Abstract
In April 2023, over 30 experts and advocates from four countries met in Rome, Italy to discuss unmet needs in endocrine and bone health care for individuals with Duchenne muscular dystrophy (DMD). Despite recent advances in muscle-targeted therapy, long-term glucocorticoids (GC) remain the backbone of treatment for the foreseeable future. This affirms the need to intensify efforts that will mitigate serious complications of GC therapy, including unexpected mortality due to fat embolism syndrome following bone injury and also unrecognized adrenal suppression, early loss of ambulation linked to excess weight and/or fragility fracture, adverse cardiometabolic effects of GC, the psychosocial impact of profound growth and pubertal delay/hypogonadism, and the burden to families arising from monitoring and treating endocrine and skeletal complications of GC therapy. Delegates discussed: 1. The impact of GC therapy on the heart, 2. Predictors of fragility fractures and experience with intravenous and oral bisphosphonates plus teriparatide, 3. The effect of hormonal therapy on muscle-bone health, 4. Adrenal suppression, 5. Weight management, 6. Puberty, sexuality, fertility and gender identity, 7. The impact of early GC initiation, 8. Emerging knowledge about vamorolone (a novel dissociative steroid) and its effects on muscle, bone and endocrine health, and 9. Experiences implementing an endocrine-bone health management strategy nation-wide (in the UK). At the conclusion of the meeting, it was agreed that an endocrine-bone working group should be struck to continue the narrative, following which the International OPTIMIZE DMD Consortium was ignited to move the dial in these important areas.
Keywords
Introduction
Pat Furlong, Parent Project Muscular Dystrophy, USA; Filippo Buccella, Parent Project APS, Italy; Leanne Ward MD, University of Ottawa, Canada
Over 30 health care professionals, patient advocates, and industry representatives from four countries (Canada, the United States of America (USA), the United Kingdom (UK), and Italy) met in Rome, Italy from the 26th to 28th of April 2023 to discuss current standards and unmet needs in the endocrine and bone health care of individuals living with Duchenne muscular dystrophy (DMD). The Agenda for the Meeting is provided in a Supplemental Figure.
Pat Furlong (Founder and Director, Parent Project Muscular Dystrophy), Filippo Buccella (Parent Project, Rome, Italy) and Dr Leanne Ward (Clinician Scientist, University of Ottawa) opened the “Endocrine and Bone Issues in Duchenne Muscular Dystrophy (DMD): an Ever-Changing Landscape” International Workshop with a word of welcome, facilitation of introductions among delegates, and a review of the workshop's goals.
Dr Ward summarized that despite significant advances in muscle-targeted therapy for DMD, long-term, high-dose glucocorticoids (GC) remain the backbone of treatment for the foreseeable future. Dr Ward emphasized that there are serious complications of long-term GC therapy combined with the progressive myopathy of DMD 1 (Figure 1). These include 1. Unexpected mortality arising from fat embolism syndrome after a fracture or sub-clinical bone injury, 2. Unexpected death due to unrecognized and therefore untreated GC-induced adrenal suppression, 3. Increased risk of premature loss of ambulation following osteoporotic fractures and/or excess weight gain, 4. Adverse cardiometabolic effects resulting from GC-driven weight gain (e.g., hypertension, dyslipidemia, impaired glucose tolerance and, more rarely, diabetes), 5. The psychosocial impact of profound growth and pubertal delay, and 6. The burden to families from the need to monitor and treat the endocrine and skeletal effects of GC and the frequent medical visits involved in such care.
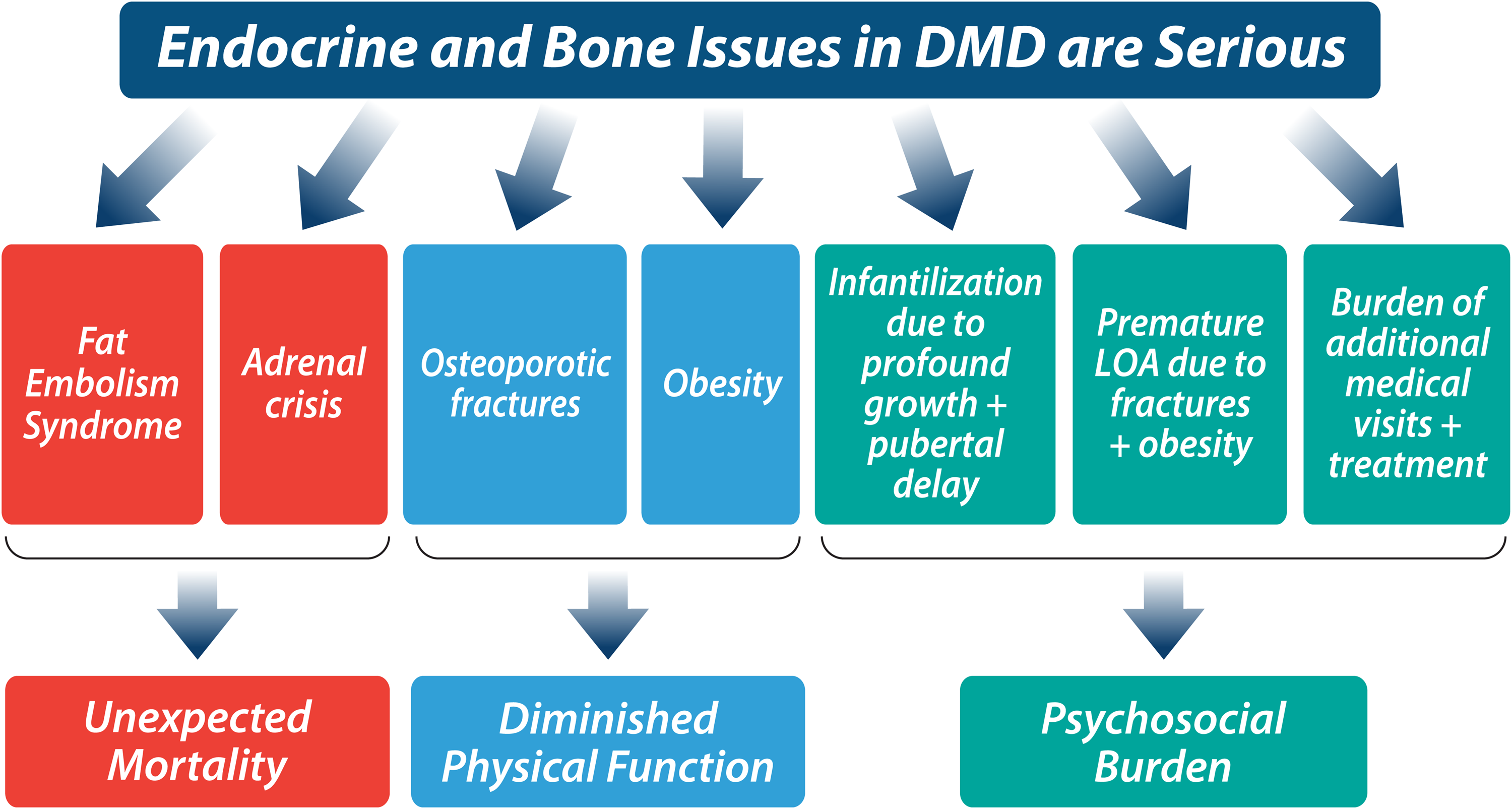
Underscores the serious complications of long-term, high-dose GC therapy combined with progressive myopathy in patients with DMD. For further details, see text.
Ms. Furlong and Mr Buccella affirmed that the serious consequences of GC therapy are a significant challenge for families living with DMD day-to-day, and that the need to educate and support health professionals in their leadership to achieve the current standards of care2–4 globally is at once tremendous and urgent. Pat Furlong also emphasized the need to “do better” than the current standards of care, with examples provided in two important areas: prevention of first-ever fracture given the links between bone fragility and both premature loss of ambulation and mortality, and the need to prevent unexpected death arising from unrecognized adrenal suppression (a classic and indeed highly preventable consequence of GC therapy in any disease setting). Pat Furlong highlighted the challenges that families face in receiving inconsistent messages from health care providers about the best course of action for optimal bone and endocrine care. Pat also reminded the delegates that families do not have time for advances to eke out slowly, and that the efforts of health care professionals in partnership with patients and the DMD community need to be nimble and quick, in order to positively impact the families living with DMD today, as well as the families of the future.
