Abstract
Deflazacort and prednisone/prednisolone are the current standard of care for patients with Duchenne muscular dystrophy (DMD) based on evidence that they improve muscle strength, improve timed motor function, delay loss of ambulation, improve pulmonary function, reduce the need for scoliosis surgery, delay onset of cardiomyopathy, and increase survival. Both have been used off-label for many years (choice dependent on patient preference, cost, and geographic location) before FDA approval of deflazacort for DMD in 2017. In this review, we compare deflazacort and prednisone/prednisolone in terms of their key pharmacological features, relative efficacy, and safety profiles in patients with DMD. Differentiating features include lipid solubility, pharmacokinetics, changes in gene expression profiles, affinity for the mineralocorticoid receptor, and impact on glucose metabolism. Evidence from randomized clinical trials, prospective studies, meta-analyses, and
INTRODUCTION
Duchenne muscular dystrophy (DMD), the most common form of muscular dystrophy, occurs in 15.9 per 100,000 live male births in the US and 19.5 per 100,000 live male births in the UK [1–3]. This lethal, X-linked neuromuscular disorder is caused by a mutation in the dystrophin gene (
The typical DMD phenotype includes significant progressive skeletal muscle weakness in early childhood with loss of ambulation (LoA) typically by age 13 [6]. Almost all individuals experience cardiomyopathy after the age of 18, and few live beyond the third decade [6].
Optimal glucocorticoid administration remains a topic of debate in the management of DMD. Although the importance of glucocorticoids in DMD is well-established, the side effects of long-term treatment are significant and force clinicians to constantly review the risks and benefits of the therapy. To this end, questions remain around the choice of agent, dosing, timing and frequency. This review aims to survey the evidence supporting glucocorticoids as standard of care in DMD, and specifically compare the mode of action, features, and outcomes associated with commonly prescribed glucocorticoids — prednisone/prednisolone and deflazacort [7]. Deflazacort was approved by the US Food and Drug Administration in 2017 for DMD in patients aged 5 years and older and the label was expanded in June 2019 to include patients aged 2 to 5 years [8]. It was approved in other countries for decades prior to this and used off-label in the US among those who were able to gain access to foreign supplies [9]. Prednisone/prednisolone continues to be alternatively prescribed off-label for the treatment of DMD in the US.
