Abstract
Background:
Duchenne muscular dystrophy (DMD) is a progressive muscular disease associated with muscle fiber degeneration and increased inflammatory responses including Interleukin-1 beta (IL-1β) and other cytokines. Canakinumab (Ilaris) is an anti-human IL-1β monoclonal antibody that neutralizes IL-1β.
Methods:
We completed an open-label, single dose pilot study of canakinumab 2 mg/kg subcutaneous injection in steroid naïve boys with DMD older than 2 years of age to determine safety and potential serum response biomarkers of efficacy at 4-weeks post treatment. Proteome profiling was performed using high throughput multiplexing aptamer SomaScan assay based technology targeting 1,500 unique serum proteins.
Results:
Three subjects completed the study with no adverse events reported and no significant changes in safety labs. Proteomic analysis within 4 weeks of treatment identified significantly decreased inflammation associated factors including plasma serine protease inhibitor, interleukin-6 receptor alpha, Lymphocyte antigen 86, Immunoglobulin D and myostatin. Significantly increased proteins included muscle-associated proteins aldolase A and lactate dehydrogenase B.
Conclusions:
Canakinumab 2 mg/kg dose is safe for children with DMD and demonstrated potential response biomarkers of efficacy in treating related muscle disease. Canakinumab did not affect the circulating levels of IL-1β but did decrease some key proinflammatory markers and myostatin. Increased muscle specific proteins could be associated with increased physical activities or damage seen in young patients with DMD. Further studies using canakinumab for a longer treatment period may demonstrate increased benefit.
Introduction
Duchenne muscular dystrophy (DMD) is a rapidly progressive form of muscular dystrophy that occurs in males and manifests prior to the age of six years. DMD affects approximately 1 in 3600 to 9300 male births worldwide. 1 DMD is caused by mutations in the dystrophin gene which codes for a protein that provides structural stability to the dystroglycan complex on muscle cell membranes. 2 Due to membrane instability, up-regulated inflammatory gene expression and activated immune cell infiltrates including interleukin 1-beta (IL-1β), at least partially mediated by nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), are evident during early disease stages and play a significant role in DMD disease progression. 3 Increased secretion of IL-1β was seen after lipopolysaccharide simulation in the dystrophin deficient mdx mouse model. 4 It was also shown that IL-1β plays a crucial role in the loss of muscle regeneration capacity of human DMD myogenic cells. 5 And blocking NF-κB activity was shown to reduce inflammatory responses and IL-1β levels in dystrophin deficient mice. 6 This NF-κB inhibiting effect is similar to glucocorticoid treatments. As such, anti- IL-1β blockade has the potential to act synergistically with the NF-κB -inhibiting effects of glucocorticoids to reduce muscle inflammation and improve muscle strength in DMD. 7
Canakinumab is a recombinant human anti-human-IL1β monoclonal antibody of the IgG1/ kappa isotype that binds to human IL1β and neutralizes its activity by blocking its interaction with IL-1 receptors. It is FDA approved for use in children with pediatric inflammatory conditions including familial Mediterranean fever, systemic onset juvenile idiopathic arthritis, and TNF-receptor associated periodic fever syndrome (see package insert). Since an abnormal immune response in muscle is part of the pathophysiology of DMD, canakinumab may be efficacious in preventing manifestations of this disease. Therefore, we completed a pilot study of canakinumab in children with DMD to demonstrate safety and changes in potential response biomarkers of inflammation.
