Abstract
Objective:
We aimed to review the available evidence on the association between vitamin B12, folate, and homocysteine levels with worse outcomes among COVID-19 patients.
Methods:
The search was carried out in ten databases simultaneously run on 10 May 2023, without language restrictions. We included cross-sectional, case-control, and cohort studies. The random-effects meta-analysis was performed using the Sidik-Jonkman method and corrected 95% confidence intervals using the truncated Knapp-Hartung standard errors. Standardized mean difference and 95% CI was used as the measure effect size.
Results:
Thirteen articles were included in this review (n = 2134). Patients with COVID-19 who did not survive had the highest serum vitamin B12 values (SMD: 1.05; 95% CI: 0.31–1.78; p = 0.01, I2 = 91.22%). In contrast, low serum folate values were associated with patients with severe COVID-19 (SMD: −0.77; 95% CI: −1.35 to −0.19; p = 0.02, I2 = 59.09%). The remaining tested differences did not yield significant results.
Conclusion:
Elevated serum levels of vitamin B12 were associated with higher mortality in patients with COVID-19. Severe cases of COVID-19 were associated with low serum folate levels. Future studies should incorporate a larger sample size.
Introduction
The majority of health systems worldwide have demonstrated their fragility in the face of the COVID-19 pandemic. 1 Approximately 770 million cases and more than 6 million deaths have been confirmed worldwide by October 2023. 2 It is devastating effect on human health would be related to the uncontrolled immune response. Indeed, a systematic review found that increased cytokine levels posed a risk factor for worse outcomes in COVID-19 patients. 3 In this scenario, due to the regulating role of vitamins on immunity, they may influence clinical outcomes among infected patients.
The effects of vitamins go beyond boosting the immune system, as they may improve the symptoms of COVID-19 or perhaps treat it. Homocysteine (Hcy) and its cofactors, vitamin B12 and folate, are involved in crucial biological processes. Vitamin B12 is involved in the regulation of the immune system and antiviral activity, inhibiting inflammation by modulating growth factors, cytokines, and other substrates with anti-inflammatory properties. 4 Folic acid (vitamin B9) inhibits furin, causing the virus to be unable to enter the cell because it prevents the binding of the angiotensin-converting enzyme 2 receptor to the SARS-CoV-2 spike protein on the cell surface. 5 Both cell-mediated and humoral immunity are affected in folate and vitamin B12 deficiency states. 6 Overall, the deficiency of either folate or B12 leads to an increase in Hcy, a sulfur amino acid. In high levels, it exerts deleterious effects on the vascular endothelium and induces excitotoxic effects in immune-competent cells.7,8 As well, it increases serum levels of inflammatory proteins and other cytokines.9,10 All in all, high and low levels of these macromolecules can drastically alter the immune response, predisposing to worse outcomes among COVID-19 patients.
Despite the vitamin supplementation in COVID-19 patients has not yet been proven to be effective in reducing the risk of infection, the basal levels may influence the clinical outcomes.11,12 As well as certain biomarkers, vitamins may predict clinical endpoints among infected patients.13–17 Indeed, two systematic reviews found that low vitamin levels aggravated the clinical status.18,19 High serum levels of Hcy, as well as low levels of vitamin B12 and folate, have been associated with worse clinical outcomes; however, the studies are not conclusive.19,20 Hence, we aimed to conduct a systematic review to evaluate the association between serum levels of Hcy, Vitamin B12, and folate in the mortality and severity among adults with COVID-19.
Methodology
Report guidelines and protocol register
The protocol of our systematic review was registered in the International Prospective Register of Systematic Reviews (PROSPERO) with registration number CRD42022361678. We followed the methodological guidelines of the Cochrane Handbook, and for writing the manuscript, we adhered to the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) statement. 21
Search strategy
The search strategy was built following the guidelines of the Peer Review of Electronic Search Strategies (PRESS). 22 We developed a formula for Pubmed using MeSH and free terms. Then, that formula was adapted for the other search engines. Supplemental Table S1 displays the search formulas used.
Systematic search and databases
The systematic search was simultaneously run in all search engines on 10 May 2023. We did not apply language restrictions for this systematic review. We searched through the following databases: Scopus, Web of Science, PubMed, Embase, and Ovid Medline. Likewise, we performed a manual search in CINAHL, China National Knowledge Infrastructure, Wangfang Data, Research Square, and medRxiv.
