Abstract
Four patients with myotonic dystrophy type 1 (DM1) and a patient with paramyotonia congenita (PMC) developed devastating painful muscle stiffness with markedly elevated serum creatine kinase (CK) levels after 20 weeks of gestation. Immediately after delivery, painful muscle stiffness completely disappeared and the CK levels returned to the baseline. In a patient with DM1, muscle biopsy at delivery and skeletal muscle MRI in six days after delivery showed inflammatory changes, which disappeared in MRI on postpartum day 41. Pregnancy-associated aggravation of myotonia has been reported in myotonic disorders, but painful muscle stiffness has been scarcely reported. Inactivation of muscle Cl channel by progesterone that is prominently increased in the middle phase of gestation is likely to be the underlying mechanism of the pregnancy-associated painful muscle stiffness in myotonic disorders.
Keywords
Introduction
DM1 is an autosomal dominant disorder characterized by myotonia, muscle weakness, smooth muscle involvement, endocrine and glucose metabolic disorders, cardiac conduction blocks, and cataracts.1,2 Pathogenic variants responsible for DM1 are abnormal expansions of a CTG repeat, (CTG)n, in the 3′ untranslated region of the DMPK gene encoding DM1 protein kinase located on chromosome 19q 13.3. 3 Pregnancy-associated aggravation of myotonia has been reported in patients with DM14–6 and myotonic dystrophy type 2 (DM2), 7 as well as in patients with non-dystrophic myotonias due to Na and Cl channelopathies.8–11 The aggravation of myotonia generally resolves after delivery. In a report of the impact of pregnancy in 122 patients with DM1, the ratio of patients with pain was gradually increased up to six months after delivery, but the organs and the nature of the pain were not indicated. 12 Similarly, in a report of the impact of 51 pregnancies in patients with non-dystrophic myotonias, muscle pain was worsened in 24% during pregnancy and resolved completely after pregnancy in about two-thirds in 2 days to 12 months. 11 We here report painful muscle stiffness with markedly elevated serum creatine kinase (CK) levels after 20 weeks of gestation in four patients with DM1 and in a patient with PMC, a Na channelopathy. 13 Immediately after delivery, painful muscle stiffness disappeared and serum CK levels were markedly reduced. Painful muscle stiffness developing in the middle phase of pregnancy may have been underdiagnosed and underestimated in myotonic disorders.
Case report
Patient (Pt) 1 (a 30-year-old primigravida) visited our hospital for prenatal care at 28 weeks of gestation. The pregnancy was uneventful up to approximately 25 weeks. Subsequently, she intermittently suffered from painful muscle stiffness of the proximal limb muscles during motion but minimally at rest. Proximal leg muscles were more affected than proximal arm muscles. At 33 weeks, she lost independent ambulation because of painful muscle stiffness and became dependent on a wheelchair. Her activity of daily living (ADL) was, therefore, significantly compromised and serum CK levels (normal ≤ 140 IU/L) were markedly elevated, both of which lasted until the end of pregnancy (Figure 1).
