Abstract
X-linked myopathy with excessive autophagy (XMEA) is an X-linked recessive hereditary disorder, characterized by childhood onset weakness of predominantly limb and trunk skeletal muscles due to progressive loss of muscle tissue. Cardiovascular system is clinically spared generally. Here, we report the clinical characteristics, muscle biopsy and genetic findings of one adult-onset individual suffering from XMEA, caused by a novel variant in a Chinese patient. Unusually, the patient developed unexplained hypertension with high left ventricular voltage and cardiac left ventricular hypertrophy at the age of 27. Muscle MRI revealed extensive fat infiltration in the lower extremities involving vastus medialis, vastus lateralis, vastus intermedius, and long head of the biceps femoris, while rectus femoris, gracilis, adductor magnus, semimembranosus, and sartorius muscle relatively preserved. A muscle biopsy indicated numerous cytoplasmic autophagic vacuoles and deposition of complement C5b-9 and also expression of MHC class I. Immunofluorescence staining showed increased immunoreactivity of lysosomal membrane protein LAMP2 and autophagosome protein LC3A/B both localized with cytoplasmic vacuoles, and autophagy carrier protein P62-positive vacuoles accumulated remarkably in the patient's muscle. VMA21 protein levels were lower than controls in skeletal muscle tissue, suggesting a possible pathogenicity related to an alteration of the splicing efficiency in intronic mutation. This is the first report of a Chinese patient with a novel hemizygous intronic c.163 + 3A > G variant in the VMA21 gene, expanding the mutational and phenotypic spectrum of this disease.
Introduction
X-linked myopathy with excessive autophagy (XMEA) was caused by mutation of the VMA21 gene, firstly reported by Kalimo in 1988. 1 All mutations are hypomorphic alleles that reduce VMA21 protein expression. Most of the mutation are intronic variants that act by reducing splicing efficiency,2,3 and a few occur in the exon region near to introns. 4 VMA21 deficiency decreases V-ATPase activity, resulting in an increase in intralysosomal pH, which impairs lysosomal action and blocks the autophagic degradation of proteins leading to accumulation of autophagosomes and autophagic vacuoles. 5 These vacuoles are rimmed by membranes containing sarcolemmal proteins.6–8 In classic XMEA muscle biopsies, membrane-bound sarcoplasmic vacuoles can be visualized. By electron microscopy, an excessive number of autophagic vacuoles with staining properties of lysosomes is also observed. Thus, weakness of limb and trunk skeletal muscles due to progressive loss of muscle tissue can be manifested clinically.
The predominant clinical presentation in patients with VMA21 mutations is skeletal muscle involvement. Most patients have onset of limb girdle weakness sometime in childhood with slow progression towards wasting and ambulation loss,3,9–11 while rarely have adulthood-onset skeletal muscle disease.4,12 Yet, whether the myocardium is affected in XMEA remains controversial. As reported no cardiac abnormalities were present with a comprehensive electrocardiography, echocardiography, and gadolinium-enhanced cardiac MRI in five patients. 13 Another study reported mild cardiac hypertrophy in echocardiography, but additional testing was unperformed and cardiac vacuolation was unknown. 14 Besides, the intronic mutation exhibited typical myocardial vacuolation in XMEA patient was also reported. 15 However, studies on the pathogenesis, genotype and clinical manifestations of XEMA with cardiac involvement are still lacking.
Here, we describe a 37-year-old Chinese man with adult-onset myopathy, and developed unexplained hypertension with high left ventricular voltage and left ventricular hypertrophy at the age of 27. Muscle biopsy specimens revealed numerous cytoplasmic autophagic vacuoles. A hypomorphic variant in introns region near exon 2 (c.163 + 3A > G) in VMA21 gene was identified in the proband. Sanger sequencing confirmed correct segregation of the detected variant in three other family members. VMA21 protein levels were lower than controls in skeletal muscle tissue, suggesting a possible pathogenicity related to an alteration of the splicing efficiency in intronic variant. Through our study, we explain the possible pathogenesis, genotype and clinical manifestations of XEMA with cardiac involvement, and notably expand the spectrum of genotype-phenotype correlation of the disease.
Materials and methods
Patient
A Chinese patient with adult-onset myopathy underwent clinical and laboratory tests, muscle imaging, histopathological examination and molecular genetics studies to identify the etiology of the disease. The patient provided informed consent for the examinations and the study was approved by the Ethics Committee of the affiliated Hospital of Institute of Neurology, Anhui University of Chinese Medicine.
Muscle MRI
Axial sections of the lower limb muscles, and T1-weighted and T2-weighted sequences were evaluated using Semans MRI scanner in the patient.
