Abstract
Background:
Dengue virus (DENV) is hyperendemic in Indonesia and of high public health concern globally. Without an effective antiviral therapy, an accurate diagnosis of the four viruses, DENV1-4, is crucial for patient management and outcome. We aim to assess the diagnostic accuracy and usefulness of a commercial DENV antigen rapid diagnostic test kit in point-of-care conditions.
Design and methods:
Six-hundred-forty-six patients with fever or history of fever, and without leukocytosis, were enrolled from Tabanan General Hospital, Bali in 2017–2018. All sera were tested for DENV NS1 antigen using the Standard Diagnostics Bioline NS1 Ag rapid test kit and Flavivirus genus specific RT-PCR. Positive specimens were further confirmed using DENV multiplex RT-PCR.
Results:
DENV was detected in 10.4% (67/646) of enrolled patients by molecular assays. Only 41 of the positives were found to be positive by the NS1 rapid test. Our findings indicate that the kit was the most sensitive in the first 2 days after disease onset with sensitivity > 75%, and declining at day 3 with a sensitivity of <60%. In addition, the kit was found to be the most sensitive to DENV-2 and least sensitive to DENV-4. Overall, the kit had a sensitivity of 59.7% [95% CI: 44.5–74.9] and specificity of 99.8% [95% CI: 99.5–100.0].
Conclusions:
Our evaluation indicated that the rapid diagnostic test is useful for both surveillance and initial detection of DENV infection in community settings. However, it should be supplemented with additional diagnostic methods to ensure accurate confirmation for patient management.
Introduction
Dengue fever, caused by one of four dengue viruses (DENV), is associated with high morbidity and mortality in tropical and subtropical regions. Indonesia is hyperendemic for DENV, with the majority of the population having antibodies against one or more DENV serotypes by the age of 14 years old. 1
Early clinical diagnosis of DENV infection can be challenging due to its non-specific symptoms, but is critical for successful disease management. Laboratory confirmation of DENV infection depends on standard assays, such as virus isolation, molecular tests, or serological assays, each with its own limitations and high logistical requirements. Enzyme-linked immunosorbent assay (ELISA) is widely used to measure anti-DENV IgM or IgG antibodies. False negative results from antibody-based assays; however, remain a concern as antibodies are not reliably detectable until 5 days after disease onset and require more complex laboratory infrastructure. Additionally, there is cross-reactivity between antibodies of closely related flaviviruses. Thus, there is an urgent need for reliable screening tests that allow timely diagnosis, especially in resource-limited primary healthcare settings where more complex diagnostic equipment is often not available.
The four closely related but antigenically distinct DENV-1, -2, -3, and -4 present with indistinguishable clinical symptoms. DENV non-structural protein 1 (NS1) is a useful diagnostic marker for early infection of all DENV serotypes. It is produced during viral replication and is secreted from infected cells into the bloodstream. Although there is high NS1 similarity between all DENV serotypes, accuracy of NS1 detection appeared to vary between regions and endemicity.2–9
Bali, a popular tourist destination in Indonesia, has a high risk for DENV transmission, which is one of the most common causes of acute fever among international visitors.10,11 Our study focused on the assessment of a locally available commercial DENV NS1 rapid diagnostic test (RDT) kit with limited information on field evaluation.
Design and methods
Patients with undifferentiated fever (axillary ≥ 37.5°C) or history of fever (≤7 days), without leukocytosis, and no clinically apparent alternative diagnosis were enrolled in the study from in-patient and out-patient departments of Tabanan General Hospital in 2017–2018. Written consent, demographic and clinical information were obtained. Sera were tested for DENV NS1 antigen at the hospital using Standard Diagnostics (SD) Bioline NS1 Ag rapid test kit (Standard Diagnostic, Australia) according to the manufacturer’s instructions. Results were interpreted by a trained laboratory technician. The lateral flow kit has a manufacturer-quoted sensitivity of 92.4% and specificity of 98.4%. The specimens were shipped to the Eijkman Institute for Molecular Biology, Jakarta, Indonesia, and stored at −80°C until further testing. RNA was extracted from the specimens using QIAamp Viral RNA Mini kit (QIAGEN, Germany), followed by a broadly reactive Flavivirus genus-specific one-step RT-PCR which exhibits a high degree of similarity among flaviviruses. 12 Positive specimens were further tested using DENV multiplex semi-nested RT-PCR.13,14 The assays were conducted by personnel who were blinded to the RDT results. Patients with incomplete clinical information were excluded from analysis.
