Abstract
Background
Duchenne muscular dystrophy (DMD), an X-linked progressive neurodegenerative disorder, is being added to required universal screening programs for newborns in the United States. It is estimated that this will result in around 880 patients presenting at clinics in infancy. Very little is known about the early gross motor abilities in infants and young boys with DMD.
Objective
Describe the early gross motor skill acquisition in boys in our clinic with DMD.
Methods
Between the years 2016 and 2024, 90 boys with DMD under the age of 6 years (2 months – 5 years 10 months at their first visit) were evaluated as part of their standard of care during regularly scheduled clinic visits using the using the Bayley Scales of Infant & Toddler Development, Third Edition (Bayley-III). Forty-seven boys were seen in clinic longitudinally with two to six follow up visits for a total of 129 assessments.
Results
Ninety four percent of the boys with DMD seen in our clinic demonstrated delays in gross motor skills across the age span when compared with normative controls. None of the boys reached a ceiling on the Bayley-III despite being older than the intended age range of the test.
Conclusions
Our clinic data showed an almost universal gross motor delay in infants and toddlers that did not diminish over time.
Keywords
Introduction
Duchenne Muscular Dystrophy (DMD) is an X-linked degenerative disorder resulting in progressive weakness. Although causative variants in the dystrophin gene are present at birth, DMD isn’t typically diagnosed until around age 5 years and the allelic milder Becker Muscular Dystrophy is diagnosed even later.1–3 A diagnosis of DMD can be delayed because children continue to develop new motor skills, although at a slower rate than healthy peers, prompting providers to take a “wait and see” approach. 4 Around 7 years of age, due to progressive weakness caused by muscle breakdown, the demands of age-appropriate motor skills exceed the child's capacity, and after a functional plateau, all boys with DMD begin to lose skills. 5
There is an increased focus on the developmental abilities of infants and young boys with DMD as clinical trials for DMD are increasingly enrolling younger patient groups. In addition, several states are implementing newborn screening for DMD which will allow for diagnosis within the first few weeks of life. Extrapolating the average incidence rate of previously published DMD-NBS programs (1 in 4087) to annual USA birth rate from 2023 (3,596,017) it is expected that 879 infants with DMD will be born each year. 6 A recent gene therapy trial for another rare progressive disorder, spinal muscular atrophy(SMA), identified the importance of early treatment. 7 Infants with SMA treated at an earlier age had significantly better treatment responses than those treated at an older age; even when older infants had higher motor skill scores at treatment initiation. Infants in the SMA gene therapy study were inititially evaluated with a disease specific scale, The Children's Hospital of Philadephia Infant Test of Neuromuscular disorders, but rapidly exceeded the utility of this test.7,8 The Bayley-3 was added as a measure of motor development to compare infants to their peers. Since severity of weakness was less predictive of long-term function than earlier age at initiation of treatment, it could be hypothesized that earlier treatment could also optimize the benefit of a new therapeutic for DMD.
Normal maturational development of the gross motor skills in young boys with DMD makes clinical trials challenging as it is difficult to determine if post-treatment improvement is attributable to the therapeutic or simply to maturation. The most used DMD specific test, the Northstar ambulatory assessment does not have items applicable below the age of 3 years, thus would not be appropriate in infants. 9 The objective of this paper is to document the gross motor development of young boys with DMD using the Bayley-III. Our hypothesis is that most boys with DMD will have a gross motor delay in infancy and despite gaining milestones over time will continue to be delayed compared to age match peers. The Bayley Scales of Infant & Toddler Development, Third Edition (Bayley-III) is a discriminative assessment tool used globally to quantify a child's development in infants from 16 days – 42 months 15 days of age. 10 The Bayley-III consists of five subscales: cognitive, receptive language, expressive language, fine motor and gross motor. Although previous work has provided information on all 5 subtests, this study looks exclusively at gross motor function.11–13 The Bayley-III provides a raw score quantifying the number of motor skills the child was able to demonstrate as well as a scaled score up to age 42 months 15 days. The advantage of the scaled score is that it compares the child to age matched peers, thus scaled scores can be compared across ages. Scaled scores range from 1–19 with higher scores indicating superior performance. A scaled score of 10 (±3 points) would be considered “typical development”. Thus, scaled scores over 13 and above would indicate above average performance, and 7 and below are indicative of below average gross motor skills.
Although the Bayley-III was designed for use in children ages 16 days – 42 months 15 days of age, the authors of the assessment suggest that the test can be used in children outside of the age range with lower functional abilities. 10 While the ability to compare directly to typically developing peers is not possible using the Bayley after 42 months 15 days of age, the test will continue to track progression of motor skills and an “age equivalent score” can be determined. Age equivalent scores provide a general estimate of a child's performance in comparison to peers when that child's age is outside of the normative sample. The age equivalent is generated by identifying the age at which the subject's raw score would be given a scaled score of ten, or when roughly 50% of the normative patient group would have mastered similar skills. Thus, even if a child is greater than 42 months 15 days of age but has not achieved all the items on the test, their age equivalent score provides an approximation of the current level of gross motor skills and can be used cautiously as a scaled score surrogate.