As the delegates embarked on two days of information sharing, active discussion, and strategizing for a better future, it was clear that this meeting would play an unprecedented role in catalyzing advances in endocrine-bone health care for individuals living with DMD, and in establishing the relationships that would fuel such advances. The range of individuals attending the workshop ensured broad-based discussions that included a total of four patient representatives (three from Parent Project Muscular Dystrophy, and one from Parent Project APS, Italy), 27 clinicians (key opinion leaders in the areas of pediatric endocrinology, osteology and neuromuscular disorders), one data scientist from The Collaborative Trajectory Analysis Project (to participate in data sharing discussions related to future research arising from the collaborations struck during the workshop), and two industry representatives (to provide perspectives on emerging therapy for the treatment of DMD, and the ways in which novel, DMD-targeted therapy may influence endocrine and bone health trajectories in this condition). This workshop was sponsored by Parent Project Muscular Dystrophy + Parent Project APS (Italy) and received financial support via an unrestricted (unconditional) educational grant from PTC Therapeutics, Pfizer, and Santhera Pharmaceuticals.
Current and novel Duchenne therapies: an overview
Hugh McMillan MD, Children's Hospital of Eastern Ontario, University of Ottawa, Canada
As stated in the introduction, classic GC therapies (deflazacort and prednisone) are well-known for their beneficial effect on skeletal muscle in DMD, 5 having been shown to prolong independent ambulation, delay scoliosis onset and progression, and improve cardio-respiratory outcomes.6,7 However, the significant co-morbidities caused by long-term, daily, high dose GC therapy have spurred interest in other therapies for DMD. 8
Dr McMillan discussed the expanding number of disease-modifying therapies for individuals with DMD. While many novel therapies are under study in clinical trials, a number have attained regulatory approval and are commercially available in various parts of the world. Some disease-modifying therapies are specific to a patient carrying a certain exon deletion or nonsense variant whereas others are applicable to any patient with DMD. All approved gene-directed therapies have been used in conjunction with GC treatment with some (i.e., gene replacement therapy) requiring higher GC dosing at the time of novel interventions. As such, GC-related morbidity is anticipated to remain a challenge for these patients for the foreseeable future, a fact that fueled this forward-looking DMD Endocrine-Bone Workshop. Overall, Dr McMillan highlighted that the DMD treatment landscape is growing, and that the endocrine and bone morbidities associated with DMD will therefore evolve, necessitating periodic updates to current “endo-bone” standards of care so that they are appropriately calibrated to advances in the treatment of the underlying condition.
The impact of glucocorticoids on the heart in DMD
Linda H. Cripe MD, Nationwide Children's Hospital and Ohio State University, USA
Dr Cripe reviewed that the heart is a muscle and should be considered when discussing the impact of therapies such as GC in those with DMD. Despite a well-established negative cardiovascular impact from exogenous GC therapy use in numerous clinical settings,9–11 the literature supports cardiac benefit in patients with DMD. 12 Several investigators have demonstrated that early initiation of GC may delay the development of left ventricular dysfunction. In addition, GC-treated patients experience improved longevity over GC-naïve patients.13–16 One of the most significant and more recent studies was a multi-center trial examining 408 patients with DMD. Thirty percent of deaths were attributable to cardiovascular causes, with lack of GC use associated with cardiac causes of death. 17 Another large multi-center study of 462 DMD patients demonstrated a significant delay in the onset of clinical cardiomyopathy, from a mean age of 13.1 years in non-GC-treated patients to 14.3 years in GC-treated patient. 18 In addition, longer duration of GC use has been associated with less of an age-related increase in myocardial fibrosis. 19 Unfortunately, the mechanism of GC therapy slowing cardiac disease remains unclear and is likely complex; in addition, GC type and dosing regimen are likely to be significant confounding variables. Prospective clinical trials are currently needed to investigate these complex issues, since the majority of studies to date have been retrospective observational studies.
Vamorolone and muscle health in DMD
Paula Clemens MD, University of Pittsburgh Medical Center, USA
Dr Clemens reiterated, as did others presenting at this workshop, that GC (prednisone and deflazacort) are the standard of care for DMD, and have been clearly shown to prolong the age of loss of ambulation if treatment is begun during childhood. 20 The desire to identify an intervention that would preserve the anti-inflammatory benefits of GC on muscle health but diminish the undesirable side effects led to the clinical development program for vamorolone.
Vamorolone is a first-in-class, dissociative steroid developed with the goal to retain the benefits of GC with fewer adverse effects. Pre-clinical studies established that vamorolone inhibits NF-κB signaling and thus acts as a potent anti-inflammatory agent, similar to GC. Unlike GC, vamorolone has reduced activation of GC response elements, is not a substrate for 11βHSD regulatory enzymes, and is a mineralocorticoid receptor antagonist. In contrast, classic GC are mineralocorticoid agonists; mineralocorticoid antagonism is predicted to be cardioprotective. 21
A Phase 1 study in healthy adult males established early safety for vamorolone. 22 A Phase 2A open-label study was conducted in males aged 4–7 years with DMD. The study had a 2-week dose-finding stage 23 that was extended at each dose level for six months of treatment. 24 This study demonstrated good safety and tolerability of vamorolone in young children with DMD and identified dose-related improvements in muscle function compared to an external, group-matched dataset drawn from the prospectively collected Cooperative International Neuromuscular Research Group (CINRG) Duchenne Natural History Study (the “CINRG DNHS”). Participants in the extension of the Phase 2A study were followed for 30 months, with published reports at 18 25 and 30 26 months. Long-term therapy demonstrated continued safety, tolerability, and maintenance of a beneficial treatment effect.
The Phase 2A study established efficacious dose levels to inform the randomized, placebo- and prednisone-controlled, double-blind Phase 2B study, which was conducted across more than 30 study sites in 11 countries. At 24 weeks of treatment, this study met its primary and first four hierarchically defined secondary outcomes. 8 At 48 weeks of treatment, there was maintenance of efficacy in the vamorolone and prednisone-to-vamorolone cross-over arms and demonstration of efficacy in the placebo-to-vamorolone cross-over arm. 27 Overall, vamorolone at a dose of 6 mg/kg daily showed better maintenance of effect compared with vamorolone at a dose of 2 mg/kg daily for most motor outcomes over 48 weeks. 27
Overall, the studies of vamorolone treatment for children with DMD ages 4–7 years, up to and including a rigorous, controlled, blinded study, showed clinically relevant functional improvements in muscle function for participants. Vamorolone was safe and well-tolerated throughout all studies. Notably, and detailed elsewhere, vamorolone preserved (for those who initiated vamorolone) and restored (for those who switch from prednisone to vamorolone) growth trajectories. The study of vamorolone has now been expanded to include a blinded pilot study of vamorolone for the treatment of adults with Becker muscular dystrophy, currently underway at two sites (USA and Italy, NCT05166109). Subsequent to the workshop, vamorolone was approved for the treatment of DMD by the Food and Drug Administration (FDA) in October of 2023 and by the European Medicines Agency (EMA) in December of 2023. A longer-term observational (registry) study of vamorolone is presently underway (NCT NCT06564974).