COMPARING DEFLAZACORT AND PREDNISONE/PREDNISOLONE IN DMD
Chemical structure
Prednisone, prednisolone and deflazacort are synthetic glucocorticoids, with deflazacort being an oxazoline derivative of prednisolone [10]. All three compounds are heterocyclic: prednisone and prednisolone having four rings and deflazacort having five rings (Fig. 1) [10]. The structural characteristics of deflazacort may explain some of its differing pharmacological activities from prednisone/prednisolone, including its lack of sodium-retaining activity and lower interference with carbohydrate metabolism in comparison with classic corticosteroids [10].
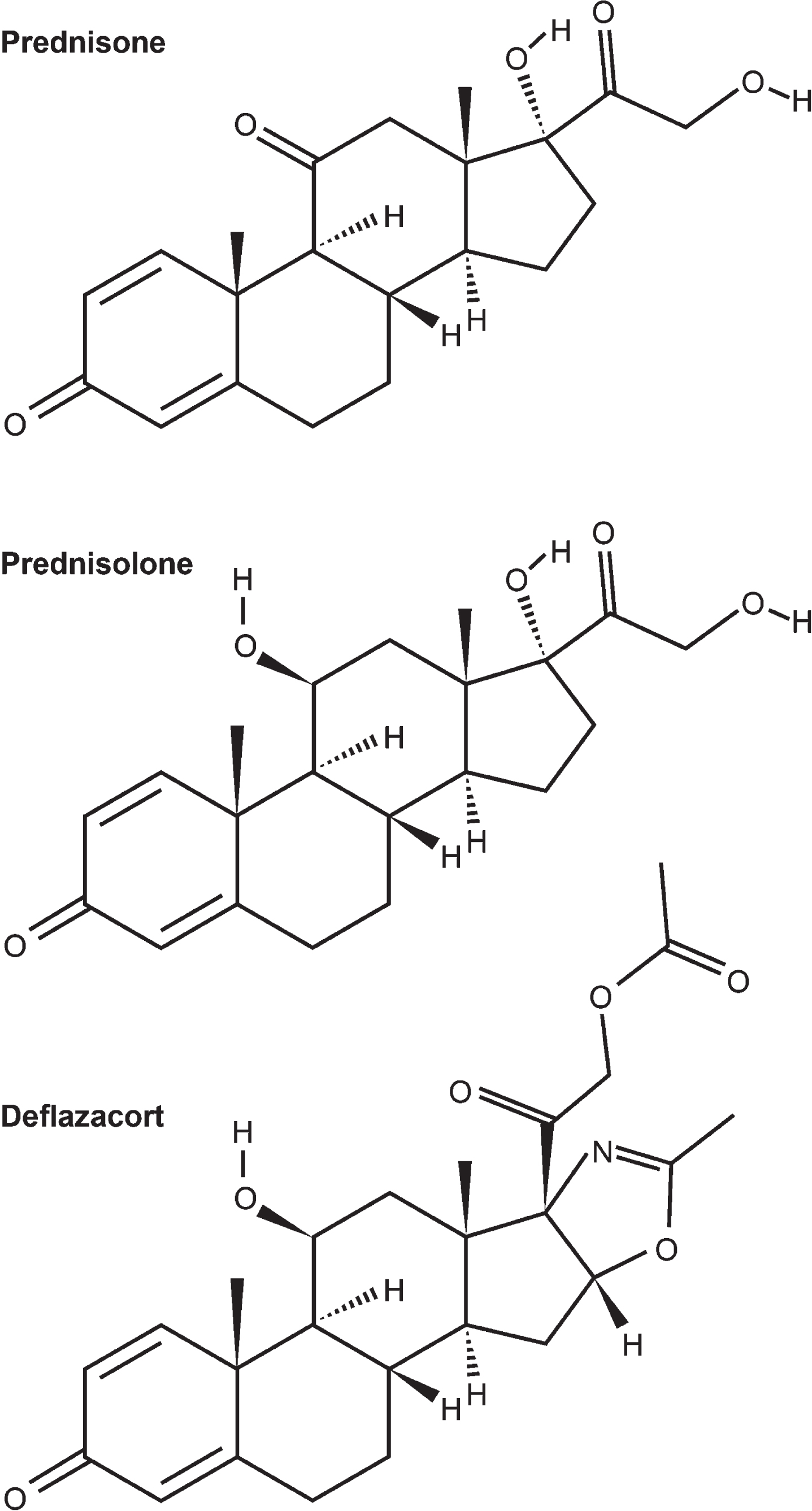
Structures of prednisone, prednisolone, and deflazacort, as listed in the PubChem database [70].
Mechanisms of action
The exact mechanisms of action of deflazacort and prednisone/prednisolone in DMD have not yet been fully elucidated but are clearly multifaceted, with both drugs affecting multiple gene expression pathways. One study evaluated mRNA transcripts from muscle biopsies from nine boys with DMD, collected before and after 3 months of deflazacort treatment, in comparison with those of eight healthy boys [11]. In dystrophic muscle, the expression of genes important for myogenesis, muscle regeneration and muscle maturation was increased towards normal ranges by deflazacort treatment, while expression of several genes with roles in inflammation was reduced towards normal ranges [11]. In a second study that compared gene expression in whole blood from 14 corticosteroid-treated children and adolescents with DMD with 20 who were corticosteroid-naïve, treatment with corticosteroids was found to upregulate genes associated with primary and secondary granules in neutrophils, iron trafficking, and chondroitin sulfate synthesis [12]. When comparing gene expression in deflazacort- and prednisone-treated individuals, there were fewer expression changes overall with deflazacort versus prednisone. Differences between the treatments centered on genes involved in adipose metabolism, potentially suggesting mechanisms by which corticosteroid-related weight gain, more typically associated with prednisone/prednisolone than with deflazacort, may be orchestrated [12].