This study was reviewed and approved by the Medical Research Ethics Committee at the Faculty of Medicine, Udayana University and Sanglah Central General Hospital, Bali, Indonesia (No. 01/UN.14.2/KEP/2017).
Results
PCR-confirmed DENV infection was seen in 10.4% (67/646) of the samples. Cohort demographic and clinical manifestations are summarized in Table 1. Aside from fever, the most common symptoms among enrollees were headache (55.7%), nausea (54.5%), and chills (44.0%). The median time of sample collection was 4 days after symptom onset.
Demographic and clinical characteristics of acute febrile study patients in Bali, Indonesia, 2017–2018.

Of 646 collected sera, 41 (6.3%) were DENV positive by the SD Bioline NS1 Ag rapid test kit. Of the 41 positives, 40 (97.6%) were confirmed to be DENV positive by RT-PCR assays. An additional 27 (4.5%) DENV were detected in 605 sera that were DENV NS1 negative by the RDT kit. Moreover, only 13.5% (19/141) of patients with thrombocytopenia (Range: 102,000–149,000 counts/μL) tested positive for DENV NS1 by the RDT kit while 19.9% (28/141) were positive by RT-PCR. The overall sensitivity and specificity of the kit was determined to be 59.7% [95% CI: 44.5–74.9] and 99.8% [95% CI: 98.5–100.0], respectively, when compared to DENV RT-PCR results. The overall positive predictive value using the kit was 97.6% [95% CI: 92.8–100.0] and the negative predictive value was 95.5% [95% CI: 93.9–97.2] (Table 2).
Sensitivity and specificity of SD Bioline NS1 Ag rapid test kit by day of onset and DENV serotype.
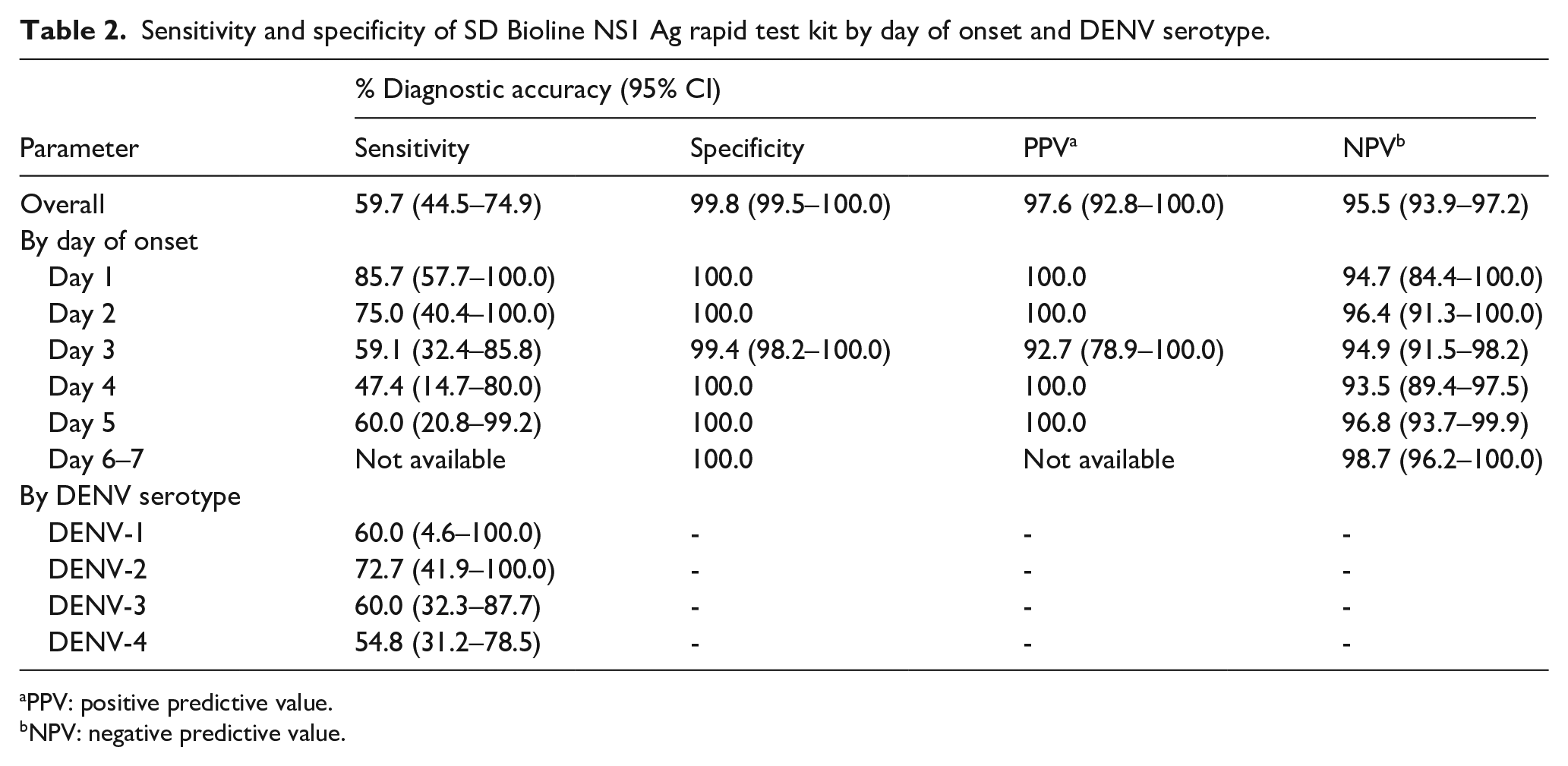
PPV: positive predictive value.
NPV: negative predictive value.