Vamorolone and bone health in DMD
Leanne Ward, Children's Hospital of Eastern Ontario, University of Ottawa, Canada
Dr Ward discussed the results of skeletal health evaluations in young, ambulatory patients with DMD who had participated in phase 2 longitudinal studies of vamorolone.8,26 Vamorolone-treated individuals showed improved linear growth compared with a natural history cohort treated with daily GC for 30 months; however, vamorolone-treated patients also demonstrated increases in weight that were comparable to the natural history, prednisone-treated cohort. 26 These results were affirmed in a 24-week, randomized, double-blind, placebo-controlled trial of vamorolone 2 mg/kg and 6 mg/kg versus placebo and prednisone; this trial provided the additional observation that vamorolone-treated trial participants experienced no changes in serum markers of bone turnover, compared with the rapid and excessive decline observed in the prednisone-treated group. 8
In a preliminary analysis, the prevalence of vertebral fractures in ambulatory children with DMD on vamorolone after years of drug exposure was benchmarked to an age-matched, external comparator, the Finding the Optimal Regimen FOR-DMD cohort. 7 Although the comparative data arose from two different studies, the central imaging radiologists were the same for both trials, the lateral spine radiographs were read according to the same method (Genant semi-quantitative) by the gold standard “triple read” approach, and central reading took place over similar time periods (May 2019 to March 2022 for both trials). This preliminary analysis showed the prevalence of vertebral fractures on vamorolone was lower compared to classic daily GC therapy (13% on vamorolone compared with close to 30% on daily deflazacort and daily prednisone after 3–3.5 years of exposure). In addition, it was shown that the Spinal Deformity Index (SDI, the sum of the Genant grades and an overall measure of spine fracture burden) on vamorolone was about 1/3 that of daily deflazacort (where the SDI was the highest), and about 2/3 that of daily prednisone. After 3 years of drug exposure, there were no vertebral fractures identified on intermittent prednisone 10 days on/off. While intermittent GC therapy appears bone-protective, 28 both observational 28 and randomized 7 studies have shown that saltatory GC exposure (prednisone 10 days on/off) is associated with reduced ability to preserve muscle strength relative to daily therapy.7,28
These data were encouraging, as they suggested that spine fracture burden may be reduced, albeit not absent, on vamorolone relative to classic daily GC. It was further highlighted that the impact of vamorolone on long bone fractures, which are not as sensitive to positive changes in bone turnover due to the small bone size inherent to DMD, remains unknown. The impact of other classic GC regimens beyond daily prednisone, daily deflazacort and intermittent prednisone 10 days on/off on skeletal health (including vertebral and long bone fractures) also remains uncertain.
Gene therapy, steroids and bone health in DMD
Susan Apkon MD, Children's Hospital Colorado and University of Colorado Health Sciences Center, USA
Dr Apkon summarized that with numerous gene therapy clinical trials underway and the FDA having approved delandistrogene moxeparvovec, understanding the impact of gene therapy on bone strength and fragility is important. Currently, there are no data available to shed light on the impact of gene therapy on bone strength. While it is speculated that clinically significant improvements in muscle strength will translate into improved bone strength (given the intimate functional relationship between the two), the magnitude of the improvement in muscle that is needed to substantially reduce vertebral and non-vertebral fracture rates in DMD is unknown. Important to the endo-bone landscape is the fact that current gene therapy trials include the use of short-term, high-dose GC with varying protocols, an approach that is designed to modulate the immune response to the investigational product. Additionally, high-dose GC therapy may also be needed if adverse events such as elevated transaminases or liver toxicity are observed following the administration of gene therapy. The impact of these high-dose GC protocols on bone strength in the gene therapy setting has not been investigated. Furthermore, the ongoing need for GC treatment as a standard of care even after gene therapy has been delivered is also under study; therefore, patients with DMD may indeed be faced with the use of GC therapy long-term despite gene therapy. Going forward, gene therapy efforts should focus on minimizing the risk of bone fragility as far as possible through timely initiation of bone protection therapy, so that the benefits of gene therapy can be fully realized without attenuation by the adverse effects of GC on bone strength.
In summary, the impact of gene therapy on bone strength remains unstudied. In addition, the deleterious impact of high-dose GC treatment following gene therapy also requires further investigation. It is imperative to collect data on bone health during and following gene therapy clinical trials and with commercially available treatment.
Experience with bisphosphonates and teriparatide for the treatment of osteoporosis in patients with DMD
Cuixia Tian MD and Nat Nasomyont MD, Cincinnati Children's Hospital and University of Cincinnati College of Medicine, USA
Dr Cuixia Tian reviewed the bone health protocol at Cincinnati Children's for individuals with DMD. Since 2010, spine radiographs have been routinely performed to detect mild and/or asymptomatic vertebral fractures, prompting early intervention with oral bisphosphonates. As practices have continued to evolve, intravenous bisphosphonates are also used in patients who develop moderate to severe vertebral or long bone fractures.
Dr Tian discussed dual-energy x-ray absorptiometry (DXA) findings from a retrospective observational study of oral bisphosphonate (alendronate) therapy in 54 patients with DMD on long term GC treatment. 29 When started at the earliest stages of osteoporosis (mild vertebral height loss), oral bisphosphonate therapy was associated with improvement in the pre- versus post-treatment bone mineral density (BMD) Z-score trajectories at the lumbar spine and lateral distal femur (region 1) in the first three years of therapy (but not in the longer term, from three to six years following initiation).
Dr Nat Nasomyont went on to summarize vertebral morphometry and fracture outcomes with oral alendronate. In a retrospective observational study of 52 patients with DMD on long term GC treatment treated with alendronate for five years, 30 the prevalence and severity (by Genant criteria) of most vertebral fractures remained stable. While about half (54%) of the patients were observed to have worsening fracture severity grading in at least one vertebra, a similar number (46%) were observed to have improvement in at least one vertebra. 30 Among those with improvement, 8/24 (33%) had complete vertebral body reshaping, defined as normalization of vertebral dimensions in those with residual growth potential. Based on the experience from Cincinnati Children's, oral bisphosphonate treatment has been well-tolerated, with gastrointestinal side effects occurring very uncommonly.
Dr Nasomyont reviewed Cincinnati Children's experience with teriparatide treatment in six patients with GC-treated DMD and severe osteoporosis (despite long-term oral bisphosphonate therapy in five out of six patients). 31 After two years of teriparatide in patients aged 13.9 to 22.1 (mean 17.9) years at initiation, stable vertebral dimensions of L1 to L4, stable or improved fracture severity grading of most vertebrae (T9 to L4), and fewer long bone fractures were observed, with modest increases in procollagen 1 intact N-terminal propeptide (a bone formation marker); however, multi-site areal BMD Z-scores (not adjusted for height due to non-ambulatory status) showed continuing decline related to the baseline trajectory. The lack of a positive effect on BMD and the attenuated treatment response relative to adults with GC-induced osteoporosis but without DMD was felt to be testament to the magnitude of the GC exposure and the aggressiveness of the osteoporosis in patients with DMD, and suggested that teriparatide may not fully over-ride the potent osteotoxicity of GC-treated DMD. From a safety standpoint, teriparatide treatment was well-tolerated with no significant adverse effects. It was noted, importantly, that the black box warning against teriparatide with respect to the agent's use in children was lifted by the FDA in 2020; 32 long-term safety data in children with hypoparathyroidism is also reassuring against theoretical concerns regarding osteosarcoma. 32 Dr Nasomyont concluded that although teriparatide did not stabilize BMD Z-scores, the observation of stable bone health from a fracture standpoint and the safety profile of this agent over two years provides rationale for study of its effect on a larger number of GC-treated patients with DMD.
Drs. Tian and Nasomyont further concluded that oral bisphosphonates may have a role in mitigating the development and progression of vertebral fractures and preventing spine BMD Z-score declines when initiated in the sub-clinical phase; for example, prior to first fracture or when there are minimal reductions in vertebral height ratios that do not meet the criteria for a grade 1 (mild) vertebral fracture (i.e., primary prevention). Teriparatide may be an effective bone anabolic treatment option for severe osteoporosis in patients with DMD. The role of oral bisphosphonates and teriparatide for primary and/or secondary prevention of osteoporosis should be further investigated in GC-treated individuals with DMD.
