Abstract
Background
The new treatment paradigm in Spinal Muscular Atrophy (SMA) has introduced novel phenotypes, changes in trajectories and clinical questions not fully addressed in clinical trials. To explore these challenges, several international initiatives have emerged. CuidAME was created as a nationwide clinical network in Spain designed to standardise SMA care, facilitate knowledge sharing, and capture data in a longitudinal comprehensive registry.
Objective
Evaluating the usefulness of the CuidAME project to capture data in a real-world setting.
Methods
This multicentric cohort study includes individuals with SMA followed at participating hospitals. Clinical examinations and multidisciplinary assessments were performed during routine clinical visits. We present a cross-sectional analysis of the registry population.
Results
As of February 2025, 543 participants from 25 hospitals were recruited: 12 were presymptomatic, 125 (23%) had SMA type 1, 208 (35%) type 2, 194 (38%) type 3, and 4 (<1%) type 4. Among the cohort, 91% (n = 495) had received at least one disease-modifier treatment, with 17 discontinuations. The registry included 5092 motor assessments, 1565 performed before treatment initiation.
Conclusions
CuidAME is an academic, longitudinal, real-world data collection project that demonstrated a fast and effective model for implementation facilitating the standardization of clinical practices and outcome measures across Spain. By aligning with core dataset used in other registries and establishing multidisciplinary working groups, the initiative will contribute to sharing knowledge to advance SMA care and improve patient outcomes.
Keywords
Introduction
Spinal muscular atrophy (SMA) is a neuromuscular genetic disorder characterised by the progressive loss of anterior horn motor neurons within the spinal cord, resulting in systemic muscle atrophy, progressive weakness, and disability. Effective management of SMA requires a multidisciplinary approach, as outlined in the consensus statement for Standards of Care published in 2017.1–3 Proactive supportive therapies, including non-invasive ventilatory support, alongside the emergence of disease-modifying therapies, have significantly improved life expectancy and quality of life for individuals with SMA.4–10
The incidence of SMA ranges from 1 in 6000 to 1 in 10,000 live births, classifying it as a rare disease.11,12 With a prevalence of 1–2 per 100,000 people, it is estimated that in Spain live between 500 and 1000 individuals with the disease. 13 The rarity of SMA poses challenges for clinical research, particularly regarding the generalization of findings from randomized clinical trials, which often apply restrictive inclusion and exclusion criteria. This has driven global efforts to collect real-world data to complement clinical trial results and provide insights into broader patient populations.
Real-world data initiatives aim to address knowledge gaps and reflect clinical and resource-related differences across countries.14–19 For instance, in Spain, access to disease-modifying treatments has been guided by national clinical protocols that establish additional criteria beyond the regulatory indications for initiating, discontinuing and monitoring response to therapy (Supplementary Figure 1).20–22 These local policies highlight the importance of analysing SMA data from a country-specific perspective to understand the nuances of disease management and treatment outcomes.
Emerging real-world data from registries has shed light on previously unresolved clinical questions, particularly as therapies are now approved for broader populations than those included in clinical trials. Furthermore, new SMA phenotypes have emerged in the context of treatment. Initiatives such as TREAT-NMD, SMArtCare and ISMAC have made notable efforts on harmonizing and standardizing datasets for SMA registries to facilitate international collaborations and foster large-scale studies capable of addressing complex clinical questions.14,15,23 These initiatives, as well as the registries led by patient advocacy groups, provide invaluable information on the natural history of the disease, treatment outcomes, and patient-reported outcomes, complementing clinical insights and guiding patient-centred care in this new era of treatments.24–27
In Spain, prior to the advent of treatments, only a limited number of hospitals were designated as reference centres for neuromuscular disorders. 28 The approval of treatments expanded the number of hospitals providing care to individuals with SMA across all regions of the country. 29 This expansion underscored the need for a coordinated effort, leading to the creation of the CuidAME project.
The CuidAME initiative is a nationwide clinical network designed to standardise SMA care practices, facilitate knowledge sharing, and create a comprehensive registry of all individuals with SMA in Spain. The primary goals of CuidAME are (i) to collect systematic, longitudinal data on the natural history of untreated individuals with SMA, (ii) to assess the impact of disease-modifying therapies, and (iii) to evaluate the impact of the implementation updating and harmonization of standards of care within the Spanish healthcare system. Through a multidisciplinary network of specialists, the CuidAME project aims to identify gaps and challenges in the disease management, and the improvement in the characterization of new phenotypes. By better understanding SMA therapies and biomarkers, the initiative seeks to enhance the stratification of individuals with SMA, adopt personalized care approaches, and provide more accurate prognosis to support expectation management and care planning.
This work evaluates the data generated in the CuidAME project through a descriptive, cross-sectional analysis of the SMA population currently included in the registry, encompassing 543 individuals.
Methods
Roles and responsibilities
CuidAME is a clinician-driven longitudinal study sponsored by the Fundació Sant Joan de Déu, which acts as the data controller, with all responsibilities related to data protection and analysis. To ensure effective project governance, a CuidAME steering committee was established, comprising 12 members, including neurologists, paediatric neurologists, and rehabilitation specialists from seven Spanish hospitals, and a patient representative from FundAME, the primary Spanish association of individuals with SMA, who acts as a link between individuals with SMA, advocacy groups, and CuidAME. The steering committee ensures compliance with the objectives of the project, plans key tasks and promote collaborative research projects within the working groups. External requests from third parties to access the data registry must be approved by the steering committee, and only anonymous, aggregate data is shared with third parties.
A lean project management team supports the initiative and includes hybrid roles such as disease experts and data managers, project managers and administrative supporters, and data stewards, that are located in centres with a high number of individuals to ensure efficient data management and oversight.