Muscle histopathology and immunofluorescence studies
The muscle biopsy of the patient's left biceps brachii was performed. Pathological biopsy muscle specimens were rapidly processed in precooled isopentane placed in liquid nitrogen and 8 μ thick sections were cut for immunohistochemistry and Immunofluorescence. Muscle specimens with a diameter of 1 mm² were placed in 2.5% neutral glutaraldehyde electron microscope fixation solution for ultrastructural pathology. Muscle biopsies were analyzed in patient, including hematoxylin and eosin (H&E), nicotinamide adenine dinucleotide (NADH), modified Gomori trichrome (MGT). For the immunohistochemistry studies, the primary antibodies used for immunohistochemistry staining were rabbit monoclonal to dystrophin C (Abcam ab275391), rabbit monoclonal to MCH-I (Abcam ab274415), rabbit polyclonal to C5b-9(Abcam ab55811). For the immunofluorescence studies, the primary antibodies used for immunofluorescent staining were rat monoclonal to Laminin 2 alpha (Abcam ab11576), rabbit monoclonal to p62 (Abcam ab207305), rabbit anti-LC3A/B antibody (bs-20414R). The ultrastructure of muscle fibers was observed using electron microscopy (Japan Electron Optics Laboratory Co., Ltd, JEM-1400). Electron microscopy was performed using standard techniques.
DNA sequencing
The exons of 251 myopathy-associated genes sequencing were undertaken for the index case on an Illumina HiSeq 2000 platform. The list of targeted genes is available upon request. Sanger sequencing of VMA21 gene was used to analyze DNA samples of family members I-1, I-2, II-1, III-1 and III-2. DNA extraction from blood was performed using DNAeasy Blood & Tissue Kit from Qiagen. Three separate PCR reactions were used to amplify the 2 exons. The primers were previously used to identify VMA21 variants in XMEA patients. The PCR reactions were carried out using a standard protocol. The PCR products were visualized on a 1.5% agarose gel and purified using micro-CLEAN (Microzone). Quantification was performed using Nano Drop ND-1000 spectrophotometer V 3.3 (Thermoscientific). PCR products (50 ng each) were used for Sanger sequencing, with the forward and reverse primers at a concentration of 5 pmol each in a final volume of 7.7 μL. The patient's variant was confirmed by sequences originating from upstream and downstream primers.
Western blot
To confirm that the variant affects the VMA21 protein, we performed Western Blot analysis on total protein extracted from skeletal muscle of our patient. Primary polyclonal antibodies for VMA21 and GAPDH (loading control) and secondary anti-rabbit antibody were from China (respectively, Affinity and Servicebio).
Results
Clinical findings
A 37-year-old man presented with a 10-year history of slowly progressive bilateral lower limb weakness, predominantly involving the proximal muscles. For the first 5 years of the disease, the patient has mild weakness in both legs. In the last 5 years, he had difficulty in getting up from a squatting position and climbing stairs. There was no history of myalgia, sensory symptoms, oculobulbar involvement, respiratory distress. There was no history of muscle disease in the family. The patient had a history of hypertension for 10 years (Maximum of BP: 165/100 mmHg), he was always taking felodipine sustained-release tablets. Physical examination revealed symmetrical predominant hip-girdle weakness [Medical Research Council score of 4/5 in deltoid, biceps and triceps, 4/5 in wrist extension and flexion, 4/5 in psoas, 3/5 in hip adductors and abductors, and quadriceps, and 4/5 in tibialis anterior, plantar flexor and extensor bilaterally]. There was no scapular winging but had mild atrophy of shoulder girdle muscles and pelvic girdle muscles. He could not rise from the floor and had a positive Gowers’ sign. Fasciculations, joint laxity, and intellectual disability were absent. The serum creatine kinase (CK) level was 859 U/L (normal range 24-170U/L), which was higher than 5 times the upper normal limit. The needle electromyography recorded myogenic lesion of right biceps brachii, right deltoid, right vastus internus, left gastrocnemius, right tibialis anterior. The motor and sensory nerve conduction velocity and amplitude of the peripheral nerve were normal. The proximal muscles of the lower extremities showed extensive fat infiltration on MRI involving vastus medialis, vastus lateralis, vastus intermedius, and long head of the biceps femoris, while rectus femoris, gracilis, adductor magnus, semimembranosus, and sartorius muscle relatively preserved (Figure 1A, B). Resting ECG voltage met the Sokolov-Lyon criteria for left ventricle (LV) hypertrophy without other abnormalities. Echocardiography showed the larger left atrium is and the thicker left ventricular, while normal in the valves, pericardium, large vessel and pulmonary artery pressures.