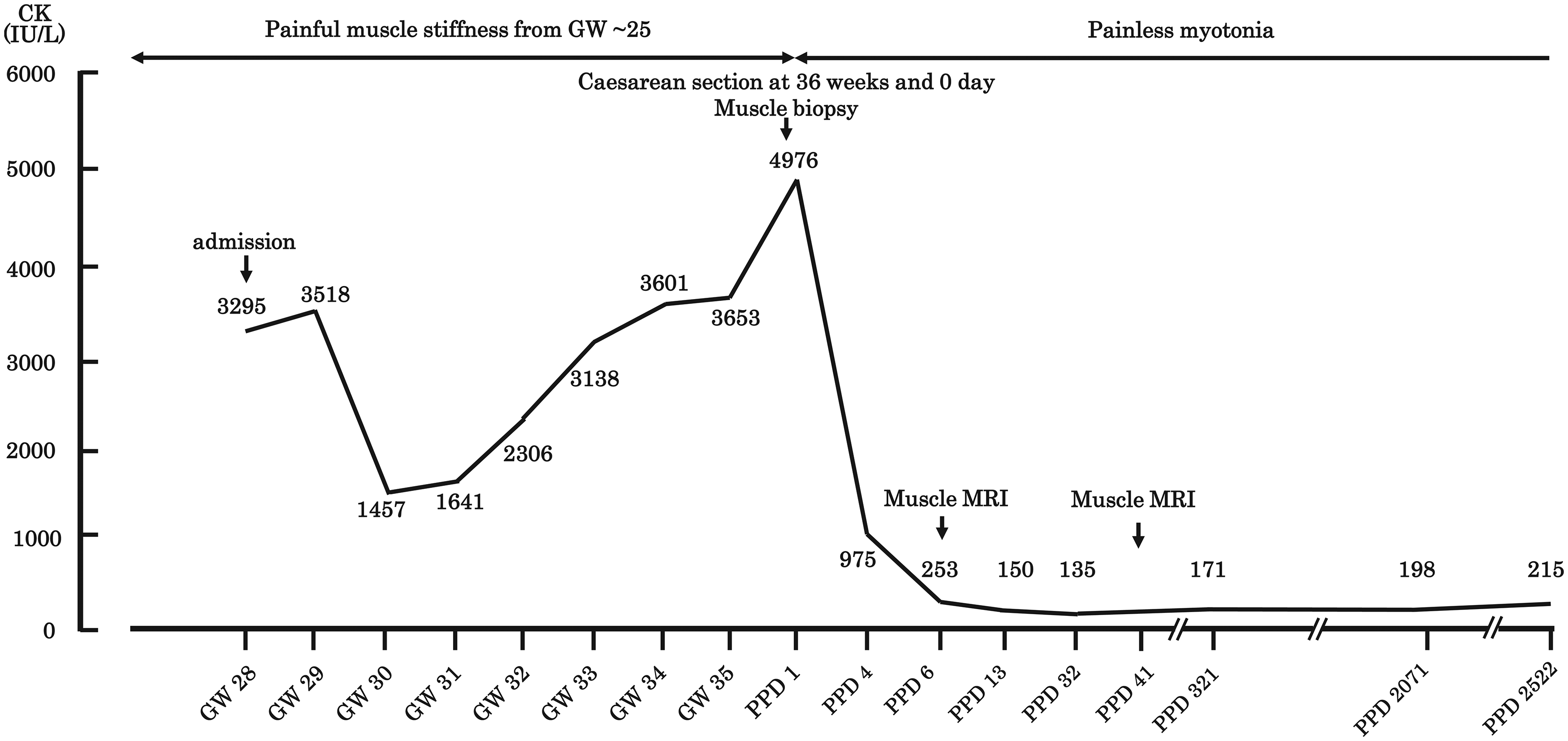
Clinical features and serum CK levels during and after pregnancy in Pt 1with DM1. Pt 1 developed painful muscle stiffness and elevated serum CK levels at approximately 28 weeks of gestation (GW). Painful muscle stiffness disappeared immediately after delivery and serum CK levels returned to the baseline in postpartum days (PPD).
The facial muscles, sternocleidomastoid muscles, and distal limb muscles showed mild weakness. Painful muscle stiffness precluded the examination of grip myotonia, whereas percussion myotonia was present. Her mother was previously diagnosed with DM1. To determine the (CTG)n expansion of DMPK, Southern blot analysis was performed as described previously. 14 Briefly, genomic DNA extracted from leukocytes was digested with a restriction enzyme EcoRI and hybridized with a 32P-labeled cDNA25 probe. The sizes of EcoRI-digested fragments were 9.8 and 11.9 kb, indicating ∼700 repeats of (CTG)n, and adult DM1 was diagnosed.