Participation in the registry is open to any Spanish hospital following individuals with SMA, subject to approval from the local ethics committee and, if necessary, a site agreement, in addition to the national approval by the Ethics Committee of Hospital Sant Joan de Déu (PIC- 188-18 and EOM-02-24). Besides, all individuals or their legal representatives must give their written consent before being included in the study. Project sustainability is based on a multi-sponsor funding model, with structural costs funded by pharmaceutical companies, who also act as third-party requestors. Agreements have been implemented to ensure independence and equitable data access for all funders.
Design and setup
The registry of CuidAME is observational, prospective and multicentric. Initially, CuidAME used the SMArtCare online platform, developed by the Freiburg University to collect data from individuals with SMA in Germany, Austria and Switzerland.14,30 This secure web-based platform complied with GDPR regulations and employed pseudonymised patient identifier codes for data protection. The use of SMArtCare facilitated a rapid project setup, enabling the start of data collection in May 2020. Later, in 2023, the CuidAME data was transferred to its own platform, following the same standards than the German one.
Data collection
Data collection is conducted via a web platform specifically designed for this purpose. It was created by an IT architect and software developer using Oracle APEX application platform, which allows for high flexibility and adaptability of the registry design. Data is obtained during routine clinical visits, captured from medical records and updated at least annually.
Given the observational nature of the registry, there is no requirement to perform tests beyond those routinely conducted. However, the registry includes comprehensive information covering standard clinical examinations and assessments performed by key professionals involved in SMA care and collects 941 variables. The collected data includes validated outcome measures used in clinical practice and/or recommended by experts, such as motor scales (HINE section 2, CHOP-INTEND, HFMSE, RULM, 6MWT, ALS-FRS, and EK2), pulmonary function, and Cobb angle, among others. Additional variables include demographic details, comorbidities, treatment data, and clinical features of SMA, such as respiratory, nutritional, cognitive, and orthopaedic status, and laboratory examinations, among others (Table 1). The study also allows for the retrospective collection of data. Multidisciplinary working groups focused on rehabilitation, respiratory care, bulbar function, neurocognition, adult population and biomarkers were formed to review and adapt the dataset to meet emerging research needs. As part of the transition to the new CuidAME platform, these groups reviewed and updated the dataset, adding new variables to the core dataset while preserving most elements, definitions and response categories from the SMArtCare platform with the aim of using standardised data and common vocabulary with them. Optional variables were also included to expand the scope of the data collection. It is ensured that this information related to the structure and the content of the dataset is contained in a data dictionary as a tool for quality data collection and analysis.
Clinical dataset captured in the CuidAME study.

WHO: World Health Organization; ENMG: electroneuromyography; HINE: Hammersmith Infant Neurological Examination; CHOP INTEND: Children's Hospital of Philadelphia Infant Test of Neuromuscular Disorders; HFMSE: Expanded Hammersmith Functional Motor Scale; Revised Upper Limb Module; 6MWT: Six Minutes Walking Test; EK2: Egen Klassifikation 2; ALSFRS: Revised Amyotrophic Lateral Sclerosis Functional Rating Scale.
Data quality
To ensure the reliability and standardization of outcome measures, CuidAME conducts regular training sessions for physiotherapists and workshops on data quality. Physiotherapy training sessions consist of face-to-face meetings led by expert physiotherapists and rehabilitation physicians, who were previously trained in clinical trial protocols. These sessions teach the standardized administration of motor scales to the physiotherapists responsible for applying them in each participating centre. Attendees are encouraged to conduct online certified courses to complement their training. Data quality workshops are conducted for data stewards, typically physicians or pharmacists familiar with SMA, to ensure consistent and standardized data entry into the platform. Regular data cleaning and analysis are performed, supported by automated checks within the capture platform. Additionally, a contract research organization has been engaged to monitor data quality, ensure regulatory compliance and farmacovigilance processes.
Statistics
Frequencies and mean were used for the calculation of this descriptive analysis. The analysis was performed using R (version 4.4.2.).
Results
As of February 2025, CuidAME had 25 active centres across Spain, having already recruited 543 individuals. Of these, 96.3% were alive, 2.4% were deceased and 1.3% were lost of follow-up. The 49.7% was under 16 years of age. Notably, the study encompassed 14 out of the 17 regions in Spain, representing 89% of the Spanish population and providing broad national coverage, as illustrated in Figure 1. Updated information can be found at http://www.registro-cuidame.org.