Connolly, et al. used the Bayley-III to show that 24 boys with DMD demonstrated delayed gross motor development compared to normative peers within the first years of life which further declined over 1 year.11,12 Connolly also used the Bayley-III to demonstrate that twice weekly corticosteroid use in 23 young boys with DMD, initiated under the age of 2 years 6 months, resulted in improved gross motor skills compared to a historical control patient group following one year of treatment. 13 Using the World Health Organization motor milestones 14 and the Northstar Ambulatory Assessment, 15 other studies have also confirmed that boys with DMD achieve gross motor milestones at a later age than typically developing peers.4,9
With newborn screening in place, the field will likely move toward treating infants and toddlers. It will be essential to document the baseline gross motor abilities of the child prior to receiving treatment. Since boys with DMD typically gain skills until around age six years, it could be difficult to determine efficacy until the child starts to lose skills. Knowing the prevalence of gross motor delay and understanding the developmental trajectory of gross motor skills in infants and toddlers with DMD could be used to plan more effective treatment trials and help differentiate changes attributable to a therapeutic intervention versus maturation alone. The aim of this paper is to present our retrospective clinic data to lay the framework for designing a future prospective research study.
Materials and methods
Participants
The data in this manuscript were collected as part of standard clinical care at the Nationwide Children's Hospital (NCH) Muscular Dystrophy Clinic. The deidentified data were reviewed retrospectively. The institutional review board at NCH as determined that informed consent was not required to analyze these de-identified data.
Between January 2016 and January 2021, ninety boys under the age of six years with DMD were evaluated with the Bayley III as part of their standard clinical care at the Nationwide Children's Hospital (NCH) Muscular Dystrophy Clinic. Boys were classified as having DMD if they had a genetic variant of the DMD gene that prematurely terminated the translational reading frame, thus preventing translation of the DMD messenger ribonucleic acid encoding the muscle isoform of dystrophin or encoding a truncated protein lacking an intact actin binding domain 1 and that variant was known to be associated with a DMD phenotype (Table 1, Supplemental Table 1). While dystrophin-related phenotypes can vary with even zero to small amounts of dystrophin (0 - ≤ 5%), the patients presented in this report are expected to not walk after 16 years of age (an acceptable clinical diagnosis of Duchenne muscular dystrophy). 16
Duchenne muscular dystrophy variant distribution at baseline (N = 90).

Boys were enrolled regardless of corticosteroid use but were excluded if they were on an approved exon skipping therapy or enrolled in a clinical trial. Forty-seven boys had at least one longitudinal visit with a range of 3- to 12-month intervals between visits. Both the average and median number of visits was 2 but subjects ranged between 1 and 7 visit. Overall, we report 187 assessments as many participants had more than 2 visits. Forty-three boys did not have longitudinal data for the following reasons: the boy turned 6 years of age (N = 19, 44%) or enrolled in a clinical trial (N = 6, 14%) prior to the next clinic visit. Eleven boys were only seen once in the reported time frame. Seven (16%), were previously established at a local clinic and came to NCH for a second opinion, while four (9%) did not have another clinic visit during the reported time window. Six boys were switched to disease specific assessments at next visit (14%) and one (2%) patient was not testable at the next visit due to temperament.
Procedures
Bayley-III
The Bayley-III gross motor subtest was administered according to the standardized administration manual. 10 The raw score and age equivalent score are reported for all participants. Norm-referenced scaled scores are reported for boys under the age of the normative sample limit of 42 months 15 days.
Statistical analysis
Statistical analysis was performed using the SAS 9.4 (SAS Institute, Cary, NC) and IBM SPSS 28.0 (SPSS Inc., Chicago IL). Descriptive statistics are used to characterize the group performance. Both one-way ANOVA and Kruskal-Wallis tests with Dunnet's T3 post hoc testing were used to determine if the developmental trajectory was continuous or plateaued. Minimum clinically detectable difference was determined using the 0.5 standard deviation (SD) definition.
Results
Cross sectional data
Ninety unique participants were evaluated as part of their clinical standard of care. Table 1 shows variant distribution of the sample (For additional details on variant see Supplemental Table 1). Our patient group had a median age at baseline of 3 years 4 months (IQR 2 years 4 months – 5 years 10 months) years with deletion being the most common variant class.