In order to explore the causes of painful muscle stiffness, the right rectus femoris muscle was biopsied during the caesarean section. In hematoxylin-eosin (HE) staining, infiltration of lymphocytes was observed around the perivascular areas in bundles of muscle fibers (Figure 2). In addition, both basophilic atrophic/necrotic fibers were observed. In contrast, central myonuclei were not conspicuous. The dystrophic changes were consistent with those observed in DM1, and an overlap of myositis was speculated. On postpartum day (PPD) 6, the proximal muscles of bilateral lower extremities were examined by MRI. Although the MRI images were taken 16 years ago and were deleted from the archived records, a neuroradiologist diagnosed high signals in T2-weighted images in left vastus lateralis and medialis muscles, bilateral semitendinosus muscles, bilateral gracilis muscles, and bilateral sartorius muscles. Both the pathological and MRI findings point to inflammatory changes in the skeletal muscles.
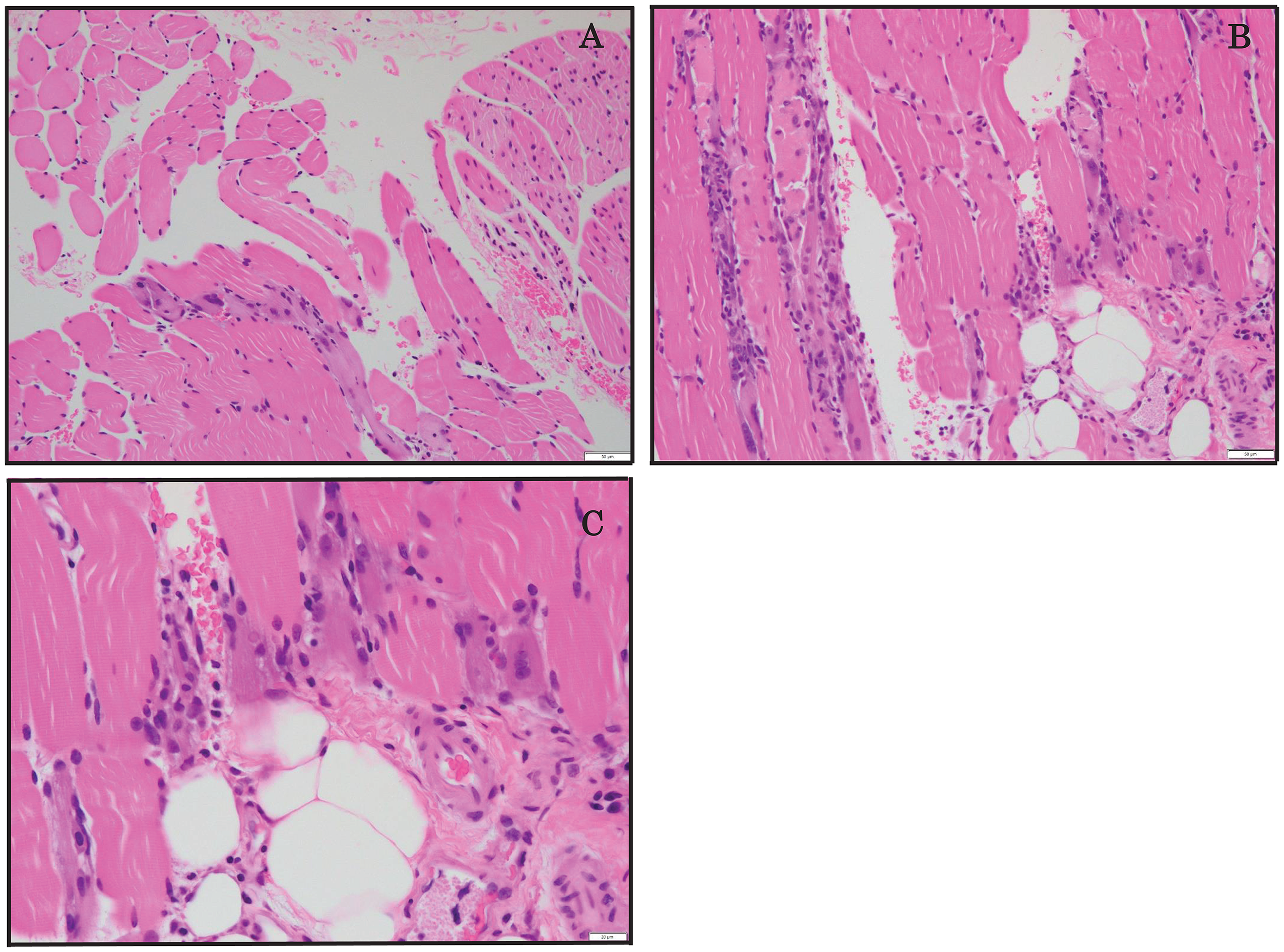
Representative hematoxylin-eosin (HE) staining of the biopsied right rectus femoris muscle in Pt 1. Lymphocytes were infiltrated around the perivascular areas in bundles of muscle fibers. Both basophilic and atrophic/necrotic fibers were present. In contrast, central myonuclei were not prominent. Scale bar = 50 µm (A, B) and 20 µm (C).