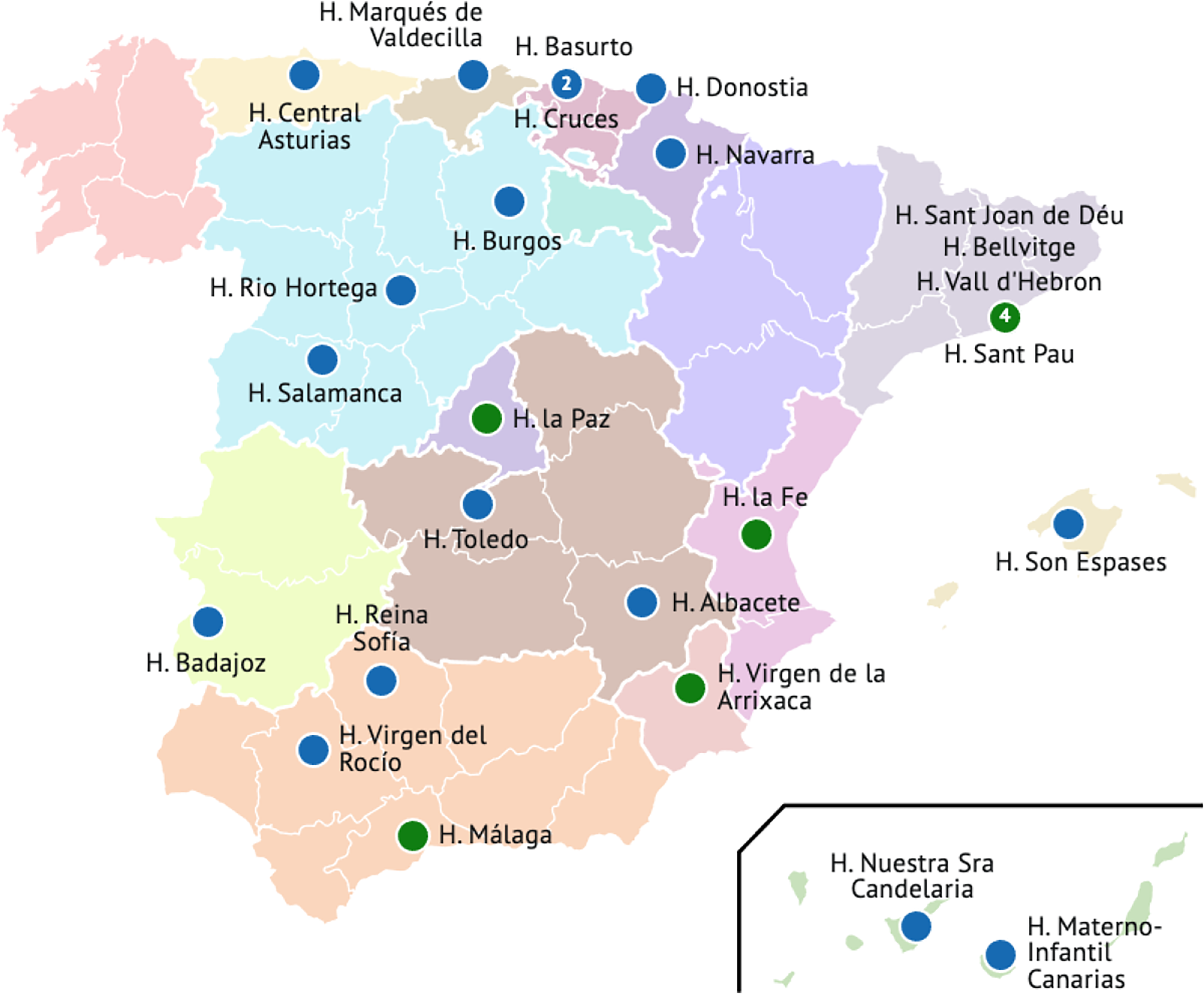
Active CuidAME centres. Centers recruiting individuals at the data cutoff of February 2025.
The distribution of SMA types was as follows: 23% (n = 125) had SMA type 1, 35% (n = 208) type 2, and 38% (n = 194) had type 3. SMA type, SMN2 copies and treatment distribution for the most prevalent types among age groups are shown in Figures 2 and 3. The distribution of individuals by SMA type reveals differences between age groups. For the purpose of the present analysis, individuals older than 16 years were classified as adults, given that in Spain, they are usually visited by adult specialists. Only twelve individuals in the registry were presymptomatic, seven with 2 SMN2 copies and five with 3 SMN2 copies. All of them were receiving treatment (50% with gene therapy, 33% with nusinersen, and 17% with risdiplam). Finally, four individuals with SMA were classified as SMA type 4.
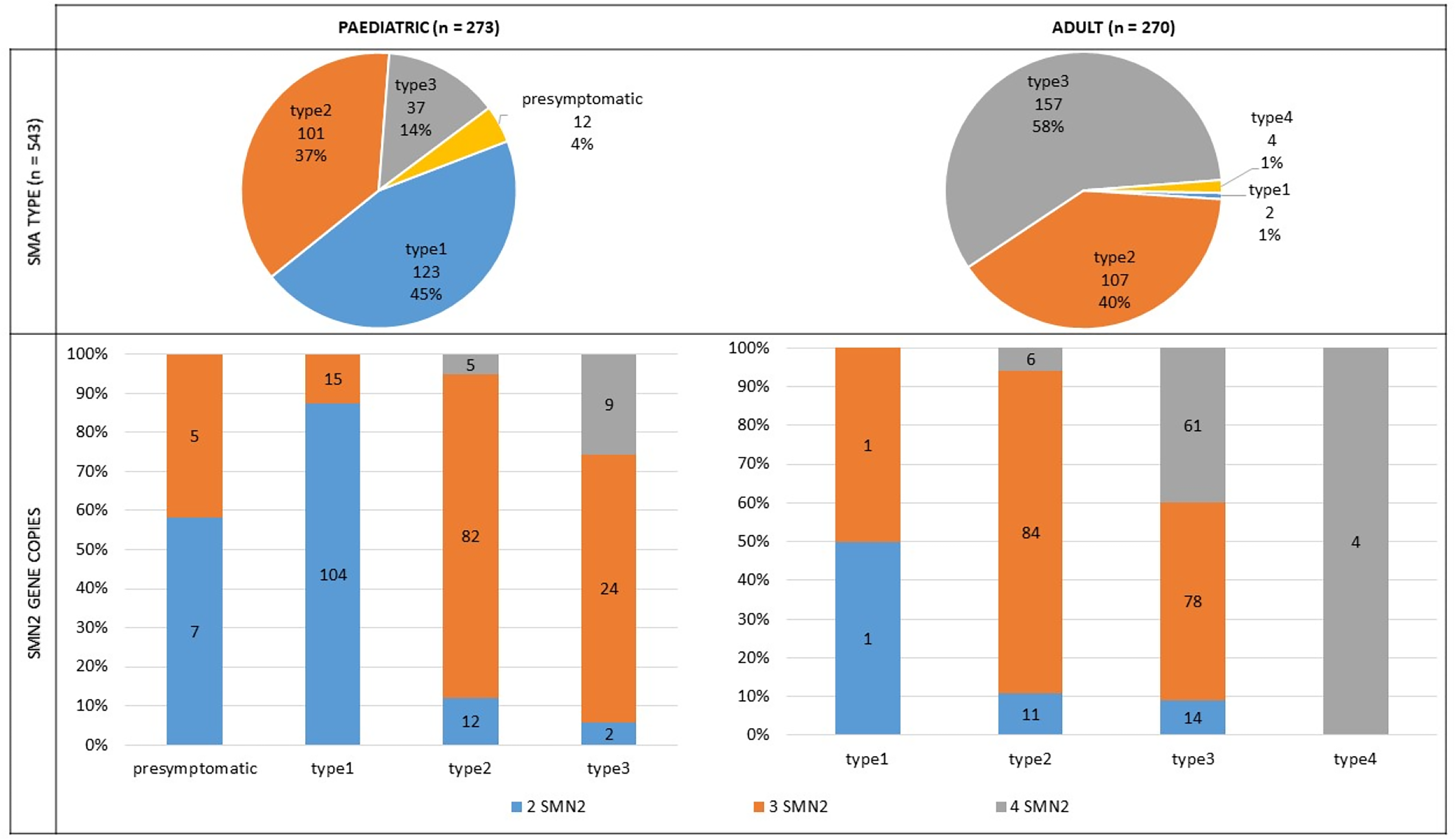
SMA type and SMN2 copies among age groups. SMA: Spinal Muscular Atrophy; SMN2: Survival Motor Neuron.