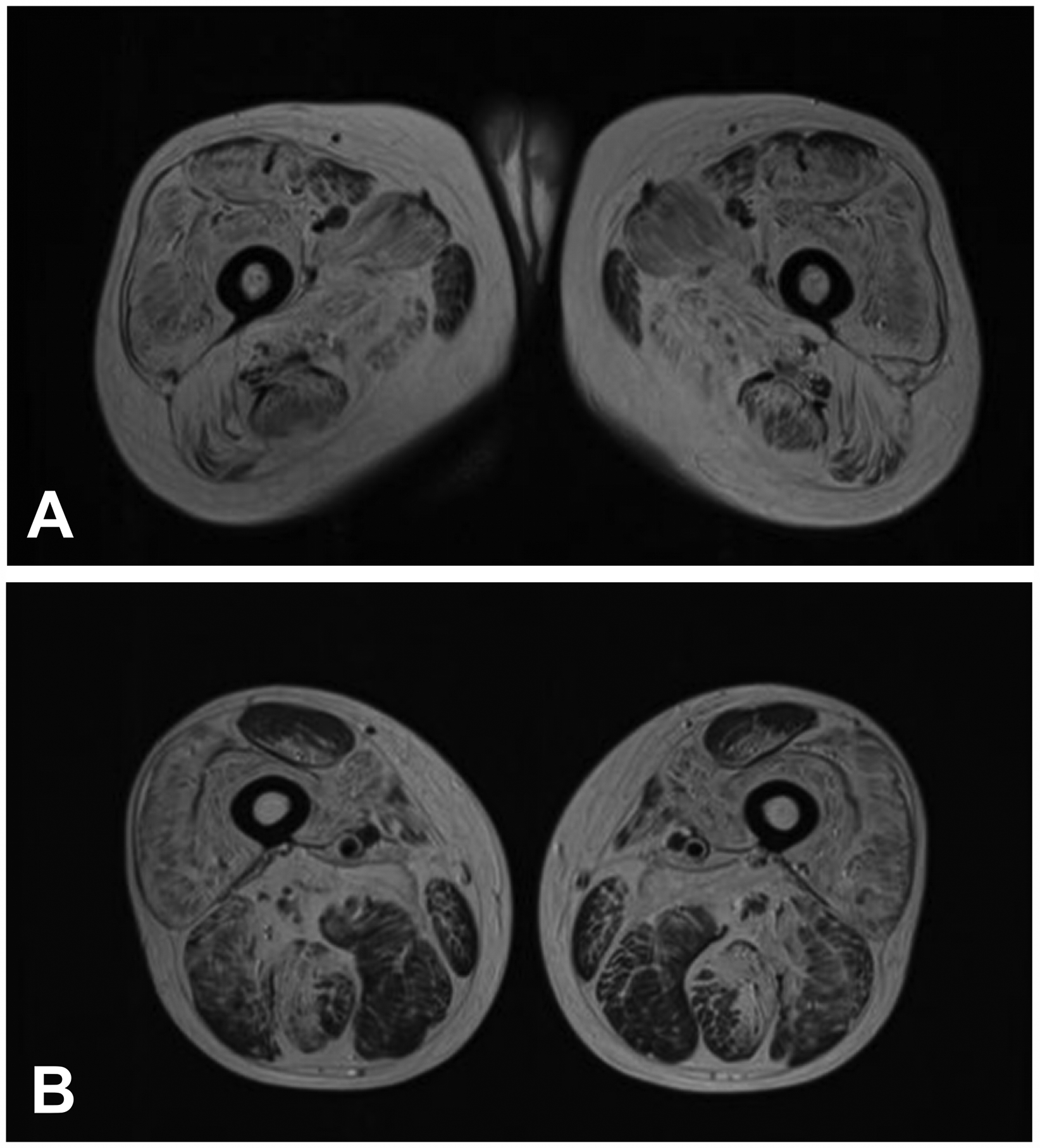
Muscle MRI in the patient (A: the proximal part of the thigh, B: the distal part of the thigh). T1-weighted imaging of bilateral thighs revealed extensive fat infiltration involving the vastus medialis, vastus lateralis, vastus intermedius, and long head of the biceps femoris (A, B), while rectus femoris, gracilis, adductor magnus, semimembranosus, and sartorius muscle relatively preserved (B).
