Abstract
The Morinda genus, part of the Rubiaceae family, is a valuable source of diverse bioactive compounds beyond the well-known anthraquinones. This review focuses on the secondary metabolites from Morinda species that do not contain anthraquinone structures, highlighting their chemical variety and biological importance. Important groups such as iridoids, phenolic compounds, coumarins, and polysaccharides are covered in terms of their distribution and effects. Numerous biological activities have been demonstrated by these compounds, notably antioxidant, anti-inflammatory, antimicrobial, antimalarial, neuroprotective, anticancer, antiviral, and immune-enhancing properties contributing to the therapeutic potential of Morinda. By reviewing recent advances in phytochemical research and pharmacological evaluation, this article identifies promising non-anthraquinone molecules for further development in medicine and biotechnology. This work aims to stimulate further exploration of Morinda’s unique natural products for their effective use in modern and traditional healthcare, including preliminary structure–activity relationship (SAR) insights.
Introduction
The Rubiaceae family is the fourth largest among flowering plants, consisting of 600–650 genera and over 13,000 species. 1 Genus Morinda includes flowering plants in the Rubiaceae family. This genus consists of 80 species, which include vines, shrubs, and trees in tropical regions of the world. Many of the Morinda species’ naturally occurring biologically active products have been extensively researched. 2 More than 200 distinct compounds have been identified and isolated from various Morinda species. 3 However, depending on the plant portion, the chemical makeup varies significantly. Traditional medicine has made use of nearly every part of these plants, including their bark, roots, leaves, stems, and fruits, because of their antibacterial, antifungal, anthelminthic, antitumor, anti-inflammatory, analgesic, and immune-stimulating qualities. 4
Anthraquinones represent the most extensively studied class of compounds in Morinda due to their established bioactivities. 3 Despite the prominence of anthraquinone compounds, non-anthraquinone metabolites in Morinda have begun attracting interest due to their promising bioactive effects, yet remain underexplored compared to anthraquinones.
Non-anthraquinone metabolites from the Morinda genus comprise a diverse range of phytochemicals such as iridoids, flavonoids, coumarins, polysaccharides, glycosides, lignans, sterols, and terpenoids. 5 These compounds have shown antioxidant, anti-inflammatory, antimicrobial, immunomodulatory, hepatoprotective, and neuroprotective effects. Some non-anthraquinone compounds, such as polysaccharides and certain iridoids, may have milder toxicity profiles, potentially broadening their applications in health supplements than the better-known anthraquinones. 6
Although anthraquinones have been identified as the predominant phytoconstituents of Morinda species, accumulating evidence indicates that non-anthraquinone metabolites play equally crucial roles in defining the pharmacological potential of this genus. A consolidated understanding of these non-anthraquinone metabolites is essential for broadening perspectives on the chemical ecology, therapeutic promise, and future applications of Morinda. Therefore, this review aims to systematically compile the non-anthraquinone constituents of Morinda species, highlighting their biological relevance and opportunities for future investigations.
Materials and Methods
Electronic literature searches were conducted in Scopus, Web of Science, Google Scholar, SciFinder, PubChem, and PubMed, using combinations of controlled terms and free-text keywords related to the genus Morinda and non-anthraquinone constituents. In addition, a variety of publications and relevant book chapters were reviewed. The core search string used in all databases was: “Morinda,” “iridoid,” “flavonoid,” “polysaccharides,” “coumarins,” “non-anthraquinone,” “phenolic compounds,” “pharmacology,” “bioactivity,” “structure–activity relationship (SAR)”.
Studies were included if they: (a) Examined non-anthraquinone secondary metabolites isolated from Morinda species; (b) presented original experimental data on chemical characterization and/or biological activity; and (c) provided sufficient information to support SAR analysis by clearly linking individual non-anthraquinone metabolites from Morinda to their corresponding biological effects. Studies were excluded if they: (a) Focused solely on anthraquinone derivatives; (b) were conference abstracts lacking adequate experimental detail, editorials, or theses; (c) failed to report unambiguous compound identification or biological activity data; or (d) were not written in English.
The search covered articles published from 1998 to 2025. Only original experimental studies were considered, including in vitro assays, in vivo models, and in silico studies, with a small number of review articles on the identified non-anthraquinone metabolites. When multiple publications from the same research group contained overlapping data, the most detailed or most recent article was selected for inclusion.
Results and Discussion
A comparative overview of the documented biological activities of non-anthraquinone metabolites from different Morinda species, grouped according to their chemical class and source species (Tables 1–4). This serves to highlight the therapeutic prospects and research relevance of these plant-derived natural products.
Biological Activities of Iridoids and Their Derivatives from the Morinda Genus.

Biological Activities of Phenolic Compounds from the Morinda Genus.

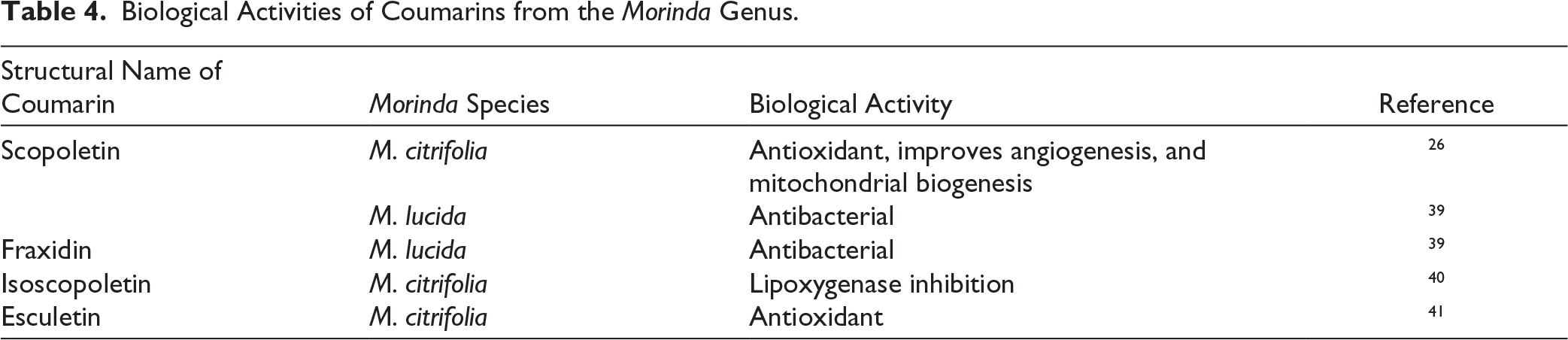
Biological Activities of Polysaccharides from the Morinda Genus.
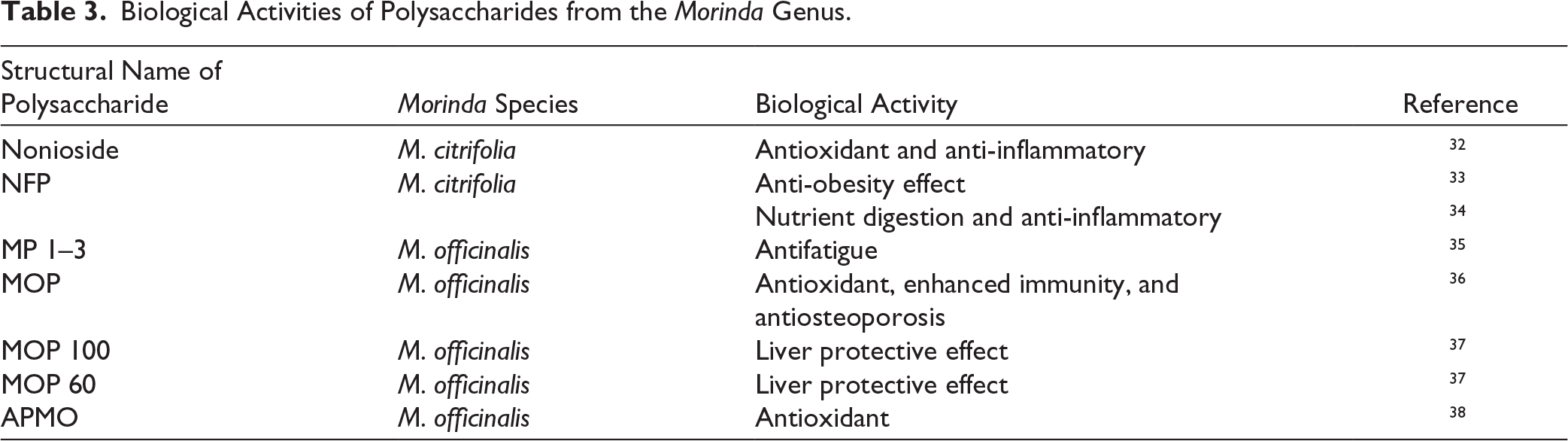
Biological Activities of Coumarins from the Morinda Genus.