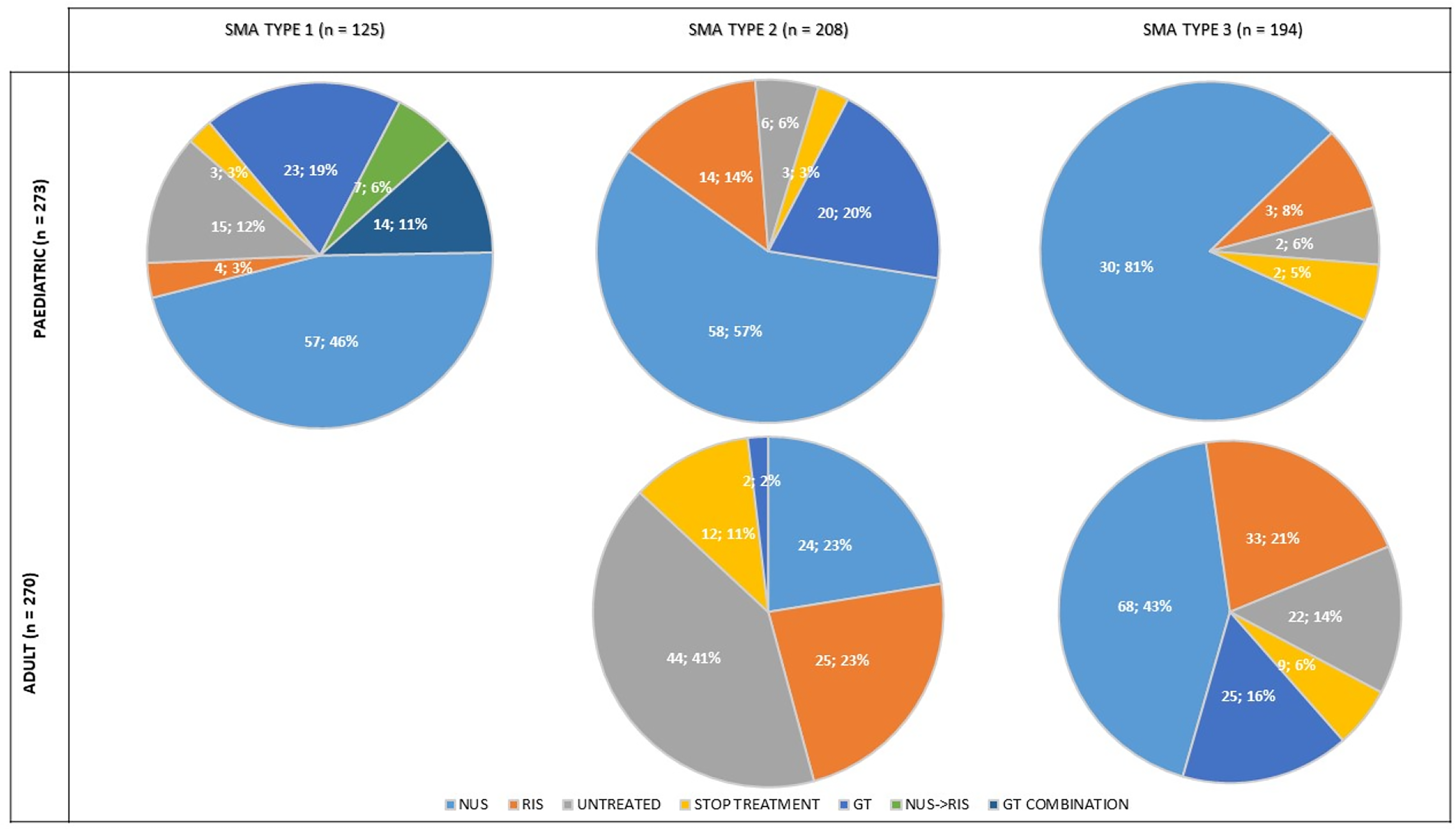
Disease-modifier treatment distribution among age groups. (1) NUS: nusinersen treatment were individuals who were treated with nusinersen only; (2) GT: gene therapy treatment were individuals who were treated with gene therapy only; (3) RIS: risdiplam treatment were individuals who were treated with risdiplam only; (4) NUS- RIS: were individuals who were treated with nusinersen initially, then stopped the medication for any reason and started their second treatment with risdiplam (note: all individuals but one switched from nusinersen to risdiplam in this group); (5) GT COMBINATION: gene therapy combination were individuals who were treated exceptionally under specific circumstances with gene therapy after or before being treated with another disease modifier treatment, mainly nusinersen; (6) STOP: individuals who were previously treated with a disease modifier treatment but discontinued treatment after a period; (7) UNTREATED: individuals who were never exposed to a disease modifier treatment. SMA: Spinal Muscular Atrophy.