Pharmacokinetics and pharmacodynamics
Both deflazacort and prednisone are prodrugs. Deflazacort is deacetylated by plasma esterases
Deflazacort exhibits linear, dose-proportional pharmacokinetics. The absolute oral bioavailability of deflazacort is 68%, and less than 40% was bound to plasma proteins after administration in healthy volunteers [13]. After a single 30 mg dose of deflazacort, maximum plasma concentrations of deflazacort-21-hydroxide were 121μg/L (oral) and 652μg/L (intravenous) [13]. The pharmacokinetics of prednisolone and prednisone are dose-dependent due to nonlinear protein binding. Protein binding of prednisolone decreases nonlinearly from 95% to about 60 to 70%, while the plasma concentration increases from 200μg/L to 800μg/L when protein binding reaches a plateau, observed with doses above approximately 20 mg [15].
The half-life of deflazacort after oral and intravenous administrations is 1.8 hours and 1.5 hours, respectively [13], whereas the half-life of prednisolone is approximately 3 hours when administered via either route
The dosing ratio of deflazacort to prednisone/prednisolone is nearly 85%; thus, 6 mg of deflazacort is roughly equivalent to 5 mg of prednisone/prednisolone [13, 16].
Key differentiating features
Deflazacort has lower lipid solubility than prednisolone [17]. It has no affinity for transcortin, binding instead to plasma protein and blood cells. Deflazacort crosses the blood-brain barrier more poorly than prednisone/prednisolone [17].
The lower lipid solubility may explain the lower crossing of the blood brain barrier with deflazacort relative to prednisone and the resultant greater behavioral side effects observed with prednisone/prednisolone [18]. The greater aqueous distribution of deflazacort may also account for the greater frequency of cataract formation with this drug versus prednisone/prednisolone due to the lens of the eye being adjacent to a fluid filled structure [19].
Glucocorticoids alter glucose metabolism, inducing a diabetes-like state in some individuals [20]. The rates of hyperglycemia and diabetes induced by glucocorticoid therapy vary depending on the population studied, but have been estimated at 32% and 19%, respectively, in patients without prior diabetes [21]. Deflazacort has less effect on glucose metabolism than prednisone/prednisolone, even in those with a family history of diabetes [10, 13]. Long-term administration of deflazacort at doses up to 50 mg per day has been shown to have no effect on glucose metabolism [13].
Deflazacort has little to no affinity for the human mineralocorticoid receptor, and the active metabolite, deflazacort-21-hydroxide, shows low transactivation [22]. This was shown in a binding assay, in which deflazacort showed no transactivation; the potencies of deflazacort and its active metabolite were not detectable [22]. By contrast, the mineralocorticoid activity of prednisone/prednisolone is about half that of natural glucocorticoids and their sodium retaining potency is about 80% that of cortisol [10]. Deflazacort is believed to have a lower impact on calcium metabolism than other synthetic glucocorticoids, with a resulting lower risk osteoporosis/osteopenia in the first decade of treatment [10, 24].
GLUCOCORTICOIDS AS STANDARD OF CARE IN DMD
Guideline recommendations and meta-analysis
The 2016 update of the American Academy of Neurology guidelines on corticosteroid treatment in DMD [25] and a Cochrane systematic review published in 2016 [10] conclude that corticosteroids improve strength, timed motor function and pulmonary function, delay LoA and onset of cardiomyopathy, reduce need for scoliosis surgery, and increase survival [25].
