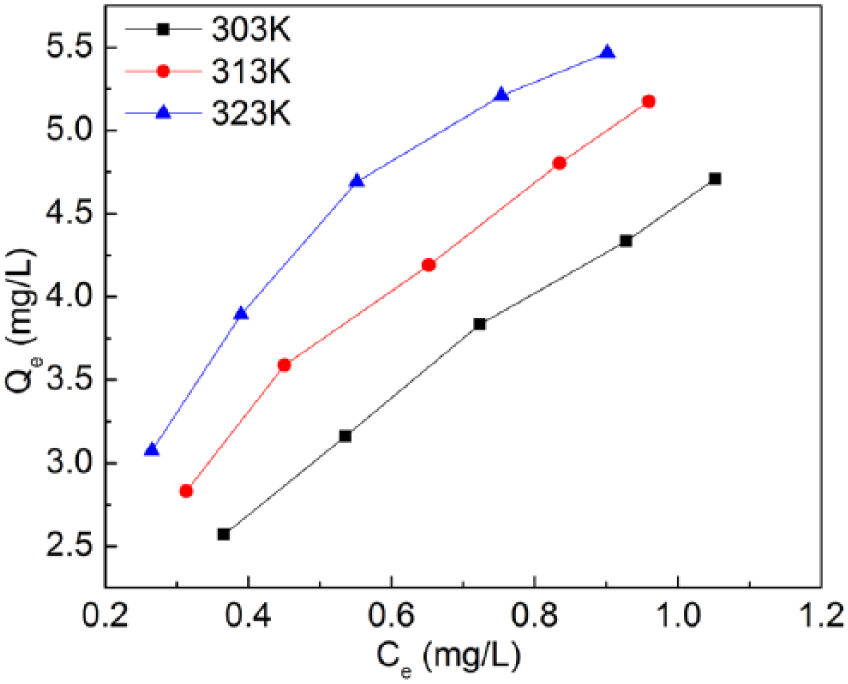
Plot of resin adsorption capacity at equilibrium, Qe, as function of Fe concentration at equilibrium, Ce, at different test temperatures.
Nomenclature in this paper.

The famous Langmuir equation is often used to correlate adsorption equilibrium data. This equation can be written in the form12,13

Other isotherm models used to correlate adsorption equilibrium data include Freundlich (equation (2b)), Temkin (equation (2c)), and Dubinin–Radushkevich (equation (2d)) models:



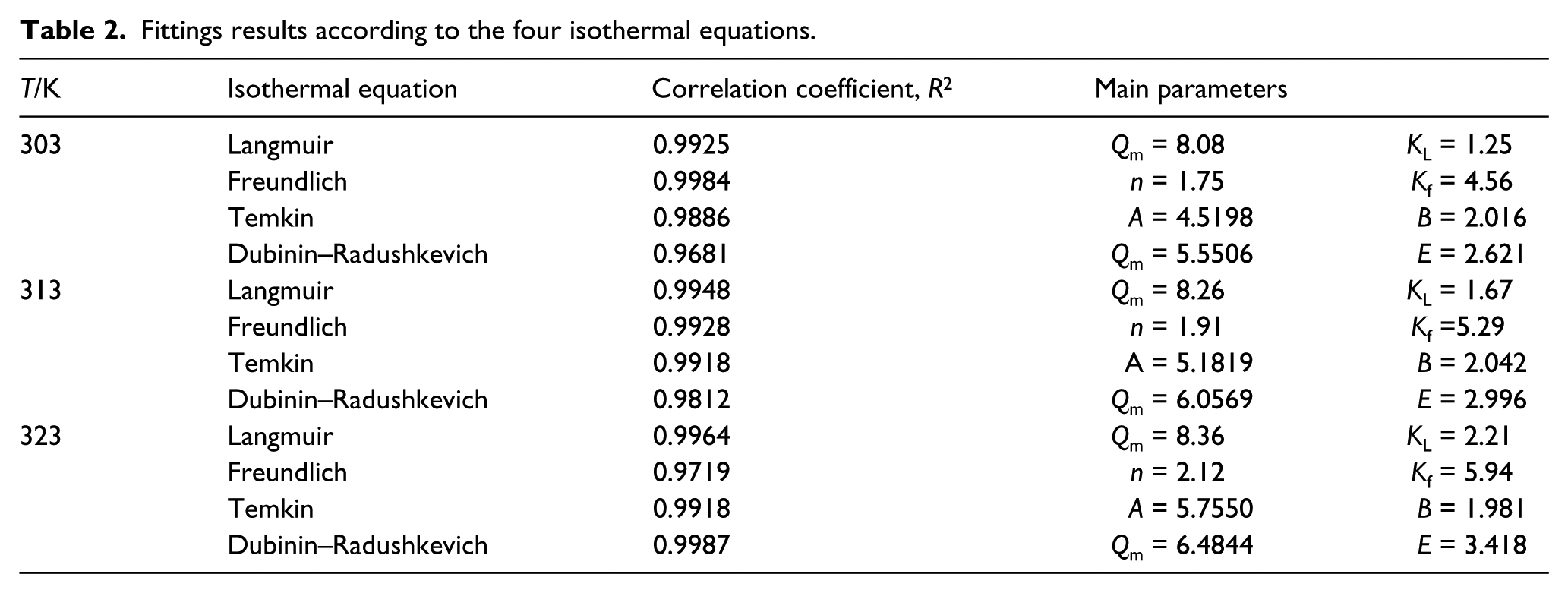
In order to determine which model applies in this study, we rearranged and plotted the data in Figure 2 according to each model to obtain graphs shown in Figure 3. Table 2 summarizes the fitting results for the four models. It can be seen from Figure 3 that the Langmuir equation correlates the best with the equilibrium data in this study, with a correlation factor