Regarding disease-modifying treatments, 91% (n = 495) of the individuals received at least one treatment at some point under varying circumstances at the time of this data cutoff.
Fifty-seven individuals (10% of the total population) were followed-up and treated under any of the ongoing clinical trials in Spain. For the purpose of this analysis, these individuals were classified based on their disease modifier treatment status before the trial inclusion. Of them, 13 individuals were treatment-naïve prior to trial participation. Excluding this subgroup, the never-treated population represented 10% of the total population (n = 48). The clinical characteristics of this untreated subgroup are shown in Table 2.
Clinical characteristics of treated with disease-modifying treatments, non treated and stop-treated individuals with SMA.
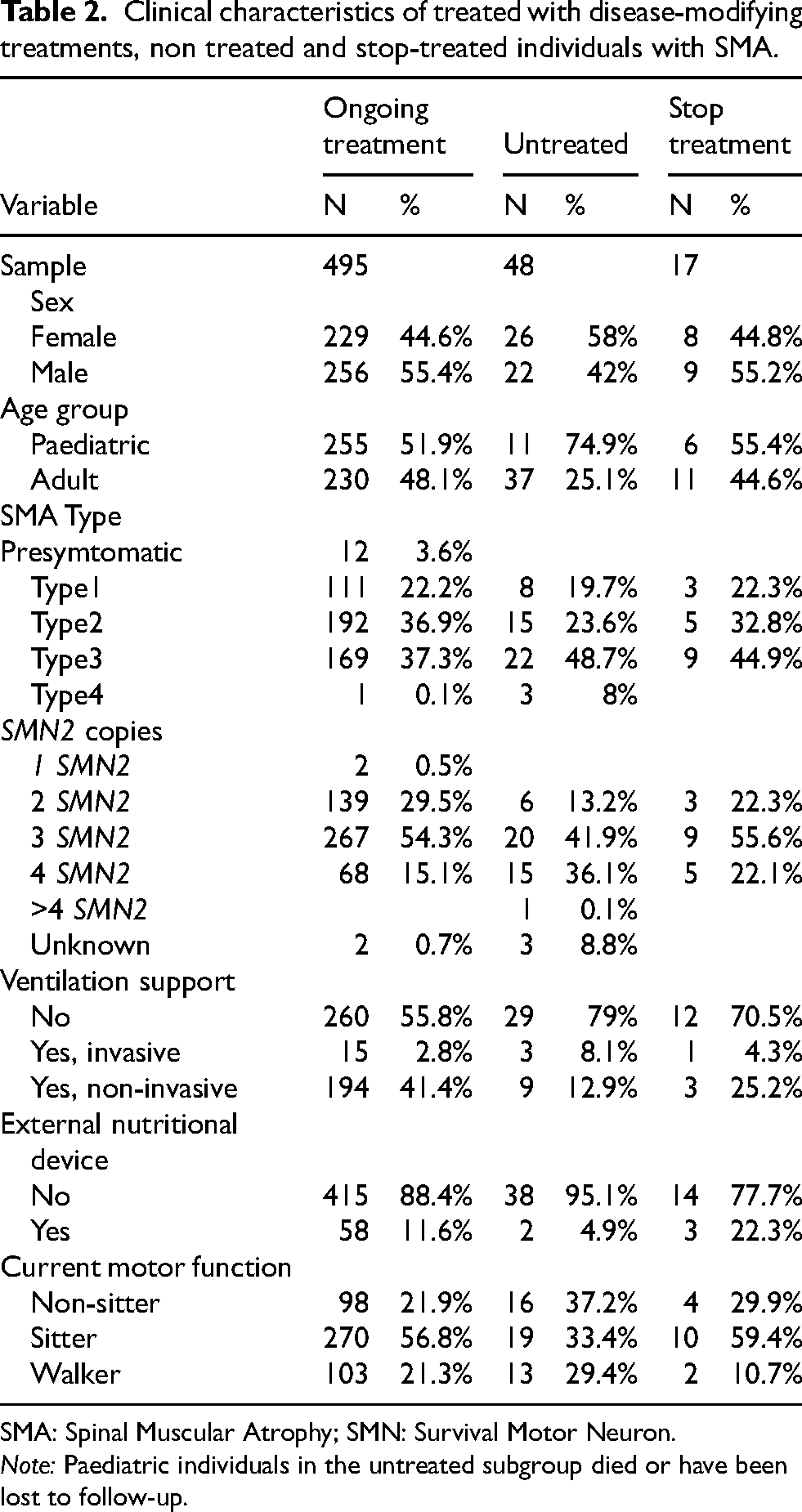
SMA: Spinal Muscular Atrophy; SMN: Survival Motor Neuron.
Note: Paediatric individuals in the untreated subgroup died or have been lost to follow-up.
Inclusion and exclusion criteria to access treatment were one of the features of the first protocol established by the Spanish Health Authorities between 2018 and 2024. Thus, some individuals with no efficacy evidence according to the protocol or safety issues were discontinued. Characteristics of the stop-treatment subgroup (n = 17) are displayed in Table 2.