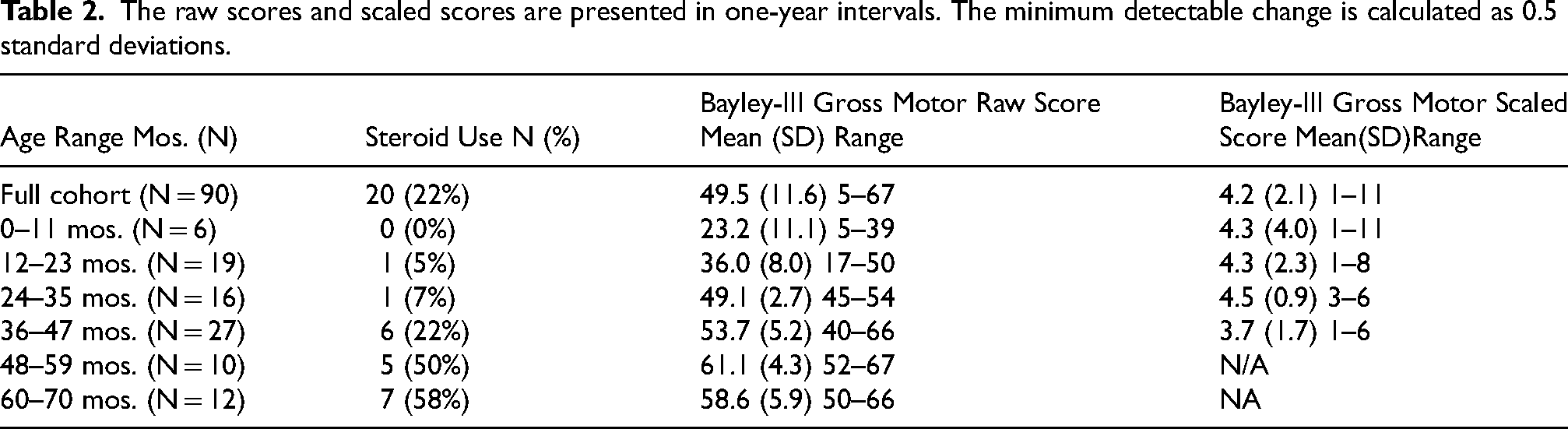
The raw and scaled scores of the cohort at the first clinic visit are listed in Table 2. Although the age of diagnosis is reported to be around 5 years of age, we evaluated 80 boys under this age in our clinic with 41 being seen for their first visit under 3 years of age. Figure 1(a) displays all of the raw scores by age. The curved lines on the graph display the range of scores that would be considered typically developing for each age. These lines were generated by plotting the average raw score for the eighth and twelfth scaled score at each age as reported in the Bayley manual. The points inside the shaded box represent boys who have not yet reached the test ceiling but who are over the age of 42 months 15 days for whom the assessment as created. This shows that the boys with DMD in this cohort do not show a ceiling effect on the Bayley despite being older than 42 months 15 days of age. Figure 1(b) shows the median raw score by 1 year age bins. Both a one-way ANOVA (F (5, 181) = [89.98], p ≤ .001) and Kruskal-Wallis (H(5) = 65.3, p < .001) test revealed that there was a statistically significant difference between the groups. However, post hoc pairwise comparisons across groups revealed there wasn’t a significant difference in raw scores between individuals 4 year and 5 years old (adj. p > 0.392).
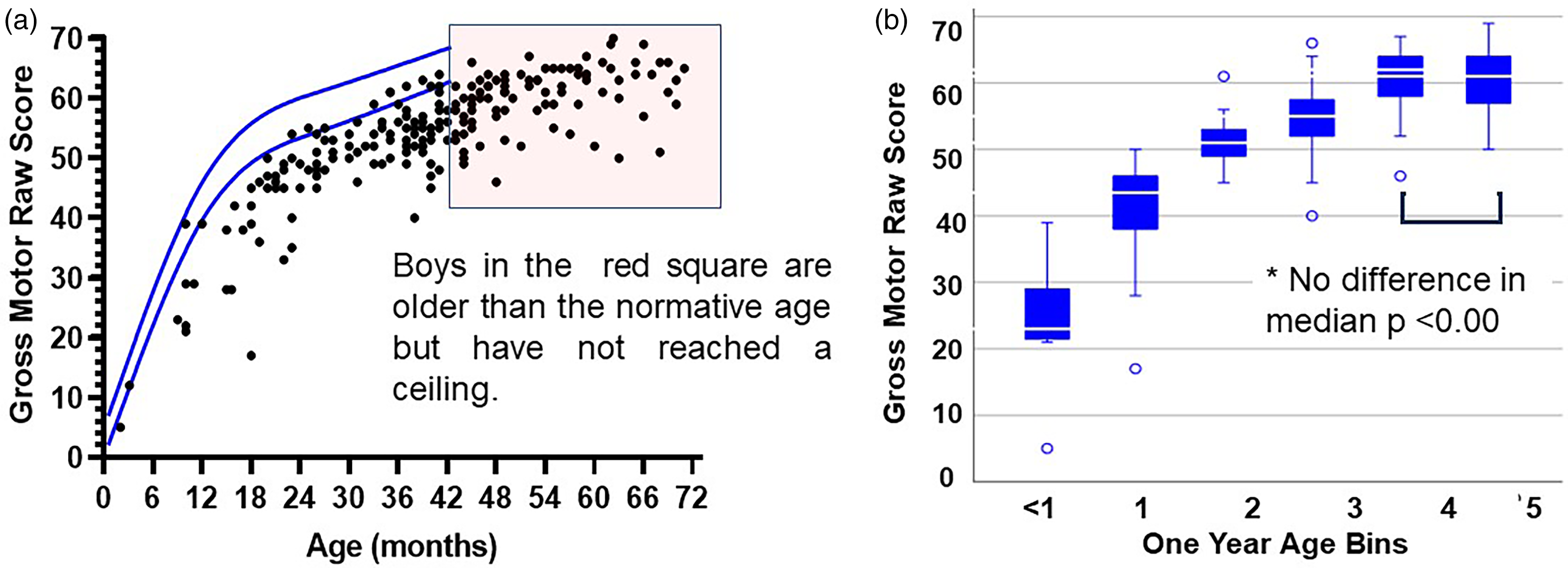
(a) The raw scores for boys with Duchenne muscular dystrophy are well below the lines that would indicate an average performance for age matched peers. The boys in the box are older than the testing age range but have not reached a ceiling. (b) median raw scores per 1 year age bins. Median scores of ages 4 and 5 years are not statistically significantly different suggeting a plateau.