Biological Activities of Non-anthraquinone Metabolites from Morinda Genus
Antioxidant Activity
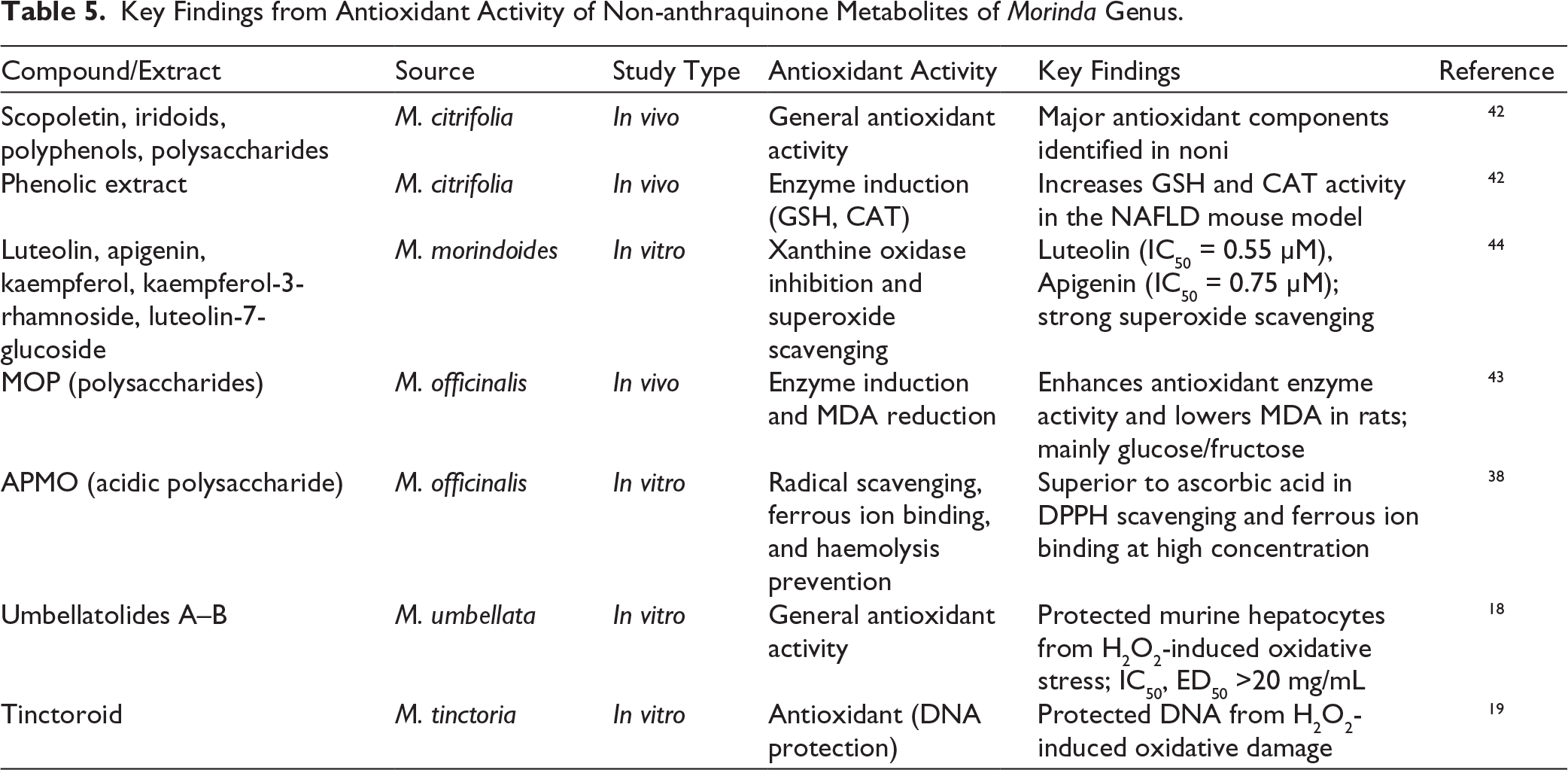
The primary antioxidants in M. citrifolia L. are known to be scopoletin, iridoids, polyphenol, and polysaccharide components. The phenolic extract of M. citrifolia fruits markedly enhances the activity of the antioxidant enzyme catalase (CAT) and elevates the level of the non-enzymatic antioxidant glutathione (GSH), thereby reducing oxidative stress in an in vivo high-fat diet (HFD) induced non-alcoholic fatty liver disease (NAFLD) mouse model. 42
A large number of polysaccharides from M. officinalis F.C. How (synonym: Gynochthodes officinalis (F.C. How) Razafim. and B. Bremer) were successively extracted with hot water after preliminary treatments, and their quality and purity were confirmed by UV scan, showing the absence of proteins. HPLC analysis revealed that MOP (M. officinalis polysaccharide) mainly consisted of glucose and fructose with a molar ratio of 1.29:2.71. The primary focus of this in vivo study was on the antioxidant activity of MOP, which was evaluated based on its chemical composition. The results demonstrated that MOP supplementation significantly enhanced antioxidant enzyme activities and reduced malondialdehyde (MDA) levels in rats, highlighting its strong antioxidant potential. 43
Ten flavonoids and their four corresponding aglycone forms that have been extracted from M. morindoides (Baker) Milne-Redh. leaves were evaluated in vitro for their ability to inhibit superoxide radicals and inhibit xanthine oxidase. Among them, luteolin and apigenin showed strong xanthine oxidase inhibition (IC50 = 0.55 µM, 0.75 µM, respectively), while all the flavonoids, except chrysoeriol, demonstrated the ability to scavenge superoxide anions. The compounds with the highest activity were luteolin, kaempferol, and apigenin, followed by kaempferol-3-rhamnoside and luteolin-7-glucoside in descending order. 44
APMO, an acidic polysaccharide derived from M. officinalis, demonstrates superior antioxidant and protective effects by efficiently scavenging DPPH radicals, binding ferrous ions, and preventing hydrogen peroxide-induced haemolysis in rat erythrocytes in an in vitro study, outperforming ascorbic acid at higher concentrations. 38
Umbellatolides A and B, isolated from the aerial parts of M. umbellata L. (synonym: Gynochthodes umbellata (L.) Razafim. and B. Bremer), exhibited protective activity against H₂O₂-induced oxidative stress injury in murine hepatocytes in vitro, with IC₅₀ and ED₅₀ values exceeding 20 mg/mL. 18 Tinctoroid, isolated from M. tinctoria Roxb., demonstrated the ability to protect DNA against H₂O₂-induced oxidative damage in vitro. 19
The key findings on the antioxidant activity of non-anthraquinone metabolites from the genus Morinda are summarized in Table 5.
Key Findings from Antioxidant Activity of Non-anthraquinone Metabolites of Morinda Genus.
Anti-inflammatory Activity
In traditional oriental medicine, M. officinalis root has been used to alleviate conditions such as impotence and rheumatoid arthritis. Monotropein and deacetylasperulosidic acid were separated from the active portion of the BuOH extract of the root of M. officinalis. Monotropein, an iridoid glycoside found in M. officinalis roots, was shown to possess substantial anti-inflammatory and analgesic effects in animal models. Monotropein exhibited anti-inflammatory activity, with effects noticeable one hour after carrageenan injection and the maximum suppression of swelling seen three hours after edema induction. When administered orally at 30 mg/kg, monotropein reduced swelling by 39.6% after three hours, while oral ibuprofen at 100 mg/kg decreased swelling by 62% at the same time point. These results from in vivo studies highlight monotropein’s capacity to alleviate inflammation and pain, supporting its role as a major bioactive compound underlying the therapeutic benefits traditionally associated with M. officinalis root preparations. 12
Ripe fruits include phenolic compounds that can reduce intestinal inflammation brought on by an HFD. In an investigation, noni fruit phenolic extract was administered to obese mice fed an HFD for a period of 10 weeks. The treatment led to reduced body weight and fat deposits in both the liver and adipose tissue. It also helped restore a healthier balance of gut bacteria by boosting beneficial, short-chain fatty acid producers and lowering harmful, lipopolysaccharide-producing bacteria. These changes alleviated gut inflammation and oxidative stress. Additionally, the extract strengthened the gut barrier by raising important protein levels such as occludin, claudin-1, and ZO-1, and also suppressed the high-fat-diet induced intestinal inflammation by repressing the LPS/TLR4/NF-κB pathway. Overall, this in vivo study showed that noni fruit phenolic extract helps guard against obesity by improving gut health, reducing inflammation, and supporting intestinal barrier function, making it promising for protecting against HFD damage. 45
Gentisic acid 5-O-β-D-(6′-O-sinapoyl) glucopyranoside, isolated from the ethyl acetate fraction of M. parvifolia Bartl. ex DC. (synonym: Gynochthodes parvifolia (Bartl. ex DC.) Razafim. and B. Bremer), showed anti-inflammatory effects in vitro by inhibiting IL-6 mRNA expression. 30 Deacetylasperulosidic acid, an iridoid glycoside highly present in M. citrifolia fruit, was shown in an in vitro study to regulate MAPK phosphorylation in human keratinocyte cells treated with inflammatory mediators TNF-α and IFN-γ, in a manner that varied with concentration. These findings suggest that deacetylasperulosidic acid helps restore balance between T helper cell 1 and T helper cell 2 immune responses and improve skin barrier function, making it a promising candidate for treating atopic dermatitis, an inflammatory skin disease. 46
Moreover, different extracts of noni have been investigated and shown to possess notable anti-inflammatory activity, primarily attributed to the presence of polysaccharides. 32 Table 6 summarizes the main findings on the anti-inflammatory activity of non-anthraquinone metabolites from the genus Morinda.
