Abstract
Background:
Deficiency of Vitamin B12 and folate may determine hematological, neurological, and metabolic alterations; therefore, an accurate quantification of their serum levels is required, especially in the presence of symptoms that might suggest a deficiency. CHORUS VIT B12 and CHORUS FOLATE are two automated immunoassays, developed to quantify vitamin B12 and folate, respectively, in human serum.
Design and methods:
This single-center, non-pharmacological, diagnostic study described the validation and characterization of CHORUS VIT B12 and CHORUS FOLATE, with a specific focus on performance, precision, and reliability. For each assay, 500 serum samples were analyzed. A comparison between CHORUS assays and commercially available kit was also performed.
Results:
For CHORUS VIT B12 the lower limit of quantification (LLoQ) was 165.0 pg/mL and the upper LoQ (ULoQ) was 1846.8 pg/mL. The assay was linear within the calibration range (150–2000 pg/mL) and the accuracy was described with the International Standard Vitamin B12, Serum Folate, HOLO TC (NIBSC code: 03/178), with a mean recovery on two lots of 111%. For CHORUS FOLATE (calibration range of 2.0–20.0 ng/mL), LLoQ was 2.0 ng/mL and ULoQ 19.6 ng/mL. The linearity was demonstrated from 2.4 to 20.0 ng/mL; the accuracy was described with the International Standard mentioned above, achieving a mean recovery on three lots of 92%. The lowest and highest values of both CHORUS and COBAS kits were similar and the median values did not significantly vary.
Conclusion:
CHORUS VIT B12 and CHORUS FOLATE performed well, accurately, and reliably in quantifying vitamin B12 and folate in human serum.
Introduction
Vitamin B12 (cobalamin) consists of a central cobalt atom surrounded by a heme-like planar corrin ring structure, with the four pyrrole nitrogens coordinated with the cobalt. 1 Vitamin B12 is needed for two metabolic reactions in human, methylation of homocysteine to methionine and conversion of methyl malonyl-CoA to succinyl-CoA.2,3
Folates comprise a family of chemically related compounds based on the folic acid structure. Folate monoglutamate transported into cells is metabolized to polyglutamate forms by the enzyme folylpolyglutamate synthetase, as the polyglutamated form can be effectively retained by tissues. 1 Folate coenzymes are involved in three major interrelated metabolic cycles in the cytosol, which are required for the synthesis of thymidylate and purines, precursors for DNA and RNA synthesis, and for the synthesis of methionine from homocysteine and the interconversion of serine and glycine.1,4
Vitamin B12 and folate deficiencies in infants and children classically present with megaloblastic anemia and are often accompanied by neurological signs, in particular if the diagnosis is delayed. 2 Depending on the cause and severity of vitamin B12 deficiency, unspecific clinical signs and symptoms may already arise in the first months of life, including failure to thrive (weight and head circumference), developmental regression, irritability, apathy, anorexia, and refusal of solid foods. 2 The main hematological abnormalities, such as megaloblastic anemia, hypersegmentation of neutrophils and (pan) cytopenia may only manifest later or not at all. Neurological deficits can occur without hematological abnormalities and include hypotonia, loss of deep tendon reflexes, tremor and movement disorders, paralysis, seizures, developmental regression, dementia, and neuropsychiatric changes.2,5
In adulthood, a relationship between metabolic syndrome and vitamin B complex, specifically vitamin B12 and folate, has been the subject of many studies worldwide. Several studies have reported a positive association between the supplementation of folate and vitamin B12 and their effectiveness in the reduction of cardiovascular disease and insulin resistance.5,6
Given the clinical importance of vitamin B12 and folate, it is necessary to accurately quantify their serum levels, especially in presence of symptoms that might suggest a deficiency. For instance, B12 testing in infants and related mothers selected at neonatal screening for B12 deficiency, it is crucial to promote an adequate B12 supplementation. 7 There are several approaches to diagnosing vitamin B12 deficiency. 8 These include tests to detect physiological correlates of vitamin B12 deficiency, for example the identification of hyper-segmented neutrophils, oval macrocytes and circulating megaloblasts in the blood film or functional tests for biochemical abnormalities, for example methylmalonic acid or total homocysteine levels. Direct tests to quantify vitamin B12 status in serum are also available. 8