Discussions about oral bisphosphonate therapy took place on a background of existing knowledge about bisphosphonates in general to treat children with and at risk for osteoporosis, including those with DMD. First, it is important to recognize that bisphosphonates remain off-label for use in children with osteoporosis in most countries, with few exceptions. Secondly, similar to adults, the bioavailability of oral bisphosphonates is less than 1%, an observation that has been demonstrated in both pediatric osteogenesis imperfecta 33 and in GC-treated children. 34 The greater potency of intravenous (IV) bisphosphonate therapy, along with the cumulative evidence supporting the use of IV bisphosphonates across myriad primary and secondary osteoporotic conditions of childhood, have led to IV bisphosphonate therapy being recommended first-line for the treatment of overt bone fragility in those with persistent risk factors and limited potential for recovery (such as children with fragility fractures and DMD). 2
Evidence for the use of IV bisphosphonates in DMD is now available, arising from two randomized controlled trials of intravenous zoledronic acid that included pediatric patients with DMD.35,36 Both trials showed that intravenous zoledronic acid was associated with significant increase in DXA-based lumbar spine BMD Z-scores. Zacharin et al. also showed an increase in radial trabecular volumetric BMD by peripheral quantitative computed tomography on zoledronic acid plus nutritional support versus nutritional support alone. 36 Data from the Zacharin et al. study also suggested that zoledronic acid may have a role in preventing incident vertebral fractures, with 24% of control subjects developing incident vertebral fractures over the 24-month duration of the study compared to 15% in the zoledronic acid cohort. Further, five of 31 children in the control arm withdrew from the study due to incident (new) grade 3 (severe) vertebral fractures compared to none with severe incident vertebral fractures in the zoledronic acid cohort. 36 Overall, there is now high-quality evidence to support the use of IV bisphosphonate therapy for the treatment of GC-associated osteoporosis in DMD, targeting increases in areal BMD Z-scores at the lumbar spine. 37
Predictors of bone morbidity in DMD
Kim Phung, Children's Hospital of Eastern Ontario, Canada
Dr Kim Phung discussed predictors of vertebral and non-vertebral fractures in DMD. Studies have shown that bone morbidity in DMD is linked to declining muscle function and increased GC exposure. Crabtree and colleagues have shown in a longitudinal observational study of 27 pediatric patients with GC-treated DMD that trabecular volumetric BMD by peripheral computed tomography was lower in patients with DMD at baseline compared to healthy controls, and declined further with loss of ambulation. 38 In 292 ambulatory children with GC-treated DMD, Tian et al. showed that total body (less head) and proximal femur areal BMD by DXA decreased, and vertebral fractures increased, with worsening functional mobility scores (highlighting the association between bone health indices and motor function in DMD). 39 A retrospective study by James et al. has also identified wheel-chair use as a risk factor for fractures in patients with dystrophinopathies. 40 The relationship between fractures and GC exposure was highlighted in a study by Joseph et al., where the likelihood of a clinical fracture was highest on daily oral deflazacort, followed by daily oral prednisone and then intermittent GC regimens. 41
Dr Phung presented her group's data on risk factors associated with vertebral fractures in DMD. In a 12-month prospective bi-center study following 60 GC-treated males with DMD (mean age: 11.5 years, range 5.4–19.5 years) who were bisphosphonate-naïve, baseline (prevalent) vertebral fractures after 4.5 years of GC exposure were observed in 32% of patients. 42 Increased spine fracture burden, as measured by the Spinal Deformity Index (SDI), the sum of the Genant vertebral fracture grades from T4 to L4, 43 was associated with markers of systemic GC exposure, including lower lumbar spine BMD Z-scores, delayed bone ages, shorter stature, and increased weight. 42 In this cohort, 24% of patients (9/38) sustained at least one incident vertebral fracture after 12 months of follow-up; this was likely an under-estimate of the total vertebral fracture incidence, since 12/60 patients (20%) were withdrawn from the study after the baseline visit due to painful vertebral fractures necessitating bone protection therapy. 44 The presence of clinically significant fragility fractures (vertebral and/or non-vertebral) at baseline were the strongest predictor of worsening spine fracture burden over 12 months in this cohort. Delayed skeletal maturation, assessed by bone age, was also a predictor of incident vertebral fracture.
The observation that the presence of any prior fracture was the strongest predictor of new vertebral fractures underlines the need for primary osteoporosis prevention efforts in DMD. It is logical that strategies to guide primary prevention of first-ever fractures in DMD may rely on risk factors associated with fractures in DMD, including markers of systemic GC exposure and objective measures of motor function decline, to assist clinicians in identifying who to prioritize for early initiation of bone protective therapies.
Prevention of first fractures in DMD: where are we now?
Leanne Ward, Children's Hospital of Eastern Ontario, University of Ottawa, Canada
The approach to the management of bone fragility due to osteoporosis in DMD has evolved considerably since the DMD Care Consideration published in 2010.
45
In the 2010 guidance, spine x-rays were recommended to detect vertebral fractures if back pain or kyphoscoliosis were present, followed by initiation of intravenous bisphosphonate therapy if vertebral fractures were present. In the years following the inaugural 2010 DMD Care Considerations, seminal studies by the Canadian
With this knowledge, the latest iteration of the international standards of care for DMD published in 2018, known as the 2018 DMD Care Considerations as previously discussed, 2 recommended routine, standardized spine imaging for early detection of vertebral fractures (according to the Genant semi-quantitative method), combined with early (rather than late) initiation of bone protection therapy in the presence of even a single low-trauma vertebral or long bone fracture. 2 As such, the 2018 guidance was predicated upon routine screening to detect both symptomatic and asymptomatic vertebral fractures in their earlier stages followed by IV bisphosphonate therapy to prevent osteoporosis progression (a more aggressive approach than screening for vertebral fractures only in the presence of back pain, and treating only in the presence of painful vertebral fractures or kyphosis as per the 2010 guidance). 45 The decision to recommend a more pro-active standard of care in 2018 was based on principles of early identification and prevention of progression, and the evidence that was available to support this change at the time.2,53 In addition, conservative measures to optimize bone strength, including diagnosis and treatment of vitamin D deficiency, were also encouraged (with annual assessment of calcium and vitamin D intake plus an annual serum 25-hydroxyvitamin D measurement).
While the 2018 DMD Care Considerations were more pro-active than the 2010 guidance, there has been concern about waiting for a first fracture to start IV bisphosphonate therapy, instead of taking steps to prevent first fractures and their associated complications in such a high-risk setting. As a result, there has been interest in anticipatory measures to prevent a “first-ever” fracture. Dr Ward reviewed different approaches that could be taken to prevent first fractures, including broad-strokes strategies that include initiating bone protection therapy at the time of diagnosis, or no later than GC initiation, in everyone with a confirmed diagnosis. However, there was concern among delegates that such an approach may be challenging to fund and/or implement for logistical reasons, given the larger volume of patients needing treatment under this paradigm as compared to the fracture-triggered approach. Dr Ward also presented a more tailored, risk-stratified strategy, one that is based on identifying children pre-fracture who were nevertheless prioritized as more (or less) likely to sustain a fracture than others. This strategy is based on the premise that the likelihood of a fragility fracture in GC-treated DMD is determined by two main driving factors: GC exposure, and progressive loss of ambulation.
Capitalizing on these themes, Dr Ward discussed the concept of generating a
The delegates concluded that it is indeed high time, if not long overdue, to implement an even more pro-active fracture prevention strategy, one that ultimately seeks to prevent BMD declines as early in the course of the disease as possible in an effort to prevent first-ever fractures. Discussions ensued around the optimal timing for early, anticipatory intervention (with sensitivity to the logistical burden to families of such an approach) and the choice of agent (including intravenous versus oral bisphosphonate therapy). It was decided that further, in-depth discussion would be needed to weigh the benefits and burden of different therapeutic choices in the sub-clinical phase of an osteoporosis prevention care pathway, and that this aspect would be debated during future discussions. Figure 2 highlights the shifting standard of care for the management of osteoporosis in DMD since the original 2010 guidelines, highlighting that intervention is moving towards an increasingly pro-active approach to fracture prevention.