For boys within the normative range (up to age 42 months fifteen days), the scaled scores were well below the expected range (Average = 4.7, SD = 2.1; Median = 5, IQR = 4,6). Figure 2 shows the distribution of scaled scores with a superimposed normal distribution. This figure illustrates that the most prevalent scaled score in typically developing children is ten, while boys with DMD cluster around a scaled score of 5 which is 1.6 standard deviations below the mean and indicates a significant motor delay.
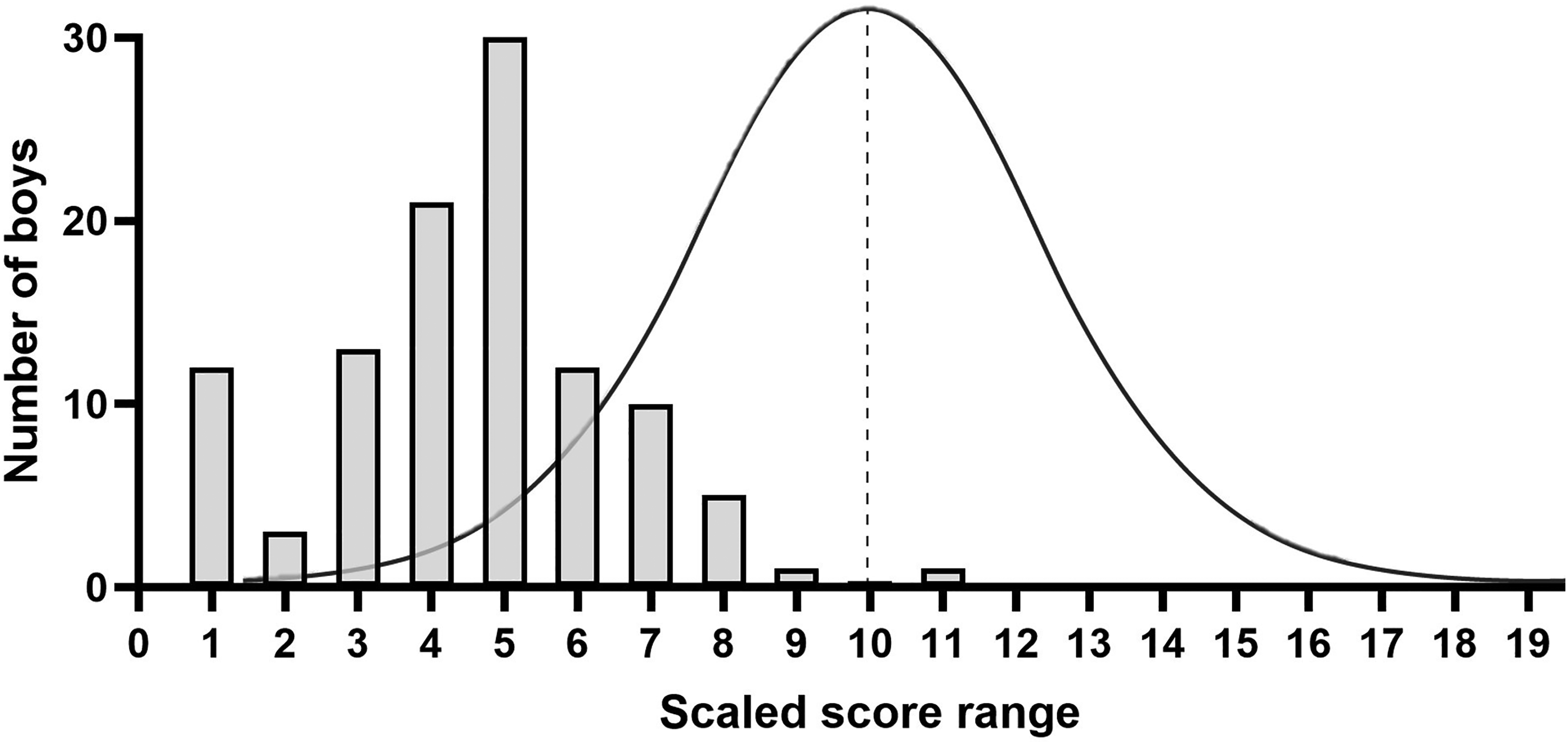
Distribution of scaled scores for DMD with superimposed expected normal distribution. 10 is the average scaled score for typically development but the DMD curve is shifted to the left with only one child (age 8 mos.) above average.