Muscle biopsy findings
Histopathological examination revealed mild hyperplasia of the endomyosium, and marked perimysial fat infiltration (Figure 2A). The fiber size variate obviously, and a large amount of vacuolar fiber and necrotic fibers can be seen. NADH revealed that the two types of muscle fibers were distributed interphase, and the structure of myofibrillar fibers was disordered with numerous vacuolar fibers (Figure 2B). MGT revealed that numerous cytoplasmic vacuoles, complex muscle fiber splitting (Figure 2C). Immunohistochemical results showed no loss of dystrophin expression in fibers, and some vesicle structures with positive dystrophin C termini expression were seen in some muscle fibers (Figure 2F). Membrane attack complex (MAC, complement C5b-9) could be seen within cytoplasmic vacuoles and at the muscle fiber surface membrane (Figure 2D). Another inflammatory marker, MHC class I expression had a similar distribution (Figure 2E). In immunofluorescence studies, various sarcolemma-associated proteins were shown in cytoplasmic vacuoles. Increased immunoreactivity of lysosomal membrane protein LAMP2 was seen both at the surface membranes and within the cytoplasm of affected fibers (Figure 2G). Autophagosome protein LC3A/B immunostaining localized with cytoplasmic vacuoles (Figure 2H). A remarkable accumulation of autophagy carrier protein P62-positive vacuoles was showed in the patient muscle (Figure 2I).
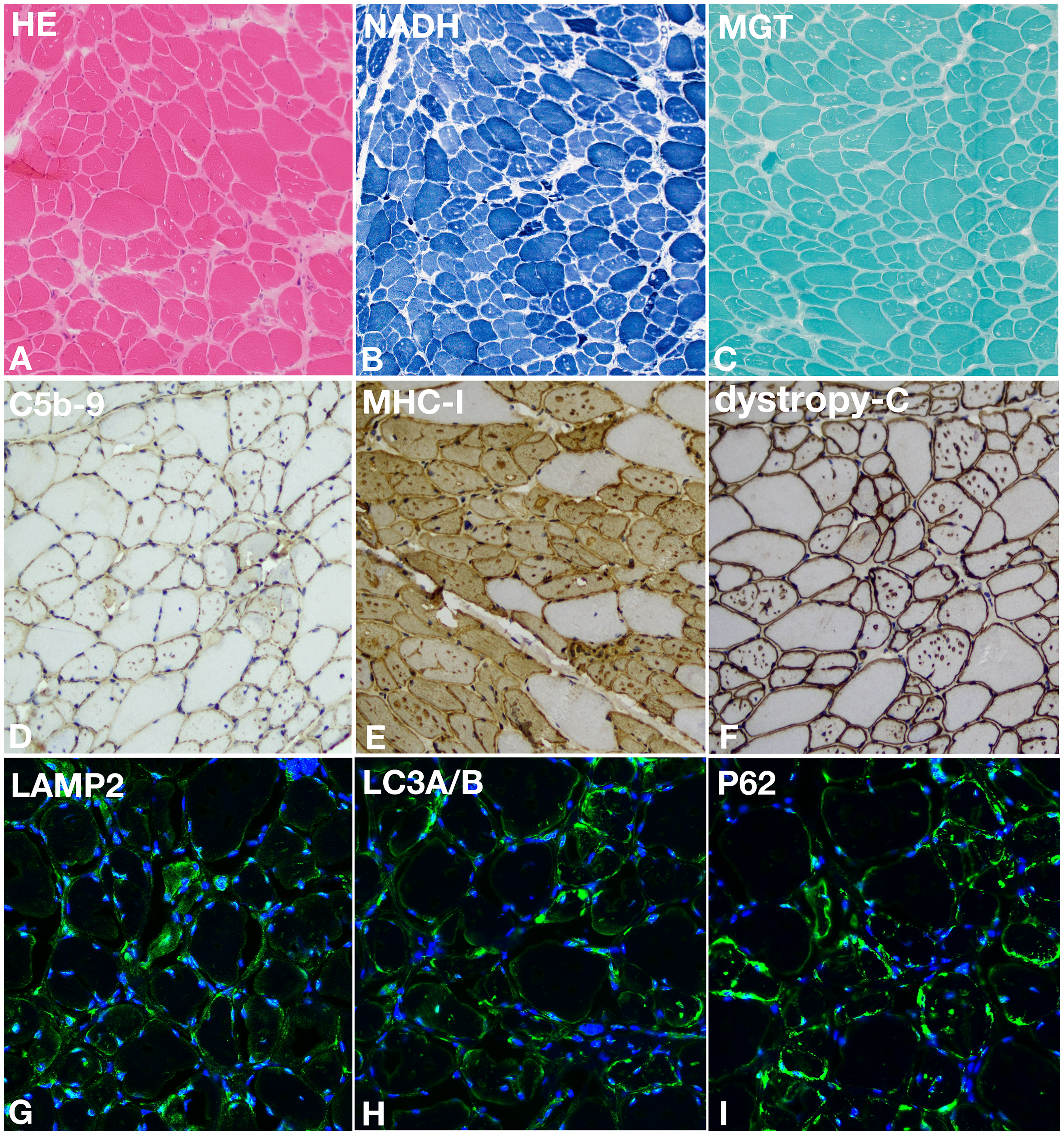
Light microscopic histopathology. Characteristic features of the patient that are readily observable by light microscopic analysis of muscle biopsy samples include numerous cytoplasmic autophagic vacuoles in HE (A), NADH (B), and MGT (C). Complement C5b-9 deposition is widespread and involves both the sarcolemma and cytoplasmic vacuoles (D). MHC class I expression has a similar distribution (E). Immunohistochemical results showed some vesicle structures with positive dystrophin C termini