Materials and methods
Clinical trial: This is an open-label, single dose pilot study to assess safety and to evaluate short-term changes in biomarkers in steroid naïve children with DMD (NCT03936894). The study was approved by the Institutional Review Board at Children's National Hospital (protocol # 10234) and enrolled subjects from 2019–2022. All participants provided written informed consent prior to enrollment in the study. This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. Children were treated with a single dose of canakinumab 2 mg/kg subcutaneously with no pretreatment. The study was comprised of a screening visit with baseline lab assessment, treatment day, phone assessment 3–5 days later, a 10–14 day post-treatment evaluation with safety labs, and a 30 day post-treatment evaluation with safety labs. Serum for biomarker analysis was obtained at baseline and 30 day visits. Enrolled subjects had genetically confirmed diagnosis of DMD, were older than 2 years of age, had not started steroid treatment or other immunosuppressive therapies, had a negative Quantiferon-Gold tuberculosis screen, had no active infections or renal/hepatic impairment, no evidence of cardiomyopathy on echocardiogram, and remained ambulatory. Baseline lab values were in the reference range for healthy individuals or if outside this range, determined to be not clinically significant by the principal investigator. Study data were collected and managed using REDCap (Research Electronic Data Capture) tools hosted at Children's National.8,9
Serum proteome profiling: Serum samples were collected and stored following an established SOP protocol (Supplemental Material 1). Aliquots of 150 μL were prepared from each sample and submitted for proteome profiling using high throughput multiplexing aptamer-based technology known as SomaScan assay (SomaLogic Inc., Boulder, CO) as previously described.10,11 In this study we used a SomaScan assay targeting 1500 serum proteins.
Statistical analysis: For each sample and protein, the difference in expression was calculated between the baseline protein expression value and the corresponding value at 4 weeks. Normality was assessed on the difference values and these difference values were compared to the null value of 0 for each protein using a one-sample t-test or Wilcoxon sign rank test as appropriate. Due to having only 3 biological samples, assessments of normality can be problematic, however due to the hypothesis generating nature of this experiment, those difference values shown to be normal via the Shapiro-Wilk normality test utilized a parametric t-test. Summary statistics for the 4 week differences were reported and p-values were not adjusted for multiple testing. With the small sample size, it is expected that few proteins will show evidence of any statistical effect, and it is understood that those that do could possibly be false positives. Therefore, the purpose of this analysis was to generate hypotheses to direct further experiments rather than explicitly test hypotheses.
