Abstract
Background:
Fatigue and fatigability are commonly reported in spinal muscular atrophy (SMA). Physical fatigability, proposed to be the most relevant to SMA pathophysiology, encompasses performance-based and perceived physical fatigability (PPF) assessments. While performance-based measures have highlighted physical fatigability as an SMA hallmark, PPF is not well characterized due to the lack of disease- and construct-specific scales.
Objective:
Our aim was to create a patient-reported outcome measure tailored for SMA, named the SMA EFFORT, to improve PPF assessment. Here, we describe the scale development process, assess scale properties, and discuss future research and application.
Methods:
SMA and scale development experts organized a comprehensive physical activity item bank, relevant across the SMA phenotypic spectrum. Activities were systematically categorized by varying intensities and durations. The SMA EFFORT was completed by an international cohort of individuals with all types of SMA. To compare PPF across demographic and clinical variables, SMA EFFORT PPF percent (PPF%) composite scoring was established.
Results:
One hundred eighteen participants completed the SMA EFFORT. Total PPF% scores were broadly distributed within functional groups, with differences between non-sitters (35.1 ± 21.0) and sitters (24.9 ± 15.1) (p = 0.006), and those with (34.4 ± 18.1) and without respiratory support (26.4 ± 17.8) (p = 0.02). Participants treated with disease modifying therapy (DMT) showed similar scores to those without treatment (p = 0.70). Further, no differences in scores were observed in participants with scoliosis surgery and those without (p = 0.71). Subscale analyses revealed differences in mean PPF% subscale scores by functional group.
Conclusions:
The novel SMA EFFORT standardizes PPF ratings by anchoring activity to intensity and duration. Item and scale data insights will inform the next iteration, which will undergo additional investigation. The SMA EFFORT aims to improve upon current measures to better assess treatment impact on physical well-being across the SMA spectrum.
Keywords
Introduction
Spinal muscular atrophy (SMA) is a recessively inherited neuromuscular disorder, resulting from a deletion and/or mutation in the survival motor neuron (SMNI) gene located on chromosome 5q13.1–3 Selective degeneration of anterior horn cells in the spinal cord and motor nuclei in the brainstem leads to manifestation of symptoms including muscle weakness, motor, and bulbar function impairments, as well as frequent patient-reported fatigue.4–6 The paralogue SMN2 gene produces variable amounts of functional SMN protein resulting in a wide phenotypic spectrum. 7 SMA type is based on age of symptom onset and highest motor milestone achieved, while current functional status is indicated by non-sitter, sitter, or walker classification.5,8 While performance fatigability is consistently observed in SMA, its connection to patient-reported fatigue remains unclear.9,10 The lack of established association has resulted in their proposed consideration as distinct concepts.9–11 Further, this suggested independence between fatigue and performance fatigability is believed to arise from differences in the physiological underpinnings that each measurement type is proposed to evaluate.9,11
Despite evidence of several mechanisms contributing to physical activity limitations in SMA, the relative impact of these variables on physical fatigability, whether assessed through patient-reported or performance-based measures, is not known.12–16 Importantly, the availability of three FDA-approved disease modifying therapies (DMT) has fundamentally altered the natural history in individuals with SMA.17–20 Clinically meaningful improvements in motor function, including on performance-based measures such as the Six-Minute Walk Test (6MWT), have been shown with DMT; however, physical fatigability (patient-reported and performance-based) persists.14,21–23 Anecdotally, some patients treated with SMN upregulating therapies have reported changes in their ability to execute repetitive physical tasks, but the SMA community has been unable to capture these changes with existing scales. Notably, a global rating scale has recently been used to evaluate the impact of adjuvant therapies targeting performance fatigability, but linking these changes to specific activities remains to be elucidated. 15 Indeed, addressing physical fatigability remains a top priority for the SMA community.10,24
Characterizing perceived physical fatigability (PPF) is essential to understanding this intractable, disease-related symptom. Motor function scales and patient-reported outcome measures (PROMs) are an important part of evaluating patients with SMA; though, an appropriate selection process is key to account for the diverse clinical phenotypes. 25 Even with the comprehensive exploration of patient reported fatigue across numerous dimensions in SMA, PPF has not been adequately studied. 26 Importantly, the use of non-specific fatigue PROMs has perpetuated inconsistencies in assessment methodology and an incomplete understanding of the underpinnings of PPF in SMA. 26 Therefore, the development of a disease-specific perceived physical fatigability PROM that considers activity intensity and duration is the focus of this work.10,26 The anchoring of activity to intensity and duration may align closer to performance-based fatigability assessments in SMA. Further, it will provide a more sensitive assessment of perceptions of physical fatigability in individuals with SMA.26,27
To address this knowledge gap, a multidisciplinary team with SMA and scale development expertise was assembled to construct a disease-specific PROM (SMA EFFORT) aimed to improve the evaluation of PPF. Our objective was to develop a scale applicable across the diverse phenotypic spectrum to help improve our understanding of physical well-being in people with SMA. In addition to outlining the iterative scale development process, we present initial results from a heterogenous sample of individuals with SMA who completed the SMA EFFORT and discuss future directions for scale validation.
Materials and methods
A comprehensive, relevant, and disease-specific item bank was compiled with patient-reported physical activity challenges as a core focus. In this incremental scale design, additional data and input from multiple sources were incorporated. Sources included a scoping review of the scientific literature and input from a clinical and scientific multidisciplinary expert panel. The panel consisted of fourteen clinicians and researchers, including research physical therapists (RRT, JM, CHK, SDY, GC, RML, AR), a neurologist (DCD), a clinical exercise physiologist (DU), an epidemiologist (NWG), an epidemiology doctoral student (EG), and biostatistical experts (MW, TC, MPM). Further, this expert panel comprised distinguished researchers who have developed and validated performance-based and person-reported outcome measures for various populations. Multiple round table discussions took place with varying group compositions.
Definition and elaboration of the construct of interest
A prevalent inconsistency in fatigue-related nomenclature has been identified as a major challenge to a unified understanding of physical fatigability in SMA. 26 As such, the initial phase of scale development involved operationalizing the construct to be assessed in SMA. The starting point for conceptualizing the physical fatigability dimension involved establishing consistent nomenclature. This included instituting a clear definition of fatigability, incorporating both performance-based and perceived physical fatigability.