Eligibility criteria
We sought studies that evaluated the values of vitamin B12, folate, or homocysteine with the mortality or severity of COVID-19 patients. Inclusion criteria were: (i) case-control, cross-sectional, and cohort studies, (ii) studies conducted in adult patients (⩾18 years), and (iii) studies that assessed the values of vitamin B12, folate, or Hcy in the mortality or severity of COVID-19 patients. We excluded narrative reviews, conference abstracts, systematic reviews, duplicated studies, and scoping reviews. Our primary outcome was the COVID-19 severity, and COVID-19 mortality was the secondary outcome.
Study selection process and data extraction
We use Rayyan QCRI (Rayyan Systems Inc©) to collect records from search engines and to remove duplicate records. 23 The Rayyan software was also used for screening by title and abstract. This process was independently performed by four authors (A.A-C, E.A.H-B, E.A.A-B, and J.R.U-B). The full text from the remaining records was independently reviewed by these authors. We resolved by consensus any conflict or discrepancy concerning the inclusion of articles. Then, two authors (J.C.C-G and M.A.H-C) extracted independently the data from each of the included studies in a pre-set Google Sheets©. In the same way, any conflict was resolved by consensus. Extracted data were: first author, publication date, study design, country, data source, sample size, sex, age, and the values of the markers in each outcome.
Risk of bias and publication bias
The Newcastle-Ottawa Scale (NOS) 24 was used for case-control and cohort studies. In addition, cross-sectional studies were evaluated using the adjusted NOS version for cross-sectional studies (NOS-CS). 25 We categorized the risk of bias as either having a high risk of bias (<7 stars) or a low risk of bias (⩾7 stars).
The Begg test and funnel plots were used for assessing publication bias. The p-values < 0.1 in Begg test were considered indicative of publication bias. There has to be a minimum of 10 articles in the quantitative synthesis for the assessment using funnel plots.
Data synthesis and analysis
We used STATA 17.0 (College Station, TX, USA: Stata Press©) for the statistical analysis. Standardized mean difference (SMD) and 95% confidence intervals (CIs) were used as the only effect size. The values expressed as medians and interquartile ranges (IQRs) were transformed into means and standard deviations (SDs) using Hozo’s method. 26 We carried out a random-effects meta-analysis using the Sidik-Jonkman method and corrected 95% CI using the truncated Knapp-Hartung standard errors.27,28 We analyzed the statistical heterogeneity in the meta-analysis using Cochran’s Q test and the I2 statistic. The p-values > 0.1 in the Cochran’s Q test were considered as no heterogeneity. 29 The values for I2 statistic ⩾60% were considered as severe heterogeneity. We conducted subgroup analyses according to continents. A sensitivity analysis was conducted, excluding studies that were previously identified as having a high risk of bias. The p-value < 0.05 was set as the cut-off point of statistical significance for the meta-analyses.
Results
Search results
The systematic search identified 2832 records. After removing duplicates, 632 records were screened by title and abstract. Next, 28 records were selected for full-text review, and only 13 were deemed eligible for the synthesis.30–42 The article by Hemdan et al. 32 presents information on vitamin B12 values in two different groups (males group and females group). The PRISMA flow chart of studies selection is shown in Figure 1.

PRISMA flow diagram.
Study characteristics
Thirteen studies were included; eight were cross-sectional, four cohorts, and one case-control study. Studies were conducted across various countries, including Turkey (three studies), Russia (two studies), Iraq (two studies), Israel (one study), Italy (one study), Iran (one study), Saudi Arabia (one study), Switzerland (one study), and China (one study). Regarding the outcomes, six studies examined only severity, and seven studies evaluated only mortality. Two thousand one hundred thirty-four adults infected with COVID-19 were evaluated (1071 men and 859 women). One study did not provide information about the sex of the participants. 40 These characteristics are summarized in Tables 1 and 2.
Characteristics of the included studies that evaluated severity.

NR: not reported; CLIA: chemiluminescence immunoassay; ECLIA: electrochemiluminescence immunoassay; ELISA: enzyme-linked immune-sorbent assay.
Characteristics of the included studies that evaluated mortality.