Results
Three patients aged 4, 4 and 5 years old completed the pilot study receiving 2 mg/kg dose of canakinumab. The initial study design included a second cohort of 3 patients at 4 mg/kg dose of canakinumab, but the study was stopped early due to slow enrollment secondary to pandemic which closed protocols to new enrollment for approximately 6 months at our institution and limited in person clinic and research visits making recruitment more difficult. For the three patients that completed the study, there were no adverse events including no local reactions at the site of injection. Subject 1 had an IL-1β level of 2143 at baseline and 2516 at 4 weeks. Subject 2 had an IL-1β level of 2828 at baseline and 2484 at 4 weeks. Subject 3 had an IL-1β level of 3437 at baseline and 3246 at 4 weeks. There were no significant changes in lab work over the one-month period (Table 1). Liver enzymes aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were increased at baseline in all three patients, consistent with DMD. 12
Lab values for 3 study participants at baseline (0 wk), two weeks (2 wk) and four weeks (4 wk) post canakinumab 2 mg/kg treatment.
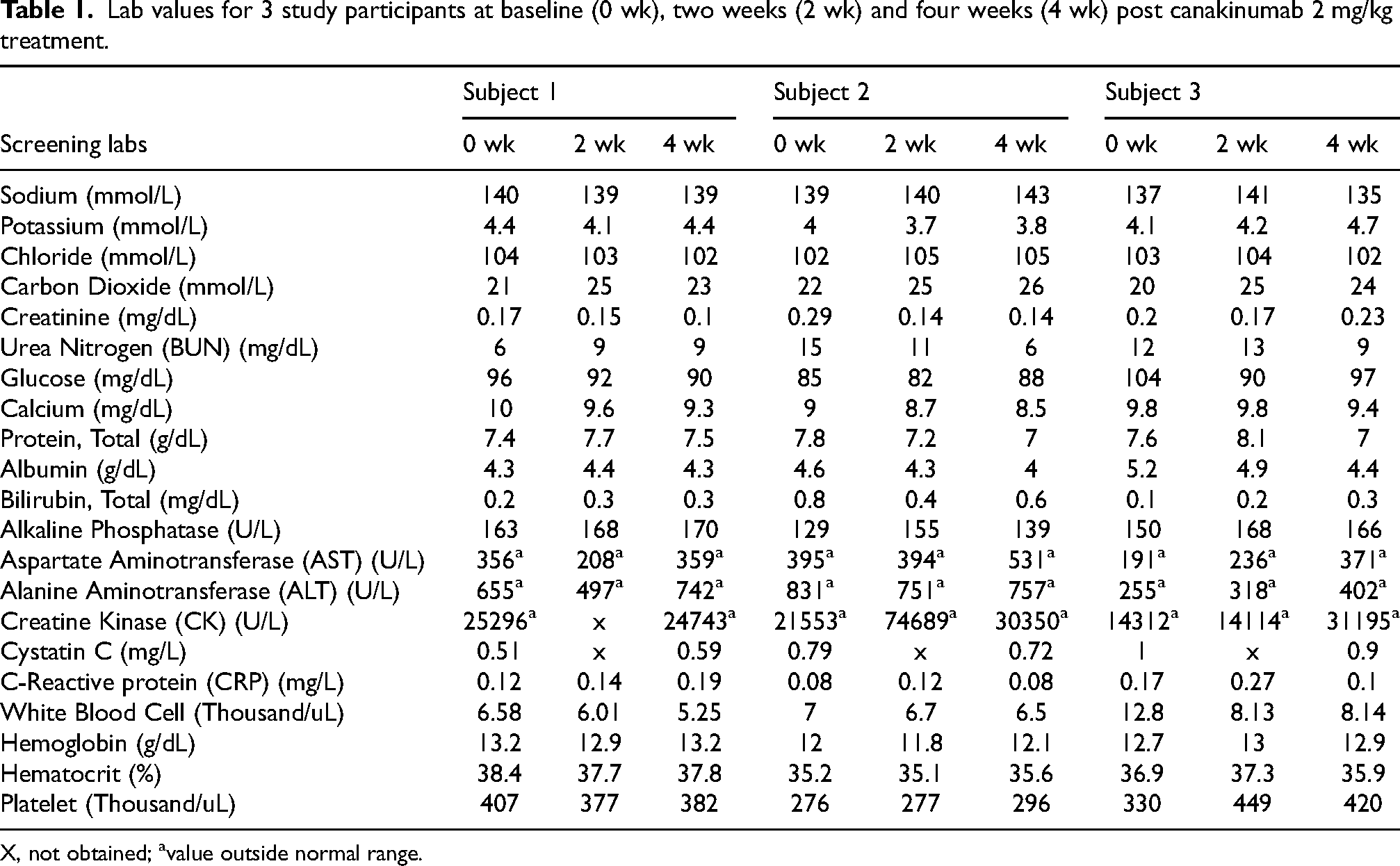
X, not obtained; avalue outside normal range.
Overall, 60 proteins significantly increased and 11 significantly decreased compared to baseline at four weeks following treatment (Table 2 and Figure 1). While a single dose treatment with canakinumab 2 mg/kg did not affect the level of circulating IL-1β after 4 weeks as measured by SomaScan, it did result in a significant decrease (0.8 fold, p = 0.002) of plasma serine protease inhibitor (PCI) and a slight but statistically significant decrease of interleukin-6 receptor subunit alpha (IL-6 sRa), Lymphocyte antigen 86 (LY86) and Immunoglobulin D (IgD) (0.9 fold p < 0.05). Another slight but significant decrease (p < 0.03) was seen for myostatin and glucosylceramidase (GLCM). Increased proteins consisted mostly of muscle specific enzymes such as fructose-bisphosphate aldolase or aldolase A (+1.5-fold, p = 0.001), L-lactate dehydrogenase B (LDH-H1), (+1.3-fold, p = 0.002) and glycerol-3-phosphate phosphatase (PGP), (+1.3-fold, p = 0.002). Another significant increase was seen for the large ribosomal subunit protein (bL12m) (+1.6-fold, p = 0.008), the mitochondria acetyl-CoA acetyltransferase (THIL), (+1.4-fold, p = 0.013) and angiopoietin-related protein 3 (+1.4-fold, p = 0.002).