Immediately after delivery, painful muscle stiffness was resolved, and the serum CK level returned to the baseline in 6 days (Figure 1). Amelioration of painful muscle stiffness unmasked grip myotonia. She became ambulatory again on PPD 2 (Figure 1). On PPD 41, MRI of lower limb muscles showed a marked reduction of high signals in T2-weighted images.
Pts 2 (a 32-year-old primigravida), 3 (a 28-year-old primigravida), and 4 (a 37-year-old secundigravida) with DM1, as well as Pt 5 (a 36-year-old primigravida) with PMC, developed painful muscle stiffness at approximately 27, 20, 18, 20 weeks of gestation, respectively (Figure 3). Patients complained of painful muscle stiffness when they moved the muscles but minimally at rest. Similar to Pt 1, proximal leg muscles were more affected than proximal arm muscles in Pts 2, 3, 4, and 5. The serum CK levels were markedly elevated in Pts 2, 3, and 4, and moderately elevated in Pt 5, after the onset of painful muscle stiffness. Additionally, ADLs in Pts 2, 3, 4, and 5 were severely compromised by painful muscle stiffness. The painful muscle stiffness lasted until the end of pregnancy. Pt 4 had an episode of preceding pregnancy with painful muscle stiffness at approximately 20 weeks of gestation at age 34 years, but serum CK levels were not available. Pt 4 had no similar symptoms between pregnancies. Again, in Pts 2, 3, and 4, muscle pain precluded the examination of grip myotonia, whereas percussion myotonia was present. Amelioration of painful muscle stiffness after delivery unmasked grip myotonia in Pts 2, 3, and 4. Southern blotting showed that the sizes of EcoRI-digested fragments in Pts 2, 3, and 4 were 9.8/11.3, 9.8/11.6, and 8.6/11.3 kb, indicating ∼500, ∼600, and ∼500 repeats of (CTG)n, respectively, which confirmed the diagnosis of adult DM1. In Pt 2, MRI of proximal lower extremities at 36 weeks of gestation showed high signals in T2-weighted images in bilateral gracilis, bilateral semimembranous, bilateral biceps femoris, right rectus femoris, right sartorius, and left adductor longus muscles, while MRI on PPD 32 showed disappearance of high signals. MRI images were taken 12 years ago and were deleted from the archived records. Pt 5 with PMC had a hemiallelic pathogenic variant in p.T1313M in SCN4A encoding skeletal muscle Na channel. 13 Pt 5 had painful myotonia that was provoked by cold or exercise. The nature and the degree of painful muscle stiffness after 20 weeks of gestation in Pt5 was different from that before pregnancy. Pt 5 took ritodrine (15 mg, orally three times daily), a short-acting selective β2 agonist to prevent uterine contractions, at 30 weeks and 4 days of gestation for 2 days, and developed transient rhabdomyolysis (Figure 3(d)). Ritodrine-induced rhabdomyolysis has been reported in nine patients with DM1. 15 The other patients did not take ritodrine. Similar to Pt 1, painful muscle stiffness and the reduced ADL were resolved immediately after delivery and the serum CK levels were markedly reduced in Pts 2, 3, 4, and 5.