A scaled score of 10 is considered average on the Bayley III. Only one boy received a scaled score above average when he was 9 months old. The average scaled score for the group was 4.2 Figure 3(a) displays the scaled scores by age and corticosteroid use from every visit in the correct age range, thus some boys are represented more than once. This demonstrates that almost all boys with DMD show a gross motor developmental delay in infancy which persists over time, suggesting that the disease process starts very early in life and the boys do not “catch-up with their peers. Only five boys scored in the normal range. Three boys had one visit in the normal range while the other 2 boys had 2 visits within the low average range for a total of 7 out of 109 patient group assessments.
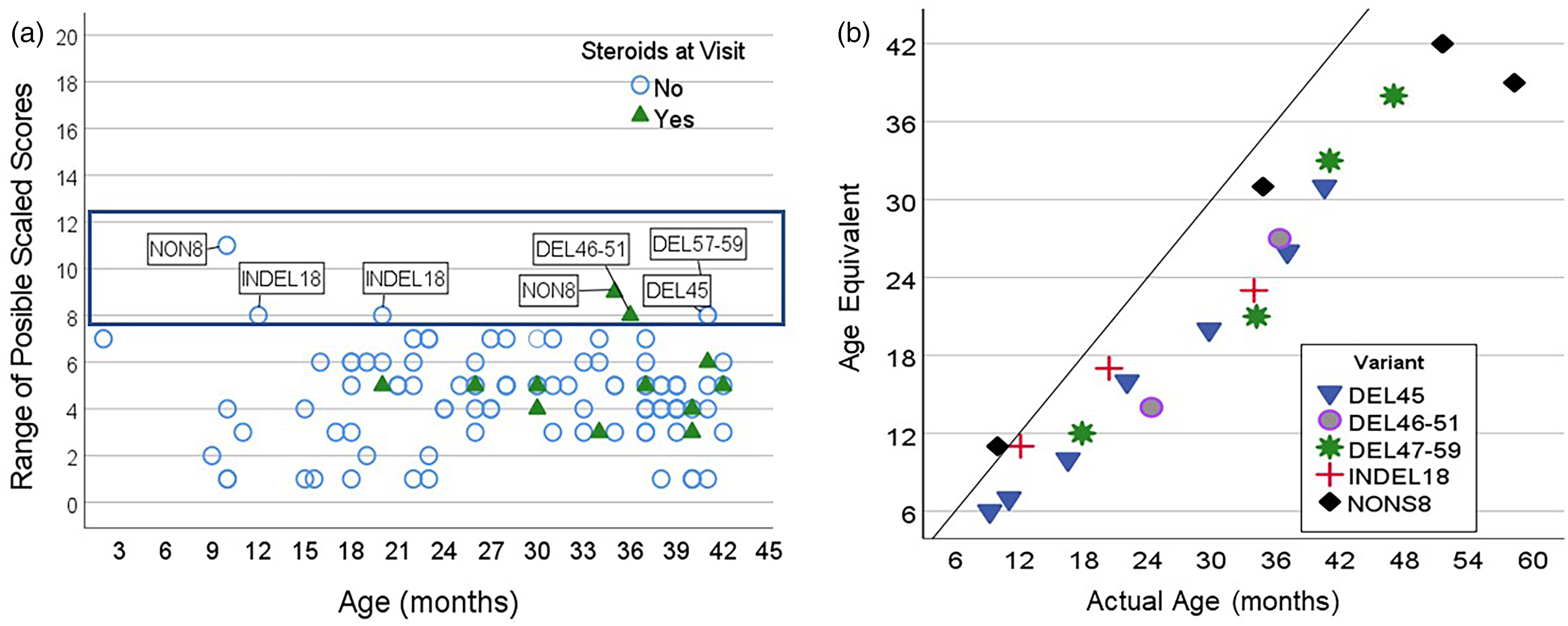
(a) The scaled scores for all visits are displayed by age. Seven visits fall with the average range illustrated by the darker box. (b) The 5 boys who scored within the typical range for at least one visit are identified by mutation. Age equivalents for the longitudinal visits for these boys are displayed to show that most of their visits were below age expectations. The solid line indicates “average development”. Visits closer to the line indicate better performance.
The individual trajectories of the five boys who received at least one scaled score in the typical range are also presented in Figure 3(b). These data are displayed as age equivalent scores as the later visits were outside the age range for determining a scaled score. The boys with deletions of exon 45, exons 46–51 and 57–59 initially presented with a gross motor delay but gained skills at a rate that brought them closer to their age expectation over time. The boy with an INDEL18 deletion stayed within the normal range until his visit at 34 months while the boy with a nonsense 8 mutation stayed within the normal until his visit at 58 months.