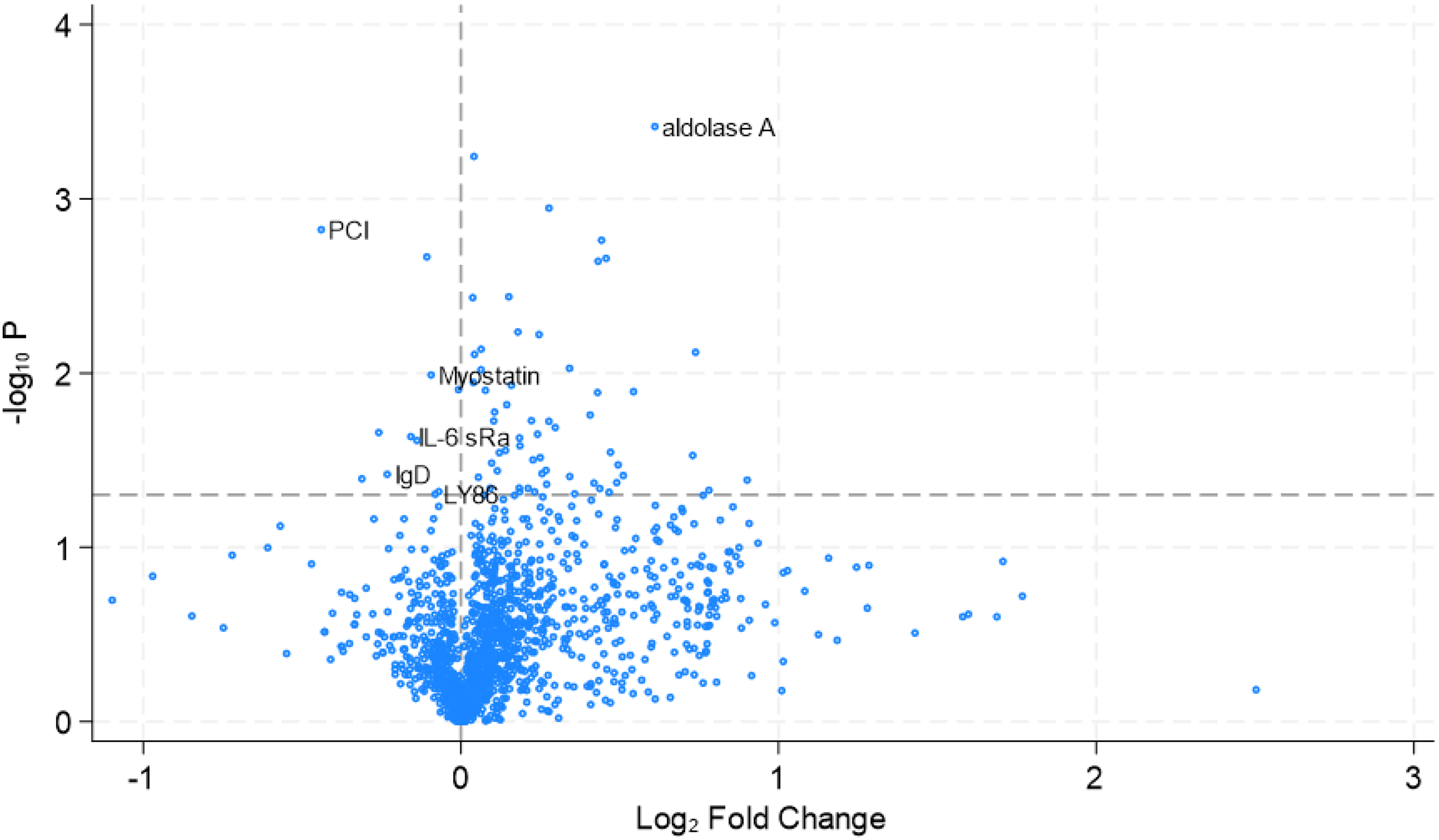
Volcano plot of protein analysis with significantly changed proteins IL-6sRa, IgD, Ly86, PCI, myostatin and aldolase A noted.
Proteins with significantly altered expression levels four weeks after treatment with canakinumab compared to baseline levels.