NR: Not reported; CLIA: Chemiluminescence immunoassay; ECLIA: Electrochemiluminescence immunoassay; ELISA: Enzyme-linked immune-sorbent assay.
Using NOS and NOS-C scales, the low risk of bias was present in ten studies, and three studies had high risk of bias (Supplemental Table S2).
Vitamin B12 values and COVID-19 severity
Four studies (n = 380) evaluated vitamin B12 values and COVID-19 severity association. The meta-analysis showed no statistical significance in vitamin B12 levels and COVID-19 severity. (SMD: 1.48; 95% CI: −2.97 to 5.91; p = 0.37, I2 = 99%) (Figure 2). The small number of articles included did not allow conducting a subgroup analysis. The sensitivity analysis further confirmed the lack of significant difference, with persistent, significant heterogeneity observed (SMD: 2.21; 95% CI: −5.03 to 9.45; p = 0.32, I2 = 98.9%) (Supplemental Figure S1).

Vitamin B12 values in severe vs non-severe COVID-19 patients.
Vitamin B12 values and COVID-19 mortality
Six studies (n = 1179) evaluated vitamin B12 values and COVID-19 mortality association. The meta-analysis showed that adults with COVID-19 who did not survive had higher vitamin B12 values compared to those who survived (SMD: 1.05; 95% CI: 0.31–1.78; p = 0.01, I2 = 91.22%) (Figure 3). In the subgroup analysis based on continents (Supplemental Figure S2), there were no significant differences in any of the subgroups. In the sensitivity analysis, the difference remained statistically significant and had high heterogeneity (SMD: 1.07; 95% CI: −0.27 to 2.42; p < 0.05, I2 = 95.47%) (Supplemental Figure S3).

Vitamin B12 values in survivors versus non-survivors COVID-19 patients.
Folate values and COVID-19 severity
Four studies (n = 380) evaluated folate values and COVID-19 severity association. The meta-analysis showed that lower folate values are present in adults with severe COVID-19 (SMD: −0.77; 95% CI: −1.35 to −0.19; p = 0.02, I2 = 59.09%) (Figure 4). The small number of articles included did not allow conducting a subgroup analysis. The sensitivity analysis confirmed the difference being statistically significant with high heterogeneity (SMD: −0.7; 95% CI: −1.69 to −0.29; p = 0.04, I2 = 63.91%) (Supplemental Figure S4).
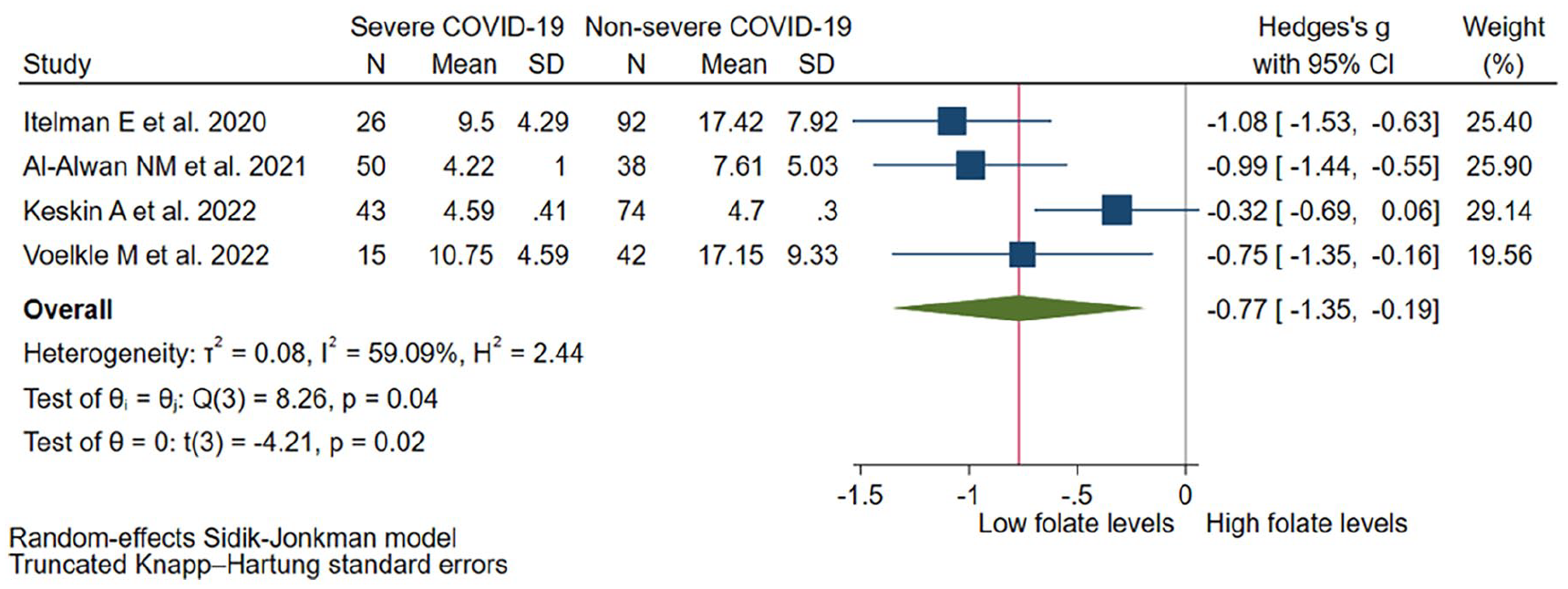
Folate values in severe vs non-severe COVID-19 patients.