The determination of folate in serum is the most common laboratory test used for folate status evaluation. The results closely reflect folate status in most patients and correlate with red blood cell folate and total homocysteine. Serum folate concentrations rise in response to folic acid/folate ingestion for up to 2 h and then decline rapidly. Fasting folate concentrations may represent reduced folate released by tissues. 9 Folate binding protein (FBP)-based assays are the most used method for the assessment of folate status. Labeled folate standard competes with endogenous folate for a folate binder, or in non-competitive binding assays, an excess of FBP is added to a sample followed by a labeled folate conjugate. Both set-ups are inexpensive and offer a quick analysis of a large number of samples, plus a high degree of automation. 9
In general, for serum vitamin B12 the capability of the test to rule out the deficiency at the individual level is low when a single threshold value, such as the lower reference limit (LRL), is adopted. Multiple thresholds have been validated by clinical studies establishing the relationship between marker concentrations, the probability of deficiency, and the need for supplementation. 10 The evaluation of the inter-assay variability of vitamin B12 measurement at serum concentrations around the established thresholds for the risk of deficiency may be useful in indicating if these cut-offs may be generalized or require an adjustment according to the employed method. For serum folate testing, a single threshold, set at LRL, is usually employed to screen for vitamin deficiency in non-fortified countries. Consequently, the prevalence of folate deficiency is strictly dependent on the correctness of cut-off establishment and the accuracy of folate assays. 11
This paper describes the validation, performances, and reliability of two automated tests for vitamin B12 and folate quantification. A comparison with a commercially available kit was also provided.
Design and methods
Study design and samples
This single-center, non-pharmacological, diagnostic study was carried out at Le Scotte Hospital in Siena to validate two diagnostic tests, CHORUS VIT B12 and CHORUS FOLATE, applied on the CHORUS TRIO instrument.
The study was conducted according to the ethical principles reported in the Declaration of Helsinki in its latest revision and the protocol was approved by the local ethics committee (protocol CHOR-VIT; Comitato Etico Regionale per la Sperimentazione Clinica della Regione Toscana).
Participation in the study did not require informed consent.
Serum from patients was collected for routine clinical analysis and all samples had been anonymized. Data were aggregated and provided for analysis without any reference to collection time, patient’s prognosis status, and ongoing treatment. After the withdrawal, the samples were frozen and kept at −80°C. Samples extraction has been performed before analysis. The extraction process allows vitamin B12 and folate to free themselves from the carrier proteins, so that they can be detected.
Reagents
Vitamins were extracted from human serum with CHORUS Extraction kit, according to manufacturer’s instructions. 100 µl of sample were mixed with 50 µl of extraction buffer (stabilizing agent and releasing agent at 1/40 dilution); after vortexing and incubating at room temperature for 15 min, 50 µl of neutralizing agent were added to the solution and the sample was ready to be applied to CHORUS TRIO.
CHORUS VIT B12 is an immunoassay kit for the automated quantitative determination of vitamin B12. The test was performed on human serum using a disposable device and the CHORUS TRIO instrument, according to the manufacturer’s instructions. Briefly, it is a competitive enzyme-linked immunoassay: samples and a biotinylated anti-B12 antibody are incubated into streptavidin-coated microwells; during the incubation, the complex between vitamin B12 and anti-B12 antibody binds streptavidin; then, a vitamin B12 conjugated with peroxidase is added to compete with the vitamin B12 present in the sample; after adding peroxidase substrate to develop the quantitative reaction, in the CHORUS TRIO instrument gives the results obtained for each sample and the amount of vitamin B12 revealed is inversely correlated to the color intensity.