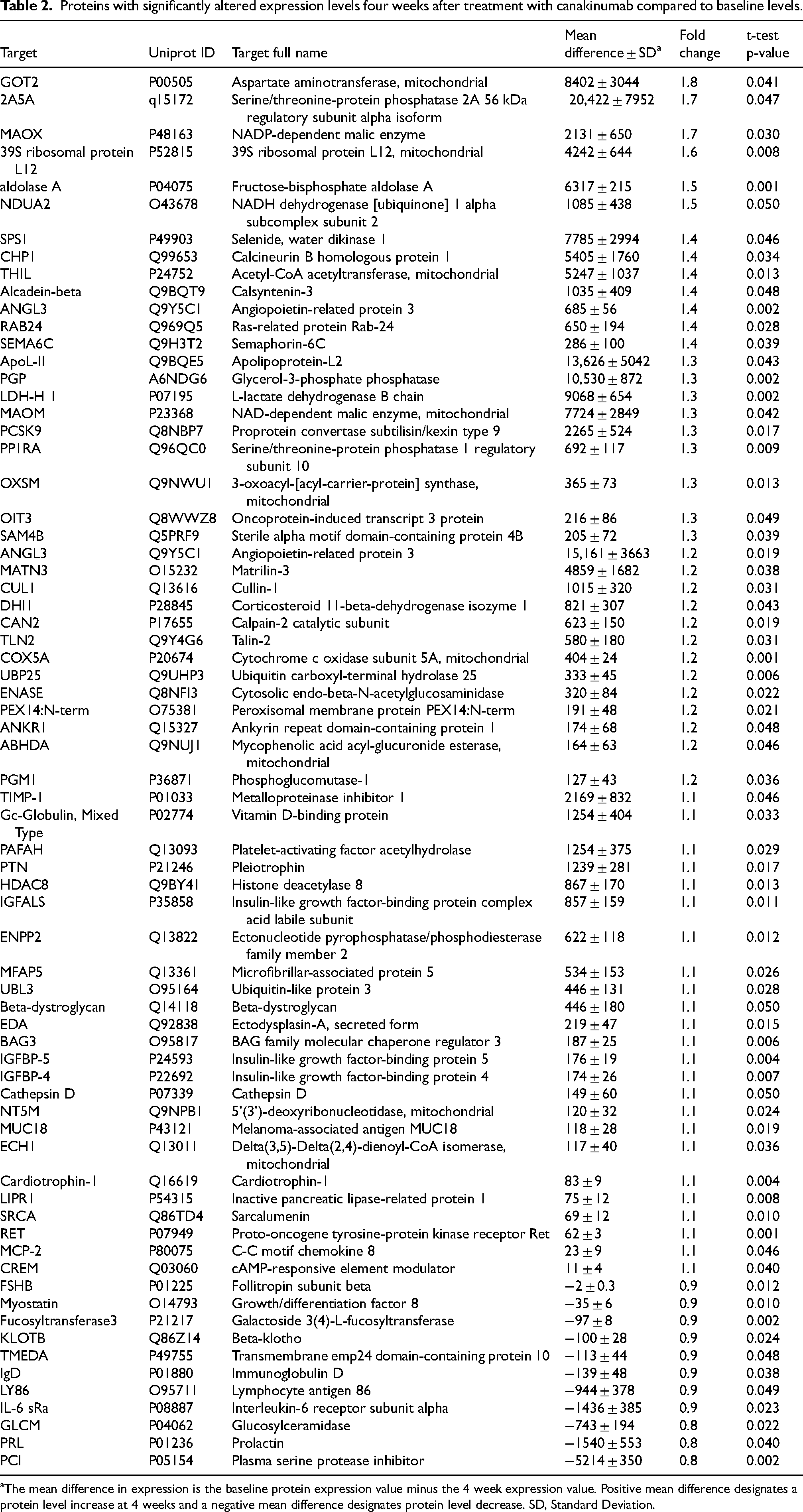
aThe mean difference in expression is the baseline protein expression value minus the 4 week expression value. Positive mean difference designates a protein level increase at 4 weeks and a negative mean difference designates protein level decrease. SD, Standard Deviation.