Highlights the shifting standard of care for the management of osteoporosis in DMD since the 2010 international “Care Considerations” by showing that intervention is moving towards an increasingly pro-active approach in childhood to fracture prevention.
The impact of testosterone therapy on muscle-bone health in DMD
Claire Wood MD, Great North Children's Hospital and Newcastle University, United Kingdom
Dr Claire Wood discussed that pubertal delay is almost universal in young people with DMD on daily GC therapy. Exogenous androgen is increasingly being used for pubertal induction in adolescents with DMD, and indeed is recommended as part of the 2018 DMD Care Considerations to start in those who are over 14 years of age (i.e., in those meeting the definition of delayed puberty). 3 Further guidance as to agents and dosing has been provided in a companion paper to the 2018 DMD Care Considerations. 53 Outstanding questions include the duration of therapy and the frequency of need to re-initiate testosterone when there are signs of endogenous puberty following pubertal induction.
Exogenous androgen can have an anabolic effect on a range of different tissues and may mitigate some of the deleterious effects of GC on muscle, bone and psychological well-being. Dr Wood's group recently undertook a controlled study where 15 boys with DMD received a 2-year incremental regimen of 4-weekly intramuscular testosterone isocaproate injections. 55 Contractile surface area in the combined muscle groups of arms and legs measured by muscle MRI significantly increased over the study period and the fat fraction remained unchanged, in contrast to natural history study data which suggested that an increase in fat fraction would have been expected. 56 Data from the same study showed an increase in quality of life in a majority of participants during pubertal induction, as assessed by the Neuromuscular module of the PedsQOL. 57 Quality of life data from a meta-analysis on testosterone replacement treatment in hypogonadal men versus placebo evaluated 23 randomized controlled trials, involving 3090 participants. Compared with placebo, treatment with any testosterone replacement significantly improved quality of life. 58
BMD also remained stable in the controlled study, suggesting a possible protective effect of androgen therapy on bone despite ongoing GC therapy. Similarly, in a case-controlled study of 36 adult men with hypogonadism, bioimpedance and peripheral quantitative computed tomography were used to demonstrate that those receiving testosterone enanthate had decreased subcutaneous fat, increased lean muscle mass, reduced bone remodeling and greater trabecular bone density compared to their age-matched controls. 59 A retrospective observational study of 16 individuals with DMD who underwent pubertal induction with oral testosterone undecanoate showed a statistically significant increase in BMD at the lumbar spine, as well as no progression in vertebral fractures during the study period in most individuals. 60 Ten of 14 patients who were assessed had no progression in vertebral fractures. Standard deviation scores for fat mass index and lean body mass index, and percentage change in these two indices, were statistically unchanged; in addition, cardiac function remained stable. Motor function in non-ambulatory individuals with Egen Klassifikation scores improved in 7 of 8. Together, these results suggested some degree of bone protection at the spine from testosterone therapy in the absence of deleterious effects on the muscle strength. 60
The longer-term (unpublished) data from the recently published study by Dr Wood's group, 55 however, did not demonstrate a sustained improvement in muscle mass in the 3 years after testosterone treatment was stopped and a significant decrease in BMD was observed during the prolonged follow-up period. Testosterone levels also fell to levels prompting long-term supplementation in 6/15 participants. This suggests that pubertal induction promotes gonadotrophin release but is not always sustained so it is important to continue monitoring testosterone levels and supplement as required. Dr Wood recommended considering prompt induction of puberty in DMD provided that the young person is psychologically ready; ideally around 12 to 13 years of age.
Puberty and awkward discussions in DMD
Leanne Ward, Children's Hospital of Eastern Ontario, University of Ottawa, Canada
Dr Wood's talk provided a segue for Dr Ward to further expand on the sensitive nature of initiating discussions about pubertal delay and examining patients for signs of physical maturation. Dr Ward highlighted the importance of an established, collaborative relationship with the pediatric patient, one that is ideally in place before discussions about pubertal delay are initiated (hence the importance of referring to an endocrinologist early, which is needed for monitoring other aspects of endocrine-bone health in any case, as discussed throughout this report). While discussions about pubertal delay can be initiated around 10 years of age, it is recognized that some individuals may be cognitively, but not emotionally, ready to discuss puberty. In addition, other individuals with DMD may be neither cognitively nor emotionally ready at this time. It is the physician's responsibility to gauge the patient's understanding and readiness, to avoid assumptions about readiness without exploration, and to engage in a conversation with the youth periodically in private, without the caregivers present, to accurately understand the depth of the individual's knowledge, concerns and feelings about puberty and its delay. Similarly, for patients who decide to proceed with pubertal induction (the majority, in the delegates’ experience), physicians need to assess not only the patients’ responses to treatment (signs and symptoms of virilization and evidence of endogenous pubertal activation, including libido and erections), but their emotional responses to the biophysical changes, and the impact of these feelings on their personal relationships.
While most patients choose pubertal induction and feel positively about the treatment (as reported in Dr Wood's study), 55 Dr Ward noted that some patients are cognitively able to understand the information related to pubertal induction but do not wish to embrace this approach. To illustrate this point, Dr Ward presented the case of a 19-year-old male with DMD who was followed in a tertiary care endocrinology-osteology clinic for endocrine and bone health care over many years. The topic of pubertal induction was embarked upon in an age-appropriate fashion, and the individual expressed an understanding of the information. The individual, who was remarkably still ambulatory at 19 years of age (with distinguishing features that included a height age of 6 years, lack of excessive weight gain, and a markedly delayed bone age), noted that he was comfortable with his pre-pubertal status and short stature, and was reluctant to “rock the boat” since he was in exceptional health for his chronological age.
When asked, he noted positive experiences with friends and acquaintances, and although he preferred to be taller, he stated that he accepted his GC-induced short stature in the interest of preserved muscle strength. On careful review of his bone health phenotype (which included bone protection (bisphosphonate) therapy at the first sign of minimal vertebral collapse), he had low but higher-than-baseline lumbar spine and hip BMD Z-score trajectories, and absence of new (incident) vertebral fractures since starting bisphosphonate therapy. Since it is presently unclear whether testosterone has a significant effect on skeletal strength beyond that which is provided by timely intervention with anti-resorptive therapy, or conversely, whether there are considerable disadvantages to not receiving testosterone in the DMD setting (not to mention the over-riding need to respect the patient's wishes), this patient has remained without testosterone therapy at 19 years of age despite Tanner stage 1 pubertal development (with the support of his physician).
Going forward, it was acknowledged by the delegates that there needs to be greater emphasis on speaking to teens about pubertal induction (and sexuality, see later) privately in an age- and developmental-stage appropriate manner during routine clinical care. In addition, there was consensus that considerable questions remain with respect to optimal timing of testosterone intervention, the psychosocial burden of pubertal delay (and short stature), and the impact of pubertal induction on the individual's overall physical and emotional health and well-being.
Sexuality, fertility, and gender identity in DMD
Nora Renthal, Boston Children's Hospital and Harvard Medical School, USA
Comprehensive care for patients with DMD includes discussions around sexual wellness, fertility, and gender identity. 4 It is crucial for healthcare providers to initiate conversations about sexual health and development, at age-appropriate levels, starting from the typical age of puberty when patients are entering or observing their peers starting this maturational phase (average age 11–13 years). It is recommended that patients be offered the opportunity to be interviewed without their parent(s)/caregiver(s) present to ensure open communication and confidentiality.
Like all adolescents, patients with DMD have the right to privacy, comprehensive sexual education, and the freedom to express their gender identity, yet patients with physical disabilities are reported to feel infantilized and desexualized by their caregivers and medical team. 61 Healthcare providers should discuss internet safety, including online dating and pornography consumption. Providers should assess the extent to which patients have received sexual education from parents, school, and their primary care providers, supplementing with additional resources if deficits are identified, including recommendations for books and online resources from organizations such as PPMD.
In addition, patients should be assessed for their desire for parenthood in an age-appropriate manner. In general, these conversations are suggested to occur earlier in adolescence, at the time of spontaneous puberty or with induction of puberty, as considerations for testosterone are discussed with patients and families. A multidisciplinary approach including genetic counseling is recommended when possible.