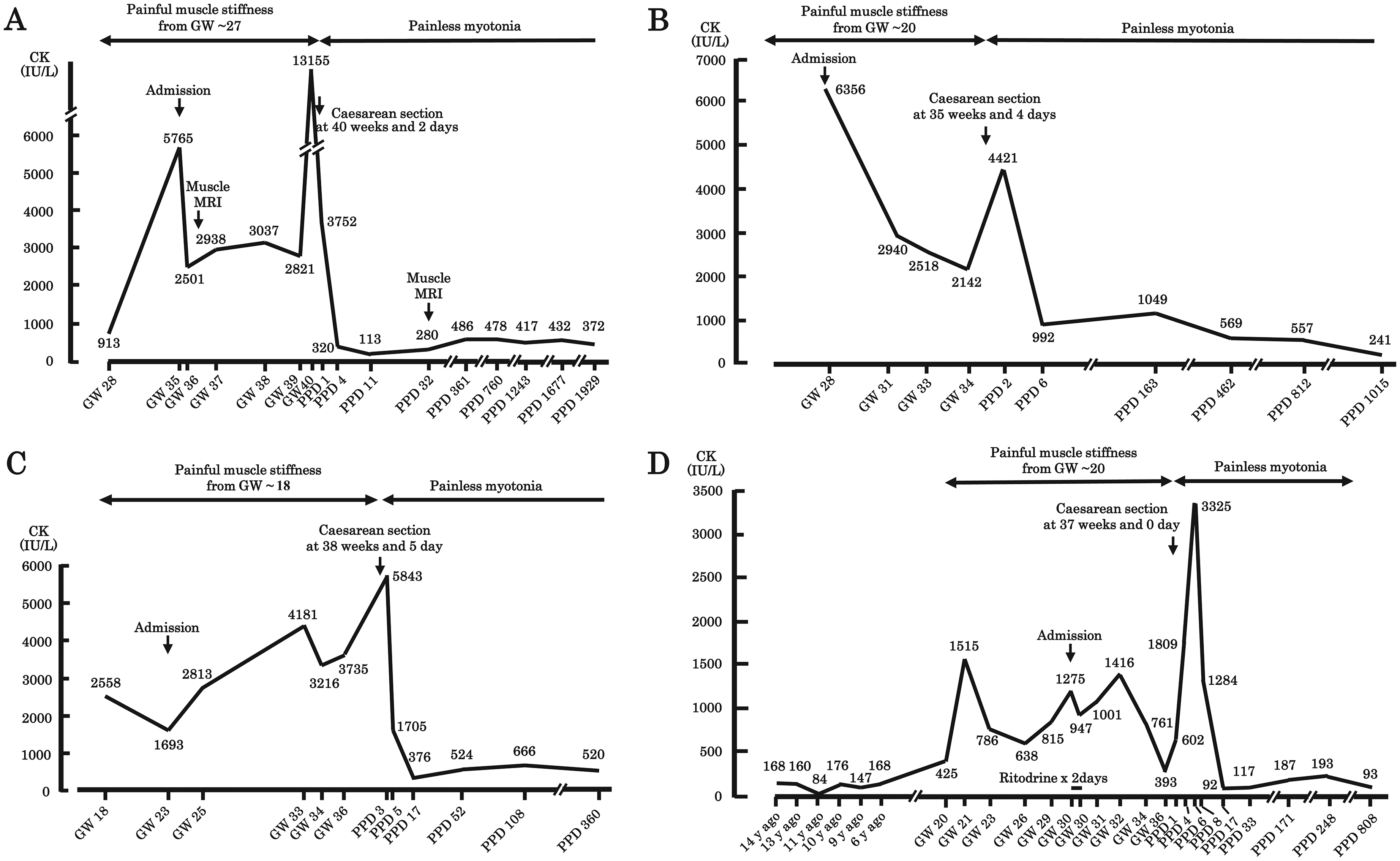
Clinical features and serum CK levels during and after pregnancy in Pts 2 (A), 3 (B), and 4 (C) with DM1 and Pt 5 with PMC (D). Pts 2, 3, 4, and 5 developed painful muscle stiffness at 27, 20, 18, 20 weeks of gestation (GW), respectively. Similarly, elevated serum CK levels were shown at 28, 28, 18, 20 weeks of GW, respectively. After delivery, painful muscle stiffness was immediately resolved and serum CK levels were markedly reduced in all the patients. In Pt 5 with PMC, administration of ritodrine at 30 weeks and 4 days of gestation deteriorated painful muscle stiffness and increased serum CK levels. However, they were relieved by the termination of ritodrine.