Key Findings from Anti-inflammatory Activity of Non-anthraquinone Metabolites of Morinda Genus.

Antimicrobial Activity
M. reticulata Gamble. (synonym: Gynochthodes ridsdalei Razafim. and B. Bremer) is rich in polyphenolic compounds with strong antimicrobial activity. Plant extracts were prepared using different solvents such as water, ethanol, acetone, and methanol. These extracts were evaluated in an in vitro study for their activity against antibiotic-resistant bacterial strains—such as Staphylococcus aureus, Enterobacter aerogenes, Salmonella typhimurium, Enterococcus faecalis, and Escherichia coli—and toxin-producing molds, including Penicillium chrysogenum, and Aspergillus species. Among the solvents, methanol extracts showed the highest antibacterial and antifungal effects. At 1,080 mg tannic acid equivalent/ml, S. typhimurium and E. coli showed no sensitivity, whereas S. aureus displayed the widest inhibition zone (22 mm), with E. faecalis and E. aerogenes showing zones of 18 mm and 21 mm, respectively. For the tested fungi, Penicillium expansum exhibited the greatest growth suppression (73%), while A. niger showed the least (15%) at the same extract concentration. Overall, all tested bacteria and molds except S. typhimurium and E. coli were significantly suppressed, emphasizing M. reticulata’s potential as a natural antimicrobial source against the diseases caused by the resistant pathogens. 47
Scopoletin isolated from the methylene chloride extract of the stem bark of M. lucida Benth. exhibited significant antibacterial activity against Salmonella enterica subsp. enterica serovar typhimurium with a minimal inhibitory concentration value of 8 µg/mL, 39 while scopoletin derived from noni fruits exhibited inhibitory activity against E. coli. It also demonstrated suppressive effects on Helicobacter pylori, the bacterium responsible for stomach ulcers. 48 These findings were from research conducted in vitro.
The chloroform fraction of M. lucida leaves yielded the tetracyclic iridoids molucidin, ML-2-3, and ML-F52, which have demonstrated efficacy against Trypanosoma brucei. The IC50 values for molucidin, ML-2-3, and ML-F52 were found to be 1.27 µM, 3.75 µM, and 0.43 µM, respectively. In an in vitro study, ML-2-3 and ML-F52 reduced the expression of the paraflagellar rod protein subunit 2 and triggered changes in the cell cycle, which occurred before the onset of apoptosis in the bloodstream form of Trypanosoma parasites. These new tetracyclic iridoids have the potential to be the primary compounds in the creation of novel treatments for human and animal trypanosomiasis. Among them, ML-F52 has shown the strongest activity and potential for therapeutic development. 49 Table 7 provides a summary of key findings regarding the antimicrobial activity of non-anthraquinone metabolites from the genus Morinda.
Key Findings from Antimicrobial Activity of Non-anthraquinone Metabolites of Morinda Genus.

Antimalarial Activity
The ethanol extract of M. morindoides displayed potent antimalarial activity in an in vitro study. Among the fractions derived from the water extract, both the ethyl acetate and ethanol fractions were notably active against the malarial parasite. Furthermore, the chloroform fractions obtained from the ethanol and methanol extracts also exhibited considerable antimalarial potential. This effect was attributed to the presence of quercetin and phenylpropanoid-conjugated iridoids identified in the methanol extract. 50 Similarly, M. lucida extracts, particularly from stem bark and leaves, also demonstrated notable antimalarial effects, possibly due to synergistic actions of its bioactive compounds, mainly high levels of phenol, flavonoid, and tannin content in the extract. 51 8-acetoxyepishanzilactone, isolated from methanol extracts of M. asteroscepa K. Schum. leaf and stem bark, displayed anti-plasmodial activity against the chloroquine-sensitive strain of Plasmodium falciparum in vitro. 23
Quercetin extracted from M. morindoides leaves could be a promising candidate for curing the NF54 strain in an in vitro study, with its IC50 value of 5.5 ± 1.8 µg/ml. Although crude leaf extracts of M. morindoides showed IC50 values up to 1,000 times higher (less potent) than control drugs such as chloroquine or quinine, certain fractions from the ethanol extract—such as isoamylic alcohol, petroleum ether, and chloroform fractions—along with flavonoids such as quercetin and chrysoeriol, demonstrated notable activity with IC50 values below 15 µg/mL. This suggests that at sufficiently high concentrations, M. morindoides leaf extracts can exhibit antimalarial effectiveness comparable to standard drugs such as chloroquine diphosphate in vitro. 52
Flavonoids and iridoids isolated from the leaves of M. morindoides demonstrated significant antimalarial activity, indicating that these compounds are likely the principal bioactive constituents responsible for the plant’s antimalarial effects. 53
Neuroprotective Activity
M. officinalis has a long history of use in traditional Chinese medicine, where its major constituent in many traditional Chinese medicine prescriptions has antidementia and neuroprotective effects. Bajijiasu and oligosaccharides from M. officinalis are the primary active compounds that have been widely researched both in laboratory and in vivo models for Alzheimer’s disease treatment. Additionally, iridoid glycosides, another bioactive component of M. officinalis, have demonstrated potent anti-inflammatory effects. 54 Pharmacokinetic studies show that iridoid glycosides are primarily found in the digestive tract, where they may contribute to Alzheimer’s treatment by modulating inflammation within the intestines. This study offers a scientific foundation for the continued use of M. officinalis by highlighting the involvement of its active ingredients, involving in anti-inflammatory and antioxidant mechanisms utilized to treat Alzheimer’s disease. 55
Anti-osteoporosis Agent
For thousands of years, the root of M. officinalis has been utilized extensively in China to treat a variety of bone diseases. 56 Two polysaccharides, MOP70-1 and MOP70-2, were isolated from M. officinalis. In an in vitro study, these two significantly improved MC3T3-E1 cell growth, differentiation, and mineralization. MOP70-2 specifically elevated the expression levels of several osteogenic marker genes, such as osteocalcin, bone sialoprotein, osterix, osteopontin, osteoprotegerin, and runt-related transcription factor 2. This indicates that MOP70-2 promotes osteoblast differentiation by upregulating genes linked to bone formation and development. 56 The study’s overall findings offer compelling evidence for M. officinalis’ potential use as an anti-osteoporosis treatment in the future.