R2 being larger than 0.99 for all three test temperatures (see Table 2). Both KL and Qm increase with increasing test temperature, indicating that ion exchange is faster and more Fe can be adsorbed by Monophos resin at higher temperature.
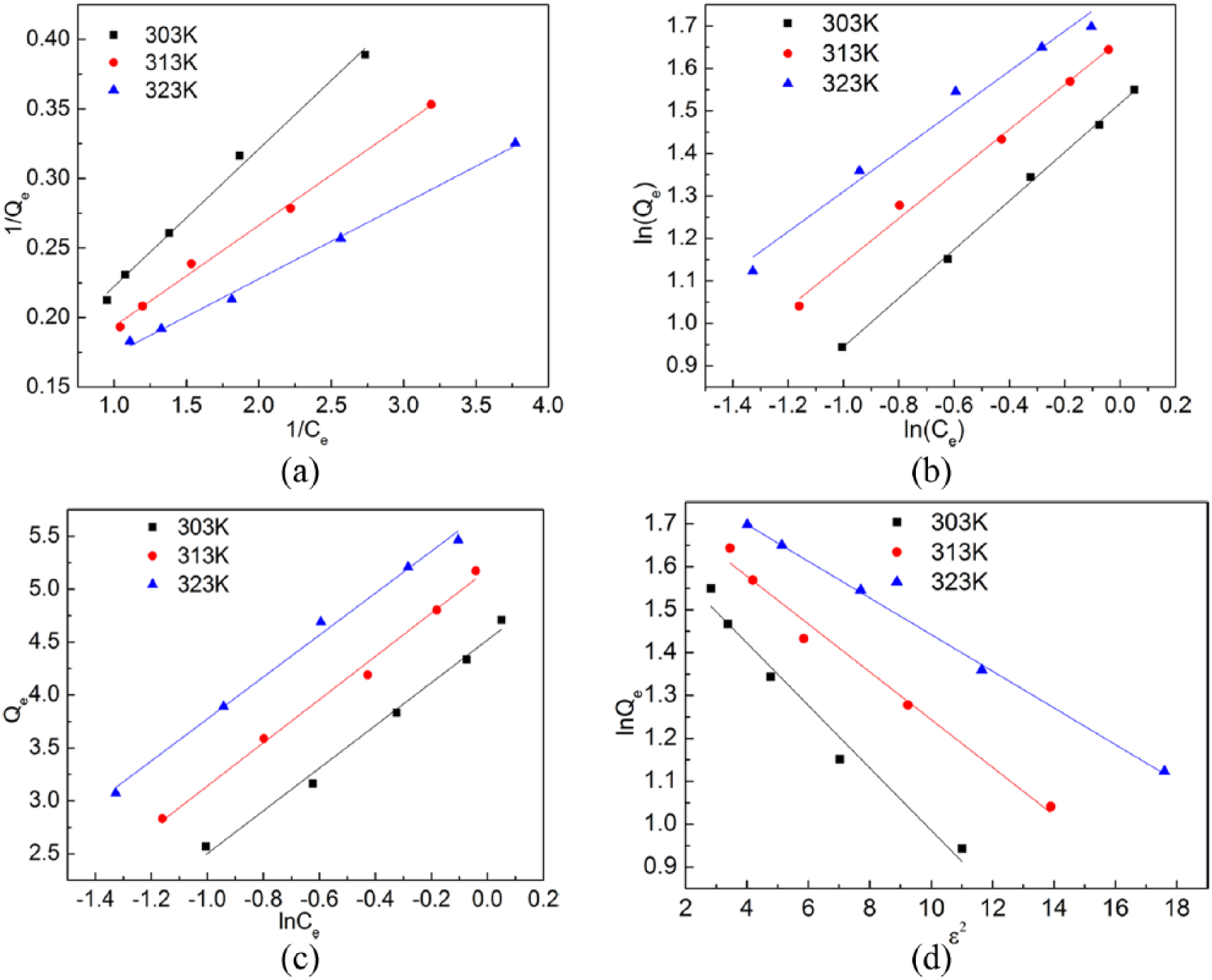
Plots of resin adsorption equilibrium data shown in Figure 2 for (a) Langmuir, (b) Freundlich, (c) Temkin, and (d) Dubinin–Radushkevich models.
Fittings results according to the four isothermal equations.
It is well known that the parameter KL is related to the change of Gibbs energy ΔG according to

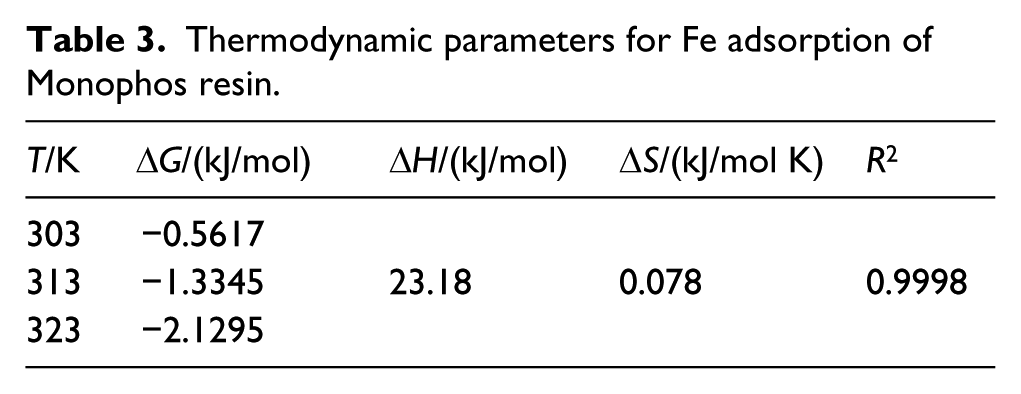
Using this equation and the KL data in Table 2, we calculated ΔG for the adsorption reactions in this study (see Table 3). For all three test temperatures, ΔG < 0, indicating that Monophos resin spontaneously adsorbs Fe in aqueous cobalt sulfate solution.
Thermodynamic parameters for Fe adsorption of Monophos resin.
It is also well known that the following equation is valid at equilibrium:

Combining equations (3) and (4), we obtain the following equation:

In this equation, KL is the Langmuir constant, ΔH is the enthalpy change of reaction (kJ/mol), ΔS is the entropy change of reaction (kJ/mol K), R is the gas constant, 8.314 J/mol K, and T is temperature (K). The temperature range studied in this paper is so small (303–323 K) that one can assume both ΔH and ΔS are constant. Then, we can obtain a straight line by plotting ln KL against 1/T (see Figure 4). Furthermore, we can determine ΔH and ΔS using the slope and the intercept of the straight line (see Table 3). The positive ΔH as well as positive ΔS suggest that Fe adsorption by Monophos resin is an endothermic reaction. An increase of temperature can therefore promote the adsorption reaction. This is in agreement with the experimental observation (see Figure 1).