CHORUS FOLATE is a ready-to-use assay for determination of folate on human serum. It is based on the same principle of a competitive enzyme-linked immunoassay. Briefly, samples and a folate-binding biotinylated protein are incubated into streptavidin-coated microwells; during the incubation, the complex between folate and folate-binding protein binds streptavidin; then, a folate conjugated with peroxidase is added to compete with the folate present in the sample; after adding peroxidase substrate to develop the quantitative reaction between folate and peroxidase conjugated folate on specific binding sites of folate binding protein. The peroxidase substrate develops a quantitative reaction, inversely correlated to human folate present in the serum.
Endpoint and validation procedures
The endpoint is to determine performance and reliability in quantifying vitamin B12 and folate on serum samples of patients the following validation procedures were performed.
The Limit of Detection (LoD) was measured according to the EP17-A2 approved guideline second edition CLSI (Clinical and Laboratory Standards Institute) 12 setting the accuracy goal at 15%. Each serum sample was run in triplicate in two CHORUS TRIO instruments for 3 days in both lots. A total of 72 total measurements (72 devices) were obtained for each tested lot. The normality of methods was verified by Skewness and Kurtosis tests to select between a non-parametric or a parametric approach to analyze the results, according to guidelines. In both cases, the LoD was set as the maximal value obtained for each reagent lot and the mOD value was, then, transformed into pg/mL.
The Limit of Quantitation (LoQ) was determined in serum samples which were run in triplicate in two CHORUS TRIO instruments for 3 days in both lots. The procedure was repeated for 3 days, with a total of 72 measurements (72 devices) for each tested lot. The greatest LoQ across the two tested lots was taken as the LoQ of the method.
To determine the linearity each sample was tested once, and the results were plotted on an XY graph where the horizontal axis represented the expected values and the vertical axis the results; the graph was visually examined to assess the presence of non-linearity and potential outliers. For accuracy, International Standard 03/178 was tested on two lots. The mean recovery compared to the expected value was calculated.
Precision and repeatability were assayed by using samples at different concentrations. For the intra-assay precision, the analysis was performed with one lot on one CHORUS instrument by one operator. Each analyte concentration was tested in six replicates. For the inter-assay precision, each analyte concentration was tested over six runs. The inter-lot precision was evaluated with three lots of the kit on one CHORUS TRIO instrument by one operator; each sample was tested over six runs; the inter-instrument precision was analyzed with one lot of the kit on 3 CHORUS TRIO instruments by one operator and each sample was tested over six runs with the three instruments. In all experiments, one control sample was added to judge the acceptability of the run. A coefficient of variability (CV) ≤15% was accepted.
The analytic specificity was evaluated in three samples at increasing concentrations, in which the following interfering components were added: bilirubin (4.5–45 mg/dl), triglycerides (250–1500 mg/dl), hemoglobin (2.5–10 mg/mL), rheumatoid factor (44–220 IU/mL). Furthermore, a sample of vitamin B12 with an increasing biotin content (0.5–1000.0 ng/mL) was tested; the normal concentration of biotin in human serum is 35 ng/mL.
CHORUS kit comparison with commercially available kit
CHORUS kit and commercially available COBAS kit (predicate device, Hoffmann-La Roche, Basel, Switzerland) were compared. Each kit was used according to the manufacturer’s instructions; each sample was tested once with the CHORUS kit and once with the COBAS Device. The comparison was intended for a single batch of CHORUS and COBAS kits.
Sample size and statistical analysis
Using the two-sided test of the angular coefficient of the univariate regression to be equal to 1 -the two tests must give the same concentration value and the regression line must result as a bisector-, with alpha = 0.05 and a power of 0.90, a sample size of 500 was estimated (G*Power software).
Qualitative variables were summarized with absolute frequencies and percentages, while quantitative ones with median and interquartile range (IQR). To compare CHORUS VIT B12 or CHORUS FOLATE and COBAS® methods, Spearman’s correlation coefficient, Passing Bablok, and Bland-Altman analyses were performed. Gwet Ac1 with their 95% confidence interval (95% CI) was used to estimate the degree of concordance. A p-value < 0.05 was considered statistically significant. All analyses were performed using MedCalc® Statistical Software version 20.115 (MedCalc Software Ltd, Ostend, Belgium).