Corticosteroids
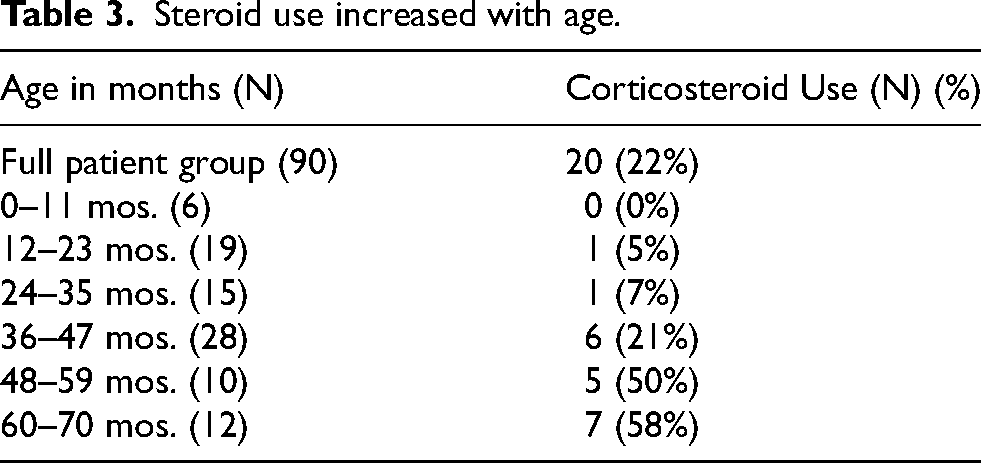
At the baseline visit, 20 (22%) participants were taking corticosteroids, either prednisone or deflazacort on either weekend or daily dosage schedules. As expected, corticosteroid use increased with age (Table 3). At the first visit, the median age of the patient group taking a corticosteroid (Median = 4 years 7 months, IQR = 3 years 7 months, 5 years 1 month) and was significantly older than the corticosteroid naïve group (Median = 2 years 8 months, IQR = 1 year 8 months, 3 years 5 months) [p < 0.001]). Therefore, most of the boys on corticosteroids at their first visit were outside of the age range in which a scaled score could be assigned. The age and size differences of the groups made a direct comparison of gross motor abilities statistically impractical, thus corticosteroid use was not factored into the analysis but is displayed graphically when appropriate. To display the entire patient group, we used the Bayley-III age equivalent scores. The age equivalent shows the age at which the boy's raw score would be considered average (scaled score of 10). Figure 4 plots each boy's age equivalent score against his actual age. The diagnonal line illustrates that older boys tend to have greater delays when compared to age expectation than younger boys.
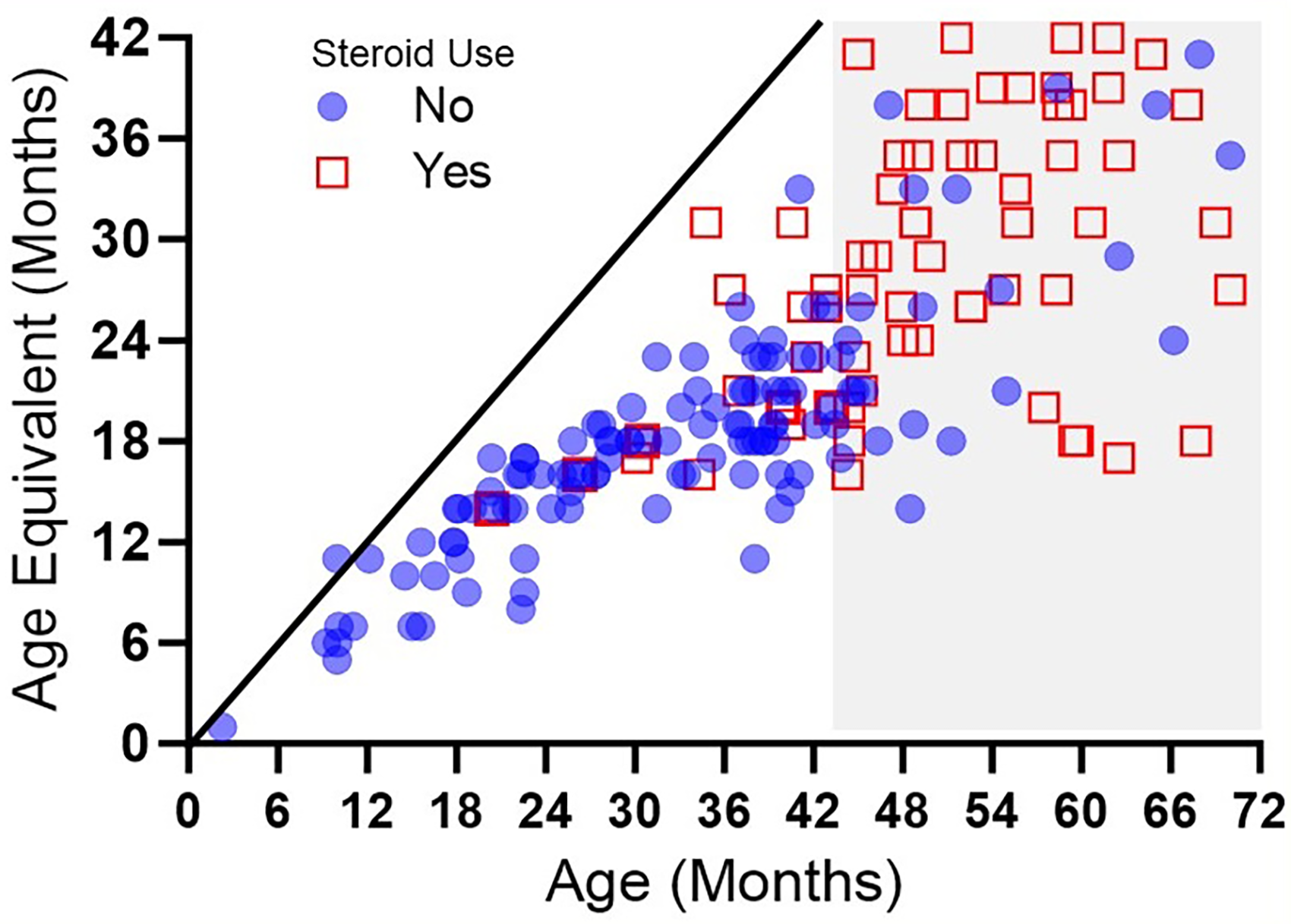
The boys age (x axis) is compared to the age at which his motor abilities would be considered typical for the age (y axis). Steroid naive boys are represented by circles. Scores that fall along the solid line would indicate that the boy's performance was average for his age. Scores further away from the solid line indicate greater delay.