As part of a comprehensive adolescent evaluation, it is crucial for providers to assess and address gender identity concerns in patients with DMD. As the incidence of adolescents identifying on the gender spectrum continues to rise, 62 we must ensure patients with DMD receive appropriate support and care for their mental health and well-being.
Overall, patients with DMD should be engaged in discussions around sexual wellness, fertility, and gender identity at age- and readiness-appropriate times, respecting the patient's right to privacy and ensuring they have access to appropriate resources. In addition to evaluation by an endocrinologist, a multidisciplinary approach involving social work, psychology, and adolescent medicine should be taken whenever possible. The evolution of a child with DMD to physical maturity and adulthood can be a challenging and emotional time for parents and caregivers. While some families may experience feelings of pride and accomplishment, others may experience grief and anxiety. 63 Therefore, it is important to provide sensitivity and resources to families during this time, acknowledging and addressing their complex emotions and needs. Nonetheless, ignoring the sexual and reproductive health of patient with DMD is a form of ableism that perpetuates health disparities and disproportionately affects minoritized communities. 64 The absence of such conversations, indeed, perpetuates stigma, which has been linked to poor quality of life. 65 More research is needed to understand and address the unique challenges faced by individuals with DMD.
The impact of rhGH/IGF-1 on muscle-bone health in DMD
Meilan Rutter, Cincinnati Children's Hospital Medical Center and University of Cincinnati, USA
Dr Meilan Rutter reviewed recombinant human growth hormone (rhGH) and insulin-like growth factor-1 (rhIGF-1) therapies and discussed clinical considerations regarding growth concerns in patients with DMD. While endocrine, and overall interdisciplinary, care for patients with DMD continues to evolve,3,66 GC-induced growth failure remains prevalent and a concern for many.
Dr Rutter summarized the Cincinnati team's experience treating 39 patients with DMD and GC-induced growth failure with rhGH for one year. 67 rhGH improved height velocity from 1.3 ± 0.2 to 5.2 ± 0.4 cm/year and stabilized height standard deviation score. Three patients had side effects, including transient intracranial hypertension (one patient), insulin resistance and impaired glucose tolerance. No detrimental effects on neuromuscular or cardiopulmonary function were observed.
The Cincinnati team also conducted a six-month randomized controlled trial of rhIGF-1 therapy in 44 prepubertal, ambulatory, GC-treated patients with DMD. 68 rhIGF-1 is a growth factor that is anabolic to muscle; 69 in pre-clinical studies, targeted expression of IGF-I or systemic administration of rhIGF-1 improved muscle regeneration and function, and prevented muscle cell death in the mdx mouse.70,71 rhIGF-1 treatment did not change motor function outcomes, but improved growth velocity (6.5 ± 1.7 versus 3.3 ± 1.3 cm/year; annualized height standard deviation score gain 0.5/year). Importantly, motor function remained stable in the setting of increased growth, with no relationship between height gain and motor function on post-hoc analyses, consistent with observations by others.72,73 rhIGF-1 improved insulin sensitivity, did not affect bone density, and increased lean mass (preliminary evidence of a pharmacodynamic response in DMD muscle). Two patients experienced transient asymptomatic papilledema and intracranial hypertension, respectively.
Dr Rutter emphasized that growth and related interventions should be integrated within the context of interdisciplinary team care. For example, appendicular lean mass, a surrogate measure of muscle strength, is affected by growth and puberty. 74 Height gain could positively impact vertebral fracture reshaping potential, illustrated by a patient who had received rhGH and oral alendronate therapies for 5 years. 30 At the same time, it was noted that there are insufficient short- and long-term data on rhGH and rhIGF-1 in patients with DMD to recommend these therapies as part of the standard of care. 3
Finally, when considering clinical care and communication regarding these issues, Dr Rutter advocated for a proactive approach (including counseling and psychological intervention),8,26 balanced with addressing concerns by informed shared decision-making around viable options. 75 Dr Rutter noted that vamorolone may be an option for some patients; in the open label and randomized, controlled trials of vamorolone in young, ambulatory boys with DMD, the deleterious effect on linear growth that is seen with classic GC therapy was not observed on vamorolone.8,26,27 Care should also be holistic, as expressed by a family living with DMD, “…growth issues are not just physical ones, but emotional and psychological as well.”
Classic steroid therapy and the adrenal axis
David Weber, Children's Hospital of Philadelphia, University of Pennsylvania Perelman School of Medicine, USA
Dr Weber presented a case of a 12-year-old male with DMD on daily deflazacort who presented to the emergency department following his first zoledronic acid infusion with symptoms of adrenal crisis triggered by the well-known, zoledronic acid “acute phase reaction”. Prednisone and deflazacort both cause central adrenal suppression at the doses used to treat DMD. The pharmacokinetics differ, with deflazacort being less potent and having a shorter half-life (1.3 vs 3–4 h) compared to prednisone. 76 Patients treated with deflazacort therefore may be more susceptible to adrenal crisis during stress, although there are no data reporting the incidence of adrenal crisis in relationship to GC type, GC dose or age among people with DMD. The 2018 DMD Care Considerations 3 and other papers,77,78 including the PJ Nicholoff Adrenal Suppression Management Protocol, 77 outline an approach to preventing adrenal crisis that includes patient education regarding the importance of adherence to daily GC administration, a slow taper if GC therapy is stopped, and the appropriate activation of a stress steroid plan.
At the workshop, participants discussed different approaches to implementing stress steroid plans. Some prescribe oral hydrocortisone for moderate stress, while others instruct patients to split or increase the regular prednisone or deflazacort dose to twice daily. Intramuscular hydrocortisone for home use was discussed. If used correctly, hydrocortisone intra-muscular injection can reverse an adrenal crisis, obviating the need in some cases for the individual to present to hospital for steroid stress dosing management. However, it is challenging to administer and can be difficult to obtain due to insurance and availability issues. Some centers prescribe Solu-Cortef on a case-by-case basis, to families willing to administer it and/or to those with difficulty accessing immediate emergency care. Other strategies to reduce risk of adrenal crisis include adding adrenal insufficiency to the electronic health record problem list, ensuring that families have a stress steroid letter for health professionals, and encouraging the use of a “medical alert bracelet” or similar alert for paramedics and other emergency personnel.
Overall, it was identified that adrenal insufficiency awareness and appropriate management remain an unmet need among patients receiving classic (and dissociative) steroid therapy. The workshop attendees identified the importance of future studies that seek to precisely capture the frequency with which a. patients present to medical attention with signs and symptoms of adrenal insufficiency, b. adrenal insufficiency is unidentified, and c. adverse events arise from both diagnosed and undiagnosed adrenal insufficiency. It was also recognized that such accounting is challenging, since clinical signs and symptoms of adrenal insufficiency are non-specific and overlap with features of the underlying disease (e.g., weakness and fatigue). On the other hand, the rapid reversal of adrenal insufficiency manifestations following “steroid stress dosing” (e.g., within 30 to 60 min) provides a “therapeutic test” that is helpful to clinicians. Given the lack of precise information on the frequency of adrenal insufficiency symptoms, crises and the need for steroid stress dosing in DMD, we have proposed that this is an area for future research, along with campaigns to raise awareness about this potentially life-threatening complication, a known cause of premature and unexpected mortality in DMD. 1
Dissociative steroid and the adrenal axis
Leanne Ward, Children's Hospital of Eastern Ontario, University of Ottawa, Canada
Dr Ward reviewed the evidence from the randomized, controlled trial of vamorolone 2 mg/kg and 6 mg/kg daily versus daily prednisone and placebo over 24 weeks in young, ambulatory children with DMD in terms of adrenal insufficiency.8,79 Dr Ward noted that unequivocal evidence for suppression was documented on prednisone and vamorolone, both based on first morning cortisol values at 12 and 24 weeks and based on standard dose ACTH stimulation testing at 24 weeks. 8 While vamorolone 2 mg/kg daily demonstrated less of an effect on the hypothalamic-pituitary axis when compared with daily prednisone or vamorolone 6 mg/kg daily (i.e., the degree of adrenal suppression was dose-dependent), it was nevertheless agreed upon by delegates that all patients on vamorolone should be considered at risk of adrenal suppression given the life-threatening nature of this complication.