We adopted the fatigue and fatigability taxonomy proposed by Kluger et al. and Enoka et al.16,28,29 Performance fatigability was defined as the decline in one or more aspects of performance (i.e., decreased motor output) during a continuous activity or prolonged task. 28 Perceived physical fatigability (PPF) referred to a comprehensive, whole-body self-assessment of one's susceptibility to fatigue with physical activity standardized to fixed intensity and duration.27,30
Review of current measurement methods
A scoping review aimed to 1) identify available evidence on SMA fatigue research, 2) clarify key concepts and definitions, 3) examine patient-reported fatigue research, and 4) analyze knowledge gaps. 26 Briefly, we learned that many PROMs used in SMA research were repurposed from other populations, with physical fatigability often considered most relevant due to the disease's pathophysiology.26,31 Despite some validation for SMA use, these scales lacked sensitivity to change and association with performance fatigability measures.9,26 Furthermore, these scales often conflate fatigue and fatigability as a single construct, which complicates efforts to link these experiences to the proposed pathophysiology. 32
The Pittsburgh Fatigability Scale (PFS) framework was used as a model for the development of a standardized tool to assess PPF in SMA. 26 It utilizes activity intensity and duration anchors to contextualize queries related to physical fatigability with various activities. The PFS has shown concurrent and convergent validity with performance-based fatigability measures, fitness, mobility, and physical function in older, non-SMA adults. 27 None of the fatigue-related PROMs currently used in SMA anchor activities to intensity or duration of a task. As a result, they may not fully capture the concept of PPF as intended. 26
Selecting and formulating items
An inclusive item bank comprising 108 items was established, encompassing 33 distinct activities with diverse intensity and duration anchors. Like the PFS, our scale was developed with the intention of using factor analysis to reduce the number of items. 27 The selection of activities was informed by the clinical expertise of the panel and patient-reported physical activities known to commonly induce physical fatigability. 16 Activities were categorized into four domains based on theorized metabolic equivalents (METs), including: 1) self-care, 2) sedentary, 3) lifestyle/light-intensity, and 4) moderate/high-intensity. Variable intensities and durations were applied to differentiate similar activities into multiple items, and to minimize floor and ceiling effects. Our deliberate approach aimed to include a broad spectrum of activities, capturing the latent experience of PPF across the SMA phenotypic spectrum. Participants were instructed to rate applicable activities only, acknowledging that not all included activities may be relevant for every individual.
Activities were initially categorized by MET level, aligning with the PFS approach. However, as there are limited energy expenditure data for individuals with neuromuscular conditions, including SMA, the expert panel deemed this method unsuitable. Instead, activities were reclassified to align closer to an International Classification of Functioning, Disability, and Health (ICF) framework spanning multiple domains of function, including: 1) exercise/recreation, 2) mobility, 3) activities of daily living (ADLs), and 4) postural control. Two items related to reading and watching TV were excluded from all analyses, as the panel deemed them not reflective of physical fatigability in SMA; this resulted in 31 distinct activities (106 total items where most activities were presented with different durations).
Study design and participants
This was a cross-sectional study of adolescents and adults with SMA. As the primary objective was to capture a wide range of responses, we did not set a specific target sample size. All individuals who met eligibility criteria were invited to participate. Participants had to be at least 12 years old and have a genetically confirmed diagnosis of 5q13-SMA. The inclusion of participants at least 12 years of age was due to our interest in understanding the experience of PPF first-hand from people living with SMA. The available literature suggests that adolescents begin to show an increased ability to engage in abstract reasoning, problem solving, and decision-making using deductive logic. 33 Additional inclusion criteria included self-reported capacity to understand English and answer questions based solely on their experience. Assistance of a caregiver to complete the questionnaire due to inability to independently navigate the electronic survey was allowed.
Potential participants were presented with the study opportunity by their treating clinician during standard of care follow-up visits or were e-mailed as part of their ongoing participation in an SMA patient registry (Cure SMA, International SMA Consortium (iSMAC) (SMA REACH UK, PNCRN USA and Italian Telethon)). 34 The SMA EFFORT questionnaire was hosted on REDCap. Individuals who chose to participate in the study remained anonymous to the research team and to other participants in the study. Of the 31 activities, 30 activities were presented as separate questions at different durations. Participants rated PPF for all items from 0 = No Fatigability to 5 = Extreme Fatigability, for activities and durations performed in the past month, or “imagined fatigability” for activities performed in the past but now lost over time (Supplemental Figure 1).
Participants’ current level of function was queried based on the following functional classifications: sitting supported; sitting unsupported; independent standing; walking with support (e.g., braces, cane, crutches, walker, etc.); walking unsupported (e.g., no assistance from a brace, device or person in the community). To align with standard nomenclature, they were then categorized as non-sitters (needing sitting support), sitters, or walkers. Only individuals who walked unsupported were considered walkers. Information regarding current DMT status and the use of respiratory support (non-invasive ventilation and tracheostomy) was gathered through yes/no responses. The highest level of education attained was also collected. Participants rated their “usual energy level” in the past month on a scale of 0 to 10, where 0 indicated no energy at all, and 10 represented highest energy level. Lastly, participants provided open-ended feedback on the scale. Data collection spanned from June 2022 to March 2023.
Statistical methods
Descriptive statistics were used to summarize demographic and clinical characteristics of the participants overall and by functional status subgroups (e.g., non-sitters, sitters, and walkers). Counts and percentages of individuals performing each of the 31 different activities within the past month were summarized along with an aggregated total count of activities; associations between activity endorsement and current level of function were also tested.
SMA EFFORT PPF percent (PPF%) total scores were calculated as the sum of activity item ratings divided by the number of items rated multiplied by five (highest rating on Likert scale), with higher scores indicating greater perceived physical fatigability. To mitigate potential recall bias, responses pertaining to activities not currently performed (in the past month) were excluded from the analyses. In addition to the PPF% total score (across all activities performed), PPF% subscale scores corresponding to the specific activities in each domain of function were similarly derived.
To provide preliminary assessment of the convergent validity of the SMA EFFORT, PPF% total score and subscale scores and their associations with current function were evaluated. Similarly, associations of the PPF% total score with age, usual energy, use of respiratory support and DMT, and history of scoliosis surgery were examined. Tests of association across levels of current function were done using Chi-square tests for categorical variables (or Fisher's exact test for cell sizes < 5), and t-tests for continuous and count variables; Pearson correlations were used to assess associations between continuous variables.
A factor analysis was used to explore alternative scoring methods. A second-order confirmatory factor analysis model was fit to averages of PPF scores for each activity (averaged across the various responses corresponding to different durations of doing the activity); Level 1 consisted of the four subscales of function (exercise/recreation, mobility, postural control, and ADLs), while Level 2 was the overall perceived physical fatigability that underlies all four subscales (Supplemental Figure 2). Model fit was assessed using the Comparative Fit Index (CFI, > 0.9 indicating acceptable fit) and Root Mean Square Error of Approximation (RMSEA, < 0.08 indicating acceptable fit), two standard goodness of fit measures. 35 Alternative models were also explored, including a bi-factor model and separate one-factor models fit to each subscale.