Folate values and COVID-19 mortality
Three studies (n = 888) evaluated folate values and COVID-19 mortality association. The meta-analysis showed no statistically significant differences between folate levels and COVID-19 mortality (SMD: −0.39; 95% CI: −1.18 to 0.39; p = 0.16, I2 = 62.8%) (Figure 5). The small number of articles included did not allow conducting a subgroup and sensitivity analysis.

Folate values in survivors versus non-survivors COVID-19 patients.
Homocysteine values and COVID-19 severity
Four studies (n = 534) evaluated homocysteine values and COVID-19 severity association. The meta-analysis showed no statistically significant differences between homocysteine levels and the severity of COVID-19. (SMD: 1.69; 95% CI: −2.39 to 5.76; p = 0.28, I2 = 99.4%) (Figure 6). The small number of articles included did not allow conducting a subgroup analysis. The sensitivity analysis did not show statistically significant differences either (SMD: 2.57; 95% CI: −3.08 to 8.23; p > 0.05, I2 = 98.67%) (Supplemental Figure S5).

Homocysteine values in severe versus non-severe COVID-19 patients.
Homocysteine values and COVID-19 mortality
Three studies (n = 295) evaluated homocysteine values and COVID-19 mortality association. The meta-analysis showed no statistically significant differences between homocysteine levels and COVID-19 mortality (SMD: 0.56; 95% CI: −1.67 to 2.79; p = 0.34, I2 = 90.5%) (Figure 7). The small number of articles included did not allow conducting a subgroup and sensitivity analysis.

Homocysteine values in survivors versus non-survivors COVID-19 patients.
Publication bias
Due to the small number of included studies (<5 studies), only the Begg test was conducted for the assessment of vitamin B12 and mortality of COVID-19 patients, and no evidence of publication bias was found (p > 0.1). Furthermore, the unavailability of sufficient studies of the outcomes had more than 10 studies for each outcome–impeded the generation of funnel plots.
Discussion
Our study aimed to synthesize the available evidence on vitamin B12, folate, and Hcy levels and their association with COVID-19 mortality and severity in adults. Non-survivor patients with COVID-19 exhibited higher vitamin B12 levels compared to survivor patients, while patients with severe COVID-19 had lower folate levels in comparison to non-severe COVID-19 patients. There were no statistically significant differences in vitamin B12 and Hcy levels concerning COVID-19 severity. Similarly, there were no significant associations found between folate and Hcy levels in relation to COVID-19 mortality. However, it is important to note that the associations identified are characterized by substantial heterogeneity, and the number of included studies was limited.
Previous systematic reviews have assessed the role of another vitamin on clinical outcomes of patients infected with COVID-19, 18 and the risk of acquiring infection.43,44 In addition, there are systematic reviews that have evaluated the supplementation of these vitamins in COVID-19 patients, although no favorable results were found.45,46 Nevertheless, to the best of our knowledge, this is the first study to systematize the available evidence of vitamin B12, folate, and Hcy on mortality and severity of COVID-19. Although the data on the efficacy of vitamins for COVID-19 treatment is inconsistent, vitamins have been proposed as viable therapeutic options with plausible mechanisms of action. 47 To date, more than 3 years after the start of the pandemic, well-designed clinical trials and even observational studies are still lacking.