Because vamorolone is associated with adrenal suppression in a dose-dependent manner and given the life-threatening nature of adrenal suppression in any disease context, Dr Ward reviewed fundamental principles for adrenal suppression management in vamorolone-treated patients, with the goal to ensure patient safety. These principles have been distilled down into five key concepts for clinicians.
The first concept is that patients on classic GC therapy should be switched to vamorolone 6 mg/kg daily and only this dose (i.e., not a lower dose). This is for two reasons. First, to maintain treatment efficacy at the muscle strength level relative to prednisone 0.75 mg/kg daily, since only vamorolone 6 mg/kg daily showed efficacy parity for muscle strength relative to oral daily prednisone at standard DMD-treatment doses. The second reason patients on classic GC therapy should be switched to vamorolone 6 mg/kg daily and only this dose is to avoid adrenal insufficiency, since all patients on long-term GC therapy are assumed to have adrenal insufficiency irrespective of dose. Therefore, patients should be switched from classic daily GC to vamorolone 6 mg/kg daily to avoid signs and symptoms of adrenal suppression during the switch from one to the other therapy. Patients should be monitored for signs and symptoms of adrenal suppression even when changing to vamorolone 6 mg/kg daily, until such time as more information is available about patients’ symptomatology during the switch to full dose vamorolone.
The second concept is that if at any time, a patient who is switching from classic steroids to vamorolone 6 mg/kg daily, or who is tapering the vamorolone dose for any reason, has symptoms that could represent adrenal insufficiency (such as fatigue, headache, abdominal pain, nausea, weakness), they should be presumed to have adrenal insufficiency and treated with classic GC stress dosing accordingly. This is the safest course of action, and therefore considered by the delegates to be in the best interest of the patient.
The third important concept was that clinicians are advised not to use vamorolone for steroid stress dosing, for two reasons. First, there is no available experience with vamorolone doses higher than 6 mg/kg daily in children and no experience with vamorolone as a stress dose agent. Secondly, vamorolone is antagonistic to the mineralocorticoid receptor; this means that vamorolone may not have the tensive effect of classic (mineralocorticoid-containing) GC therapy in an adrenal crisis. Therefore, patients on vamorolone who require stress dosing should be treated with stress doses of hydrocortisone in the usual manner, or prednisone if hydrocortisone is not available.
The fourth concept was that care must be taken to avoid symptoms of steroid withdrawal and adrenal insufficiency when reducing the dose of, or discontinuing, vamorolone. Because vamorolone causes adrenal suppression, gradual tapering is required for any patient reducing the dose of, or discontinuing, vamorolone (similar to the approach when reducing or discontinuing classic GC therapy).
The fifth important concept in adrenal suppression management is that if a patient switches from vamorolone to classic GC therapy, the patient should receive full DMD treatment doses of prednisone (0.75 mg/kg/day) or deflazacort (0.9 mg/kg/day) due to the adrenal suppressive effects of vamorolone (in order to prevent adrenal suppression side effects).
Finally, the delegates discussed that going forward, it will be crucial to communicate with health care professionals and patients that adrenal insufficiency signs and symptoms are “the great mimickers” of other diseases (given their frequency in the general population, and lack of specificity for adrenal insufficiency per se including fatigue, weakness, headache, nausea, vomiting and abdominal pain, among others). One could argue that this is particularly problematic in those with DMD, since fatigue and weakness are germane to the condition, catapulting the importance of adrenal insufficiency awareness even more to the fore.
It is for this very reason that decision-making around the interpretation of possible signs and symptoms of adrenal insufficiency must always err on the side of caution, with a low threshold to activate steroid stress dosing protocols.53,77 These principles were recently summarized in a slogan contest sponsored by Defeat Duchenne Canada. The winner's catchy jingle, developed by a member of the Defeat Duchenne Canada community and shown in Figure 3, captured the spirit behind adrenal suppression management – for decision-making, always err on the side of caution, period.

Shows a catchy jingle developed by a member of the Defeat Duchenne Canada community that was presented at the meeting, and that captures the spirit behind adrenal suppression management – always err decision-making on the side of caution, period (found at Emergency Care – Defeat Duchenne Canada, last accessed 7 November 2024).
Weight management in DMD
Phil Zeitler, Children's Hospital Colorado, USA
Excessive weight gain and obesity are common problems in DMD and other neuromuscular disorders, due to a combination of GC therapy, reduced energy expenditure, and increased caloric intake associated with limited activity options. 53 This excess adiposity is often accompanied by metabolic disturbances, such as diabetes, hypertension, and dyslipidemia. Historically, excessive weight gain in individuals with DMD has been addressed through nutritional interventions to reduce calorie and carbohydrate intake, though these have generally been met with limited success. Significant changes were made to the American Academy of Pediatrics guidelines for the evaluation and management of obesity in 2023 (for children in general, not specific to DMD) that promote recognition of obesity as a chronic problem and encourage earlier use of more intensive interventions rather than relying solely on lifestyle changes that have been repeatedly shown to lack effectiveness in youth. 80
The update to these guidelines and support for more aggressive interventions provide an opportunity to reconsider the approach to management of excess adiposity in medically complex individuals. The rapidly expanding pharmacologic options for effective management of excess adiposity and the ongoing approval of these medications for youth, along with increasing understanding of their efficacy and side effects, provide an opportunity to consider which, if any, treatments may be optimal for individuals with neuromuscular disorders. This may require a change of perspective on the part of both families and providers regarding the relative risks and benefits of pharmacologic treatment versus leaving excess adiposity untreated in medically complex children, adolescents, and young adults, such as those living with DMD.
Strategies for implementing endocrine-bone health management nation-wide: the UK experience
Sze Choong (Jarod) Wong, Royal Hospital for Children, University of Glasgow, UK
Dr Jarod Wong presented a national project, DMD Care UK (www.dmdcareuk.org) that aims to implement the 2018 DMD Care Considerations2–4 in the UK national health service system. The project is a joint initiative between Newcastle University, Duchenne UK and clinicians in the UK NorthStar network.
Following publication of the 2018 Care Considerations for DMD, it was noted that there was variability in the delivery of all aspects of health care for people with DMD in the UK, with myriad potential reasons for this observation. A survey of practices among the UK NorthStar sites (2019) revealed that 21% of sites were not performing routine spine imaging for vertebral fracture assessment and 21% do not treat fractures with bisphosphonate therapy, despite the 2018 DMD Care Considerations. A UK-wide patient survey in 2020 identified that 32% of GC-treated patients had an oral steroid sick day plan, with five different plans prescribed clinically for those on daily GC and eight different plans prescribed clinically for those on intermittent GC. Only 46% of patients had access to hydrocortisone for injection out of the hospital for severe illness.
In an attempt to address this variability and in so doing reduce health inequities, a UK guidance for endocrine-bone monitoring and management based on the 2018 Care Considerations was formally launched in 2020. The guidance was developed by the bone and endocrine working group of DMD Care UK, with official endorsement by the British Society of Paediatric Endocrinology and Diabetes (BSPED), and included detailed guidance on oral stress dosing sick day plans. Other outputs include patient information leaflets on delayed puberty, adrenal insufficiency, a DMD steroid-dependent bracelet, and an “in case of emergency application” tool developed by the Emergency Working Group. An example of early success arising from this project was shown by a recent patient survey (2021), where 81% of parents/carers and patients stated they have received stress steroid guidance for sick day management. For the next phase of this working group, the development of an online educational platform targeting adrenal insufficiency management for families, patients, carers and new clinical staff will be prioritised. The project will also align its adrenal insufficiency advice with the first national paediatric emergency adrenal insufficiency guidance that was launched in 2022. 81 Infographic videos on the need for assessment of puberty and expected changes with testosterone therapy addressing issues like emotional changes will also be developed.