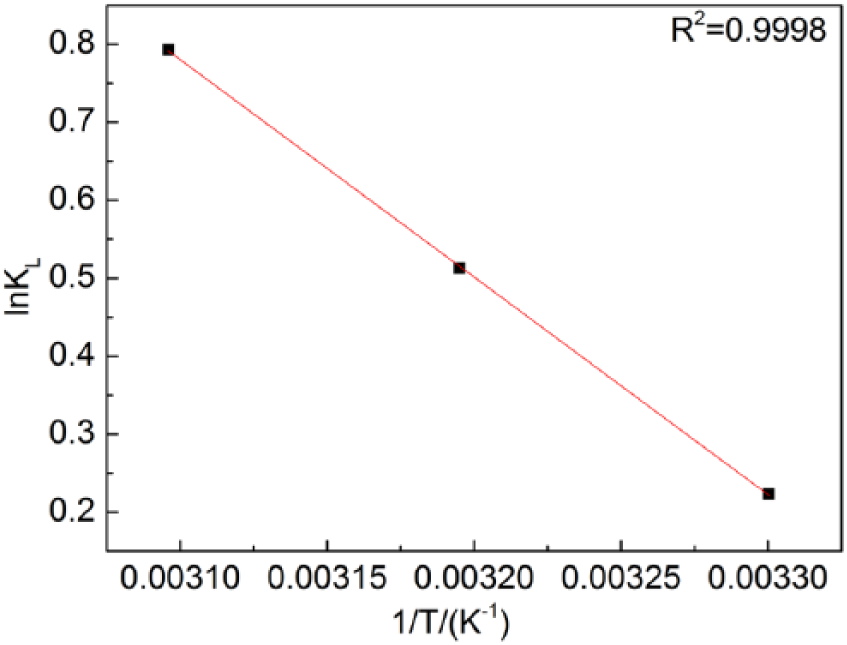
The ln KL vs.1/T plot.
Kinetic investigation
Kinetic tests were done with 50 mL of Monophos resin and 250 mL of cobalt sulfate solution of [Co] = 60g/L at 40℃. Figure 5 shows how Fe concentration changes with increasing contact time. The resin adsorption capacity at any given time, Qt, mg/L, was then calculated using

and plotted in Figure 6 as function of adsorption time. It can be seen that the Fe adsorption capacity increases with increasing time. The adsorption reaction is quite rapid in the first 30 min and it reaches an equilibrium state after about 210 min, with an equilibrium Fe adsorption capacity being about 4.2 mg/L.
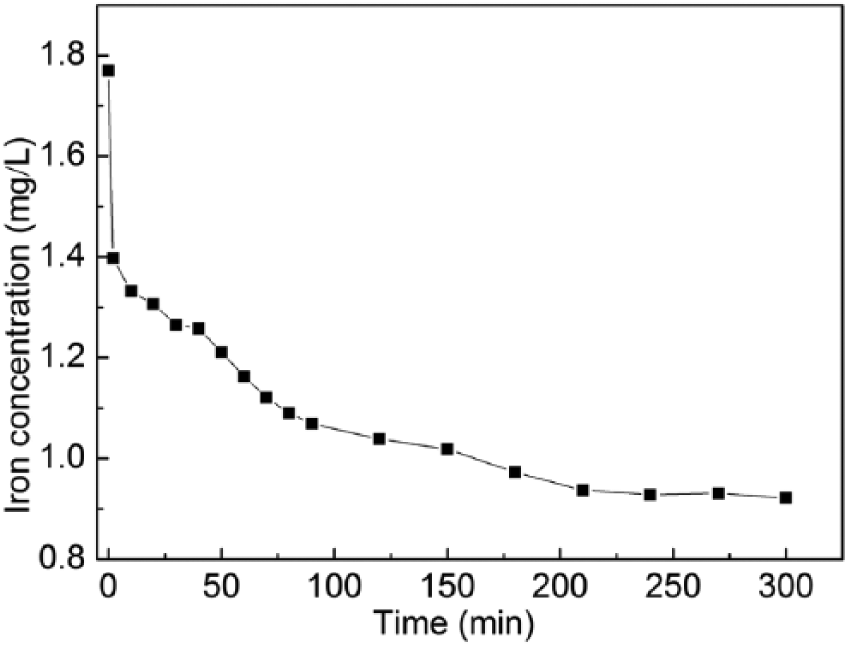
Change of Fe concentration in cobalt sulfate solution of [Co] = 60 g/L.