Results
Validation and determination of precision and reliability
The reference range of CHORUS VIT B12 was 237.5–764.8 pg/mL and LoD was 147.5 pg/mL, with a LLoQ of 165.0 pg/mL and an ULoQ of 1846.8 pg/mL. Since the CHORUS system (reagent and instrument) gave a result within the calibration range between 150.0 and 2000.0 pg/mL, LoD reported by CHORUS TRIO was <150.0 pg/mL. Testing three positive samples with the CHORUS VIT B12 kit demonstrated the linearity of the method from 150.0 to 2000.0 pg/mL. Testing the International Standard vitamin B12, Serum Folate, HOLO TC (NIBSC code: 03/178), which has a target value for vitamin B12 of 480 pg/mL, we demonstrated test accuracy considering that the mean recovery on two lots was 111%.
Table 1 summarizes the results of precision tests. All the obtained values were within the expected range, meeting the specification of CV% ≤15%. The values obtained during the precision evaluation experiment confirmed the device/method precision and repeatability of the CHORUS VIT B12 kit. The presence of interfering substances (Bilirubin, Triglycerides, Hemoglobin and Rheumatoid factor) was tested on three samples with increasing concentration of vitamin B12.
Precision and repeatability of CHORUS VIT B12.
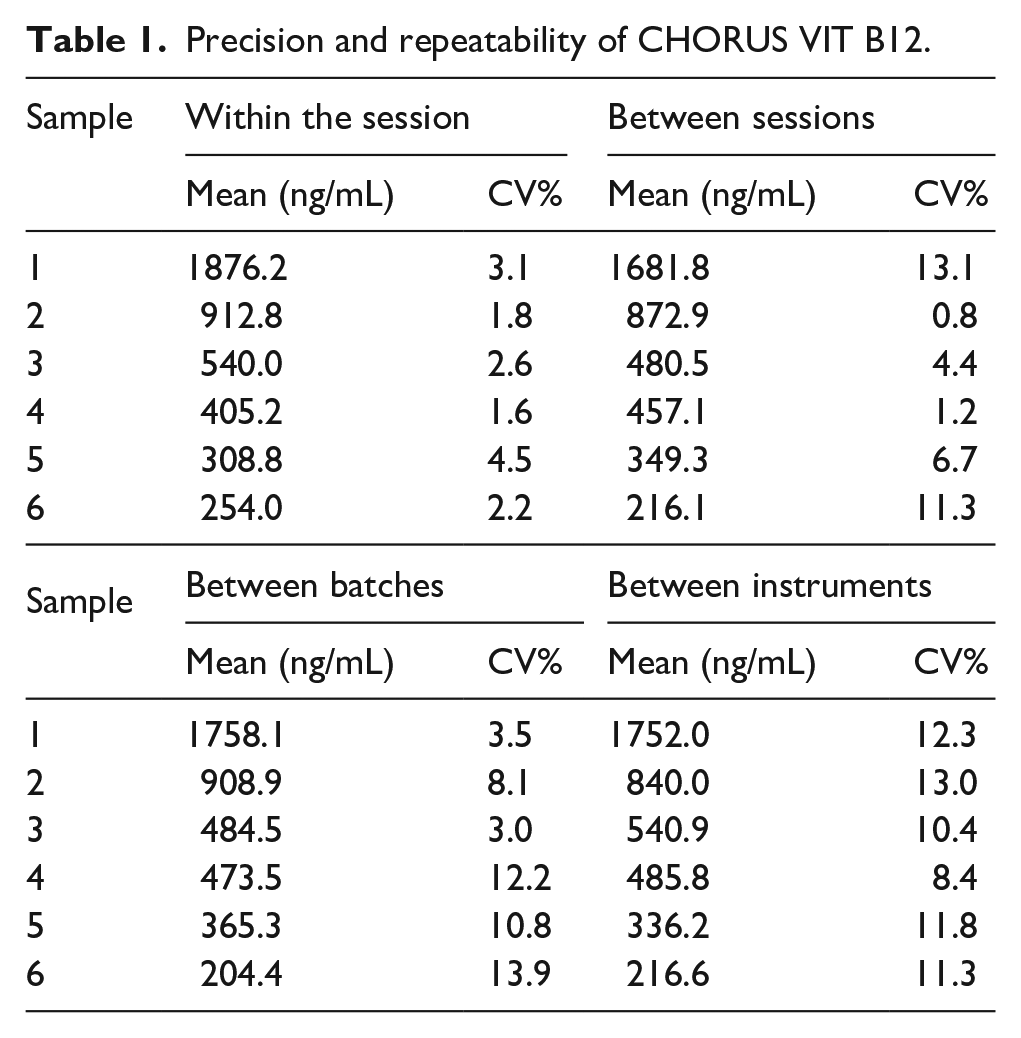
The presence of the above listed interfering substances in the serum under examination does not alter the test result, with the exception of hemoglobin. One sample with a vitamin B12 concentration in the normal range was tested, to which increasing concentrations of biotin (0.5–1000.0 ng/mL) were added. The presence of biotin in the test serum did not alter the test result for concentrations up to 35 ng/mL; higher concentrations of biotin lead to incorrect results.