Non-survivor COVID-19 patients showed significantly elevated levels of vitamin B12 compared to those who survived. The mechanisms by which excess vitamin B12 is associated with increased mortality in patients with COVID-19 are not fully understood; however, these results are not surprising. Similar results have been found in patients in the general population 48 and in patients in an intensive care unit. 49 In these patients, various explanations have been proposed that could also explain our results. High vitamin B12 plasma concentrations may represent a response to increased release of vitamin B12 from liver storage, decreased clearance, upregulation of haptocorrin and transcobalamin synthesis, or diminished affinity of vitamin B12 for transporter proteins.50,51 Those situations are often present because of liver damage or chronic kidney disease, which could be represented by the baseline association of high plasma concentrations of vitamin B12 with elevated concentrations of hepatic enzymes.50,51 Likewise, increased plasma cobalamin might be related to functional deficiency. The high cobalamin levels are not a guarantee for normal physiological function, so decreased intracellular cobalamin levels may parallel increased plasma levels. 52 In this sense, evaluation of the functional cobalamin status can be achieved by measuring the activities of the cobalamin-dependent enzymes. 52
Patients who experienced severe COVID-19 exhibited lower folate levels compared to those with non-severe COVID-19. However, it is important to note that this conclusion is based on a pooled estimate derived from the analysis of only 380 subjects. As a result, the generalizability of these findings is limited; the study is prone to inflated type 1 errors and imprecise estimation of heterogeneity between studies. 53 Consequently, careful consideration is necessary when interpreting our results. Nevertheless, some mechanisms could explain this association. Some studies have demonstrated that the binding of SARS-CoV-2 spike proteins, which facilitate the entry of the virus into cells, can be hindered by folic acid.54–56 All in all, to confirm our findings, future studies should incorporate a larger sample size.
Homocysteine is a biomarker related to many diseases; its high serum levels have been mainly related to cardiovascular, neurological, and metabolic diseases. 57 Homocysteine is produced as part of the metabolism of methionine. Elevated serum levels of homocysteine are associated with vitamin B6, B12, and folate deficiency, genetic polymorphisms, kidney failure, chronic inflammatory diseases such as rheumatoid arthritis or inflammatory bowel disease, and the intake of certain medications such as metformin or methotrexate. 58 Various studies relate high serum homocysteine values to mortality from all causes. 59 The toxic effect of hyperhomocystinemia is related to damage to the vascular endothelium. Cellular dysfunction in the endothelium causes a state of hypercoagulability due to platelet activation and thrombus formation. 60 It is believed that hyperhomocysteinemia may limit the bioavailability of nitric oxide by changing the properties of the elastic wall due to smooth cell proliferation. 61 In relation to SARS-Cov-2 infection, it is well known that the spike protein binds to angiotensin-converting enzyme 2 (ACE2) and uses it as a receptor to enter cells, especially lung cells. 62 This binding with ACE2 would lead to a pro-inflammatory state and activation of a cytokine cascade. 63 Likewise, SARS-Cov-2 would be involved in the activation of the angiotensin type 1 (AT1) receptor, which has opposite biological effects to ACE2. It has been postulated that hyperhomocysteinemia could generate activation of AT1. Likewise, homocysteine has three biologically active forms (reduced, oxidized, and free) that could interact differently with the AT1 receptor and generate different cardiovascular effects.
In our meta-analysis of four studies, we found no association between serum homocysteine levels and COVID-19 mortality or severity and showed critical heterogeneity. Studies such as that of Yang et al. 38 and Kenkin et al. 30 found higher serum homocysteine values in patients with severe disease; in the other two studies evaluated, one shows opposite findings, and the other shows no association.37,42 Yang et al. 38 explains their findings because homocysteine values are related to progression on chest computed tomography (CT). However, attributing only the progression of chest CT to disease severity, given the presence of multiple related factors, may be controversial. In relation to mortality, the meta-analysis of three studies shows no association between serum homocysteine levels and mortality and presents critical heterogeneity, and only Ali et al. 39 study does not show an association between homocysteine and mortality. Despite these findings, the evidence is not conclusive due to the small number of studies found and the heterogeneity. Likewise, studies are necessary that can define a cut-off point for hyperhomocysteinemia to establish better comparisons with mortality and severity.