Anticancer Activity
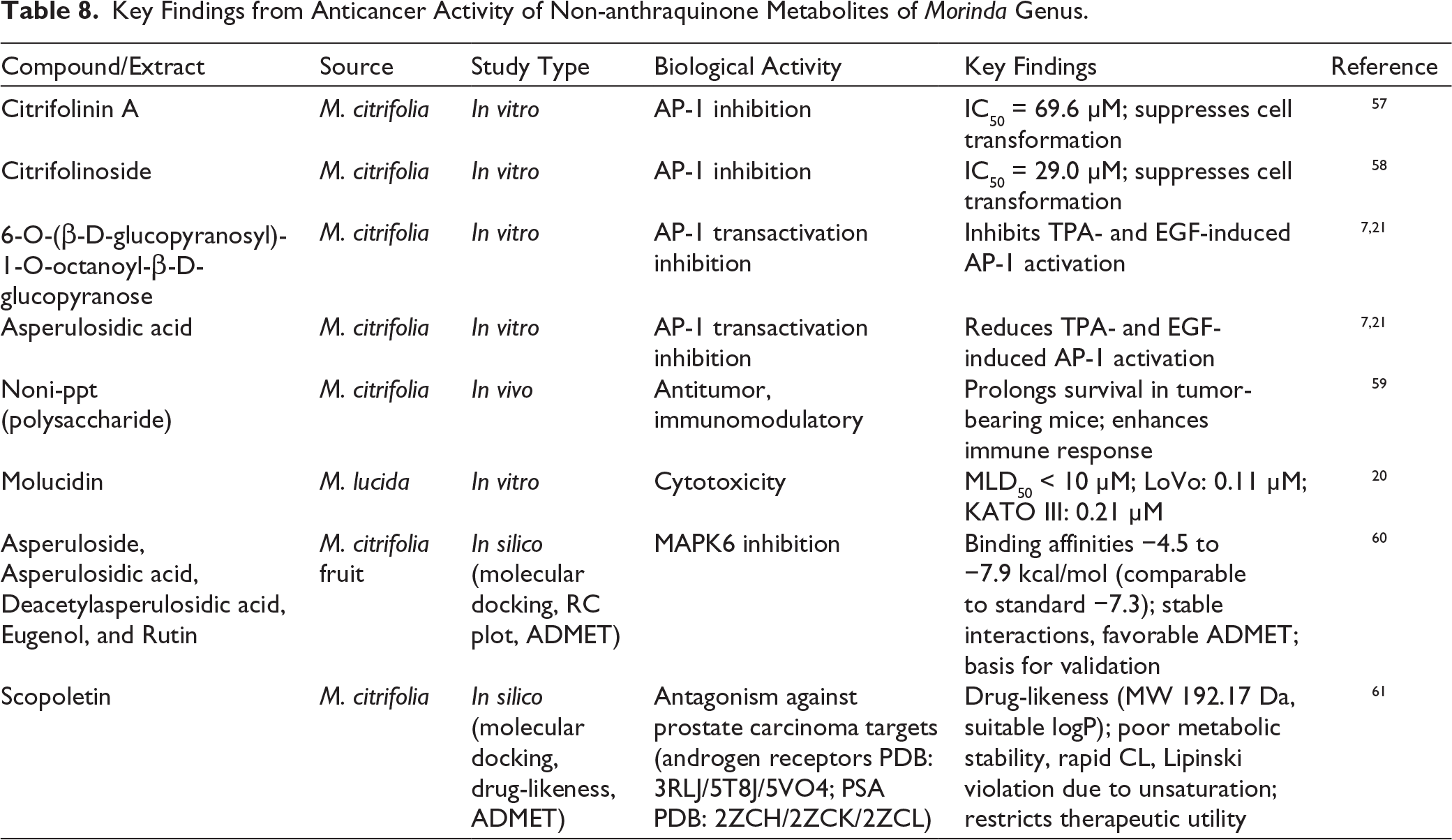
The transcriptional activator protein-1 (AP-1) is a critical factor in UVB-induced skin cancer development. Suppressing AP-1 activity has been found to reduce both cell transformation and tumor promotion. In an in vitro study, citrifolinin A and citrifolinoside isolated from M. citrifolia demonstrated notable inhibitory effects, with IC₅₀ values of 69.6 μM and 29.0 μM, respectively.57,58
In an in vitro study, two newly identified glycosides— 6-O-(β-D-glucopyranosyl)-1-O-octanoyl-β-D-glucopyranose and asperulosidic acid—from M. citrifolia showed significant inhibition of AP-1 transactivation and cell transformation in mouse epidermal JB6 cells. The findings demonstrate that both compounds significantly suppress TPA- or EGF-induced cell transformation and the associated AP-1 activity. Notably, the compounds selectively inhibited the phosphorylation of c-Jun, while having no effect on the activation of extracellular signal-regulated kinases or p38 kinases. This suggests that c-Jun N-terminal kinases are pivotal in mediating TPA- or EGF-induced AP-1 activation and subsequent cell transformation in JB6 cells.21,7
Molucidin, a compound isolated from the leaves of M. lucida, has demonstrated potent cytotoxic activity in in vitro studies, showing median lethal dose (MLD₅₀) values below 10 μM against various cancer cell lines. Notably, it exhibited remarkable potency against LoVo (colon cancer) and KATO III (gastric cancer) cell lines, with MLD₅₀ values of approximately 0.11 μM and 0.21 μM, respectively, along with high selectivity indices across multiple cancer models. 20
Studies revealed that the polysaccharide-rich fraction of M. citrifolia fruit juice, known as noni-ppt, exhibits notable antitumor potential. Experiments using the Lewis lung carcinoma model demonstrated that administration of noni-ppt significantly extended the survival of tumor-bearing mice in an in vivo study. Additionally, in an in vitro study, the ethanol-insoluble fraction of dried noni juice showed strong activity against mononuclear leukocytes. These findings suggest that noni-ppt acts as an immunomodulatory compound capable of enhancing the host’s immune response, thereby contributing to tumor suppression. Consequently, noni-ppt has been proposed as a promising adjunct in cancer therapy due to its ability to potentiate the efficacy of conventional anticancer drugs. 59
An in silico molecular docking study evaluated twenty-three bioactive phytochemicals from M. citrifolia fruit for their anticancer potential using MAPK6 as the target protein. Docking analyses revealed binding affinities ranging from −4.5 to −7.9 kcal/mol, with several compounds—including asperuloside, asperulosidic acid, deacetylasperulosidic acid, eugenol, and rutin—showing strong interactions comparable to the standard MAPK6 inhibitor (−7.3 kcal/mol). These lead compounds were further examined using RC plot analysis, density functional theory, and ADMET profiling, confirming stable protein–ligand interactions, favorable structural stability, and acceptable pharmacokinetic and safety parameters. Overall, the findings highlight the therapeutic potential of M. citrifolia constituents and provide a basis for subsequent in vitro and in vivo validations in cancer drug development. 60
In silico analysis identified six inhibitory phytochemicals from M. citrifolia with potential antagonistic affinity against prostate carcinoma targets, including human androgen receptors (PDB: 3RLJ, 5T8J, 5VO4) and prostate-specific antigens (PDB: 2ZCH, 2ZCK, 2ZCL). Scopoletin from M. citrifolia displayed favorable drug-likeness traits, including low molecular weight (192.17 Da), suitable logP, moderate solubility, and high predicted gastrointestinal permeability. However, extensive unsaturation in its coumarin structure led to poor metabolic stability, rapid clearance (CL), and violation of Lipinski’s Rule of Five, ultimately failing oral bioavailability criteria. Despite docking affinity against SARS-CoV-2 main protease, these pharmacokinetic limitations—consistent with coumarin class challenges—restrict its therapeutic utility. 61
Table 8 presents the main results concerning the anticancer activity of non-anthraquinone metabolites from the Morinda genus.
Key Findings from Anticancer Activity of Non-anthraquinone Metabolites of Morinda Genus.