The reference range of the CHORUS FOLATE kit was 2.8–14.9 ng/mL, with LoD of 1.4 ng/mL. LLoQ was 2.0 ng/mL and ULoQ was 19.6 ng/mL. The linearity of the method was demonstrated from 2.4 to 20.0 ng/mL, by testing three positive samples; the accuracy was described with the International Standard vitamin B12, Serum Folate, HOLO TC (NIBSC code: 03/178), considering that the mean recovery on three lots was 92%.
Table 2 describes the result of the precision and repeatability of the method, and all the obtained values were within the expected range, with the specification of CV% ≤15%.
Precision and repeatability of CHORUS FOLATE.

The presence of the interfering substances in the test serum did not alter the result, except for hemoglobin and triglycerides, assessed at higher concentrations than that physiologically present. Two samples were tested with folate concentrations in the normal range, to which increasing concentrations of biotin (0.5–1000.0 ng/mL) were added. The presence of biotin in the test serum did not alter the test result for concentrations up to 10 ng/mL; higher biotin concentrations may lead to incorrect results.
Comparison with the COBAS kit
CHORUS VIT B12 kit and CHORUS FOLATE kit was compared with the COBAS kit on 500 human sera. Table 3 summarizes the results. The lowest and highest values of both CHORUS and COBAS kits were similar and the median values did not significantly vary.
Comparison between CHORUS and COBAS kits.

For CHORUS VIT B12, the Passing Bablock equation was: y = −15.693 + 1.089x (Figure 1) with a Spearman’s correlation coefficient of 0.865 (95% CI: 0.841–0.885); in the Bland-Altman test the mean difference was −31.500 (95% CI: −42.963 to −20.038) with upper and lower limits of 224.190 and −287.192, respectively. Gwet Ac1 was 0.85 (95% CI:0.814–0.886) in this way the concordance was adequate.

Passing Bablock (a) and Bland Altman (lower limit −287.1887 and upper limit 224.1931) (b) plots of the comparison between CHORUS VIT B12 and COBAS kits.
For CHORUS FOLATE, the Passing Bablock equation was y = −0.694 + 1.093x (Figure 2) with a Spearman’s correlation coefficient of 0.866 (95% CI: 0.843–0.887); in the Bland-Altman mean difference was −0.114 (95% CI: −0.274 to 0.047) with upper and lower limits of 3.473 and ─3.700, respectively. Even in this case, some outliers within the highest values were present. Gwet Ac1 was 0.92 (95% CI:0.888–0.943) in this way the concordance was adequate.