expression (F). Immunofluorescence results showed lysosomal membrane protein LAMP2 immunostaining localizes with cytoplasmic vacuoles (G), Autophagosome protein LC3A/B immunostaining localizes with cytoplasmic vacuoles (H), A remarkable accumulation of autophagy carrier protein P62-positive vacuoles in the patient muscle (I). A, B, C bar = 200 μm; D, E, F bar = 100 μm. Green, LAMP2 (G), LC3A/B (H), P62 (I); Blue, DAPI, Magnification 20×, confocal microscopy. G, H, I bar = 50 μm.
Electron microscopic analysis of the biopsied muscle showed large and small autophagic vacuoles interspersed near the sarcolemma (Figure 3A). Exocytosis (extrusion) resulted in a serrated surface contour to some muscle fibers and lysosomal debris in the extracellular space was seen between multiple layers of duplicated basal lamina (Figure 3B). Autophagic vacuoles interspersed between the myofibrils, membrane-bound autophagic vacuoles containing granular debris, small vesicles, degenerating mitochondria, and membrane whorls (Figure 3C, D).
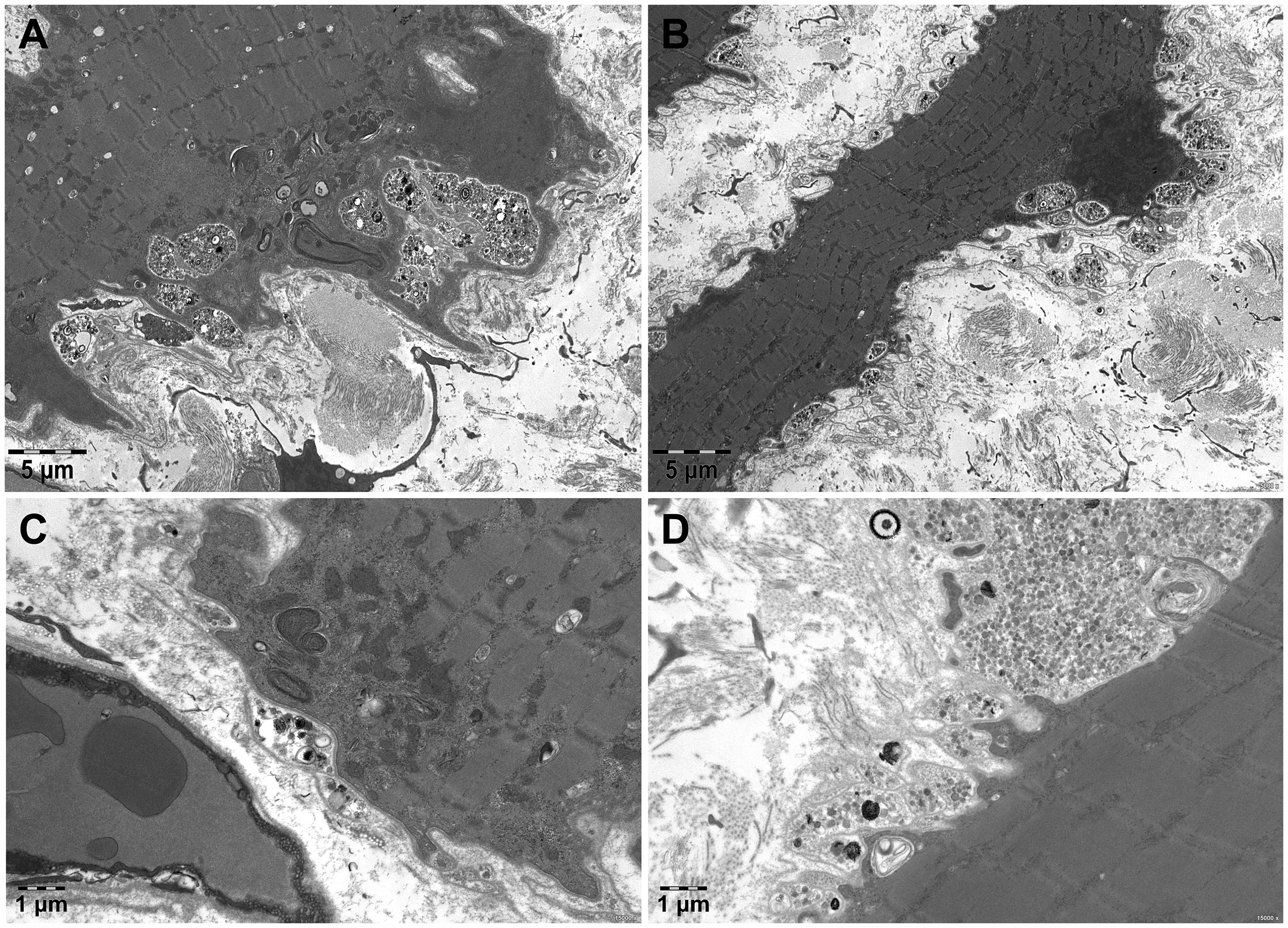
Ultrastructural pathology of the patient. Muscle biopsy electron microscopy shows large and small autophagic vacuoles interspersed near the sarcolemma (A). Exocytosis (extrusion) results in a serrated surface contour to some muscle fibers and lysosomal debris in the extracellular space is seen between multiple layers of duplicated basal lamina (B). Autophagic vacuoles interspersed between the myofibrils, membrane-bound autophagic vacuoles containing granular debris, small vesicles, degenerating mitochondria, and membrane whorls (C, D). A, B bar = 5 μm; C, D bar = 1 μm.
Genetic findings and expression of VMA21 protein