Antiviral Activity
Seven new iridoid compounds, named officinaloside A–G, were isolated from the aerial parts of M. officinalis, with officinaloside C showing anti-HSV-1 activity. Functional studies revealed that officinaloside C significantly inhibits HSV-1 plaque formation, viral gene expression, protein synthesis, and fluorescent virus replication. The antiviral effect was assessed in Vero cells using a plaque reduction assay, where treatment with increasing concentrations of officinaloside C (0–50 μM) resulted in a dose-dependent reduction in plaque numbers compared to untreated controls. A significant decrease was observed at 25 μM, demonstrating its concentration-dependent inhibition of HSV-1 replication. At this concentration, officinaloside C also markedly suppressed the expression of HSV-1 immediate early (UL54), early (UL52), and late (UL27) genes, confirming broad antiviral activity in this in vitro study. Additionally, HSV-1 GFP fluorescence was reduced in a dose-dependent manner, with strong inhibition seen at 50 μM, further supporting its antiviral efficacy. 62
Metabolic Studies
Rutin and kaempferol were identified as the major flavonoids in the M. citrifolia leaf extract. Among these, rutin exhibited strong anti-adipogenic activity by inhibiting 3T3-L1 cell differentiation in a dose-dependent manner. During the early phase of adipogenesis, rutin suppressed glycerol-3-phosphate dehydrogenase activity and downregulated key adipogenic transcription factors, including PPAR-γ and C/EBPα. In in vivo experiments, rutin supplementation (25–50 mg/kg body weight) for four weeks significantly reduced weight gain in HFD-fed C57BL/6 mice. Similarly, in obese Wistar rats, rutin administration (50–100 mg/kg body weight) for eight weeks lowered body weight and adipose tissue mass, improved lipid and hormone profiles, and mitigated oxidative stress and hepatic lipid accumulation, indicating its protective role against HFD-induced metabolic disturbances. 63
Noni and its bioactive constituents, including scopoletin, quercetin, rutin, and polysaccharides, have shown potential in managing systemic metabolic disorder, a condition marked by insulin resistance, type 2 diabetes, fatty liver, hypertension, dyslipidaemia, inflammation, and organ damage. Noni helps combat obesity by influencing gut microbiota and regulating lipid metabolism in in vivo studies. It enhances insulin sensitivity and glucose control through pathways such as Nrf2/ARE. Additionally, it protects the liver by reducing fat accumulation and oxidative stress. Noni also lowers blood pressure by inhibiting ACE activity and activating eNOS. Furthermore, it improves lipid profiles and reduces inflammation by suppressing NF-κB. By mitigating oxidative stress and preventing cell death, noni provides protective effects on both the heart and kidneys. 64
Flavonoids have been widely studied for their therapeutic potential in metabolic disorders, owing to their ability to modulate various cell signaling pathways involved in carbohydrate digestion, fat deposition, and insulin secretion, or glucose uptake in insulin-responsive tissues. Catechin, epicatechin, kaempferol, rutin, and quercetin are major bioactive flavonoids isolated from M. citrifolia that show beneficial effects against obesity and obesity-related metabolic dysfunction in in vivo models. One of the key coumarins found in M. citrifolia is scopoletin (6-methoxy-7-hydroxycoumarin), which has demonstrated significant activity in managing obesity and associated metabolic disturbances. Asperulosidic acid, a prominent iridoid isolated from M. citrifolia, has been reported in in vivo studies to improve blood fluidity, thereby contributing to better health outcomes in obese individuals and those with obesity-linked conditions such as hypertension, diabetes, and dyslipidaemia. 65
Toxicity Studies
Non-anthraquinone compounds, including iridoids such as monotropein and asperulosidic acid, flavonoids such as quercetin and rutin, and polysaccharides, are generally regarded as safe. Toxicological studies indicate minimal toxicity even at high doses, and these compounds are widely considered safe for human use. 66 Monotropein isolated from the roots of M. officinalis, when subjected to an acute toxicity test using mice, no lethality was observed up to a 2,000 mg/kg dose. This finding suggests that monotropein is extremely safe for use in clinical settings. 12
The root of M. officinalis, which is frequently used in health foods, showed reduced toxicity and no appreciable negative effects. Preliminary investigation showed that MOIG (iridoid glycoside from M. officinalis) at 2–10 g/kg did not show any toxic reaction. Therefore, to assess its toxicity, the maximum possible dose approach was employed. In a study, MOIG was administered to mice at a maximum feasible dose of 22.5 g/kg to observe its acute toxicity; no toxic reaction or death of mice was observed during the 14-day observational period. These findings demonstrated that MOIG had good safety and minimal toxicity. 54
The available data indicate that noni juice shows only slight mutagenic activity in bacterial tests, which is associated with quercetin (identified as a bacterial mutagen), but this does not translate into genotoxicity in mammalian systems. In both the V79-HPRT assay and the ex vivo UDS assay in rats, even very high exposures to noni juice did not produce evidence of DNA damage or mutations. In addition, the concentration of potentially harmful anthraquinones or related compounds in the juice is below 1 ppm, a level for which no toxic effects have been demonstrated, supporting the conclusion that properly manufactured noni juice is safe for human consumption. 67
Non-anthraquinone rich preparations from M. citrifolia fruit and seeds—such as fruit puree (PNF), water extract (EDNF), dried fruit powder (PDNF), and dried seed powder (PDNS)—contain iridoids such as deacetylasperulosidic acid (4.1–9.4 mg/g) and asperulosidic acid (2.2–5.2 mg/g) and passed all standard safety tests for DNA damage with no issues in both in vitro and in vivo studies. They showed negative results in the Ames test (bacterial reverse mutation assay using S. typhimurium strains TA98, TA100, TA1535, TA1537, and E. coli WP2 and AurA up to 5,000 μg/plate, with or without liver enzymes). They caused no chromosome damage in Chinese hamster lung cells (IU) up to 5,000 μg/mL (PNF/EDNF), 4,100 μg/mL (PDNF), or 2,100 μg/mL (PDNS) for 6–24 hours (±liver enzymes). No clastogenic or aneugenic effects in a mouse micronucleus test at 2,000 mg/kg/day orally (two doses) with normal survival, body weight, and red/white blood cell ratios. Fruit and seed powders were safe, with lethal dose 50% values over 15,000 mg/kg and 5,000 mg/kg, indicating iridoids offer protective effects against DNA damage even with minor seed anthraquinones. 68
Commercial M. citrifolia fruit products (puree, juice, powder) prepared from fully ripe seedless fruits without skin showed no detectable anthraquinones (lucidin, alizarin, rubiadin) via HPLC/LC-MS/MS, verifying their non-anthraquinone composition. Preparations lacking leaf fragments and fruit skin consistently tested negative across all processing techniques (fermentation, drying, lyophilization), whereas those containing skin/leaf material displayed alizarin levels ranging from 0.05 to 8.6 mg/kg. These findings affirm the safety of non-anthraquinone fruit pulp products and recommend excluding seeds/skin from ripe fruits to avoid genotoxic anthraquinone contamination. 69