Discussion
This open label pilot trial of canakinumab in steroid naïve children with DMD showed that a single 2 mg/kg dose was safe and well tolerated after one month. There were no significant changes in safety labs and no reported adverse events. Due to poor enrollment, we were unable to study additional subjects at a dose of 4 mg/kg. Further studies of canakinumab in DMD are needed.
We performed exploratory serum protein biomarker analysis to identify potential acute changes related to canakinumab treatment. Although sample size was very low and the dosing was for a short period of time, potential candidate response biomarkers for canakinumab were identified. Perhaps one of the relevant inflammation biomarkers that acutely decrease by canakinumab treatment is IgD, a known potent inducer of interleukins including IL-1β. 13 Its decrease in canakinumab treated DMD subjects could indicate efficacy but further studies using higher dose and longer time are required to clinically validate this biomarker. Hathout et al. studied protein biomarkers in a younger cohort of DMD patients (aged 4–10 years) including steroid naïve subjects compared to healthy controls. 10 While they showed an increase in interleukin 6 (IL-6) in the steroid naïve DMD cohort compared to healthy controls, our results showed a significant decrease in IL-6 receptor subunit alpha in steroid naïve DMD subjects at 4 weeks after canakinumab treatment. IL-6 was shown to modulate skeletal muscle disease in dystrophin and utrophin deficient mice. 14 Serine protease inhibitor (PCI) is part of a family of proteins that inhibit serine proteases. Also called plasminogen activation inhibitor 1 (PAI1), excessive levels are related to muscle inflammation, increased fibrosis and decreased regeneration. 15 Inhibition of PAI1 was shown to improve muscle regeneration in a diabetic mouse model. 16 LY86, also known as myeloid differentiation 1 (MD-1), is a marker of innate immunity inflammation and toll like receptor activation. 17 Toll-like receptors are known to contribute to DMD related muscle pathology. 18 Myostatin was found to be significantly decreased at 4-weeks post treatment. Benefits of myostatin inhibition were demonstrated in animal models of dystrophin deficiency, but clinical trials have not demonstrated a clear benefit.19,20 These decreased response biomarkers provide potential measures of canakinumab and other anti-inflammatory therapies.
Our results share some common proteins with previous DMD studies. Hathout et al. performed a SomaScan assay analysis of two cohorts of patients with DMD and age matched normal controls. They identified 44 proteins that differed significantly between DMD and controls. 21 Spitali et al. 22 studied 15 DMD subjects with a mean age of 9 years. Comparing their results to the two cohorts used by Hathout et al., 21 they identified 34 proteins that discriminated between DMD and normal controls. 22 Similar to these, our study demonstrated increases in proteins related to muscle disease including aldolase A, lactate dehydrogenase and glycerol-3-phosphate. These are related to baseline muscle pathology but changes due to increased muscle activity related to canakinumab treatment are possible.
Limitations of this study include a small subject number and short period of follow up. Further studies are necessary to validate these proposed biomarkers.
In summary, a single dose of canakinumab at 2 mg/kg is well tolerated in children with DMD after one month. Potential serum response biomarkers of canakinumab included a decrease in IgD, IL 6 receptor subunit, PCI, Ly86 and myostatin. Further long term studies regarding the benefits of canakinumab treatment in addition to corticosteroid treatment in DMD are supported by this pilot trial.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602251319177 - Supplemental material for Pilot study of canakinumab (Ilaris) in steroid naïve children with Duchenne muscular dystrophy demonstrates safety and exploratory changes in potential serum protein response biomarkers
Supplemental material, sj-docx-1-jnd-10.1177_22143602251319177 for Pilot study of canakinumab (Ilaris) in steroid naïve children with Duchenne muscular dystrophy demonstrates safety and exploratory changes in potential serum protein response biomarkers by Christopher F. Spurney, Jessica Chong, Heather Gordish-Dressman and Yetrib Hathout in Journal of Neuromuscular Diseases
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Foundation to Eradiate Duchenne.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