Statistical significance was determined at α < 0.05 throughout without any correction for multiple comparisons due to the preliminary nature of the study. Analyses were done using SAS 9.4 (SAS Institute Inc., Cary, NC) and R version 4. 36 Factor analyses were performed using Mplus 8.6. 37
Standard protocol approvals and patient consents
All participants or guardians of participants completed informed consent and assent forms, where applicable. The study protocol was approved by the IRB at Columbia University Irving Medical Center (New York, NY); reference code: AAAU0036.
Results
The clinical and demographic characteristics of the 118 study participants are summarized in Table 1. Respondents had a mean age of 41.3 years (range 14–78), with the majority (95%) being ≥18 years. Participants were mostly female (51%), and were classified as non-sitters (40%), sitters (45%), or walkers (15%). Of all participants included, 61% reported being on DMT; data on type of therapy or length of treatment time were not collected. Fifty-seven percent of non-sitters and 19% of sitters reported the use of respiratory support. Scoliosis surgery was reported by 48% of the participants.
Participant clinical and demographic characteristics by current functional status.
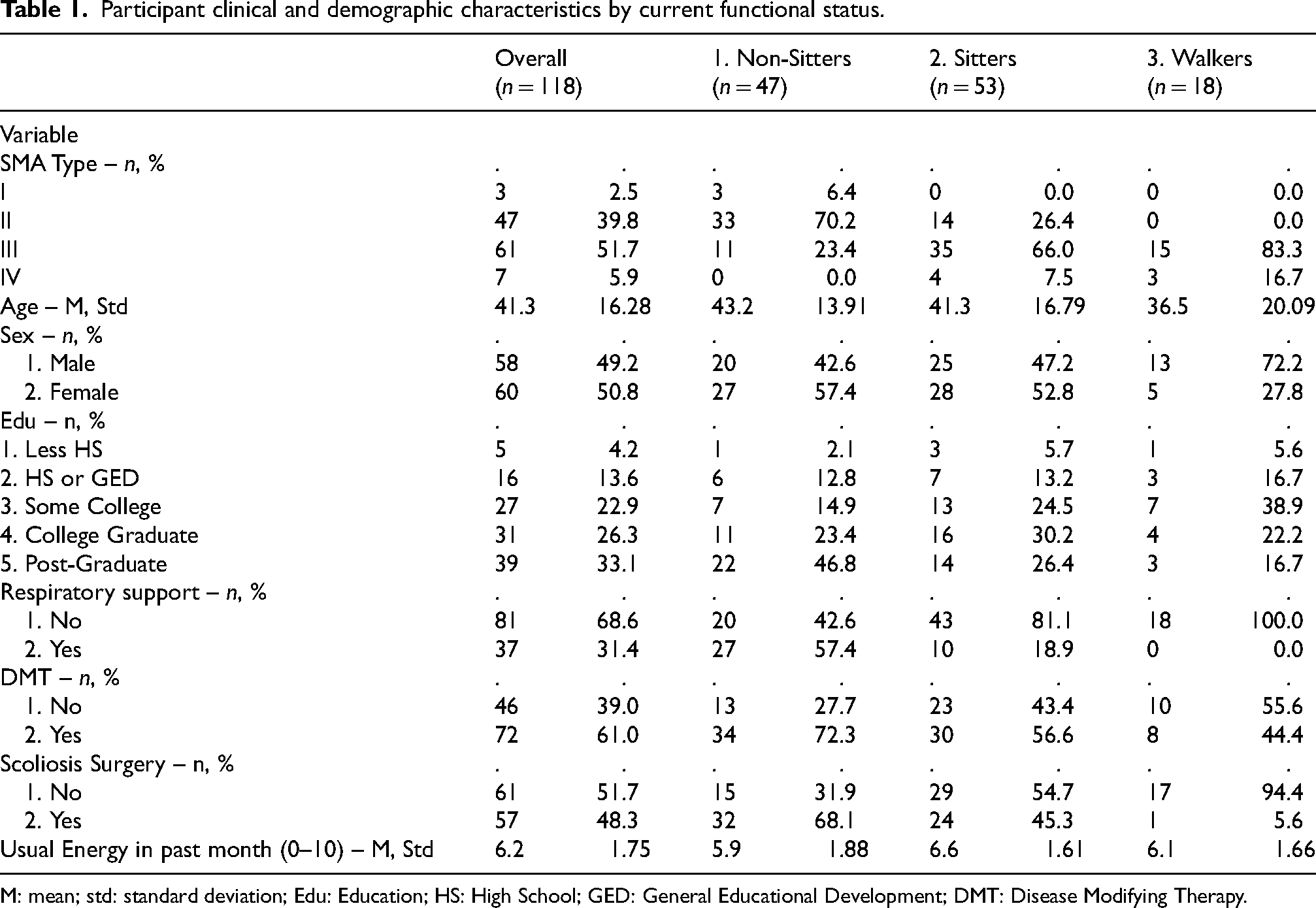
M: mean; std: standard deviation; Edu: Education; HS: High School; GED: General Educational Development; DMT: Disease Modifying Therapy.
The number of activities performed in the past month was different across all functional groups (p < 0.001). In general, the mean number of activities (±standard deviation) reported was greater for walkers (23 ± 4.0) followed by sitters (17 ± 3.8) and non-sitters (13 ± 2.8) (Table 2). Overall, functional status subgroups tended not to differ in terms of activity participation for activities that were easier.
Activity prevalence in the last month, overall and by current functional status.