Anthraquinones, such as alizarin, rhein, and lucidin, have been linked to possible toxic effects, particularly when consumed in high amounts. Research indicates that certain anthraquinones may cause genetic damage and promote cancer development in animal models, raising safety concerns for human health, especially concerning potential liver and kidney toxicity. 68 In contrast, non-anthraquinone compounds such as iridoids, flavonoids, and terpenes are typically less toxic and frequently demonstrate beneficial antioxidant and anti-inflammatory properties. 70 Consequently, the safety of Morinda-based products depends heavily on their chemical makeup, with preparations rich in anthraquinones posing greater health risks compared to those dominated by non-anthraquinone constituents. 71
Pharmacokinetic Studies
Monotropein, extracted from M. officinalis radix, showed good permeability across Caco-2 cell monolayers in an in vitro study, with higher apparent permeability at lower concentrations than at moderate or high levels. The minimal change in permeability after verapamil treatment suggests that its intestinal uptake does not rely on carrier-mediated transport. Overall, monotropein crosses membranes mainly by passive diffusion and exhibits concentration-dependent self-inhibition. 72
In an in vivo intestinal perfusion model, monotropein and deacetylasperulosidic acid showed optimal absorption in the small intestine after oral dosing, with higher apparent permeability (Papp) and absorption rate constants (Ka) at pH 7.4 compared to pH 6.8 or 5.4; self-inhibition increased with perfusion concentration, indicating dose-dependency. Monotropein absorbed best in the jejunum > duodenum > ileum, while deacetylasperulosidic acid favored ileum > duodenum > jejunum, and overall, deacetylasperulosidic acid exhibited superior Ka across segments. With Papp exceeding 0.2 × 10⁻⁴ cm/s, both compounds displayed good absorption primarily in the jejunum and ileum, respectively. 73
Precise in vivo pharmacokinetic evaluation of monotropein after oral doses of 10, 20, and 40 mg/kg in rats revealed rapid plasma accumulation, as indicated by plasma concentration-time profiles. Peak concentrations (Tmax) occurred between 1.3 and 1.4 hours, while elimination half-lives (T1/2) varied from 2.9 to 4.5 hours across the escalating doses, with maximum plasma concentrations (Cmax) spanning approximately 11.2–327.6 ng/mL. Both Cmax and area under curve (AUC0-t) demonstrated dose-linearity within the 10–40 mg/kg range, consistent with linear pharmacokinetic behavior. 74
Monotropein pharmacokinetics showed clear sex differences in rats in an in vivo study. Females displayed elevated Cmax, AUC0-t, and bioavailability, coupled with reduced volume of distribution (Vd) and clearance (CL) versus males, signifying slower elimination. Co-administration of major iridoid glycosides (50 mg/kg, equiv. 20 mg/kg monotropein) enhanced monotropein’s Cmax, T1/2, AUC0–t, and AUC0–∞ while accelerating Tmax compared to monotropein monotherapy, promoting better absorption, exposure, and systemic retention. In contrast, M. officinalis ethanol extracts (1,650 mg/kg, equiv. 20 mg/kg monotropein) yielded lower Cmax, AUCs, and bioavailability than iridoids alone, alongside increased Vd and CL, due to rapid tissue distribution and CL influenced by co-existing extract components. 75
After oral dosing at 20 mg/kg in rats, monotropein rapidly distributed widely across tissues in the order kidney > stomach > small intestine > liver > heart > lung > spleen, with quick elimination, and no prolonged buildup. Levels remained notably low in the spleen, while high kidney concentrations pointed to primary renal excretion. 74
An in vivo pharmacokinetic study found that noni fruit extract products are widely sold globally for health benefits, yet lack standardization and clinical evidence on efficacy, safety, and optimal dosing. Scopoletin was identified as a bioactive marker of noni exposure, suitable for product standardization and pharmacokinetic studies. Freeze-dried noni fruit capsules were then given orally to nine healthy participants (n = 3 per group) at 1,500 mg (3 × 500 mg), 2,000 mg (4 × 500 mg), or 2,500 mg (5 × 500 mg). Plasma and urine samples were collected pre-dose and at 0.5, 1, 2, 4, and 8 hours post-dose. Scopoletin levels were measured using HPLC-PDA-MS (200–700 nm scan). After noni intake, scopoletin quickly appeared in plasma at 0.5–5 ng/mL, persisting for at least eight hours, though with low bioavailability and high intersubject variation. Scopoletin thus qualifies as a reliable exposure biomarker in plasma and especially urine, accounting for its pharmacokinetic profile. 76
Pharmacodynamic Studies
Scopoletin effectively combats oxidative stress via DJ-1/Nrf2 pathway activation, mitigating rotenone-induced Parkinson’s disease (PD) symptoms. M. citrifolia extract reduced protein aggregates such as nitrotyrosine and α-synuclein in in vivo studies, while in vitro assays confirmed scopoletin’s protection of SH-SY5Y cells from rotenone-triggered apoptosis by curbing oxidative damage. It boosted DJ-1 expression to facilitate Nrf2 nuclear translocation and antioxidant gene activation, plus lowered Keap1 to block Nrf2 export, bolstering neuronal defenses. These results position scopoletin as a promising PD therapeutic candidate through DJ-1/Nrf2 signaling. 77
An ex vivo study used isolated rat vas deferens to assess how a methanolic M. citrifolia fruit extract (MMC) and its constituents scopoletin and rutin affect contractions triggered by dopamine and noradrenaline. Across tested ranges, all three (MMC 1–40 mg/mL, scopoletin 1–200 μg/mL, rutin 0.6–312.6 μg/mL) produced a dose-dependent inhibition of these catecholamine-induced contractions, and this inhibition was reversed by the D2 antagonist haloperidol and the α1-adrenoceptor antagonist prazosin, confirming dopaminergic and adrenergic receptor involvement. At higher MMC concentrations (60–100 mg/mL), the extract itself evoked contractions that were only partly blocked by high-dose haloperidol and were resistant to prazosin, revealing biphasic dopaminergic behavior: Predominantly antidopaminergic at lower doses (<40 mg/mL) and dopaminergic agonist-like activity at higher doses (>60 mg/mL). 78
Nonioside E from M. citrifolia fruit conditionally retained despite failing Lipinski’s rule in a computational in silico study, exhibited strong ADMET properties, including excellent blood-brain barrier penetration (0.0471) and oral bioavailability (F20% 0.2919). In network analysis, it ranked among the top 10 key compounds by degree centrality, targeting anxiety-related proteins such as PRKCA in glutamatergic synapses and neuroactive ligand-receptor pathways. Molecular docking showed favourable binding to PRKCA (Vina score −7.0 kcal/mol), surpassing controls staurosporine (−6.6) and 14(R)-hydroxy-retro-vitamin A (−6.8), via multiple hydrogen bonds with residues such as THR251, ASP254, ASP248, and others, suggesting potent modulation of PKC-α activity for anxiolytic effects. 79
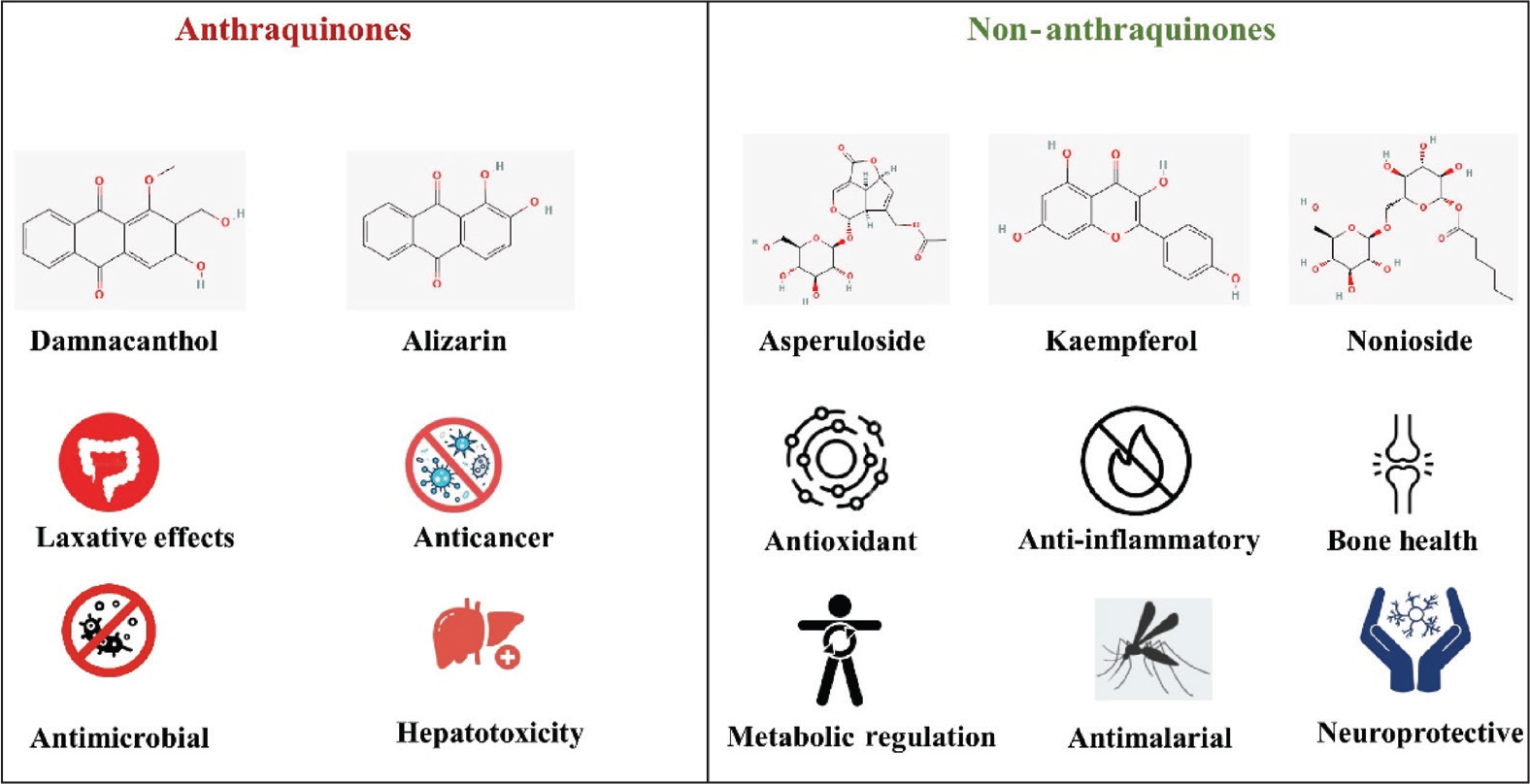
Figure 1 presents a conceptual diagram contrasting the distinct bioactivity profiles of anthraquinone versus non-anthraquinone metabolites from the Morinda genus.
Contrasting Bioactivity Profiles of Anthraquinone and Non-anthraquinone Compounds in the Genus Morinda.
Structure–Activity Relationship (SAR) of Non-anthraquinone Metabolites in the Morinda Genus
Less than 5% of medicinally significant plants have undergone chemical investigation, despite the fact that plants constitute a rich source of lead bioactive compounds. Investigating the structure–activity relationships of moderately active compounds may provide insights for enhancing their biological efficacy. 80 Structures of non-anthraquinone metabolites of the Morinda genus are given below.