Of the 67 confirmed DENV infections, five were DENV-1 (7.5%), 11 DENV-2 (16.4%), 20 DENV-3 (29.8%), and 31 DENV-4 (46.3%). Of the 27 false negatives by DENV NS1 Ag RDT kit, DENV-4 (51.9%) was the most common followed by DENV-3 (29.6%). The kit was the most sensitive in the first 2 days after disease onset, with a sensitivity of >75%, but, declined to 59.1% [95% CI: 32.4–85.8] on day 3. The kit performed better in detecting DENV-2 with a sensitivity of 72.7% [95% CI: 41.9–100.0] and less well for DENV-4 with a sensitivity of 54.8% [95% CI: 31.2–78.5; Table 2]. No sensitivity data were available for days 6 and 7, where all results by the RDT kit and RT-PCR were negative. The majority of PCR-confirmed positive cases were classified by the attending physicians as dengue fever (37.3%) compared to dengue hemorrhagic fever (28.4%).
Discussion
Diagnosis of DENV infection can be challenging due to its nonspecific clinical manifestations and limited laboratory capacity in developing regions. Early management of patients with DENV infection is critical to ensure favorable disease outcomes. DENV NS1 antigen is a well-established biomarker for early phase DENV infection. In our study, the evaluation of the SD Bioline DENV NS1 Ag RDT kit was conducted on samples collected from a cross-sectional study of undifferentiated acute fever. Our study indicates that the RDT kit has an overall sensitivity of 59.7% [95% CI: 44.5–74.9] and specificity of 99.8% [95% CI: 99.5–100.0]. The sensitivity of the kit was similar to other field studies using molecular assays as a reference from Cambodia 2 and Vietnam 3 ; but varied from other field studies from Indonesia at 73.0%, 4 Taiwan at 89.7%, 5 Malaysia at 52.4%, 6 Singapore at 68.5%, 7 Sri Lanka at 48.5%, 8 and Angola, Yap, and Fiji at 65.9%. 9 Conversely, the kit exhibited comparable specificity to other field studies, barring one by Hunsperger et al. 9 at 80.9%.