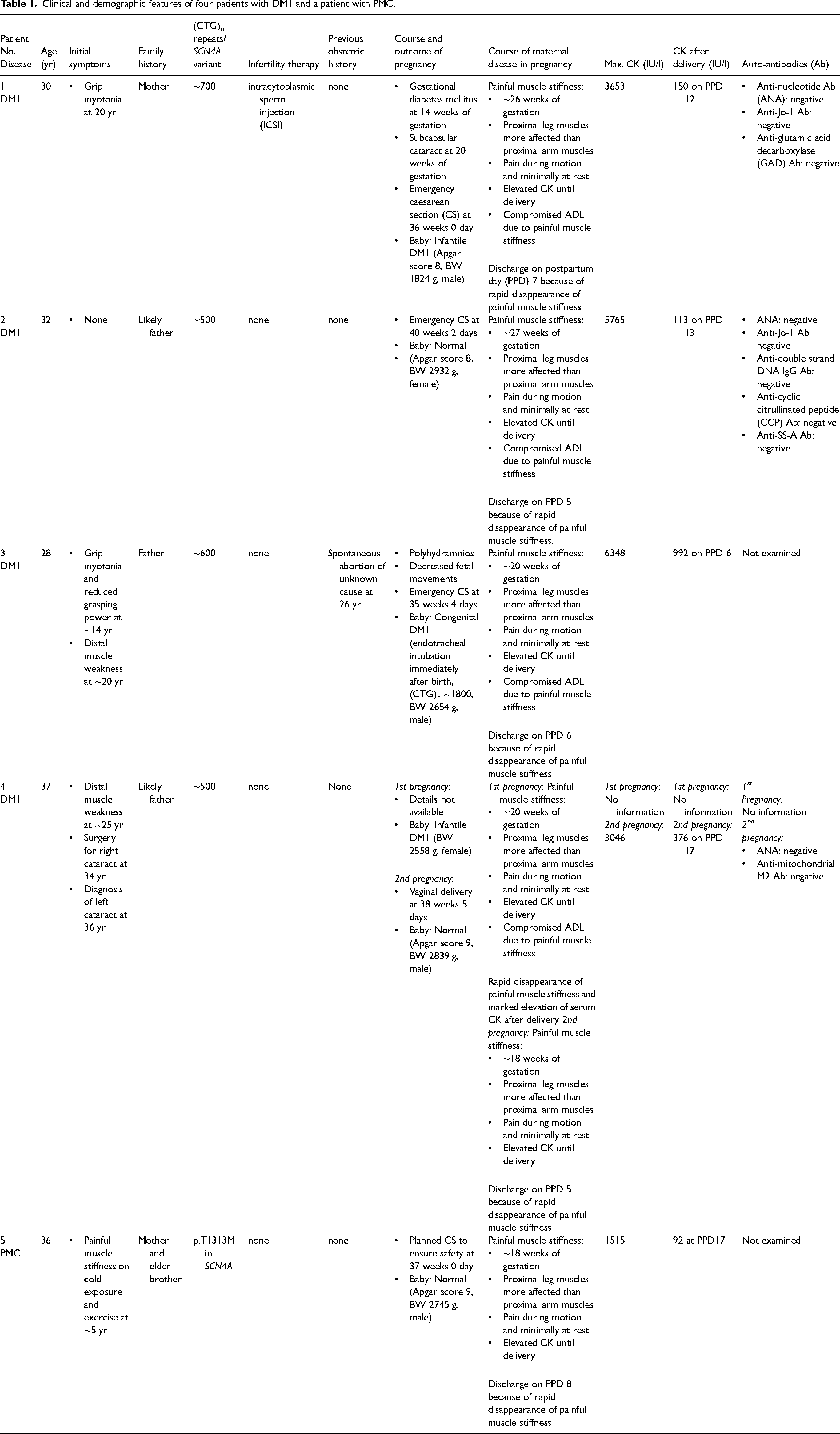
Pts 1 to 4 had DM1, but none of them were diagnosed with DM1 before pregnancy. Pt 5 was diagnosed with PMC at age 14 years, 22 years before pregnancy. Pt 1 had intracytoplasmic sperm injection (ICSI). The other four patients did not receive any assistive reproductive technologies including hormone therapy or in vitro fertilization. Pt 3 showed polyhydramnios and decreased fetal movements. Babies of Pts 1 and 3 had infantile DM1 and congenital DM1, respectively. The other patients and babies had no pregnancy-related problems including decreased fetal movements; peripartum complications like hemorrhage, anesthetic problems, pre-eclampsia, or prolonged labor; and neonatal issues like a stillborn child. Serum calcium levels during pregnancy and after delivery and parathyroid hormone levels after delivery were within normal ranges. Mild elevation of free T4 and mild suppression of TSH occur in normal pregnancy. 16 Although we did not examine thyroid functions during pregnancy, thyroid functions were within normal ranges after delivery in all the patients. In all the patients, serum CRP levels were within normal range and leukocytosis was not recognized during pregnancy or after delivery. Serum auto-antibodies were examined in Pts 1, 2, and 4, and were all negative (Table 1). Before the onset of painful muscle stiffness, no medication was initiated and no recognizable infection or fever was documented. Viral serological examination was not performed in any patients. None of the patients were treated for myositis after the onset of painful muscle stiffness. We followed up Pts 1 to 5 for 16.5, 12.2, 9.3, 4.8, and 5.6 years, respectively, after the onset of painful muscle stiffness. After the delivery, none became pregnant again and serum CK levels were always less than 500 IU/L. Clinical and demographic features of the five patients are summarized in Table 1.