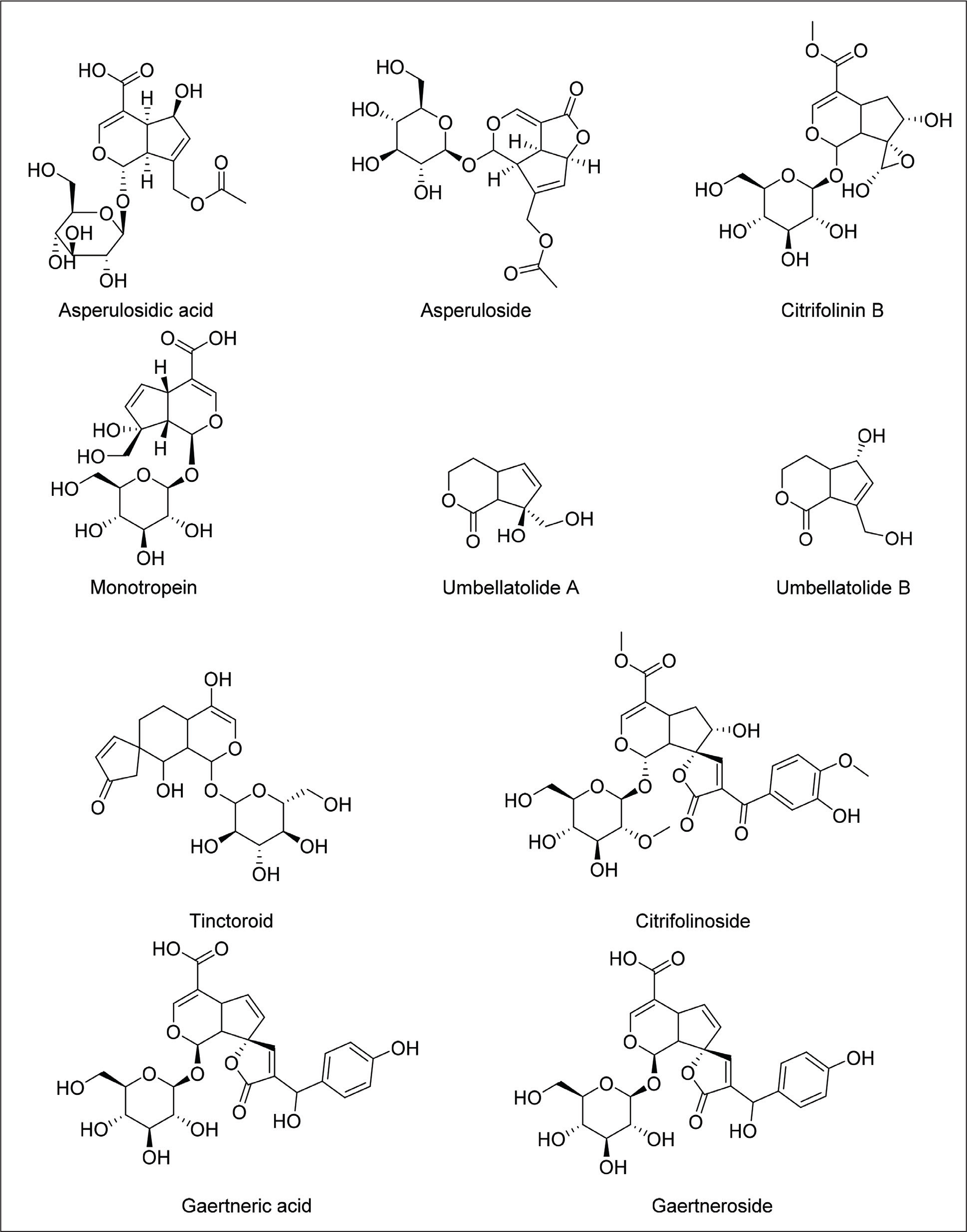
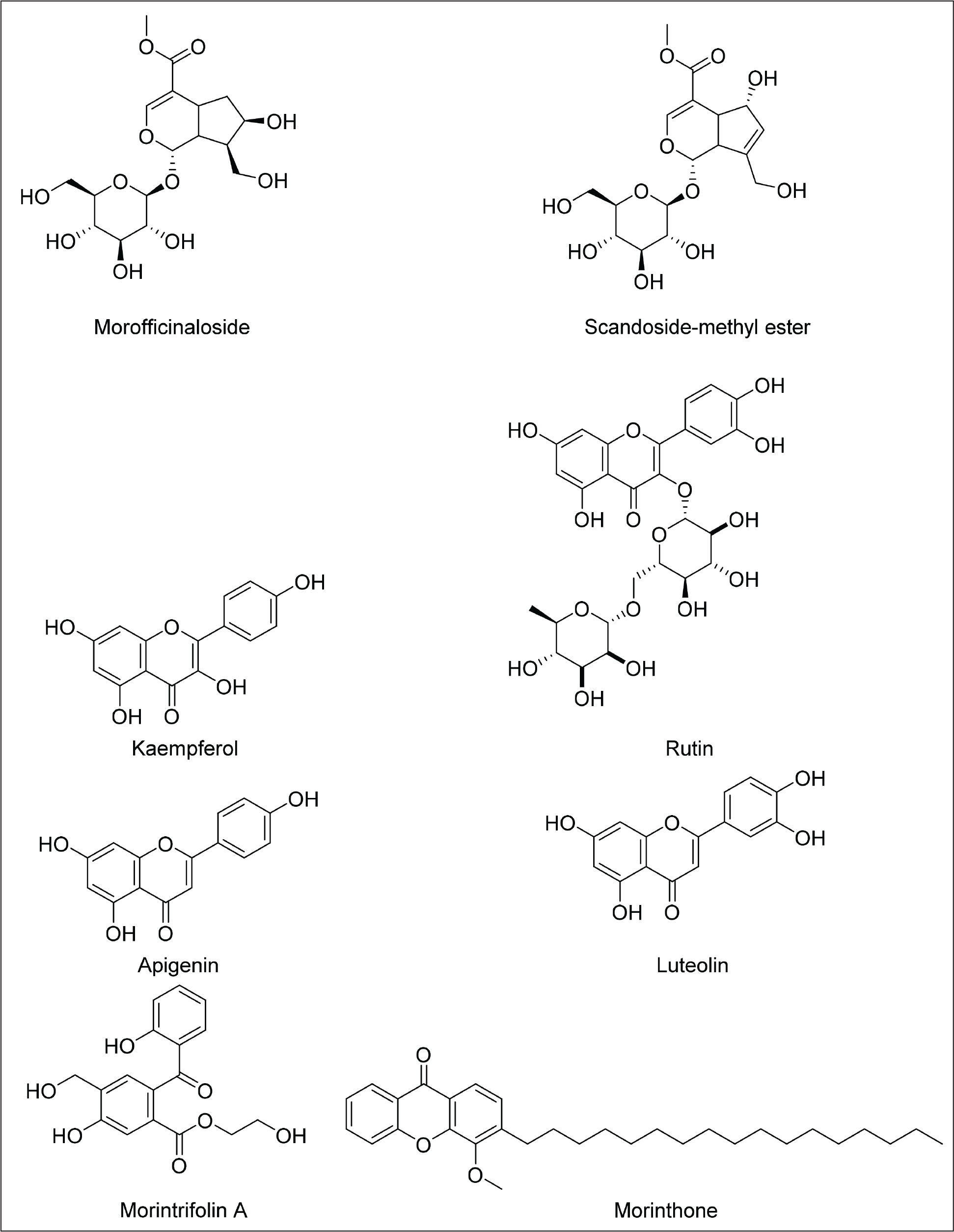
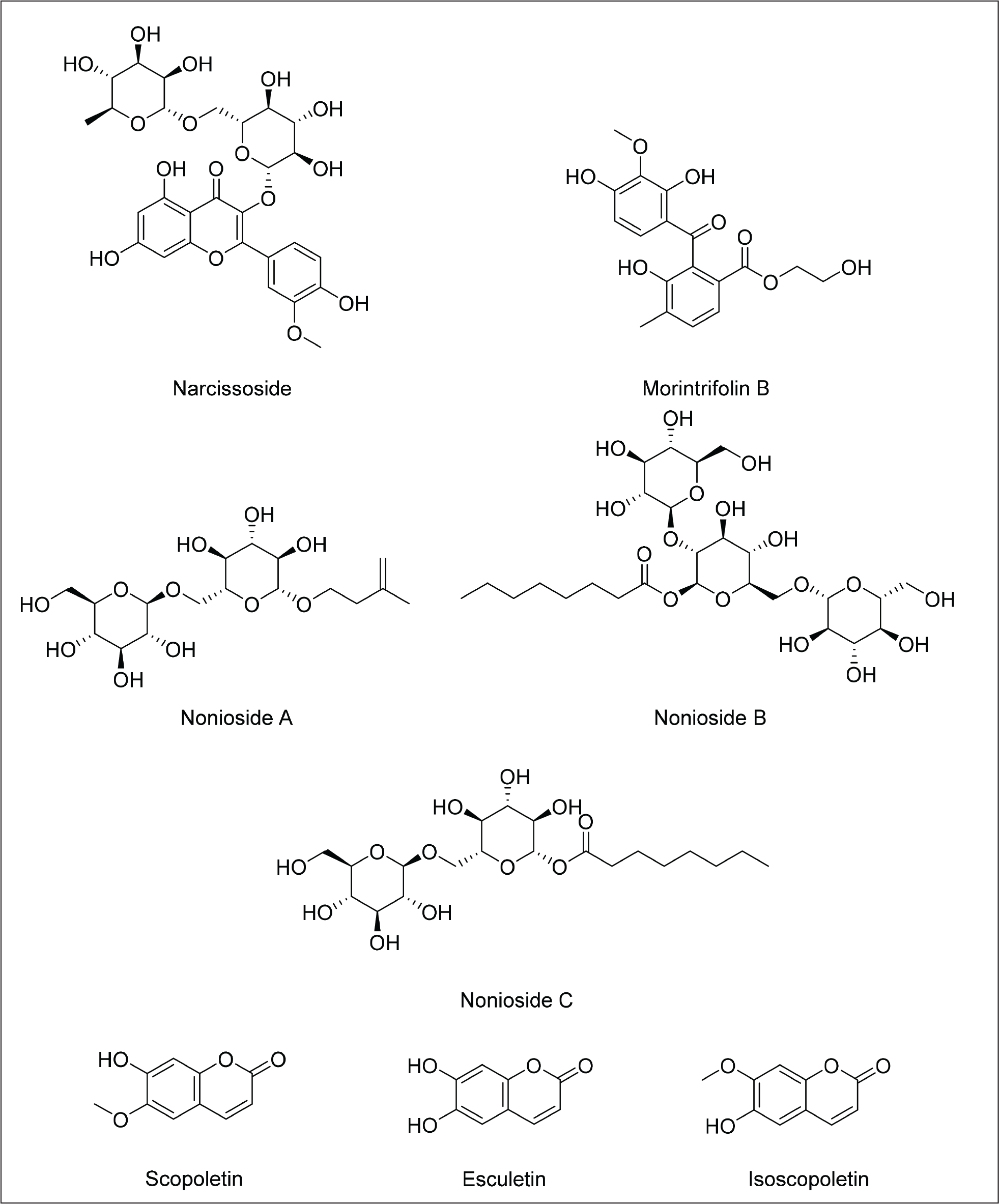
Iridoids
Nearly 40 terpenoid compounds reviewed from M. morindoides demonstrated good to excellent antimalarial activity. Their antimalarial efficacy may be explained by the presence of lactone, ether, ketone, ester, and hydroxyl groups in their structures. 50 Particularly, dehydromethoxygaertneroside, an iridoid glycoside, isolated from M. morindoides, exhibited potential inhibitory effects on the proliferation of malarial parasite P. falciparum, without cytotoxicity. The anti-malarial activity appears to be significantly improved by functionalities of 3’-methoxy, 6”-acetyl, 7’ ketonic carbonyl groups, which are not affected by cytotoxicity against host cells. 3
Multispectral and molecular docking techniques were employed for examining the SAR and interaction mechanisms of iridoid glycosides from M. citrifolia with α-glucosidase. Findings indicated that the inhibitory potential of iridoid glycosides was enhanced by the deacetylation of the C-10 of the B-ring. Removing the acetyl group creates hydrophobic interactions that strengthen deacetylasperulosidic acid’s ability to inhibit α-glucosidase enzymes. Furthermore, the interaction between iridoid glycosides and enzymes leads to the formation of non-covalent complexes, primarily stabilized by hydrogen bonding and van der waals forces. 81
The anti-inflammatory activity is consistently associated with the iridoid glycoside framework, characterized by a cyclopentanoid monoterpene nucleus conjugated with a glycosidic moiety. Notably, deacetylated iridoid glycosides derived from M. officinalis, such as monotropein and deacetylasperulosidic acid, exhibited pronounced inhibition of NF-κB and MAPK signaling pathways, suggesting that increased molecular polarity and the presence of free hydroxyl groups may enhance biological efficacy. 54
Phenolic Compounds
The antioxidant activity of phenolic compounds from M. citrifolia is strongly influenced by the number of phenolic hydroxyl groups they contain; compounds with more hydroxyl groups generally exhibit greater antioxidant effects. Specific examples include 3,3-bisdemethylpinoresinol, americanol A, americanin A, americanoic acid A, morindolin, and isoprincepin, which have been shown to inhibit LDL oxidation in a dose-dependent manner. Their antioxidant potency is comparable to established antioxidants, highlighting the importance of hydroxyl group count in enhancing their biological activity. 82
The flavonoid glycoside narcissoside, which contains a methoxy group at the C-3’ position, showed no activity in the DPPH assay. In contrast, several quercetin derivatives with a hydroxyl group at the same position exhibited strong antioxidant effects. This suggests that the hydroxyl group at C-3’ in the flavanol ring is essential for free radical scavenging. 13
Polysaccharides
Chemical modifications of polysaccharides from M. citrifolia fruits, including acetylation, phosphorylation, carboxymethylation, and sulfation, markedly enhance their antioxidant capabilities compared to native forms, underscoring key SAR trends where substituent introduction modulates bioactivity profiles. 83
Coumarins