DENV NS1 antigenemia fluctuates throughout the illness with detectable levels demonstrated at the start of illness; it peaks on days 4–5 after fever onset during primary infection, but wanes earlier in secondary infections. 15 We observed the RDT kit sensitivity was the highest on day 1 and gradually declined over the course of the disease. Our findings were in line with the previous studies.5–7,16 Moreover, DENV NS1 based RDT sensitivity was reported to be affected by primary or secondary infections; the presence of DENV-IgG was linked to a significant reduction in sensitivity. 17 Our study was conducted in a DENV-endemic setting where patients may have been exposed to multiple DENV infections 18 ; however, anti-DENV IgM/IgG tests were not performed to determine the patient immune status and confirm the association. Inclusion of anti-DENV IgM test has been reported to also improve overall sensitivity of DENV RDT for dengue diagnosis with slight reduction in specificity. Additional anti-DENV IgG tests only improve detection sensitivity slightly but incur a great loss in specificity.5,6 Caution should be taken when using anti-DENV IgM test alone for dengue diagnosis, especially in DENV-SARS-CoV-2 overlap areas, as anti-SARS-CoV-2 cross reactivity with anti-DENV IgM has been reported. 19
In our study the RDT kit demonstrated varying sensitivities across different DENV serotypes, with the highest sensitivity to DENV-2 and the lowest to DENV-4, which was also the most frequently detected serotype. Previous studies indicated that sensitivity of the kit differed according to serotype, with a reduced sensitivity observed against DENV-4,5,16 a finding that has also been reported in Indonesia. 20 Another regional study in Cambodia also showed that the sensitivity was highest in DENV-4 and lowest in DENV-2 infection. 2
Our study has some limitations. First, the absence of comprehensive data on the patient immune status (i.e. primary vs secondary) prevented stratification, which would have provided deeper insights into its relationship with NS1 positivity. Second, the study was conducted in a single hospital in Bali, which may limit the generalizability of the findings to other regions of Indonesia with different circulating DENV strains. Third, the study included only patients who sought medical attention, potentially excluding milder cases that are not typically documented in a hospital setting. Fourth, the study design involved collecting a single sample at each time point, which precluded determining the duration of positivity. Lastly, for this study, we utilized only one RDT that was locally available; however, future evaluations should include multiple brands as well as NS1 ELISA confirmation for a more comprehensive assessment.
Conclusion
Our evaluation of the SD Bioline NS1 Ag RDT on samples collected from patients with undifferentiated fever showed similar sensitivity and specificity to the previous studies conducted in other endemic DENV regions. The findings indicate that the RDT kit, performed on single blood specimens, can serve as a valuable supplementary tool for DENV surveillance in resource-limited settings, despite its lower sensitivity.
Footnotes
Acknowledgements
We thank the patients and their families for agreeing to participate, and the staff of Tabanan General Hospital for their support. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the U.S. Centers for Disease Control and Prevention.
Author contributions
Writing-original draft: NPDW, AF, KSAM; Investigation: AF, EJ, YPD, IYS, RD; Formal analysis: AF, EJ, RD; Resources: YPD, IYS, AL, IGAAR; Data curation: NPDW, AL, IGAAR; Project administration: UA, FAY; Supervision: AL, KSAM, AMP; Funding acquisition: IMA, KSAM; Conceptualization: IMA, KSAM. All authors revised and improved the manuscript. All authors read and approved the final manuscript
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Ministry of Research, Technology, and Higher Education of the Republic of Indonesia and the U.S. Centers for Disease Control and Prevention.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Significance for public health
This study highlights the role of the NS1 RDT as a valuable tool in resource-limited settings for effective surveillance and early detection of DENV infection with prompt clinical treatment. However, it also points out that the dengue NS1 antigen kit did not prove sufficiently reliable with regard to sensitivity.