Strengths and limitations
This study has limitations. First, due to the limited number of included studies, subgroup and sensitivity analyses were only performed when evaluating the association between vitamin B12 and COVID-19 outcomes. Moreover, the statistical power was low, and the studies were conducted in Asian and European countries. In this regard, it is not possible to extrapolate our findings to other populations. Second, due to the cross-sectional nature of the majority of the articles included, establishing causality was not possible. Thus, our findings are merely constrained. Third, there was high heterogeneity in the assessment of the association between vitamin B12 values and COVID-19 severity and mortality, which may introduce potential sources of bias and reduce the overall reliability and generalizability of the findings. Future studies should perform subgroup analyses according to the study design and measurement method in an attempt to reduce heterogeneity. On the other hand, our study has some strengths. Various databases were searched, ensuring a comprehensive synthesis of the existing evidence. To our current knowledge, this study represents the initial attempt to systematically gather and analyze the existing evidence on the association of vitamin B12, folate, and Hcy levels on the mortality and severity of COVID-19 patients.
Conclusion
In COVID-19 patients, non-survivors had higher vitamin B12 levels compared to survivors, and severe cases exhibited lower folate levels compared to non-severe cases. Nevertheless, the severity of COVID-19 did not yield significant differences in the levels of vitamin B12 and Hcy in the bloodstream. Similarly, the levels of folate and Hcy did not demonstrate significant associations with COVID-19 mortality. Further studies are warranted to explore other potential factors influencing vitamin B12, Hcy, and folate levels in relation to COVID-19 severity and mortality. Possible factors to consider include genetic variations, dietary patterns, and the impact of comorbidities. Additionally, longitudinal studies examining the dynamics of these vitamins throughout the course of COVID-19 infection will larger sample sizes would provide valuable insights into their clinical significance and potential therapeutic interventions.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241253957 – Supplemental material for Association of vitamin B12, folate, and homocysteine with COVID-19 severity and mortality: A systematic review and meta-analysis
Supplemental material, sj-docx-1-smo-10.1177_20503121241253957 for Association of vitamin B12, folate, and homocysteine with COVID-19 severity and mortality: A systematic review and meta-analysis by Juan R Ulloque-Badaracco, Ali Al-Kassab-Córdova, Esteban A Alarcon-Braga, Enrique A Hernandez-Bustamante, Miguel A Huayta-Cortez, Juan C Cabrera-Guzmán, Pamela Robles-Valcarcel and Vicente A Benites-Zapata in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241253957 – Supplemental material for Association of vitamin B12, folate, and homocysteine with COVID-19 severity and mortality: A systematic review and meta-analysis
Supplemental material, sj-docx-2-smo-10.1177_20503121241253957 for Association of vitamin B12, folate, and homocysteine with COVID-19 severity and mortality: A systematic review and meta-analysis by Juan R Ulloque-Badaracco, Ali Al-Kassab-Córdova, Esteban A Alarcon-Braga, Enrique A Hernandez-Bustamante, Miguel A Huayta-Cortez, Juan C Cabrera-Guzmán, Pamela Robles-Valcarcel and Vicente A Benites-Zapata in SAGE Open Medicine
Footnotes
Acknowledgements
None.
Author contributions
Conceptualization, V.A.B.-Z and J.R.U-B; Data curation, M.A.H-C, J.C.C-G, A.A-K-C, E.A.H-B, E.A.A-B, and J.R.U-B; Formal analysis, V.A.B.-Z, E.A.A-B, and J.R.U-B; Methodology, V.A.B.-Z, E.A.H-B, E.A.A-B, and J.R.U-B.; Writing—original draft, V.A.B-Z, A.A-K-C, M.A.H-C, E.A.H-B, E.A.A-B, P.R-V, and J.R.U-B.; Writing review and editing, V.A.B.-Z, P.R-V, J.R.U-B, and A.A-K-C. All authors have read and agreed to the published version of the manuscript.
Availability of data and materials
All data generated or analyzed during the current study are included in this published article and its supplemental information files.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Not applicable.
Informed consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