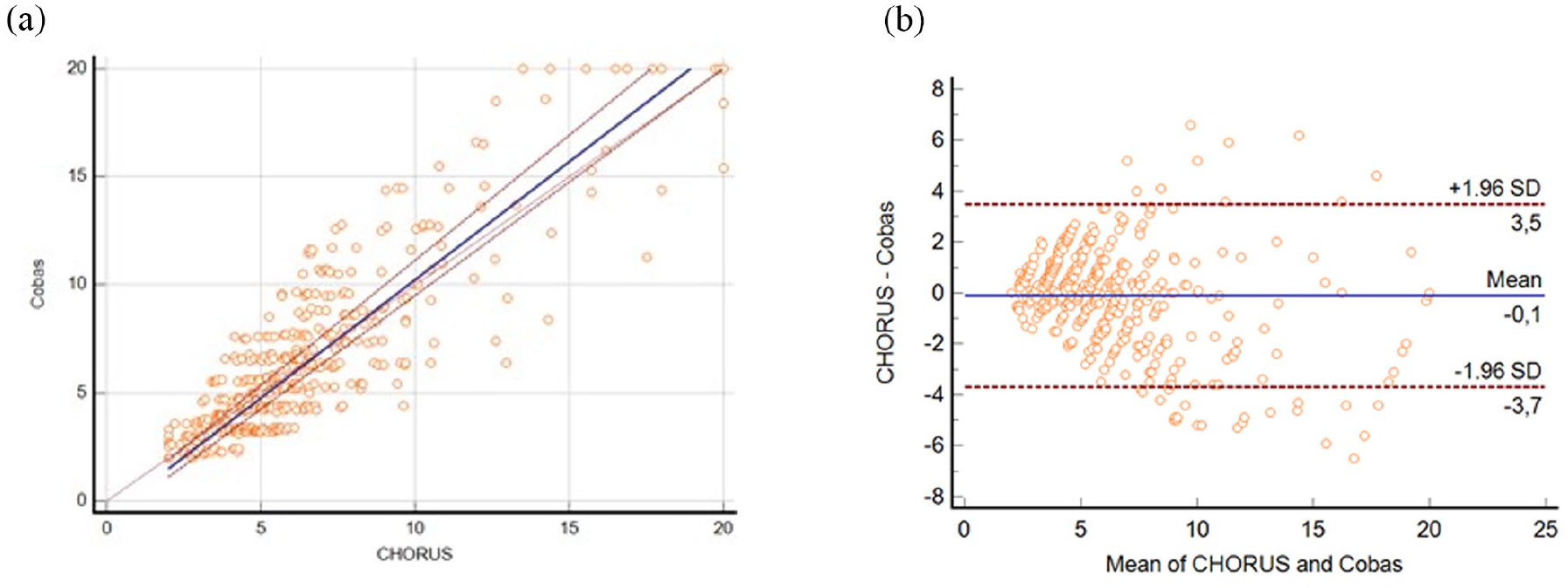
Passing Bablock (a) and Bland Altman (lower limit −3.7002 and upper limit 3.4710) (b) plot of the comparison between CHORUS FOLATE and COBAS kits.
Discussion
This work was aimed at describing the validation, performances, and reliability of two automated tests for vitamin B12 and folate assay. The results demonstrated that CHORUS VIT B12 and CHORUS FOLATE kits were accurate and reliable to quantify vitamin B12 and folate, respectively.
For CHORUS VIT B12, both lower and upper LoQ were within the calibration range of the instrument and reagents, thus ensuring that CHORUS TRIO could accurately determine the concentration of serum vitamin B12. The assay was linear within the calibration range and specific and hadn’t been affected by other serum components -bilirubin, triglycerides, and rheumatoid factor- that may interfere with the assessment of vitamin B12; only hemoglobin concentrations higher than physiological levels altered the results.
For serum folate testing, a single threshold, set at a lower reference limit, is usually employed to screen for deficiency. Therefore, the prevalence of folate deficiency is strictly dependent on the correctness of cut-off establishment and the accuracy of folate assays. CHORUS FOLATE assay was linear within the reference range and accurate.