Clinical and demographic features of four patients with DM1 and a patient with PMC.
Discussion
Four patients with DM1 and a patient with PMC showed painful muscle stiffness with markedly elevated serum CK levels after 20 weeks of gestation. In all the patients, delivery immediately relieved painful muscle stiffness and significantly lowered serum CK levels. In contrast to our patients, serum CK levels are not changed during normal pregnancy in normal subjects. 17
Worsening of myotonia during pregnancy has been reported in DM1,12,18 myotonic dystrophy type 2 (DM2),7,12 Na channelopathy (PMC 8 and Na channel myotonia 19 ), and Cl channelopathy (Thomsen’ s disease9,10). Myotonia is usually painless in DM1,20,21 but painful muscle stiffness during pregnancy has been reported in two patients with DM1.22,23 The first patient was a 26-year-old primigravida with DM1, who developed intermittent painful dystonic postures of the hands and feet, which was expressed as myotonic crisis, after 23 weeks of gestation. 22 The symptoms lasted until the 41st week of gestation when caesarean section was performed, and the symptom resolved after delivery. Serum CK levels were not documented. Muscle biopsy and skeletal muscle MRI were not performed. The second patient was a 44-year-old woman with DM1, who developed symmetrical proximal myalgia with elevated serum CK up to 9040 IU/L. 23 Lower limb MRI showed increased T2 signals suggesting myositis. Targeted muscle biopsy showed muscle fiber regeneration subsequent to a minor recent necrotizing myopathic process, but lymphocyte infiltration was not as conspicuous as that in Pt 1.