The pharmacological properties of coumarins are largely determined by their substituent groups. The presence of orthophenolic hydroxyl groups on the benzene ring is known to boost both antioxidant and antitumor activities. Substitution with aryl groups at the C4 position leads to strong anti-HIV, antitumor, anti-inflammatory, and analgesic effects. Coumarins with a C3 phenyl moiety are linked to notable antioxidant and anti-HIV actions. Additionally, hydroxylated derivatives such as esculetin and fraxidin exhibit strong antioxidant activity, primarily through scavenging reactive oxygen species and stimulating the body’s own antioxidant enzyme systems. 84 The presence of multiple double bonds in the coumarin structure causes poor oral bioavailability of scopoletin. 61
Analyzing the relationship between a compound’s chemical structure and its biological effects reveals the key structural elements driving its activity. Understanding this connection is vital for effectively designing new plant-derived medicines with optimized therapeutic properties. 85 While the elucidated SAR trends for non-anthraquinone metabolites from the Morinda genus provide valuable insights into substituent effects and pharmacophore requirements, clinical data remain scarce, and the overall literature is limited, underscoring the need for advanced in vivo validation and translational studies.
Conclusion and Future Prospects
The Morinda genus presents a wide range of non-anthraquinone compounds that exhibit diverse biological effects. Among them, iridoids, flavonoids, and polysaccharides have demonstrated the greatest potential for clinical translation. Iridoids and flavonoids are recognized for their potent antioxidant, antimalarial, anti-inflammatory, and anticancer properties, with several showing significant cytotoxicity against cancer cell lines and the ability to inhibit important signaling pathways such as AP-1 and NF-κB. Polysaccharides, especially noni polysaccharides, are valued for their immunomodulatory effects and have been shown in preclinical studies to boost the effectiveness of standard anticancer treatments. Evidence from in vitro, in vivo, and in silico studies supports these compound classes as promising candidates for advancing clinical research and nutraceutical development.
Although notable progress has been made in isolating and testing these compounds, future research should focus on the molecular actions and pharmacokinetic profiles of these compounds and standardize the isolated pure substances for therapeutic applications. Since there is limited literature on the SAR in the Morinda genus, understanding SAR can accelerate the improvement of drug candidates by guiding chemical modifications to enhance efficacy, reduce toxicity, and optimize pharmacokinetics and pharmacodynamics. Additionally, comprehensive preclinical and clinical evaluations are essential to validate their safety and effectiveness, supporting their use in pharmaceuticals, nutraceuticals, and functional foods. This integrated approach can enable novel health-related applications and promote sustainable healthcare solutions worldwide.
Footnotes
Acknowledgements
Not applicable.
Authors’ Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis, and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be authors as per the International Committee of Medical Journal Editors’ requirements/guidelines.
Consent to Participate
Not applicable.
Consent for Publication
Applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study does not involve experiments on animals or human subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI) tools for writing and editing of the manuscript, and no images were manipulated using AI.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors, or the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