Once the accuracy and performance of both CHORUS VIT B12 and CHORUS FOLATE assays were determined, the kits were compared with a commercially available assay to assess the consistency of results and inter-assay variability. Indeed, as the standardization of vitamin B12 and serum folate assay methods has not been achieved yet, it is important to compare the different assays to identify potential variability between them. Furthermore, evaluating the inter-assay variability of B12 measurement at serum concentrations around the established thresholds for the risk of deficiency may be useful in indicating if the established cut-offs may be used or require an adjustment according to the employed method. 11 According to guidelines 12 and knowing that some substances as biotin could interfere with the assays, both CHORUS VIT B12 and CHORUS FOLATE were tested in presence of interfering components which, however, did not alter the clinical significance of results.
A comparison study of vitamin B12 assay included Roche COBAS and indicated an agreement between Roche COBAS and other immunoassay autoanalyzers, specifically DxI 800 Unicel (Beckman Coulter, USA), ADVIA Centaur XP (Siemens Diagnostics, Tarrytown, NY, USA), Architect i2000sr (Abbott Laboratories, Abbott Park, IL, USA). 13 In our comparison we observed high agreement between CHORUS kits and COBAS; some outliers were present, prevalently within the highest values and in the case of hypervitaminosis and these results did not affect the clinical utility of the assay; in the study we did not include visually haemolysed samples.
Although World Health Organization (WHO) Expert Committee on Biological Standardisation established 03/178 as an International Standard for serum vitamin B12 and serum folate to standardize serum vitamin B12 and folate assays 14 and the agreement between some assays had been demonstrated, laboratory professionals and clinicians should be aware that some disagreement between assay methods and threshold value differences may be present. 13
CHORUS VIT B12 and CHORUS FOLATE have been shown to perform well, accurately, and reliably in quantifying vitamin B12 and folate in human serum. Linearity within the calibration range and lack of interference from other substances in serum make these assays reliable in clinical practice for determining vitamin deficiencies. The easy-to-use completely automated system, which reduces operator intervention minimizing the possibility of manual errors, may represent a further advantage to reduce the variability of the assays.
Significance for public health
Both in childhood and in the adulthood, vitamin B12 and folate deficiencies are associated with pathological manifestations, such as neurological signs in children and metabolic syndrome in adults. Therefore, given the clinical importance of vitamin B12 and folate, it is necessary to accurately quantify their serum levels, especially in presence of symptoms that might suggest an alteration. Vitamin B12 and folate quantification is a commonly used test; the availability of an automated test, fast, and easy-to-use provides a significant advantage for public health, reducing the possibility of manual errors and assay variability. CHORUS VIT B12 and CHORUS FOLATE kits are two automated tests for vitamin B12 and folate quantification. These tests resulted accurate and reliable to determine vitamin B12 and folate, respectively, and may become a useful tool for clinical practice.
Footnotes
Acknowledgements
The authors thank Content Ed Net for editorial support, with the helpful contribution of medical writer Elisa Sala, Ph.D.
Author contributions
Simone Bianciardi, Giulia Tesi, Helena Cerutti, Alessandra Cartocci, Tommaso Bandini, Roberto Guerranti, Alessandra Brogi, Roberto Leoncini contributed to the conception and design of the study; Simone Bianciardi, Caterina Silvestrini, Sabrina Gori were involved in clinical evaluation; Simone Bianciardi, Giulia Tesi, Helena Cerutti, Alessandra Cartocci, Roberto Guerranti, Alessandra Brogi, Roberto Leoncini interpreted the results; Giulia Tesi, Helena Cerutti, Alessandra Cartocci, performed the statistical analysis; Simone Bianciardi, Giulia Tesi, Helena Cerutti, Alessandra Cartocci, Roberto Guerranti, Tommaso Bandini, Alessandra Brogi, Roberto Leoncini drafted the manuscript; and Alessandra Brogi, Roberto Leoncini supervised the study. All authors read and approved the final manuscript.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Cerutti H, Tesi G, Bianciardi S, Bandini T, Brogi A declare to be “DIESSE Diagnostica Senese S.p.A. Società Benefit” employees. No other conflict of interest has been reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DIESSE Diagnostica Senese S.p.A. Società Benefit funded the study and the editorial assistance.