Steroid use increased with age.
The raw scores and scaled scores are presented in one-year intervals. The minimum detectable change is calculated as 0.5 standard deviations.
Longitudinal data
Fourty-seven patients completed the Bayley-III gross motor scale during at least 1 follow up visit (mean follow-up interval: 8 months, range: 2–25 months) for a total of 128 longitudinal visits. Twenty-five out of the 47 longitudinal participants (53%) were on corticosteroids for at least one of their visits. The small sample size and significant age difference between steroid/steroid naïve group prevent valid statistical analysis.
The minimum detectable change (MDC) was calculated using the 0.5 standard deviation method. 17 When using the whole sample, the MDC for the raw score was 4.81 points and for the scaled score 1.01 points. We hypothesized that there might be a difference between younger and older children, but this was not the case. For raw scores the under 36-month MDC was 4.7 and the 36–42-month group was 4.85. Similarity for the scaled score the difference was extremely small 1.07 and 1.0 respectively.
Discussion
This paper quantified the gross motor performance on the Bayley-III in 90 boys with DMD from 2 months to 6 years of age. Gross motor delays were present in 94.5% of the boys evaluated and were detected across the age span. Only five boys achieved a scaled score within the typically developing range during at least one clinic visit. When considering all visits 94.5% of the assessments were below the expected range. When looking at the overall trajectory across all visits of these boys we saw that the boys with deletions of exon 45 (last visit at age 41 months), exons 46–51(last visit 36 months) and exons 57–59 (last visit 47 months) appeared to decrease the gap between themselves and their peers over time. The boy with the nonsense variant maintained his skills in the typical range until age 58 months. This boy participated in the Connolly 13 infant steroid study and thus started steroids prior to 1 year of age which may explain his early and sustained gross motor skills.
No boy in our patient group (evaluated up to 6 years of age) achieved the maximum raw score of 72, as would be expected for a typically developing child of 42 months 15 days. This means that none of the boys achieved the skills that would be expected in a typically developing child of 42 months 15 days. The starting point is obtained based on the child's age. If the child cannot perform the first 3 items, the evaluator moves back to the starting point for the next lower age and again determines if the child can perform 3 sequential items. In boys with DMD this frequently results in starting the test considerably below age matched peers. The test is stopped after the child is unable to complete 5 sequential tasks. Since the boys with DMD were delayed, most reached a ceiling well below age expectations and were not asked to perform the highest-level activities. We can assume that the child would not be able to complete a higher-level skill like jumps 24 inches if he cannot do an easier skill of jumps 4 inches, but many of the upper items were not administered. Skills that boys with DMD over the age of 42 months 15 days should be able to do but this patient group could not, include standing on one foot for 8 s, jumping forward 24 inches, and hopping 5 feet.
Using our data and clinical expertise, we identified several items that provided disproportionate difficulty for boys with DMD in our sample. For example, item 54 asks the child to jump down from the bottom step of a set of stairs. This is the first item at the basal level for kids ages 33 months to 38 months 30 days, so it is administered frequently. We administered this item 80 times and it was only able to be completed 14 times for a 17% success rate. In comparison, the items on either side; walks sideways without support and kicks ball, were performed at around an 80% success rate.
These data also show that walking up and down 3 stairs using a reciprocal pattern without handrails was also a particularly difficult activity for this patient group. Percentages are not provided for stair skills as the Bayley requires administering this item on custom-made steps. We administer the items on our standard rehabilitation steps have a .5-inch higher rise and 2.5-inch longer run. This increase in size could have affected our results. Two additional higher level items were quite difficult for this patient group: walking heel to toe on a line and imitating postures. Both activities involve attending to the task and motor planning. There is a higher incidence of attention disorders in boys with DMD which likely contributes to the difficulty with these items. 18 It would be interesting to see if a community gross motor activity such as karate or tumbling would help with a boys gross motor skills by improving motor planning.