Recent studies of corticosteroid use in DMD
Several new studies, published since the guidelines mentioned above, shed additional light on the impact of corticosteroids in DMD. One randomized placebo-controlled trial of 196 patients was included in the Cochrane review in abstract form only but has since been published in full [26]. Patients were aged 5 to 15 years, with onset of weakness at an age below 5 years, and were randomized to deflazacort (0.9 or 1.2 mg/kg/d), prednisone/prednisolone (0.75 mg/kg/d) or placebo. At 12 weeks, compared with the placebo group, patients who received prednisone/prednisolone (
Long-term observational data of value have emerged in recent years, notably from the Cooperative International Neuromuscular Research Group (CINRG) Duchenne Natural History Study. An analysis from this prospective dataset included a cohort of 440 patients with DMD aged 2 to 28 years from 20 centers in nine countries, with follow-up for up to 10 years [27]. In order to assess the impact of glucocorticoid therapy, no glucocorticoid treatment or cumulative treatment of less than 1 month was compared with glucocorticoid treatment for 1 year or more. Glucocorticoid treatment protocols included daily dosing and other less frequent schedules, most commonly alternate-day dosing. Outcomes in terms of nine disease-related and clinically meaningful mobility and upper limb milestones were assessed. Time to all disease progression milestones was significantly longer among patients treated with glucocorticoids (prednisone, prednisolone or deflazacort) for at least 1 year, compared with those treated for less than 1 month or never treated (
The TREAT-NMD global DMD database combines clinical data from patients with DMD from 31 countries [29]. A cross-sectional study analyzed TREAT-NMD data from 5345 patients with DMD to investigate the impact of corticosteroids on major clinical outcomes [30]. Almost half (49.7%) of the study population were concomitantly using corticosteroids, 37.7% had never used corticosteroids and 9.8% reported past use (use for the remaining 2.8% was unknown). Median age at LoA was 10 years for non-corticosteroid treated patients and 13 years for patients receiving corticosteroids (
A prospective observational study from centers in Italy, the UK and USA followed 153 boys with DMD aged 3–6 years at baseline, for a minimum of 1 year and documented functional changes using the North Star Ambulatory Assessment (NSAA) scale [32]. NSAA scores progressively increased with age and multilinear regression analysis showed that use of corticosteroids significantly contributed to this change (
A US population-based study of 477 patients identified by the Muscular Dystrophy Surveillance Tracking and Research Network (MDSTARnet) stratified glucocorticoid treatment duration into short (0.25–3 years), long (>3 years) and no treatment [33]. Patients who received long-term treatment lost ambulation 2 years later, and had an 82% lower annual risk of losing ambulation, than untreated patients up to age 11. Unexpectedly, patients who received short-term treatment lost ambulation 0.8 years earlier and had a 77% higher annual risk of losing ambulation than those who were untreated.
Intermittent, rather than daily, corticosteroid treatment is a regimen used by some patients, and a recent UK retrospective longitudinal study suggests similar outcomes with these two regimens [34]. The study included 270 patients (mean age, 6.2± 2.3 years), 182 on intermittent steroids, 66 on daily steroids and 22 steroid-naïve patients [34]. Yearly FVC% predicted declined similarly from 9 years of age for patients receiving daily or intermittent corticosteroids (5.9% and 6.9% per year, respectively;
A small, single-center retrospective analysis compared 12 patients receiving daily corticosteroids with 12 patients (matched for age, BMI, and treatment duration) receiving weekend higher dose pulse corticosteroids [35, 36]. Cardiac and functional measurements did not differ between the two groups but the pulse weekend group showed improved metabolic and bone health measures compared with the daily steroid group (whole body fat mass, 36.3% vs 50.4% [
Adverse events associated with glucocorticoids in DMD
Side-effects associated with long-term corticosteroid use are well documented. Particular concerns include weight gain, effects on linear growth, behavioral effects, pubertal delay and increased risk of vertebral fractures [37–39]. Other common adverse events (AEs) include Cushingoid features, hirsutism, acne, increased insulin resistance, and frank glucose intolerance [38]. Long-term administration of corticosteroids may lead to suppression of the hypothalamic-pituitary-adrenal (HPA) axis such that the adrenal glands no longer produce endogenous cortisol [40]. If corticosteroids are stopped abruptly, adrenal insufficiency can result which may progress to adrenal crisis [37]. Some AEs associated with glucocorticoid therapy can be mitigated by dose reduction or alternative dosing regimens, if warranted (Table 1), although the potential impact of reducing dose on treatment efficacy should be considered [37].
Assessment of glucocorticoid efficacy and nonphysical AEs by patients and their caregivers may be useful to ascertain maximum benefit of therapy and to determine whether these AEs change over time. To this end, the 23-item SIDECORT (SIDe Effect of glucoCORTicoids) questionnaire was developed. It was designed to address perceptions by patients and their parents with regard to cognition, emotional functioning, and behavior [41]. SIDECORT can be regarded as having four components: general information, potential benefit from steroid therapy, putative AEs from steroid therapy, and overall impression regarding treatment [41]. In the study reporting the development of the SIDECORT questionnaire, a cohort of 86 patients and 125 of their parents completed the questionnaire. Results regarding treatment duration and AEs showed that parents reported more negative and significant correlations between treatment duration and getting upset easily, distraction, restlessness, and other psychosocial parameters, but no correlation with physical side effects. In contrast, patients reported positive correlations between treatment duration with physical side effects, especially with being hungry often and having Cushingoid features [41].