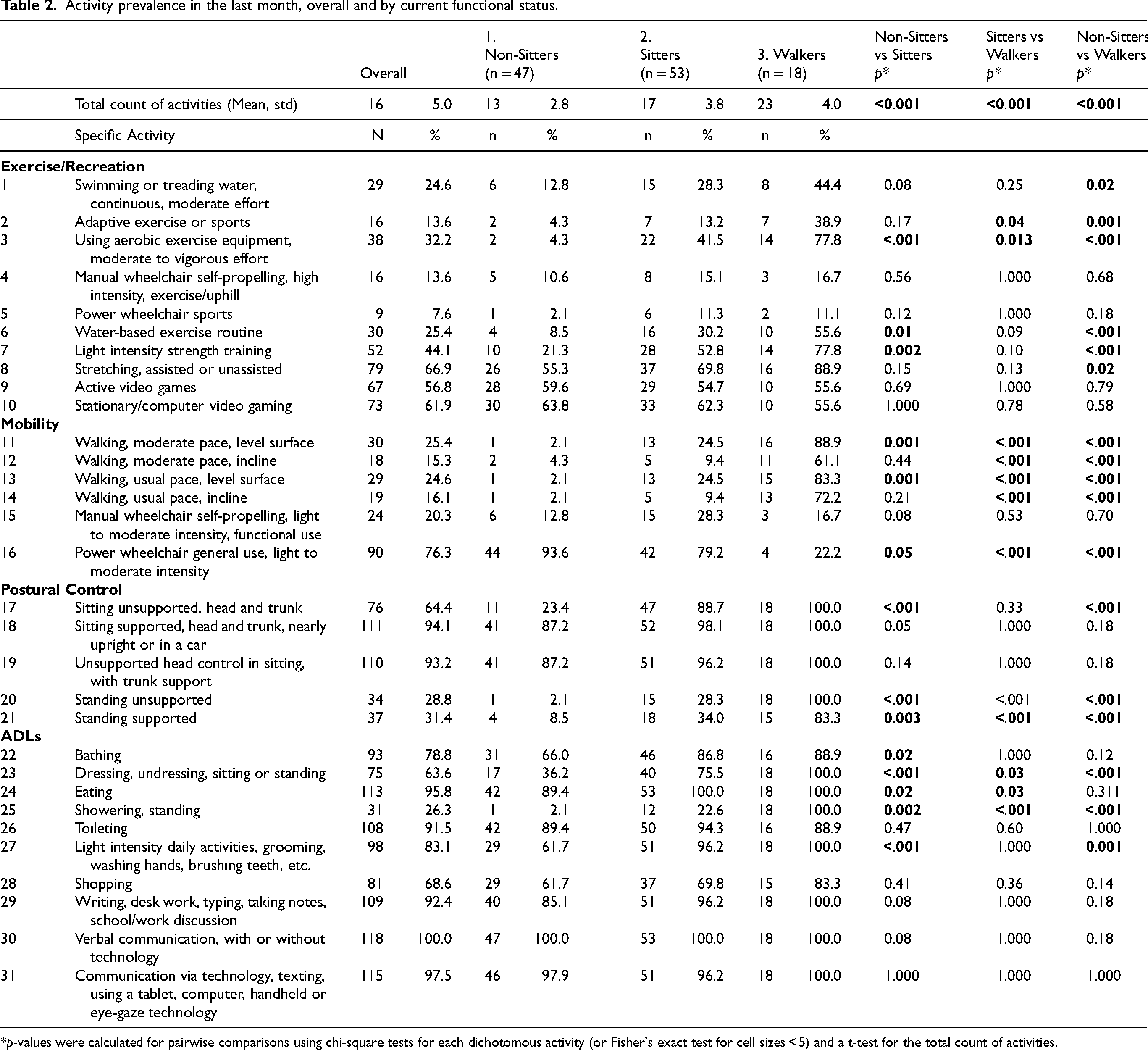
*p-values were calculated for pairwise comparisons using chi-square tests for each dichotomous activity (or Fisher's exact test for cell sizes < 5) and a t-test for the total count of activities.
The mean PPF% total score differed between Non-Sitters (35.1 ± 21.0) and Sitters (24.9 ± 15.1) (p = 0.006) (Figure 1), while no statistically significant differences were observed in mean PPF% total score between Sitters and Walkers and Non-Sitters and Walkers (p > 0.05). Mean PPF% subscale scores differed by functional group in three of the four activity categories (Supplemental Figure 3, Table 3). The mean ADLs subscale score differed between Non-Sitters (34.6 ± 21.2) and Sitters (21.2 ± 17.1) (p < 0.001), and between Non-Sitters and Walkers (14.5 ± 14.4) (p < 0.001). The mean postural control subscale score differed between Non-Sitters (31.5 ± 22.8) and Sitters (16.6 ± 16.7) (p < 0.001), and between Non-Sitters and Walkers (18.9 ± 16.1) (p = 0.04). The mean mobility subscale score differed between Non-Sitters (33.1 ± 26.5) and Walkers (48.1 ± 22.8) (p = 0.04), and between Sitters (27.7 ± 25.6) and Walkers (p = 0.004). The exercise/recreation subscale did not differentiate between groups. Moderate to strong positive correlations were found among the PPF% total and subscale scores. Associations among the subscale scores were positive and ranged from r = 0.48 between mobility and postural control, and r = 0.83 between ADLs and postural control. The ADLs subscale had the highest correlation with the PPF% total score (r = 0.95) (Supplemental Figure 4).
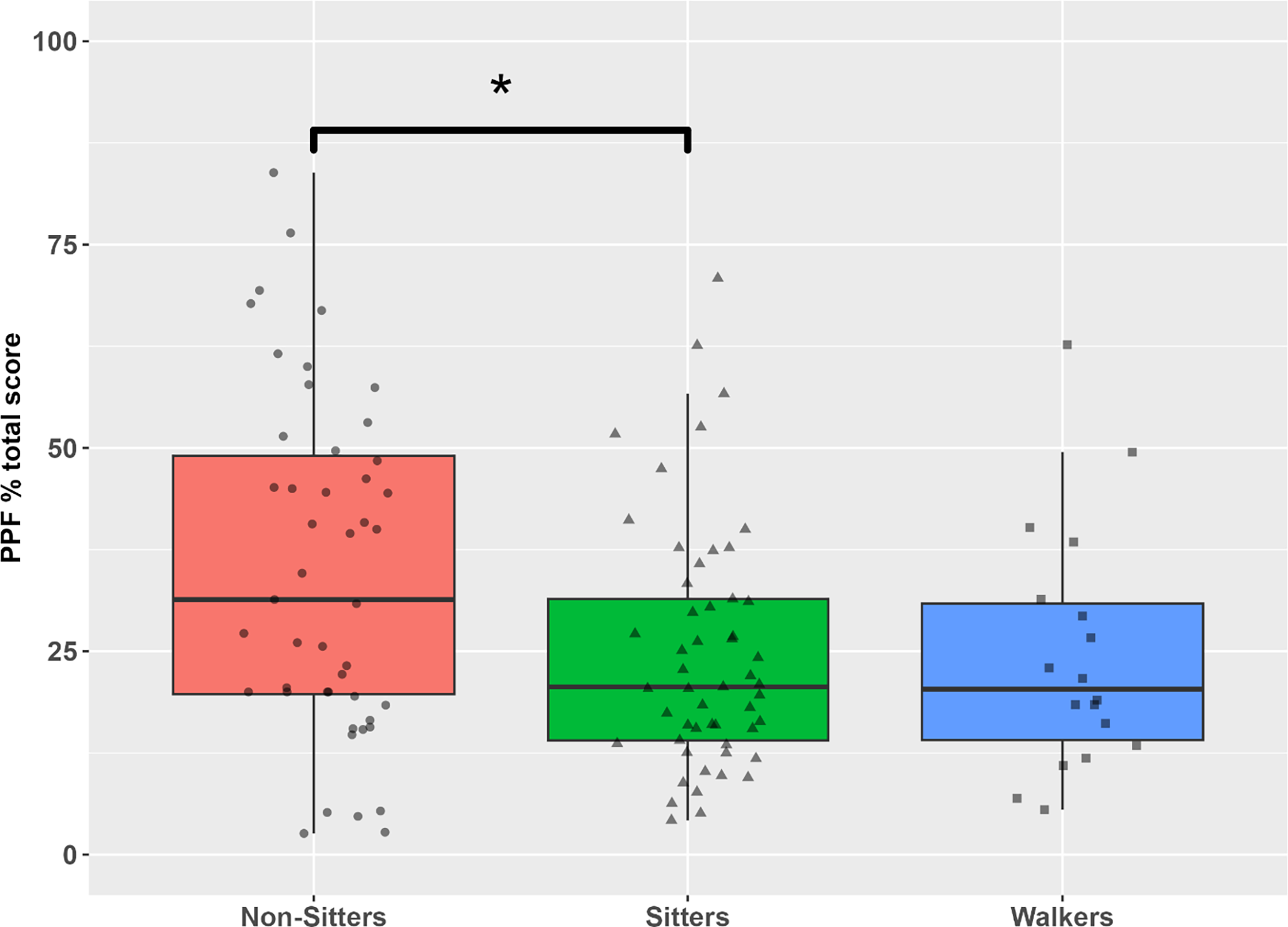
Distributions of PFF% total scores by functional group. The boxes represent the interquartile range (25th and 75th percentiles) with the horizontal line indicating the median. The means (SD) are: Non-Sitters, 35.1 (21.0); Sitters, 24.9 (15.1); Walkers, 24.6 (15.2); p = 0.006 between Non-Sitters and Sitters, p = 0.06 between Non-Sitters and Walkers.