Serum free calcium is a critical determinant of muscle stiffness. Although total serum calcium levels are decreased during pregnancy due to hemodilution and subsequent decrease of serum albumin concentrations, the serum free calcium levels remain unchanged during pregnancy.24,25 Although we did not measure serum free calcium levels, calcium deficiency was unlikely to have accounted for painful muscle stiffness in our patients. Pregnancy-associated painful muscle stiffness with elevated CK in our patients, as well as in previously reported patients with DM1 23 and DM2, 7 were likely to be related to modulations of hormones including human chorionic gonadotropin, estrogen, progesterone, human placental lactogen, relaxin, prolactin, and oxytocin. In addition, the symptoms might be related to elevation of growth hormone, free T4, ACTH, deoxycorticosterone, cortisol, and aldosterone during pregnancy.16,25,26 Among these hormones, progesterone markedly suppresses openings of human muscle Cl channels artificially expressed in Xenopus laevis oocytes 27 and mouse muscle Cl channel nascently expressed in mouse skeletal muscles. 28 In pregnancy, progesterone is initially secreted by the corpus luteum, and later by the placenta. Although we did not measure serum progesterone levels in our patients, serum progesterone levels are markedly elevated after the 22nd to 24th weeks of gestation with maturation of placenta, and rapidly drop after delivery. The increasing serum progesterone levels and the subsequent inhibition of muscle Cl channels are likely to account for the worsening of myotonia after the middle phase of pregnancy. In DM1, aberrant splicing of CLCN1 encoding muscle Cl channel reduces the expression and the conductance of Cl channel, 29 but this should not be observed in PMC. Thus, Cl channels are expected to be more suppressed in DM1 than in PMC. Indeed, in Pts 1 to 4 with DM1, as well as in a previously reported patient with DM1, 23 serum CK levels were increased to more than 5000 IU/l during pregnancy, whereas, in Pt 5 with PMC, the maximum serum CK level was 1515 IU/l. Thus, the degree of Cl current suppression was positively correlated with the degree of elevated CK levels. Similar mechanisms are likely to be operational in pregnancy-associated worsening of painful muscle stiffness in previously reported patients with DM2,7,12 Na channelopathy (PMC 8 and Na channel myotonia 19 ), and Cl channelopathy (Thomsen’ s disease9,10). In addition, myositis was likely to be overlapped in our patients, as observed in muscle biopsy in Pt 1 and high T2 signals in muscle MRI in Pts 1 and 2. Four following observations are in accordance with the notion that pregnant DM1 patients are predisposed to develop abnormal inflammation. First, progesterone is chemically more irritating than other steroids. 30 Indeed, there is an intriguing case report showing that intramuscular injection of progesterone caused severe thigh myositis in a patient with in vitro fertilization, which was confirmed by the ultrasound examination and the microscopical examination of aspirated muscle pus. 31 The ultrasound examination on day 53 after cessation of progesterone treatment showed no abnormality in this patient. Thus, increased progesterone might have enhanced muscle inflammation in our patients. Second, we observed ritodrine-induced rhabdomyolysis in Pt 5. Ritodrine causes rhabdomyolysis in pregnancy even in normal subjects, but DM1 patients are prone to develop ritodrine-induced rhabdomyolysis more than normal subjects. 15 Third, transcriptomic analysis of glial cell model of DM1 revealed that inflammation and immune responses were the major dysregulated cellular processes. 32 Fourth, in DM1 skeletal muscles, the TWEK/Fn14/NFκB pathway causing inflammation was activated. 33 However, as painful muscle stiffness is not always observed in DM1 or PMC, additional unidentified factor(s) are required to develop pregnancy-associated painful muscle stiffness.
Footnotes
Acknowledgements
We would like to acknowledge Shinji Itoyama, MD and Jyunichi Tamaru MD at Department of Pathology of Saitama Medical Center, Saitama Medical University for their pathological contributions to muscle biopsy. We also would like to acknowledge Takenori Okada, MD at Department of Radiology of Saitama Medical Center, Saitama Medical University for his neuroradiological contributions to muscle MRI.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This report was financially supported by the Ministry of Health, Labour and Welfare of Japan (Grant No. 23FC1014) and the National Center of Neurology and Psychiatry (Grant No. 5-6).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