Many individuals with DMD have been treated with glucocorticoids for more than 10 years; however, at the time of writing, we are not aware of any data on the prevalence and management of adverse effects in these young men. This is an important gap in our knowledge, particularly with regard to bone health, bowel and bladder function, nutrition, cataracts, and psychosocial wellbeing.
Direct comparisons of efficacy and safety –randomized trials
Table 2 summarizes the key studies providing data on head-to-head comparisons of deflazacort and prednisone/prednisolone. Griggs et al describe the largest randomized head-to-head trial of deflazacort and prednisone in DMD yet performed [26]. The multicenter trial enrolled 196 patients aged 5 to 15 years, with onset of weakness at an age of less than 5 years. Patients were randomized to deflazacort (0.9 or 1.2 mg/kg/d), prednisone (0.75 mg/kg/d) or placebo. At 12 weeks, the active treatment groups had small but significant improvements in average muscle strength compared with placebo, but there were no significant differences between deflazacort and prednisone at this time point (least squares [LS] mean change versus placebo for: deflazacort 0.9 mg/kg/d, 0.25 vs 20.1,
Studies comparing deflazacort and prednisone/prednisolone
*Patient numbers given for daily dosing regimen; various intermittent deflazacort and prednisone regimens were also observed. †At least daily. ‡Regimen details not given. ACT DMD, Ataluren Confirmatory Trial in Duchenne Muscular Dystrophy; BMI, body mass index; DFZ, deflazacort; DMD, Duchenne muscular dystrophy; LoA, loss of ambulation; PRED, prednisone/prednisolone; TEAE, treatment-emergent adverse event.
In a small single-blind study in Iran, 34 patients, aged 3 to 10 years, were randomized to deflazacort (0.9 mg/kg daily) or prednisone (0.75 mg/kg daily) for 18 months [42]. Motor function was assessed using an index that scored ability to get up from the ground, walking on flat ground for 10 meters and going up four steps, in which lower scores indicated better ability to perform the task. At 1 year, mean motor function score had decreased by 11.19 (% CI, –19.8––2.5) in patients receiving deflazacort and increased by 5.20 % CI, –9.9–20.3) in patients receiving prednisone (
Another very small study, conducted in Italy, included only 18 patients aged 5 to 14 years [43]. This was a double-blind, randomized, multicenter equivalence study in which patients received deflazacort 0.9 mg/kg/d (
Two of the three randomized clinical trials showed differences in selected motor skills favoring deflazacort over prednisone at 12 months, and the largest of these studies also demonstrated an improvement in strength; and two of the three trials showed that participants taking deflazacort experienced less weight gain than those taking prednisone.
Direct comparisons of efficacy and safety –observational studies
Observational studies comparing outcomes with deflazacort and prednisone/prednisolone include two analyses from the CINRG Duchenne Natural History study. The first was an analysis of data from 340 patients, with average follow-up of 3.8 years [7], and the second was an extension of the same CINRG natural history study that included an additional 100 patients with up to 10 years of follow-up (described earlier in the current review) [27].
In the initial analysis of 340 patients, deflazacort was associated with later LoA than prednisone/prednisolone (HR 0.294±0.053 vs 0.490±0.08;
In the extension study, compared with the prednisone/prednisolone group, patients in the deflazacort group had significantly increased age at loss of supine-to-stand (
Median age of transition to ambulatory and upper limb function milestones
*
Deflazacort (relative to prednisone) use was significantly associated with a longer time to loss of ability to rise from supine in an analysis of data from 316 ambulatory boys from four natural history databases (UZ Leuven, CureDuchenne, iMDEX and ImagingDMD) [45]. The median age of the patients was 7.9 years (range, 4.4–19.4 years); 64% of patients were receiving deflazacort and 36% were receiving prednisone. Overall, 119 boys (38%) lost stand from supine ability over 900 patient-years of follow-up. Baseline rise velocity, better 4-stair climb performance and younger age at baseline were also associated with a longer time to loss of rise from supine ability. A sensitivity analysis incorporating data from placebo arms of clinical trials provided similar results.