Average PPF% total and subscale scores across the overall cohort and by current functional status.
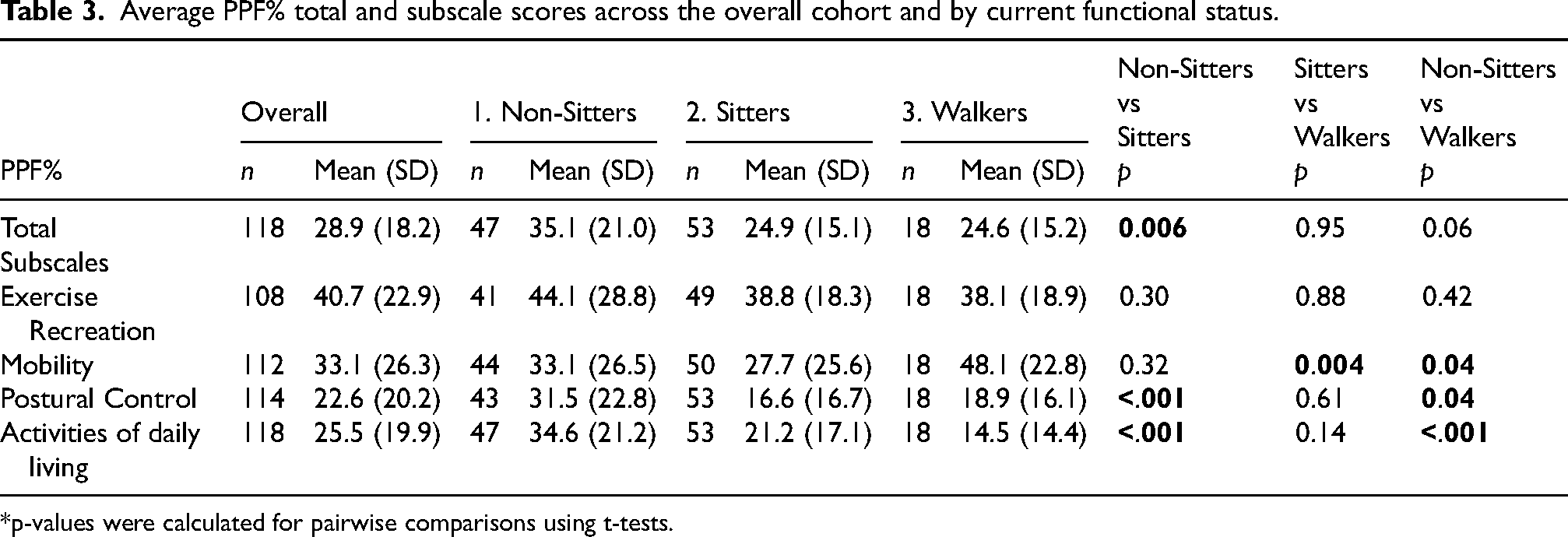
*p-values were calculated for pairwise comparisons using t-tests.
The PPF% total score was found to be weakly associated with older age (r = 0.19, p = 0.04) and negatively associated with energy level (r = −0.45, p < 0.001) (Figure 2(a) and (b), respectively). A difference in mean PPF% total score was observed between individuals with respiratory support (34.4 ± 18.1) and those without such use (26.4 ± 17.8) (p = 0.02) (Figure 3). No differences were detected in mean PPF% total score between individuals receiving versus not receiving DMT (p = 0.70) nor between those with and without scoliosis surgery (p = 0.71) (Figure 3). In the whole cohort, there was a weak negative association between PPF% total score and the total number of activities performed in the last month (r = −0.20, p = 0.03).