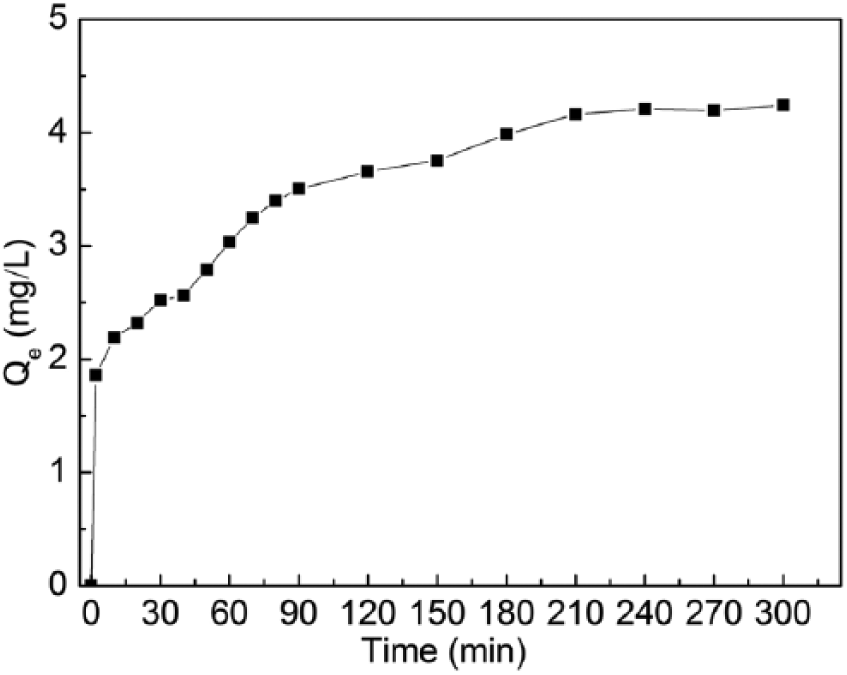
Fe adsorption capacity of Monophos resin as function of adsorption time.
In order to predict the adsorption behaviour of Monophos resin, first-order kinetics and second-order kinetics were used to fit the adsorption process. The Lagergren equation (see equation (7a)) is one of the commonly used first-order kinetic equations, whereas the McKay equation (see equation (7b)) is often used as second-order kinetic equation.12–14 In addition, other kinetic models, such as the Elovich equation (see equation (7c)), Webber–Morris equation (see equation (7d)), and Bangham equation (see equation (7e)) were also applied.

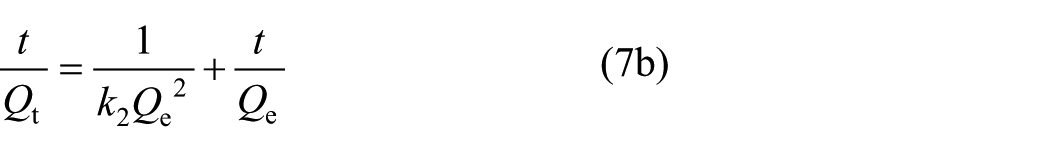



Figure 7 shows fitting plots for these five models. From the fitting results (see Figure 7 and Table 4), one can see that the McKay equation describes the Fe adsorption behavior of Monophos resin (R2 > 0.99) better than the other equations. A closer look at Figure 7(b) reveals that four data points in the beginning of the adsorption test (t < 30 min) apparently deviate from the fitting line 1. Applying the McKay equation for these four data points, we obtain the fitting line 2. The slopes of the two straight lines in Figure 7(b) are quite different, indicating that two different controlling mechanisms must exist for the two sections t < 30 min and t > 30 min.