Despite being only three years into the project, with collaboration between clinicians, patients and DMD Care UK, the group has developed strategies to address implementation of care at a national level by agreeing on care recommendations, delivering education to patients, families, and clinicians, and developing resources to help implement care. Future ambitions include developing strategies to address endocrine and bone health issues even more pro-actively (such as primary osteoporosis prevention), linking closely with international colleagues on such matters.
The impact of early glucocorticoid initiation on endocrine & bone health: sightline to the future
David Weber, Children's Hospital of Philadelphia, University of Pennsylvania Perelman School of Medicine, USA
The average age at diagnosis (4.4 years) and the time from symptom onset to diagnosis (2.2 years) in DMD has not changed in recent decades. 82 This fact, coupled with the FDA approval of the creatine kinase (CK)-MM blood spot assay, have re-invigorated the push to implement DMD newborn screening with the hope that earlier diagnosis and initiation of disease-modifying therapies will improve outcomes. 83 With earlier diagnosis comes the potential for GC initiation at younger ages. Infancy and early childhood are times of rapid growth and bone accrual; 84 with this in mind, it is expected that earlier GC initiation will amplify height and bone mass reductions and cause earlier fractures. However, there is limited data regarding the use of long-term, high dose GC in infants and young children with DMD or other conditions. Kim et al. reported a higher risk of a fracture when GC were initiated prior to five years of age in DMD. 85 High dose dexamethasone treatment for 21 days was given to premature infants in the past. Small studies have shown that these infants had slower growth on treatment, did not catch up to untreated individuals by 50 months, 86 and had lower lumbar spine BMD at 5–8 years of age. 87 A recent, one-year study by Connolly, et al., found that infants with DMD treated with twice weekly prednisolone demonstrated improved gross motor function without an adverse effect on growth. 88 Currently, GC therapy is given for eight weeks following gene therapy, and while exon-skipping therapies do not necessitate GC therapy per se, all exon-skipping studies to date have added exon-skipping therapy to GC treatment.
The challenges of bone health monitoring in young children were discussed. Reference data for DXA in young children exist,89,90 but are not widely available for clinical use. Some centers use BoneXpert, which provides a “Bone Health Index assessment” from a hand radiograph (scored by measuring the length, width and cortical thickness of the three middle metacarpal bones); this automatic surrogate for bone strength may be simpler to obtain in young children, 91 one that capitalizes on an x-ray evaluation that is already obtained as part of routine clinical care.
The path forward: clinical care, research & advocacy
Leanne Ward, Children's Hospital of Eastern Ontario, University of Ottawa, Canada; Pat Furlong, PPMD, USA
Pat Furlong and Dr Ward led a group discussion to wrap up the meeting proceedings. Together, the group decided that there was sufficient need for ongoing clinical care pathway development, for addressing research questions, and for furthering patient education and advocacy in DMD endocrine and bone care that the post-Rome meeting dialogue should continue. The group further agreed that the post-Rome narrative should proceed collaboratively among the individuals in attendance at the meeting, in partnership with Parent Project Muscular Dystrophy and other international DMD patient advocacy groups (including, but not limited to, Duchenne Parent Project Italy, DMD Care UK and Defeat Duchenne Canada, among others). To achieve these goals, it was agreed that a DMD endocrine-bone consortium of working groups would be established. Goals of the working groups would be to develop an endocrine- and bone-specific patient registry, the goal of which would be to better understand the endocrine and bone co-morbidities in DMD on a background of different muscle-targeted therapeutic regimens. In addition, the DMD endocrine-bone consortium of working groups plans to develop collaborative clinical care pathways, identify synergies to enable research initiatives that answer critical questions, and create patient and health professional educational materials that address the contemporary issues opined by the delegates and speakers in this report.
With GC regimens ensconced in the DMD therapeutic landscape for the foreseeable future, efforts to optimally manage their co-morbidities are spurred, embraced, and enabled by the DMD community, for which the network of health professionals working in this domain are immensely grateful. In October 2023, a follow-up meeting sponsored by Parent Project Muscular Dystrophy took place in Ottawa, Canada and the DMD endocrine-bone consortium was officially labeled the international “OPTIMIZE DMD Consortium”. At the October 2023 Ottawa OPTIMIZE DMD meeting, concrete plans were laid to move forward with the goals identified at the Rome 2023 meeting in the areas of clinical care, advocacy, education, and research. An OPTIMIZE DMD Steering Committee plus five Working Groups in the areas of Osteoporosis Prevention, Sexuality and Fertility, Growth and Puberty, Weight Management and Adrenal Insufficiency (including Parent Project Muscular Dystrophy representatives) have been struck, and the International OPTIMIZE DMD Consortium is now hard at work to move the dial on endocrine and bone health care for individuals with DMD.
Supplemental Material
sj-pdf-1-jnd-10.1177_22143602241303370 - Supplemental material for A Parent Project Muscular Dystrophy-sponsored International Workshop Report on Endocrine and Bone Issues in Patients with Duchenne Muscular Dystrophy: An Ever-changing Landscape
Supplemental material, sj-pdf-1-jnd-10.1177_22143602241303370 for A Parent Project Muscular Dystrophy-sponsored International Workshop Report on Endocrine and Bone Issues in Patients with Duchenne Muscular Dystrophy: An Ever-changing Landscape by Leanne M Ward, David R Weber, Sze Choong Wong, Susan Apkon, Paula R Clemens, Linda H Cripe, Hugh J McMillan, Eugenio Mercuri, Nat Nasomyont, Kim Phung, Nora E Renthal, Meilan M Rutter, Cuixia Tian, Claire L Wood, Philip S Zeitler, Filippo Buccella, Kathi Kinnett, Pat Furlong and on behalf the International OPTIMIZE DMD Consortium in Journal of Neuromuscular Diseases
Footnotes
Abbreviations
Acknowledgements (Workshop Participants)
Luca Bello, Università degli Studi di Padova; Paolo Bettica, Italfarmaco; Elena Carraro, Centro Clinico NEMO, Milan Italy; Janet Crane, Kennedy Krieger Institute, Baltimore MD, USA; Adele D’Amico, Ospedale Pediatrico Bambino Gesù, Rome, Italy; Danilo Fintini, Ospedale Pediatrico Bambino Gesù, Rome, Italy; Melissa Fiscaletti, Sainte Justine University Hospital Center, Montreal, QC, Canada; Giovanni Iolascon, Università degli Studi della Campania “Luigi Vanvitelli”, Naples, Italy; Nasrin Khan, The Ottawa Pediatric Bone Health Research Group, Children's Hospital of Eastern Ontario Research Institute, Ottawa, ON, Canada; Kathi Kinnett, Parent Project Muscular Dystrophy, USA; Nadia Merchant, Children's National Hospital, Washington, DC, USA; Stefan Nicolau, Nationwide Children's Hospital, Columbus, OH, USA; Chiara Panicucci, Istituto G. Gaslini di Genova, Genova, Italy; Annie Sbrocchi, Montreal Children's Hospital, Montreal, QC, Canada; Mena Scavina, Parent Project Muscular Dystrophy, USA; Susan Ward, Collaborative Trajectory Analysis Project (cTAP), USA; Raoul Rooman MD, PhD, Consultant to Santhera, Putte, Belgium.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This meeting was jointly funded by Parent Project Muscular Dystrophy and Parent Project APS. In addition, unrestricted (unconditional) grants were provided to support this meeting by PTC Therapeutics, Pfizer and Santhera Pharmaceuticals. Dr Kim Phung was supported by an Advanced Training Fellowship sponsored by Parent Project Muscular Dystrophy and Defeat Duchenne Canada. Dr Ward is supported by a Tier 1 (Senior) Clinical Research Chair in Pediatric Bone Disorders from the University of Ottawa and the Children's Hospital of Eastern Ontario Research Institute. Dr Claire Wood was supported by an NIHR Advanced Clinical Lectureship.
Author contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