Using a Gowers’ maneuver is a well-known hallmark of DMD. The Bayley III item “stands up mature” asks if the child can rise from lying to standing without rolling into prone or quadruped and without using their hands. We expected this item to be particularly difficult for our patient group. As expected, it was challenging skill with very few boys receiving credit for this item, however, a small percentage rose from the floor without demonstrating the Gowers’ sign movement pattern (6 out of 56 administrations). One must interpret these results cautiously, however, as this item is below the basal starting point for older boys. Any boy who can complete the basal items for 22-month 16-day old boy (walks backwards steps, walks downstairs with a railing and runs with coordination) will automatically get credit for this skill as the Bayley III scoring gives credit for all items below the boys basal. Thus, it is highly likely that older boys erroneously received a point for this item in their total score.
One major advantage of the Bayley is the large normative database that accompanies the test that enables a comparison of gross motor skills of boys across different ages. Our clinic data and others have documented that most boys with DMD have a gross motor limitation from infancy. Based on our sample we identified a minimum detectable change of 1.01 scaled points. Connolly et al. reported that initiating twice weekly steroids increased the patient group's scaled score mean by 1.3 over 12 months which differed significantly from a historical untreated patient group where scaled score declined. This difference (+1.3 over 12 months) also exceeds the MDC generated from our clinic data (MDC 1.01), thus signaling achievement of meaningful change.
The major limitation of using the Bayley III in infants with DMD is that a scaled score is only available until age 42 months 15 days. Although the authors suggest that the test is appropriate outside of this age range in children with a motor delay, it is no longer possible to compare the child's performance to age-matched peers so an external control group would be needed. However, comparison of a child's age equivalent score to the raw score may enable interpretation of older children over time.
Newborn screening for DMD is slowly being added to the universal screening panel in states in the United States of America. The Bayley III or the well-correlated Bayley-4 which replaced it could be used to identify and monitor development overtime in this patient group. There is one potential limitation for use in the youngest infants, as there are very few test items for these infants and most items focus on head control. At the youngest age of 16 days, a raw score of 0 in which the infant does not perform any of the test items will still equate to a gross motor scaled score of 6. Thus, we recommend caution when evaluating infants under 3 months of age with the Bayley III. Furthermore, Bayley scoring is stratified by age range, and at youngest ages, a change in raw score corresponds to a larger change in scaled score as compared to older age ranges; thus, scaled scores are disproportionately impacted by changes in fewer items. For example, a 2-month-old child is only required to demonstrate 2 items to fall within the average range, thus scaled score could change based on small changes in behavior and cooperation.
Our results are consistent with previous reports in smaller patient groups, providing further credence to their accuracy. DMD is a heterogeneous disease, and our sample size did not allow for variant-specific analysis of outcomes. Initiation of corticosteroids at an early age is standard of care in our clinic; therefore, we do not have sufficient data to present the corticosteroid naïve patient group separately. It is possible that older corticosteroid naïve boys may present to our clinic with a later age at diagnosis due to a milder disease course. This has the potential to confound results as scores could be artificially inflated this group.
While we believe this manuscript describes the largest sample of gross motor skills in young boys with DMD, we acknowledge the limitation of using a sample of convenience. Timing of baseline and follow up visits was not standardized as patients were evaluated at their regularly scheduled clinic appointments. Some subjects traveled to NCH for a one-time second opinion and returned to their local DMD clinic thus limiting follow-up. A formal study enrolling subjects under the age of one year with scheduled longitudinal timed follow up is required to provide scientific rigor to our anecdotal findings. It would also add the scientific literature if all the Bayley subtests were included in future studies. This would provide information on fine motor skills, receptive and expressive language, and cognition in addition to our work on gross motor development.
Another limitation with our paper is that we used the Bayley version 3, and a fourth edition of the Bayley (Bayley-4) was published in 2019. 19 While the Bayley-4 includes revised items and introduced a polytomous scoring system, it retains the five-subtest framework of the Bayley-III and was developed to allow comparability between the two editions.20,21 While raw scores are no longer directly comparable, scaled scores (as a measure of performance compared with age-matched peers) are, with the mean and standard deviation the same across the two tests and a correlation coefficient of 0.73. 21
In conclusion, the Bayley-III can be effectively used in young boys with DMD to quantify gross motor delays. Our data provide a historical control patient group for clinical counseling about gross motor development in DMD and for clinical trial interpretation by establishing whether treated patient groups perform differently than typical DMD maturational expectation.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602241289223 - Supplemental material for Gross motor delays in infants and young boys with Duchenne muscular dystrophy
Supplemental material, sj-docx-1-jnd-10.1177_22143602241289223 for Gross motor delays in infants and young boys with Duchenne muscular dystrophy by Linda P. Lowes, Natalie F. Reash, Megan A. Iammarino, Anne M. Connolly, Lindsay Pietruszewski, Melissa A. Smith, Jing Peng, Christopher L. Steiner, Chang-Yong Tsao, Megan A. Waldrop, Kevin M. Flanigan, Shannon Chagat, Alayne P. Meyer, Jerry R. Mendell and Lindsay N. Alfano in Journal of Neuromuscular Diseases
Footnotes
Abbreviations
Acknowledgements
The authors would like to thank the staff, patients and families at the Nationwide Children's Hospital MDA and Neuromuscular Clinic.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