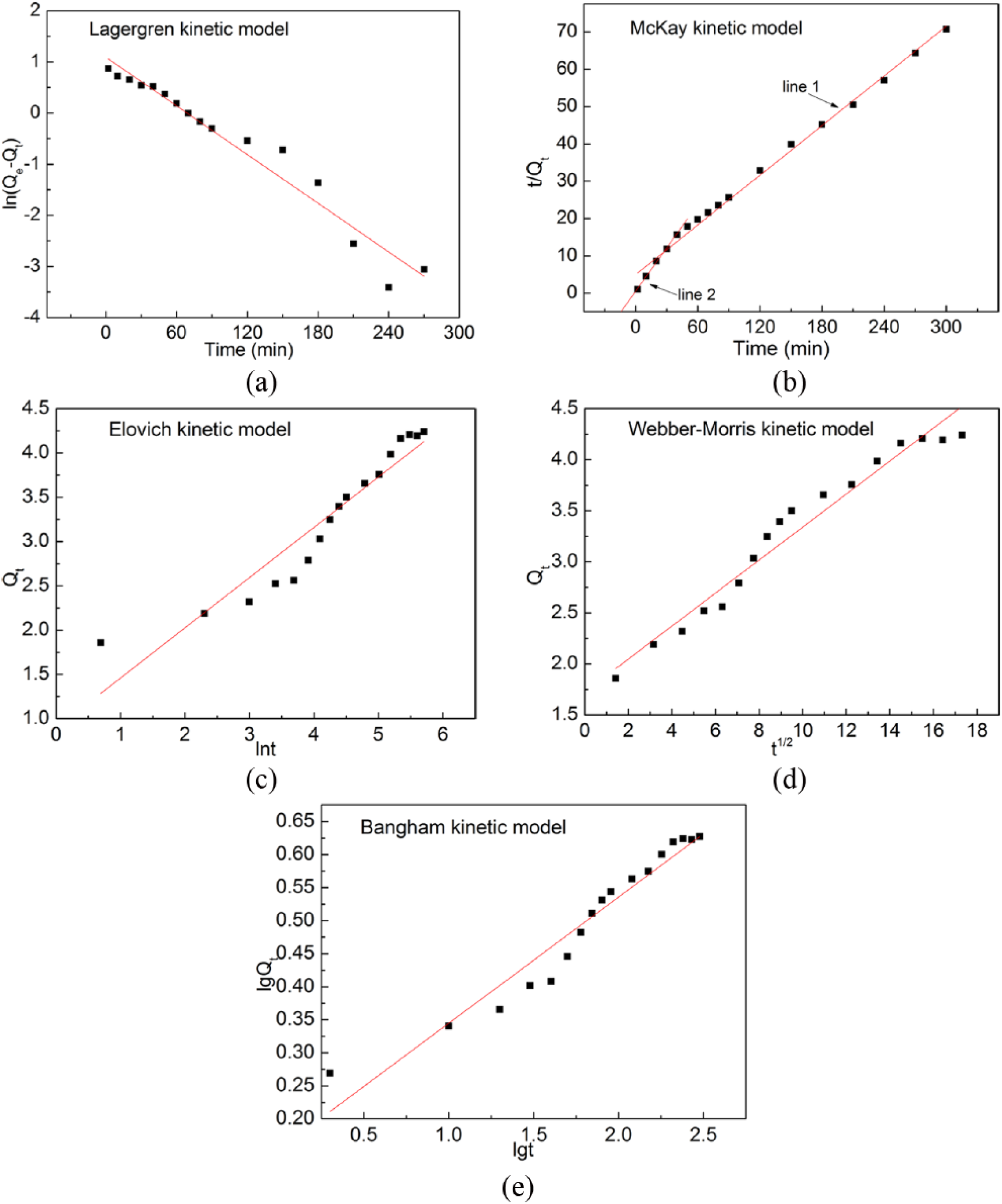
Data fitting for (a) Lagergren, (b) McKay, (c) Elovich, (d) Webber–Morris, and (e) Bangham models.