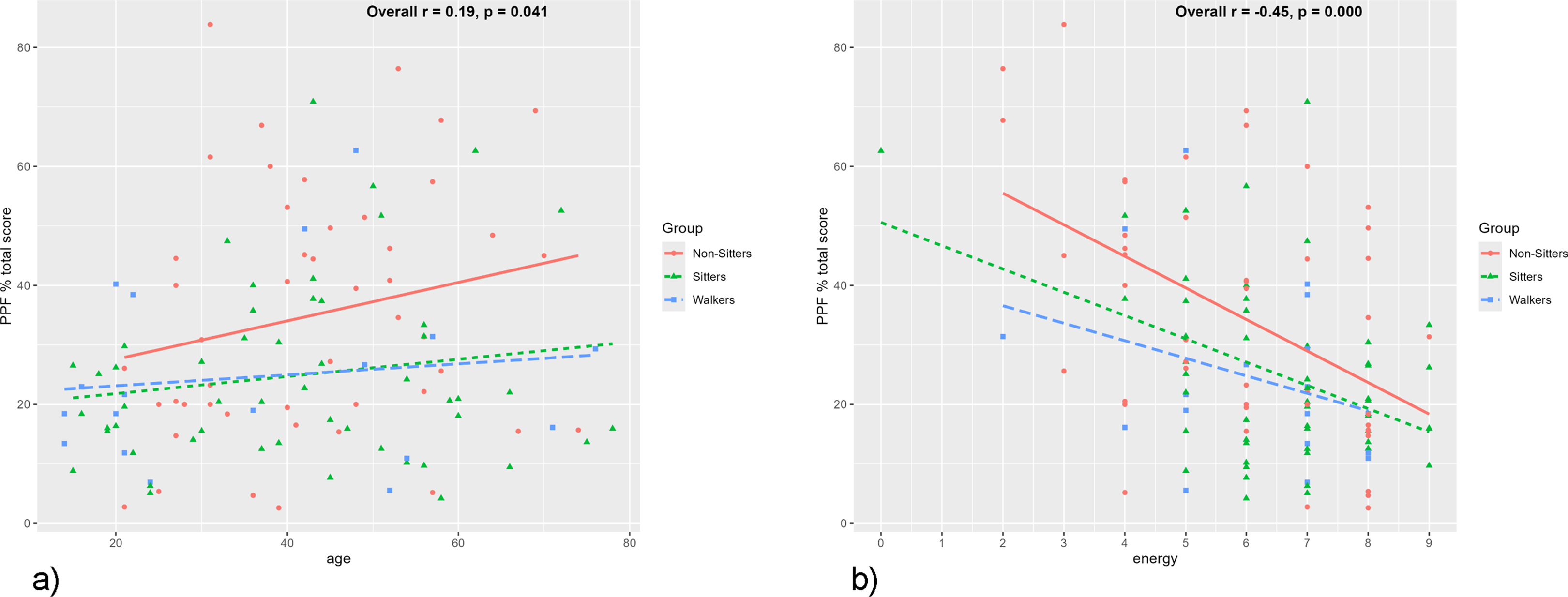
(a) Correlations between PPF% total score and age by functional status. PPF% total score demonstrated a weak association with age across the sample, r = 0.19, p = 0.04. Correlations within each functional group: Non-Sitters r = 0.21, p = 0.15; Sitters r = 0.16, p = 0.25; Walkers r = 0.12, p = 0.63. (b) Correlations between PPF% total score and energy by functional status. PPF% total score demonstrated a negative association with the single item Energy (“Usual energy level in the past 30 days”). Correlations within each current functional status group were negative: Non-Sitters r = −0.48, p < 0.001; Sitters r = −0.42, p = 0.002; Walkers r = −0.32, p = 0.19.
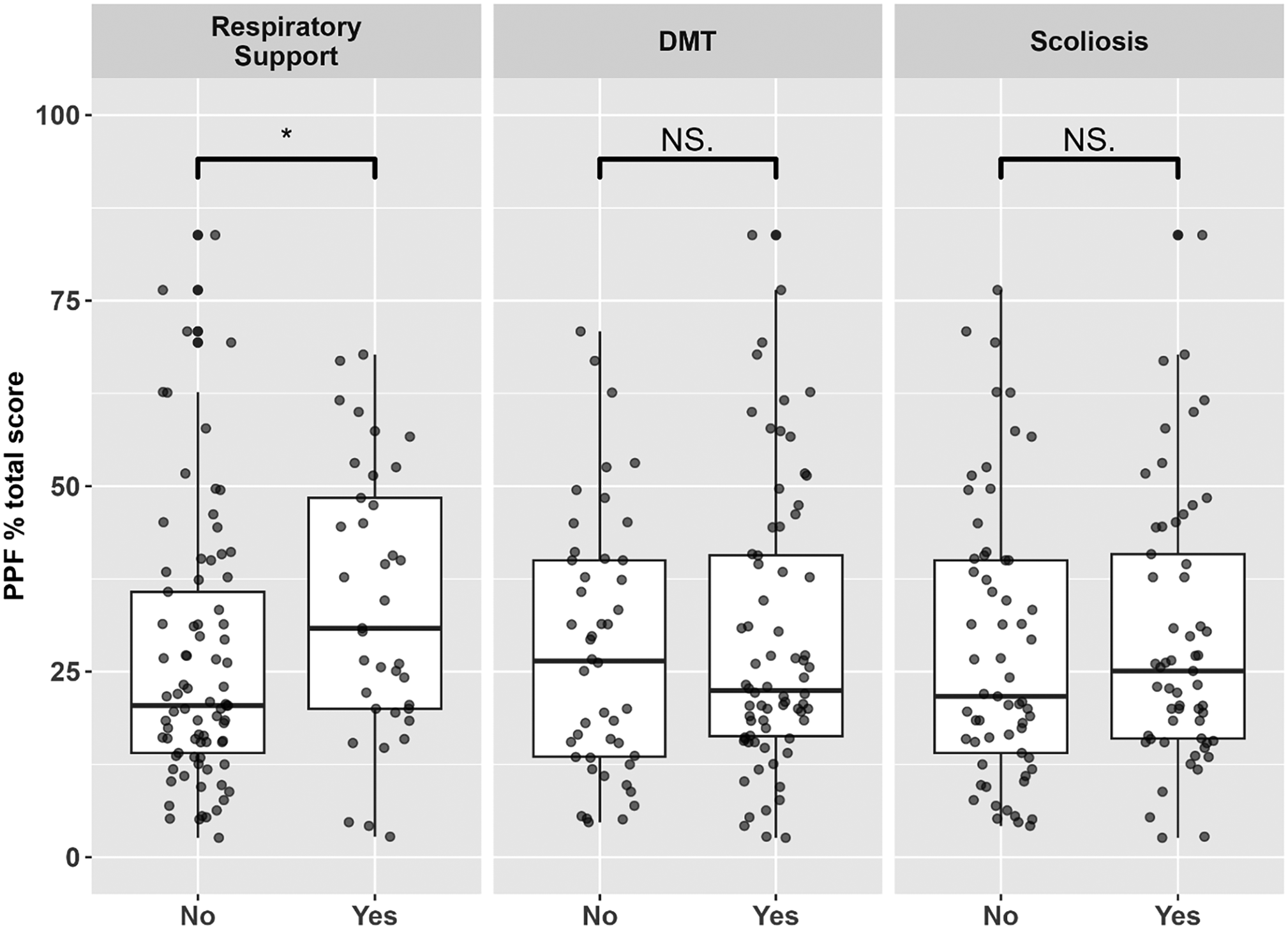
Associations between PPF% total scores and clinical variables. Mean PPF% total score was associated with respiratory support (p = 0.02), but not with disease-modifying treatment (DMT) (p = 0.70) or scoliosis surgery (p = 0.71). Information on DMT type was unavailable, as was length of time of treatment and time of treatment initiation.