Genetic analysis of VMA21 gene was performed for proband after informed consent was provided as a part of the diagnostic procedure, a hemizygous variant was revealed (c.163 + 3A > G) (Figure 4A). Sanger sequencing of the parents revealed that came from his mother (Figure 4A), who had a heterozygous variant. Both of his two daughters were verified at the corresponding sites, which had heterozygous variants in the intronic region adjacent to exon 2 of VMA21(c.163 + 3A > G) (Figure 4A). The chromosome location of this variant is chrX:150572215 (genome version: he19), and the transcript is NM 001017980.
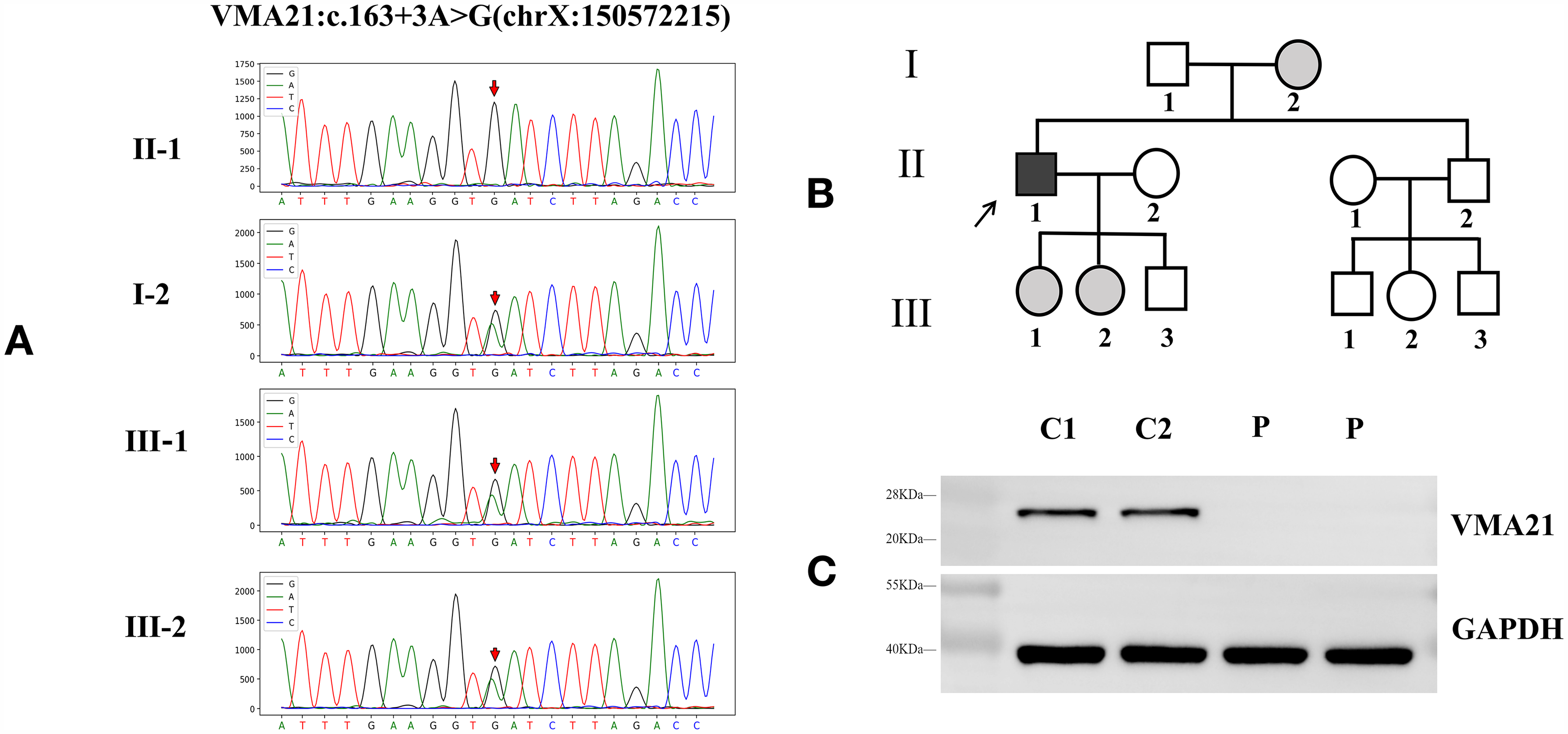
Pedigree of the family and Western blot of VMA21 protein. Pedigree and VMA21 sequences of the proband and his mother and his two daughters (A, B).
To determine whether the identified variants affected VMA21 protein expression, we performed Western Blot analysis on total protein extracted from the patient's muscle tissue. We observed a single 25-kDa band, which is the molecular weight of VMA21 in healthy control tissues. The muscle biopsy specimen of the patient contained a weaker VMA21 band than that of an age-matched healthy control (C1 and C2) (Figure 4C). VMA21 protein was dramatically decreased in our patient.
Discussion
In our study, we reported an adult-onset phenotype of individual with myopathy and cardiac involvement suffering from an VMA21-asscociated XMEA caused by a novel variant in A Chinese patient. Muscle MRI showed extensive proximal muscle fat infiltration of the lower extremities including vastus medialis, vastus lateralis, vastus intermedius, and long head of the biceps femoris (A, B), while rectus femoris, gracilis, adductor magnus, semimembranosus, and sartorius muscle relatively preserved. Muscle biopsy indicated numerous cytoplasmic autophagic vacuoles and deposition of complement C5b-9, also expression of MHC class I. Increased immunoreactivity of lysosomal membrane protein LAMP2, and autophagosome protein LC3A/B immunostaining localized with cytoplasmic vacuoles. VMA21 protein levels were lower than controls in skeletal muscle tissue. Our study explained the clinical characteristics, possible pathogenesis, and genotype of XEMA with cardiac involvement, and notably expanded the mutational and phenotypic spectrum of the disease.