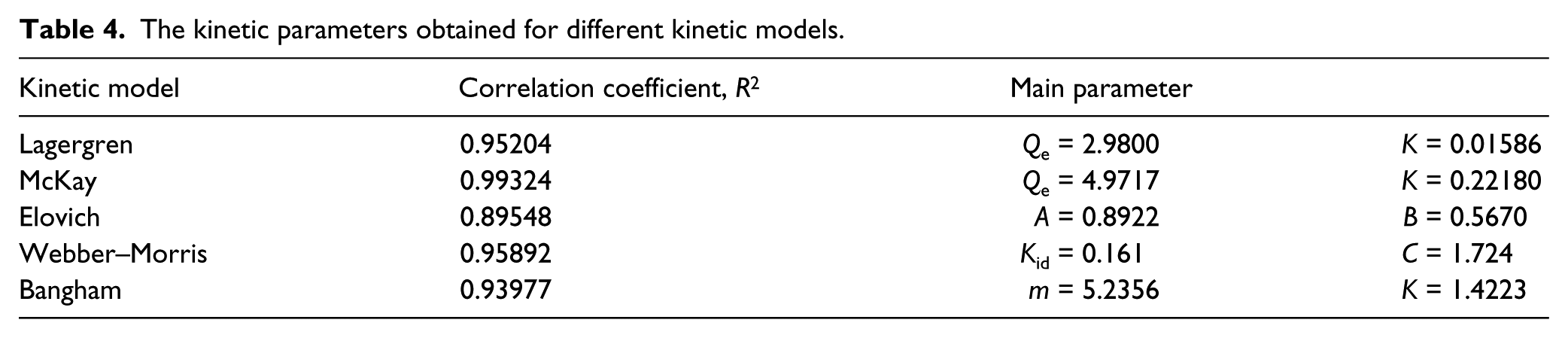
The kinetic parameters obtained for different kinetic models.
During an ion exchange process, there are three main steps: (1) diffusion of species through a liquid film around resin particles (liquid film diffusion); (2) transfer of species through resin pores to places of ion exchange (particle diffusion); and (3) chemical reaction between species and resin functional groups (chemical reaction). Equations (8), (9), and (10), respectively can describe these three steps:12–14

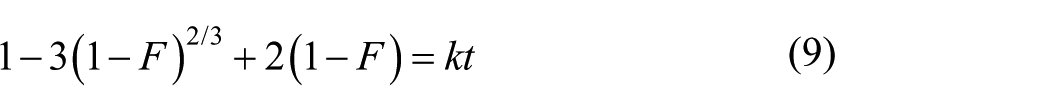

In these equations, F = Qt/Qe, k is the coefficient of mass transfer, and t is time. In order to determine which step is controlling the ion exchange process in this study, a data fitting method was employed and results obtained are shown in Figure 8(a) to (c), respectively. It is clear from Figure 8 that liquid film diffusion may not be the controlling step (Figure 8(a), R2 = 0.8715), whereas particle diffusion is probably controlling the reaction rate of ion exchange in this study. Chemical reaction fits experimental data (Figure 8(c), R2 = 0.9791) nearly as good as particle diffusion (Figure 8(b), R2 = 0.9823), indicating that both of them may act together to influence the reaction rate of ion exchange. As described above, the kinetic curve in Figure 7(b) can be divided into two sections: t < 30 min and t > 30 min. For t < 30 min, cobalt sulfate solution gradually fills up resin pores. Both Fe3+ and Co2+ react with H+, occupying all the resin active sites. Due to the complexity of resin pore structure, it is highly possible that particle diffusion acts as the controlling step for the section of t < 30 min. Because the resin has a larger affinity with Fe3+, Co2+ absorbed on resin active sites will be gradually replaced by Fe3+ . This may take place and control the Fe adsorption process in the section of t > 30 min.