Over a mean follow-up period of 4.1 years (mean 49.9, max. 105.8 months) from the first assessment until the last assessment collected, a total of 5092 motor scale assessments were registered. On average, each individual underwent four assessments for the 6MWT and five assessments for the other motor scales, with a maximum of 11 assessments for the 6MWT and 14–18 assessments for the other scales. One-third of these assessments (1565) correspond to data collected prior to treatment initiation, defined as the time from the first scale and the last scale performed during the time the patient was without treatment (scales performed the same day as the start of treatment were considered pre-treatment). The remaining 3508 assessments were performed after treatment initiation, defined as the time from the first scale and the last scale performed during the time the patient was under treatment, during a mean follow-up of 3.5 years (mean 42.3, max. 101.6 months). Detailed information for each motor scale is provided in Table 3.
Number of motor scales assessments collected.
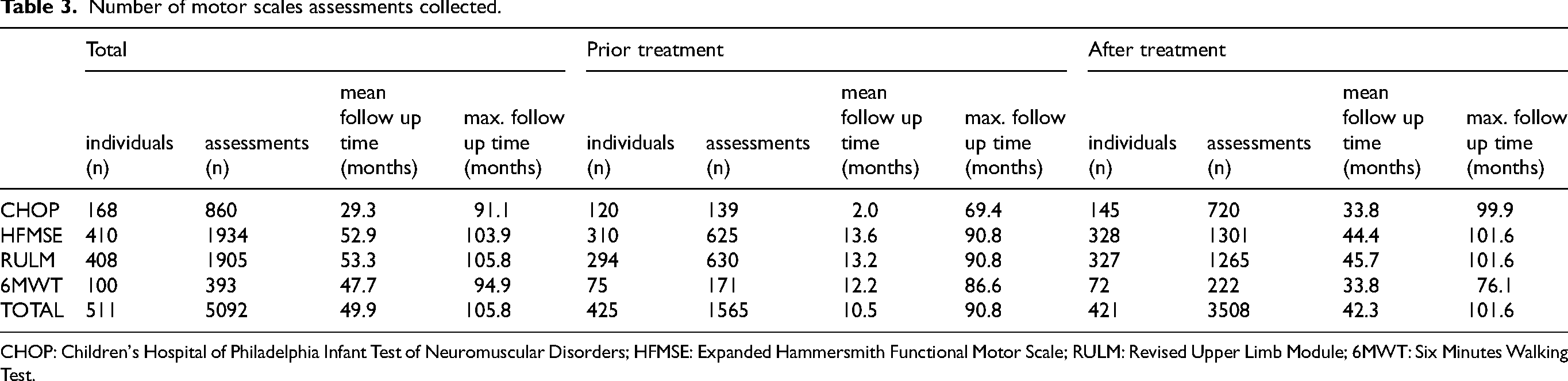
CHOP: Children's Hospital of Philadelphia Infant Test of Neuromuscular Disorders; HFMSE: Expanded Hammersmith Functional Motor Scale; RULM: Revised Upper Limb Module; 6MWT: Six Minutes Walking Test.
Discussion
The arrival of novel therapeutic options for SMA has posed scientific challenges and raised questions that clinical trial data alone cannot fully address. This underscores the critical need for initiatives that assess the response in populations with expanded access to these therapies, incorporating a diverse range of individuals with varying clinical profiles and ensuring standardized, long-term follow-up. Such projects should strive to standardize measures of disease progression and foster national networks of specialists from multiple disciplines involved in SMA care. These efforts are essential for maintaining updated treatment protocols tailored to each healthcare system, with the ultimate goal of optimizing patient outcomes and stratification.
Furthermore, these networks can facilitate the discovery of biomarkers, aid in managing expectations, and enable collaboration with similar international projects. Establishing robust national and international frameworks for SMA care not only allows for shared expertise but also enhances understanding of disease evolution and responses to therapies. Collecting long-term, real-world data to inform and refine therapeutic strategies, with the ultimate goal of achieving more adapted care for individuals with SMA across different healthcare settings is now indispensable.31,32
Despite its benefits, creating disease registries involves challenges, particularly concerning budget constraints and human resource allocation. 33 In Spain, the healthcare system´s fragmentation across 17 regions introduced administrative hurdles during the setup phase. To address these, collaboration with academic groups and patient advocacy associations has been crucial. For example, leveraging an existing legal entity dedicated to research helped streamline personnel management, data collection and processing, and resource allocation. The support of Spanish medical and scientific societies, such as the Neurology and Paediatric Neurology societies, further legitimized the project. As a result, CuidAME is widely implemented, with centres located in regions accounting for 89% of the country's population.
If we take into account epidemiological information on rare diseases in Spain where it is reported that 633 individuals with SMA lived on 1 January 2022, 34 CuidAME has recruited 83.7% of the identified SMA population in Spain, with expectations of encompassing the entire population in the near future. The type and treatment distributions observed are in line with reports from other countries, such as France or Italy.35,36 However, variations exist regarding presymptomatic individuals, which are influenced by the presence (or absence) of newborn screening programs.37–42 The CuidAME investigators emphasise the urgent need to include SMA in national newborn screening programs, given the well-documented benefits of early treatment. As of the time of this writing, only four regions in Spain have integrated SMA into neonatal screening protocols, with some pilot programs launched in recent years that lack sustained implementation.43,44
In terms of treatment, 91% of the CuidAME population have been treated, a proportion slighlty greater than the Italian population (83%) 45 Notably, CuidAME includes a subgroup of untreated individuals (n = 45) with a pool of motor scales assessments before the administration of a disease modifier treatment (1462 assessments), providing a valuable natural history comparator for future studies. Another subgroup, the “stop treatment” individuals (n = 17), will offer insights into individuals who discontinued treatment due to insufficient improvement in motor scales, technical difficulties or adverse events. It should be noted that the Spanish previous protocol, controversial criteria that were not adapted to the patient's age at treatment initiation or baseline functional level were applied, leading to the withdrawal of treatment for a group of patients who did not show improvement on the scales but maintained stability. Consequently, this was one of the most criticized aspects by the SMA community which led to a publication calling for an update of the protocol. 46 Thus, this experience laid the foundations for the new protocol in force, in which stability is considered to be a favourable response. This situation partially explains the number of treatments withdrawn during that period.