Regarding the onset age of XEMA in clinical, most patients have been reported in children, while late-onset forms of the disease have also been described. 16 Crockett reported a patient with onset of slowly progressive proximal weakness of the lower limbs after age 50 who maintains ambulation with the assistance of a cane at the age of 71. 12 Our patient had no symptom in childhood and developed clinical symptoms at the age of 27. Previous reports presented childhood onset and with a preponderance of the skeletal myopathic phenotype. However, it remains unclear what exactly determines the onset age and clinical performance. As for whether the age of onset was related to different gene loci, a reasonable explanation will be given in the future with the development of genetic research.
The patient also showed other extra muscular system involvement, by an intronic variant in the VMA21 gene. Usually, XMEA is considered a skeletal muscle disease without any systemic involvement. In cardiac autopsy of one of the original XMEA family, no significant numbers of autophagic vacuoles was detected. 13 However, a later study demonstrated that the VMA21 mutation, c.164-6 T > G, does result in XMEA-typical autophagic vacuoles in the cardiac muscle. 15 Munteanu 17 described two adult cases where resting ECG voltage met the Sokolov-Lyon criteria for left ventricle hypertrophy, and echocardiography showed mild enlargement of the left atrium. Similar to these studies, our patient also had unexplained hypertension with high left ventricular voltage and left ventricular hypertrophy. Due to the lack of extensive cardiac pathology, the question remains unclear why the cardiac and other extra muscular system are so poorly affected with VMA21 variant. We speculate that different VMA21 variant sites or acquired environmental factors may be involved in the process of myocardial involvement.