Factor analysis modeling showed high correlations between PPF% total score and total latent factor score (r = 0.96), and range (r = 0.65–0.98) for the subscales (Supplemental Figures 5 and 6). Fit statistics and factor loadings are found in Supplemental Tables 1 and 2. None of the factor analysis models showed good fit to the items, perhaps due to the large proportion of missing data for activities that participants had not performed in the past month.
Discussion
A significant barrier to assessing PPF in SMA has been in identifying PROMs that are practical and meaningful for this specific patient population. The SMA EFFORT introduces a promising and feasible method for quantifying perceived physical fatigability in SMA. Its straightforward scoring method enabled the characterization of PPF across a diverse cohort of individuals with all types of SMA, focusing solely on relevant tasks anchored to intensity and duration.
Fatigue and physical fatigability have been reported in a substantial proportion of individuals with all types of SMA.6,10,24,26 While muscle strength and motor function have been shown to be associated with physical fatigability, they are not a surrogate for physical fatigability in SMA.11,14 When assessed in a manner appropriate for the individual's current capacities, physical fatigability has been demonstrated across the SMA phenotypic spectrum.38–40 Still, despite the lack of association observed between existing global fatigue PROMs and performance fatigability, disease-specific PROMs, targeting PPF, may offer additional insights into treatment-related effects because of their ability to reflect how the disease impacts one's well-being and current physical function. 25
This study described the framework for the development of a novel, disease-specific PROM to assess PPF in SMA, emphasizing its applicability across the rapidly evolving phenotypic spectrum. Strengths of this study include our broad recruitment strategy which allowed us to capture a representative sample of SMA. Additionally, we employed an iterative process to systematically develop a scale that can capture PPF in individuals of differing abilities. As reported in the original PFS development manuscript, we believe that PPF can be better understood from a pathophysiological perspective only when intensity and duration of fatigue-inducing physical activity is considered. 27 Here we demonstrate encouraging evidence for an SMA-specific PROM that employs this approach.
While factor analysis is commonly used in social science research, it did not yield satisfactory results in terms of item fit for the SMA EFFORT. In this study, factor analysis accommodated missing data using maximum likelihood estimation based on participant functional status and responses to other items. However, other factors beyond functional status likely contribute to PPF. Moreover, this method extrapolates PPF ratings on activities that the participant does not perform. Until personal factors contributing to PPF are better understood, factor analysis may not be applicable.
Challenges to characterizing PPF
Some studies indicate a correlation between self-reported physical fatigability and disease severity.41,42 Contrasting findings suggest that milder phenotypes are more likely to report physical fatigability, 43 while others propose that impact is similar across phenotypes. 9 Regarding the notion that milder phenotypes report greater levels of physical fatigability, this is often attributed to the fact that most PROMs predominately reflect the experience of stronger individuals. 26 The findings of this study demonstrate that while PPF collectively differs based on functional status, there is a wide range of PPF within groups. Nevertheless, acknowledging the significance of self-reflection with activities relevant to each person is essential for gaining a deeper understanding of the nuances of this disease characteristic.
In this study we collected information about education and generalized energy. Although these personal factors can complicate global fatigue assessment when using non-disease-specific, multidimensional PROMs, 44 here we emphasized the evaluation of perceived physical fatigability. We aimed to direct participants to concentrate exclusively on their PPF by incorporating relevant physical activities with intensity and duration anchors. The participant-reported level of usual energy showed a negative association with the PPF% total score, highlighting the influence of overall well-being on the experience of PPF. However, characterizing PPF based merely on energy levels remains insufficient.
Limitations
Though participation in this study was completely anonymous, a biased sample may have resulted from self-selection. Indeed, most of the participants included in this study were adults over the age of 18. Future scale validation efforts will aim to recruit larger and more diverse cohorts, including more individuals between the ages of 12–18 years. The SMA EFFORT framework was derived from the PFS, originally tailored for an older, non-clinical population. 27 In the PFS validation study, participants were presumed capable of engaging in each included activity; hence, asking participants to imagine their fatigability based on past experiences was logical. However, due to SMA's progressive nature, asking individuals to assess activities pertaining to an ability that may have been lost posed numerous challenges. For example, a person with SMA type III who had lost the ability to walk might rate their “imagined fatigability” based on past walking experiences. Yet, without precise information on when this ability was lost, evaluating past experiences without current capacity likely measured a different construct than intended. The uncertainty surrounding the potential recovery of lost functional abilities in SMA further complicates matters. While including assessment of activities not currently performed was strategic in the PFS to minimize selective bias, 27 this approach proved challenging in the context of SMA, unlike its negligible impact on the PFS scale validity.
In contrast, while we did observe differences in PPF in participants based on reported use of respiratory support, similar limitations resulted from the anonymous nature of this study. Detailed information regarding the type and use of respiratory support was not collected. Interestingly, among the 43% (n = 20) of current non-sitters who reported no use of respiratory support, the average age was 47 years. Of these, 90% (n = 18) had at some point been able to at least sit independently, and 80% (n = 16) reported using DMT. Although we cannot definitively determine why these individuals were not currently using respiratory support, possible reasons may include non-adherence, underdiagnosis of respiratory insufficiency, or other contributing factors.
Further limitations arose from the deliberate anonymous nature of this study, a measure taken to encourage increased participation and data collection from more individuals. Owing to this commitment to anonymity, we encountered constraints in concurrently characterizing other vital aspects, such as outcomes on motor function scales and performance-based fatigability measures. Though results revealed no disparity in PPF% total score between individuals with and without DMT, the data were cross-sectional and lacked comprehensive details on the type and history of treatment, including time of treatment initiation. Furthermore, a causal connection between DMT and PPF% total score is not possible in the absence of longitudinal data and careful consideration of confounding factors. To better understand PPF in SMA, it is essential to examine the role of SMN upregulation therapies and their impact on the proposed central and peripheral factors underlying fatigability in SMA. This also encompasses collecting information regarding whether individuals are undergoing treatment with investigational therapies. Although no differences were observed between participants with and without DMT use, the relevance of treatment information cannot be underestimated. Given the diverse international composition of this cohort, it is conceivable that such variations existed, reflecting the differing standards of care in different countries.
Future directions
This iterative scale development process will continue with formal cognitive interviews with individuals living with SMA, an essential step in ensuring the alignment of language with intended meanings. Beyond that, this process serves as a valuable tool to identify redundancy and assess the face validity of the scale. Our objective is to refine the SMA EFFORT by optimizing the 31 items while ensuring acceptability and comprehensiveness.