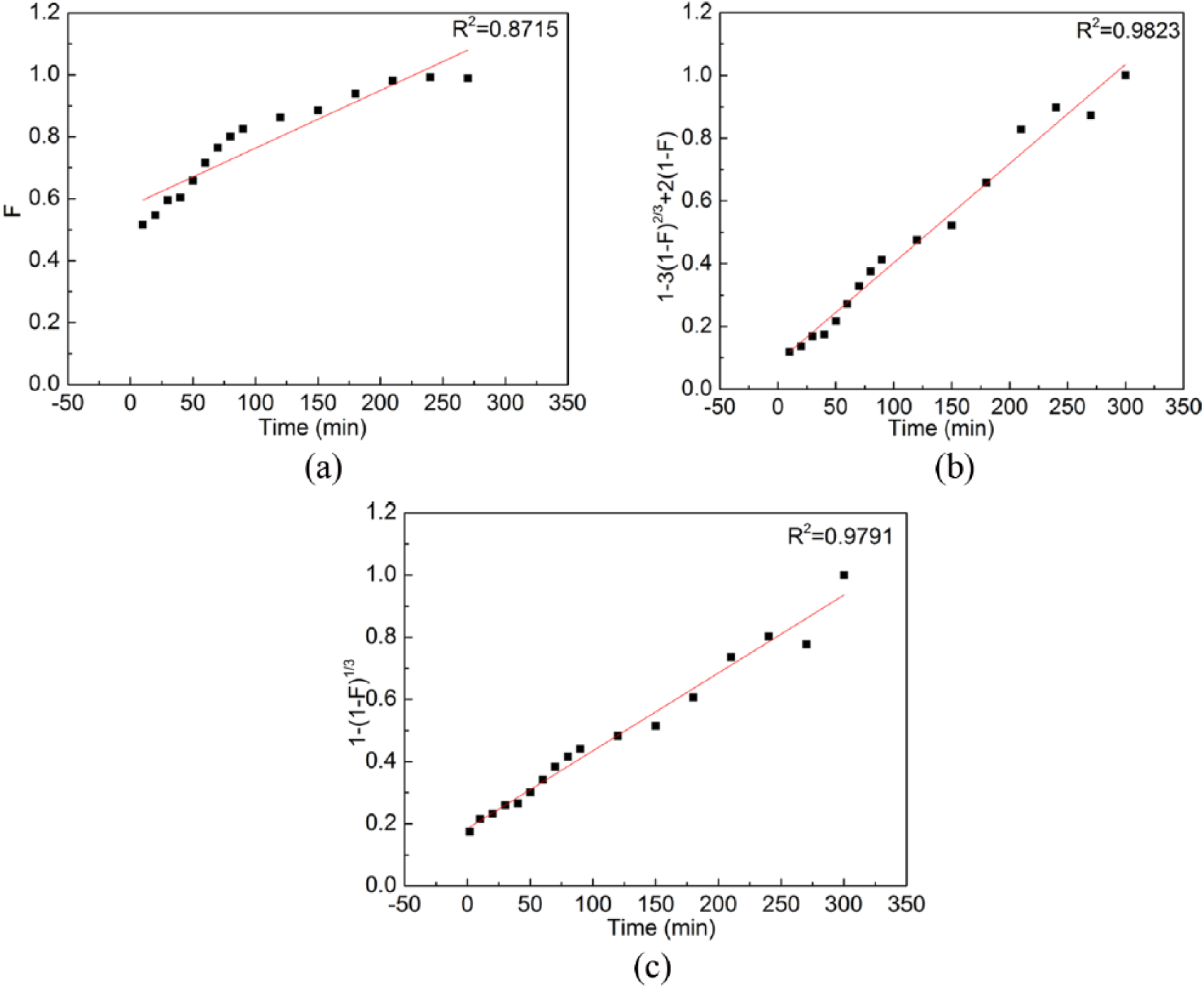
Fitting results for (a) liquid film diffusion, (b) particle diffusion, and (c) chemical reaction.
Conclusions
The thermodynamics and kinetics of the Fe adsorption process of Monophos resin have been investigated. Based on results obtained, the following conclusions can be drawn:
(1) The Langmuir isothermal adsorption model applies well to the process.
(2) The Fe adsorption reaction is endothermic. An increase of temperature is thus beneficial to Fe adsorption process.
(3) A second-order kinetics based on the McKay equation fits better with experimental data than other kinetic models.
(4) The kinetic curve can be divided into two sections. For t < 30 min, particle diffusion is probably the controlling step, whereas chemical reaction may control the adsorption process for t > 30 min.
Footnotes
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Joint Fund of the Chinese National Natural Science Foundation and Henan Province (grant number U1504516) as well as the Chinese 1000 Plan for High Level Foreign Experts (grant number WQ20154100278).