Our findings need to be contextualised within the current regulatory framework in Spain, where combinations of disease-modifying treatments or even prescribing an SMN regulator before or after gene therapy are not permitted. 47 Consequently, bridge therapy – the immediate initiation of an SMN2 regulator following diagnosis to address the delay between diagnosis and gene therapy initiation – is not authorized, unlike in other countries where it is used as good practice to shorten the time to initiate treatment in patients identified through newborn screening. This restriction likely contributes to the slower adoption of gene therapy in Spain compared to other countries. 48
A significant effort has been made to design a multipurpose registry. Through CuidAME, a national network of centers now uniformly collects and analyses SMA data. This effort has expanded the number of centres with trained personnel from three (involved in SMA clinical trials) to 25, thanks in part to CuidAME´s training initiatives. Clinical practices have been standardised nationwide, especially regarding outcome measures and follow-up frequencies. In parallel, the network facilitates smoother transitions to adult care and among hospitals, fostering collaboration and knowledge sharing even across disciplines.
In light of the transformative impact of new treatments, particularly for early-onsetindividuals with SMA, CuidAME has established working groups to address identified gaps in SMA management. Six priority areas have been identified, including biomarkers, respiratory care, cognition, gastrointestinal, rehabilitation, and adult population. Regular meetings among collaborators enable the sharing of best practices, discussion of findings, and prioritization of projects. Future efforts will focus on updating national care standards, tailoring them to the evolving needs of individuals with SMA on the one hand. On the other hand, there is an interest in advancing SMN2 gene characterisation and biomarker research, and efforts to coordinate national research in this sense are ongoing.
Like any registry, CuidAME has its limitations, along with those inherent to observational, real-world data studies. While it was designed to centralize SMA data for research and care purposes, other stakeholders – such as individuals with SMA, regulators, or payers - could benefit from this high-quality, long-term data. It is fair to mention that to guarantee sustainability, it would be desirable to diversify funding sources beyond private entities. Long-term funding is crucial for maintaining and updating the registry, as well as responding to internal and external data requests in a timely manner. Legal, ethical and administrative tasks remain time-consuming but are critical for successful project implementation and external collaborations. A well-dimensioned dedicated team is necessary to uphold quality standards across all processes.
Another key area for improvement is incorporating health-related quality-of-life assessments using patient reported outcomes (PROMs). 32 In this sense, the RegistrAME, owned by the Spanish Association of individuals with SMA (FundAME), offers complementary data, particularly in PROMs. 45 Collaboration between CuidAME and RegistrAME is essential, as standardized clinical outcomes often fail to capture subtle changes in certain patient profiles. Patient-reported outcomes can provide valuable insights into disease progression, modifying factors, and treatment responses. 49 By leveraging both registries, the SMA community in Spain can enhance data collection efficiency and gain a deeper understanding of SMA. Establishing a dedicated working group on this topic is warranted.
Finally, while CuidAME has probably already registered the majority of individuals with SMA in Spain, this is not a population-based registry and there might be selection bias. Individuals visiting small centres without neuromuscular units or those not visiting any centre are probably underrepresented in CuidAME. Therefore, the results might overestimate the use of disease-modifying treatments. However, they probably reflect accurately the characteristics of individuals with SMA being treated in Spain. Therefore, future analyses of the efficacy and safety of the disease-modifying treatments administered in the cohort are in progress.
Conclusions
CuidAME is an academic, longitudinal real-word data collection project for SMA, initiated in 2020. It currently covers the majority of the Spanish territory and follows most of the identified SMA population. This registry has demonstrated a quick start-up model that has helped standardize clinical practices and outcome measures across hospitals nationwide.
Its academic ownership and alignment with core dataset used in other international registries position CuidAME for future collaborations in research and regulatory purposes. Additionally, the creation of multidisciplinary working groups within the CuidAME network promotes knowledge sharing and addresses the scientific questions raised with the emergence of treatments that changed the paradigm of SMA care management. These collective efforts are instrumental in advancing SMA care and improving patient outcomes.