To further evaluate its psychometric properties, the SMA EFFORT will undergo reliability assessment using test-retest measurements. In the original PFS, the anchoring of activity to intensity and duration proved to be reliable across a four-week period (ICC = 0.86). 27 Additionally, convergent validity will be evaluated against SMA performance-based fatigability measures as was done with the PFS, which demonstrated the ability to discriminate older adults with high versus low performance fatigability. Still, it is important to note that in SMA, performance-based fatigability assessments differ based on functional ability and are therefore difficult to compare between individuals with differing physical capacities. Moreover, like other functional measures assessed in the clinic, performance-based fatigability assessments may not accurately reflect physical fatigability's real-life impact. 26 In such cases, the SMA EFFORT could potentially serve as a valuable complement. Looking ahead, longitudinal studies will focus on characterizing PPF using the SMA EFFORT in a fully described cohort of individuals with SMA to assess the impact of treatment on mitigating this experience. For example, in individuals treated with nusinersen, wearing-off phenomena is regularly reported between doses, often manifesting as changes in endurance and mobility, though these changes have not been objectively measured. 45 Thus, the SMA EFFORT may provide a valuable opportunity for the evaluation of current and future treatment impact on PPF.
Conclusions
The SMA EFFORT aims to improve upon and overcome deficiencies in existing PROMs applied to SMA. Many of these scales are not disease-specific and are multidimensional, capturing several fatigue-related events that are not always relevant to physical fatigability. Even when PROMs do focus on physical fatigability, they include activities that people with SMA do not perform. Fatigue self-report is sensitive to context and meaning is influenced by individual experiences, and PPF inquiries should reflect the SMA experience.31,46
Effective outcome assessments include those that measure the patients’ experience well. These outcomes should be supported by objective evidence of an intervention's influence on results that are significant to patients. 47 The ultimate requirement is that PROMs provide a valid and reliable quantitative measure of subjective elements, such as PPF. To establish the adequacy of a self-report physical fatigability PROM, it must successfully capture PPF in a manner pertinent to individuals with SMA, prioritizing the perspective of individuals living with SMA throughout the entire process, from conceptualization to validation. 31
A critical first step to achieving this aim is to establish scales that emphasize current function and experience. This is especially significant when considering progressive neuromuscular conditions as functional abilities can evolve over time. Currently, a knowledge gap remains regarding how approved DMTs modify PPF in individuals with SMA. Furthermore, adequate characterization of PPF is vital to identifying targets beyond SMN-focused therapies.
As the SMA field advances towards combination treatment approaches with adjuvant muscle and neuromuscular junction targeted-therapies to better address the unmet needs of individuals living with SMA, PROMs that capture how one feels and functions are becoming increasingly necessary and important to regulatory agencies. 48 The SMA EFFORT introduces an innovative approach to measure PPF in SMA and may provide additional perspective on long-term treatment outcomes, regardless of disease status. Importantly, it aims to enhance the assessment of a disease characteristic whose standardization has been elusive thus far and offers an additional tool to characterize physical fatigability more comprehensively in SMA. Future studies will allow for scale refinement and further evaluation of the psychometric properties of the SMA EFFORT.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602241313326 - Supplemental material for Development of the SMA EFFORT: A new approach to characterize perceived physical fatigability in spinal muscular atrophy
Supplemental material, sj-docx-1-jnd-10.1177_22143602241313326 for Development of the SMA EFFORT: A new approach to characterize perceived physical fatigability in spinal muscular atrophy by Rafael Rodriguez-Torres, Cara H Kanner, Emma L Gay, David Uher, Thomas Corbeil, Giorgia Coratti, Sally Dunaway Young, Annemarie Rohwer, Robert Muni Lofra, Michael P McDermott, Darryl C De Vivo, Melanie M Wall, Nancy W Glynn and Jacqueline Montes in Journal of Neuromuscular Diseases
Supplemental Material
sj-docx-2-jnd-10.1177_22143602241313326 - Supplemental material for Development of the SMA EFFORT: A new approach to characterize perceived physical fatigability in spinal muscular atrophy
Supplemental material, sj-docx-2-jnd-10.1177_22143602241313326 for Development of the SMA EFFORT: A new approach to characterize perceived physical fatigability in spinal muscular atrophy by Rafael Rodriguez-Torres, Cara H Kanner, Emma L Gay, David Uher, Thomas Corbeil, Giorgia Coratti, Sally Dunaway Young, Annemarie Rohwer, Robert Muni Lofra, Michael P McDermott, Darryl C De Vivo, Melanie M Wall, Nancy W Glynn and Jacqueline Montes in Journal of Neuromuscular Diseases
Footnotes
Acknowledgements
The authors thank all study participants and their families for their willingness to participate. The authors would also like to acknowledge Dr Michio Hirano, Columbia University, Department of Neurology, for his guidance and support and for his leadership role within the Pediatric Neuromuscular Clinical Research Network.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded, in part, by the Neuromuscular Study Group and the Pediatric Neuromuscular Clinical Research Network (PNCRN) infrastructure grant from Cure SMA.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R. Rodriguez-Torres has received research support from the Neuromuscular Study Group and received honoraria for serving as a consultant for Scholar Rock. The institution of RRT has received research support from Roche, Avexis/Novartis, Biogen, Scholar Rock, and Genentech; E.L. Gay is supported by the Epidemiology of Aging Training Program (NIA T32 AG000181) at the University of Pittsburgh, School of Public Health; C.H. Kanner has no disclosures to report; D. Uher has received research support from Genentech and Cure SMA; T. Corbeil has no disclosures to report; G. Coratti has received personal fees for consultancy, advisory board and steering committee membership from Biogen, Roche, Scholar Rock, and Solid; S. Dunaway Young has received research or grant support from Biogen, National Institute of Child Health and Human Development, and Cure SMA. SDY has served as a consultant or on advisory boards for Biogen, Scholar Rock, F.Hoffman LaRoche, and Cure SMA; A. Rowher has no disclosures to report; R. Muni-Lofra has received grant funding from Sarepta and Roche. RML has been involved in research projects supported by Novartis, Biogen and Roche. RML has received consultancy fees from Roche, Biogen and Novartis; M.P. McDermott has no disclosures to report; M. Wall has no disclosures to report; D.C. De Vivo has no disclosures to report; N.W. Glynn has no disclosures to report; J. Montes has received research or grant support from Genentech, Scholar Rock, Biogen, National Institute of Child Health and Human Development, Muscular Dystrophy Association, and Cure SMA. JM has received honoraria for non-CME events from Genentech and Scholar Rock and served as a consultant or on advisory boards for Biogen, Scholar Rock, and F.Hoffman LaRoche.
Data availability
The data supporting the findings of this study are available within the article and/or its supplementary material. Anonymized data are available upon reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