A retrospective review of 832 patients with DMD in the UK NorthStar database (2006–2015) focused on fracture morbidity and growth, stratifying outcomes by type of corticosteroid used [24]. The median age at baseline was 6.9 years (interquartile range 4.9–7.2 years) and most boys were ambulant. The median follow-up was 4 years (interquartile range 2.0–6.0). The fracture incidence was 682 per 10,000 person-years (95% CI, 579–798). In total, 564 participants had complete information about glucocorticoid use. The majority of patients received prednisolone, either daily (
There is some evidence that long-term deflazacort increases the likelihood of cataracts. A retrospective review of 596 patients with DMD found that the prevalence of cataracts was 22.4% among the 514 patients receiving corticosteroids [50]. The odds of cataract development were 2.4-fold higher among patients taking deflazacort than those receiving prednisone (95% CI, 1.3–4.5;
There is also some evidence that deflazacort and prednisone/prednisolone may have different impacts on behavior. A questionnaire-based study of 67 ambulant patients and their parents indicated that patients taking deflazacort exhibited more withdrawn behavior than those taking prednisone/prednisolone, whereas patients taking prednisone/prednisolone exhibited more aggressive behavior than those taking deflazacort [18].
The varying doses of corticosteroids reported in real-world, prospective natural history studies complicate interpretations of safety and efficacy. However, the DMD care considerations do not mandate that doses of deflazacort and prednisone need to have weight-based adjustments over time [37]. The real-world evidence from such prospective natural history studies does provide insights into how these corticosteroids compare with real clinical practice. If one corticosteroid regimen is typically adjusted downward due to side effects, there is still value in considering such real-world data obtained in large cohorts over multiple years.
Post-hoc analyses in placebo arms of industry-sponsored clinical trials
The placebo arms of clinical trials in patients with DMD, in which participants continue their pre-trial glucocorticoid treatment, can potentially offer an additional, low-level evidence insight into use of deflazacort vs prednisone/prednisolone. Limited conclusions can be drawn from these analyses because they were not originally designed to compare these treatments and hence bias is introduced because of the lack of randomization to glucocorticoid treatment. However, the increased age ranges of patients included in these trials gives insights into experiences with more prolonged dosing beyond the ages of 7 to 10 years.
A non-prespecified
A
A second
Finally, a recent
Limitations to studies comparing deflazacort with prednisone/prednisolone
Clinical studies of boys with DMD present many challenges. It is difficult to find well-matched cohorts of patients owing to differences in the age of symptom onset and the broad spectrum of disease severity, which may be due, at least in part, to different mutations within the dystrophin gene [55]. Owing to limitations in our understanding of DMD, the most appropriate methods to evaluate skeletal muscle strength and function are still being elucidated [56, 57]. Furthermore, the use of BMI, to indicate if a person has a healthy weight, is of limited value when boys are treated with glucocorticoids and have delayed growth, and has been shown to underestimate body-fat mass [58]. It is also difficult to match cohorts of patients because boys with significant developmental delays are often excluded from trials owing to unreliable test results. There is considerable variability in individuals’ response to glucocorticoids, which is complicated by the different dosing for deflazacort and prednisone/prednisolone and dose adjustments. Given all these variables and challenges, it is difficult to get perfectly matched cohorts for randomized and controlled trials.
Most of the data from the
DISCUSSION
Glucocorticoids are the standard of care for DMD, although they are ultimately non-curative. Their long-term use improves skeletal muscle strength; preserves upper extremity, cardiac, and pulmonary function; delays LoA and scoliosis; and maintains quality of life.