Supplemental Material
sj-pptx-1-jnd-10.1177_22143602251361190 - Supplemental material for Real-world data on spinal muscular atrophy in Spain: Insights from over 500 individuals in the CuidAME project
Supplemental material, sj-pptx-1-jnd-10.1177_22143602251361190 for Real-world data on spinal muscular atrophy in Spain: Insights from over 500 individuals in the CuidAME project by Cristina Puig-Ram, Sonia Segovia, Rocio Garcia-Uzquiano, Nancy Carolina Ñungo Garzón, Karolina Aragon-Gawinska, Mar García Romero, Jesica María Expósito-Escudero, Laura Carrera-García, Mercedes López-Lobato, Carmen Paradas, Laura González Mera, Mireia Álvarez Molinero, David Gómez Andrés, Esther Toro, Joaquín Alejandro Fernández Ramos, Maria Antonia Grimalt, Laura Toledo Bravo de Laguna, Desire González Barrios, Eduardo F Tizzano, Maria Grazia Cattinari, Julita Medina, Rocío Calvo Medina, Francina Munell, Javier Sotoca, Eduardo Martínez-Salcedo, Antonio Moreno Escribano, Mónica Povedano Panadés, Miguel A. Fernández-García, Inmaculada Pitarch-Castellano, Juan F. Vázquez-Costa, Daniel Natera-de Benito and Andres Nascimento in Journal of Neuromuscular Diseases
Footnotes
Acknowledgments
CuiAME Investigators Group: Maria Cols (Hospital Sant Joan de Déu), Oscar García Campos (Hospital de Toledo), Sergio Aguilera Albesa and Ivonne Jericó (Hospital de Navarra), Ricard Rojas Rojas (Hospital de Sant Pau i la Santa Creu), Andrea Sariego and Ana Lara Pelayo (Hospital Marqués de Valdecilla), Itxaso Martí and Roberto Fernández Torrón (Hospital de Donostia), Hilario Gómez Martín (Complejo Asistencial Universitario de Salamanca), Mario Urbano Anido (Hospital Rio Hortega), Cecilia Gil Gil and Virginia Navarro Abia (Hospital de Burgos), Maria Dolores Sardina González (Hospital de Badajoz), Jorge García García and Marta Martínez González (Complejo Hospitalario Universitario de Albacete), Solange Kapetanovic (Hospital de Basurto), Maria Jesús Martínez González and Ainhoa García Ribes (Hospital de Cruces), Raquel García Blanco and Ignacio Málaga (Hospital Central de Asturias), Sandra Roca and Obdulia Moya (Hospital Sant Joan de Déu), César Rodríguez Sánchez and Pepe Fernández-Cuesta Peñafiel (Hospital Universitario La Paz), Ana Pareja (Hospital Universitario Virgen del Rocío), Cristina Terrafeta and Carla Marco (Hospital Universitari de Bellvitge), Ana Ballester (Hospital Universitario Virgen de la Arrixaca), Oihanne Rodríguez (Biocruces). Several author of this publication are members of the European Reference Network for Rare Neuromuscular Disorders. We also thank David Hervas and EDAHULA for their contribution.
Ethical considerations
The study obtained national approval by the Ethics Committee of Hospital Sant Joan de Déu (PIC- 188-18 and EOM-02-24) on January 2019 and May 2024.
Consent to participate
Patient Information Sheets and Informed Consent Forms had been developed and approved by the ethics committee. All participants provided written informed consent prior to participating. In case of paediatric patients, parents or their legal representatives had to provide informed consent. Besides if the patient was older than twelve years, specific informed consent was required.
Author contributions
Cristina Puig-Ram has participate in the design, acquisition and data analysis, and drafted the work.
Sonia Segovia has participate in the design, acquisition and data analysis, and reviewed the content.
Rocio Garcia- Uzquiano has participate in the design, acquisition and data analysis, and reviewed the content.
Nancy Carolina Ñungo Garzón has participate in the data acquisition and reviewed the content.
Karolina Aragon-Gawinska has participate in the data acquisition and reviewed the content.
Mar García Romero has participate in the data acquisition and reviewed the content.
Jessica María Expósito-Escudero has participate in the data acquisition and reviewed the content.
Laura Carrera-García has participate in the data acquisition and reviewed the content.
Mercedes López-Lobato has participate in the data acquisition and reviewed the content.
Carmen Paradas has participate in the data acquisition and reviewed the content.
Laura González Mera has participate in the data acquisition and reviewed the content.
Mireia Álvarez Molinero has participate in the data acquisition and reviewed the content.
David Gómez Andrés has participate in the data acquisition and reviewed the content.
Esther Toro has participate in the data acquisition and reviewed the content.
Joaquín Alejandro Fernández Ramos has participate in the data acquisition and reviewed the content.
Maria Antonia Grimalt has participate in the data acquisition and reviewed the content.
Laura Toledo Bravo de Laguna has participate in the data acquisition and reviewed the content.
Desire González Barrios has participate in the data acquisition and reviewed the content.
Eduardo Tizzano Ferrari has participate in the data acquisition and reviewed the content.
Maria Grazia Cattinari has participate in the design, and reviewed the content.
Julita Medina has participate in the design, data acquisition and reviewed the content.
Rocío Calvo Medina has participate in the design, data acquisition and reviewed the content.
Francina Munell has participate in the design, data acquisition and reviewed the content.
Javier Sotoca has participate in the design, data acquisition, and reviewed the content.
Eduardo Martínez-Salcedo has participate in the design, data acquisition and reviewed the content.
Antonio Moreno Escribano has participate in the design, data acquisition and reviewed the content.
Mónica Povedano Panadés has participate in the design, data acquisition and reviewed the content.
Miguel A. Fernández-García has participate in the design, data acquisition and reviewed the content.
Inmaculada Pitarch-Castellano has participate in the design, data acquisition and reviewed the content.
Juan Francisco Vázquez-Costa has participate in the design, data acquisition and reviewed the content.
Daniel Natera-de Benito has participate in the interpretation of the data, and reviewed the content.
Andres Nascimento has participate in the design, interpretation and acquisition of the data, and reviewed the content.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this Project has been provided by Biogen, Novartis and Roche. Funders are not involved in determining article content, CuidAME registry management, or centers involved.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel Natera-de Benito, Andrés Nascimento, Francina Munell, Joaquín Alejandro Fernández Ramos and Miguel A. Fernández-García have received honoraria for speaking engagements, participation in advisory boards, and compensation for attending conferences from Biogen, Novartis, and Roche.
Nancy Carolina Ñungo has received honoraria for speaking engagements, participation in advisory boards, and compensation for attending conferences from Roche.
Karolina Aragon-Gawinska received lecture honoraria, travel and congress funding from Biogen and Roche.
Juan Francisco Vázquez-Costa and Javier Sotoca has received honoraria for speaking engagements, participation in advisory boards, and compensation for attending conferences from Biogen and Roche.
Mireia Álvarez Molinero has received honoraria for speaking engagements from Biogen and Roche.
Laura Carrera-García has received honoraria for speaking engagements and travel and congress funding from Biogen and Roche.
The rest of authors have no conflict of interest to report.
Data availability statement
Data will be available under reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