In the basis of typical clinical features, pathological findings are decisive clues to the correct diagnosis. The ultrastructural pathology of XMEA was classic, and excessive number of autophagic vacuoles with staining properties of lysosomes was observed. 1 When observed in biopsies of skeletal muscle, the bounding membranes of these vacuoles stain positively for sarcolemmal proteins, and their presence is pathognomonic for of XMEA. The vacuoles were also intensely stained for C5b-9 and MHC class I, as described in a previous comprehensive report of pathological finding in muscle biopsy in XMEA. 18 Our patient showed numerous cytoplasmic autophagic vacuoles, and also C5b-9 deposition and MHC class I expression is widespread and involves both the sarcolemma and cytoplasmic vacuoles, consistent with typical pathological features in XMEA. There is an accumulation of aggregated proteins in association with the accumulation of autophagic proteins such as microtubule-associated protein 1A/1B-light chain 3 (LC3) and the autophagic substrate p62.5,7,19 Colocalization of the vacuoles with LC3 was demonstrated in a vacuolar disease, possibly XMEA. 19 In our patient, sarcoplasmic LC3A/B and P62 positive vacuoles were significantly accumulated in the muscle biopsy, confirming an autophagy defect.
Additionally, increased numbers of cytoplasmic lysosomes may be demonstrated by immunostaining for lysosomal proteins such as LAMP2, which can distinguish XMEA from Danon disease. 20 The entire LAMP2 protein is lost in Danon disease, 21 while VMA21 is never lost in XMEA, merely diminished. 3 Our patient showed lysosomal membrane protein LAMP2 immunostaining localizes with cytoplasmic vacuoles in immunofluorescence and decreased VMA21 protein expression in Western Blot, supporting the diagnosis of XMEA. Indeed, while most XMEA patients have a similar degree of decreased VMA21 protein, recently cases were described as more pronounced VMA21 diminution accordance with an earlier and more severe disease course. The extensive muscular fat infiltration and more decreased VMA21 protein level in our patient may be related to the specific novel variant in Chinese.
The relationship between genotype and clinical phenotype in XMEA was unelucidated. To date, all known XMEA mutations are single-nucleotide substitutions, usually in introns, specifically in the splicing. The study of intron included the affected splicing branch point (c.54-27A > C; c.54-27A > T; c.54-16_54-8del),2,9,22 the affected splicing donor site (c.163 + 4A > G), 3 and the affected splicing acceptor site (c.164-6T > G; c.164-7T > G).12,15,23 Non-coding VMA21 deletions cause XMEA was also reported. 22 The mutation (c.164-7T > G) was shown to cause a late-adulthood onset variant of the disease. 12 One mutation was identified in an exon (c.272G > C), but this point mutation was predicted to affect a splice-enhancer site. 4 All these mutations lead to a reduction in VMA21 mRNA and VMA21 protein levels. XMEA mutations, most of which affect splicing, possibly do so to a lesser extent in extra muscular tissues allowing higher levels of VMA21 expression. So far, although the point variant (c.163 + 3 A > G) have been reported only in a European family, no VMA21 protein detection has been performed. 24 Compared with previous reports, our patient variant was located in the intronic region adjacent to exon 2 (c.163 + 3 A > G), which might affect the splicing similarly according to the dramatically diminished VMA21 protein expression. It is possible that the novel variant in a Chinese patient may lead to late onset or cardiac involvement, but the specific pathogenesis needs further verification.
Conclusion
We report a novel variant in the VMA21 gene for the first time in a Chinese patient. Our patient had late onset myopathy with cardiac involvement uncommon to previously studies. The current report expands the phenotypic and genotypic spectrum of XMEA caused by VMA21 variant.
Footnotes
Acknowledgments
We thank the patient and his families for placing their trust in us. We are grateful to members of our neuromuscular laboratories for constructive comments on this report.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
This study was funded by the Natural Science Foundation of Anhui Province (2308085QH290), High-level talent support plan of Anhui university of Chinese medicine (Grant No. DT2300000270) and the Key Project of Natural Science Research Project of Universities in Anhui Province (2024AH050984, KJ2021A0550, KJ2021A0551).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