Compared with prednisone/prednisolone, deflazacort has lower lipid solubility, causes less sodium retention, and has fewer effects on glucose metabolism. The body of evidence from randomized clinical trials, prospective studies, meta-analyses, and
The choice of corticosteroid extends beyond the clinical data as healthcare costs are also important. A detailed discussion surrounding costs is out of the scope of this review, but with governments and other payers increasingly struggling to cover the costs of expensive new treatments [59], this subject is important to acknowledge. Deflazacort is markedly more expensive than prednisone in the US [60], but it is also associated with pharmacological and clinical benefits, as described above. Before initiating glucocorticoid treatment, it is important for physicians, patients, and parents to discuss benefits and risks of various regimens, including costs [61], but ultimately the choice of treatment will be decided by third party payers in many cases. There are initiatives in place to help make therapies for DMD more affordable and accessible, such as Project Hercules [62], a UK-based consortium of pharmaceutical companies, charities, academics, patient organizations, experts and clinicians –stakeholders whose interests are often difficult to align. Deflazacort has a manufacturer-supported patient assistance and access program [63].
Because deflazacort was only recently approved in the US, real-world comparative data for deflazacort versus prednisone/prednisolone in the US are still emerging [44], but one observation from the deflazacort Early Access Program is that when lower doses than recommended were given, the variance between recommended dose and administered dose increased as patient weight increased [64]. Most clinicians do not appear to be making weight-based increases in doses of either prednisone or deflazacort. This may have implications for long-term outcomes, side effects, and optimal management. Optimizing dosing regimens is the goal of the ongoing, phase 3 Finding the Optimum Regimen for DMD (FOR-DMD; NCT01603407) trial [65]. This trial is comparing three regimens: daily prednisone/prednisolone at 0.75 mg/kg/d; intermittent prednisone/prednisolone at 0.75 mg/kg/d for 10 days, then 10 days of drug holiday; or daily deflazacort at 0.9 mg/kg/d [66]. The hypothesis is that daily treatment with either glucocorticoid will be more beneficial than intermittent dosing. A secondary outcome will be to compare AE profiles [65, 66]. The trial focuses on a population of patients with a narrow age range of 4 to 7 years with a 36-month follow-up, so its results cannot be extrapolated to patients older than 10 years old, who are in the decline phase of the disease. Thus, the FOR-DMD trial will not address the safety and efficacy profiles of deflazacort and prednisone/prednisolone in older patients where there may be different bioavailability of the drugs to muscle fibers due to fatty replacement of musculature with increasing age.
It should also be noted that alternative options to deflazacort and prednisone/prednisolone for the management of DMD are now reaching the market or are progressing through clinical development. New drugs targeting NFκB include the dissociative steroid vamorolone [67, 68], while other therapies aim to restore the function or expression of dystrophin, including eteplirsen, casimersen, golodirsen and viltolarsen, which are approved exon skipping drugs for use in the US, and ataluren, a premature stop codon read-through medication approved for use in the EU and other countries [68]. These therapies may in future prove to be useful alternatives to deflazacort and prednisone/prednisolone.
Footnotes
ACKNOWLEDGMENTS
Medical writing and editorial support were provided by Rosalyn Blumenthal, PhD, Beverly Barton, PhD, and Brandy Weidow, MS, ELS of BrightestSolutions, LLC, and Richard Claes, PhD and Tim Ellison, PhD, of PharmaGenesis London, London, UK, and were funded by PTC Therapeutics, Inc.
CONFLICT OF INTEREST
W. Douglas Biggar, MD has served as a consultant for PTC Therapeutics and Marathon.
Andrew Skalsky, MD has no conflicts of interest to report.
Craig M. McDonald, MD has served as a consultant for clinical trials for PTC Therapeutics, and outside the submitted work with Astellas Pharma, Avidity Biosciences, Capricor Therapeutics, Catabasis Pharmaceuticals, Edgewise Therapeutics, Entrada Therapeutics, Epirium Bio (formerly Cardero Therapeutics), FibroGen, Italfarmaco, Pfizer, PTC Therapeutics, Roche, Santhera Pharmaceuticals and Sarepta Therapeutics. He has received research support for clinical trials from Capricor Therapeutics, Catabasis Pharmaceuticals, Italfarmaco, Pfizer, PTC Therapeutics, Santhera Pharmaceuticals and Sarepta Therapeutics. He serves on external advisory boards related to Duchenne muscular dystrophy for PTC Therapeutics, Edgewise Therapeutics, Eli Lilly, Sarepta Therapeutics, Santhera Pharmaceuticals, and Capricor Therapeutics.
The authors received no compensation for writing this article.
