Abstract
Acute lymphoblastic leukemia (ALL) is a blood cancer that is characterized by the overproduction of lymphoblasts in the bone marrow. Treatment for pediatric ALL typically uses combination chemotherapy in phases, including a prolonged maintenance phase with oral methotrexate and 6-mercaptopurine, which often requires dose adjustment to balance side effects and efficacy. However, a major challenge confronting combination therapy for virtually every disease indication is the inability to pinpoint drug doses that are optimized for each patient, and the ability to adaptively and continuously optimize these doses to address comorbidities and other patient-specific physiological changes. To address this challenge, we developed a powerful digital health technology platform based on phenotypic personalized medicine (PPM). PPM identifies patient-specific maps that parabolically correlate drug inputs with phenotypic outputs. In a disease mechanism–independent fashion, we individualized drug ratios/dosages for two pediatric patients with standard-risk ALL in this study via PPM-mediated retrospective optimization. PPM optimization demonstrated that dynamically adjusted dosing of combination chemotherapy could enhance treatment outcomes while also substantially reducing the amount of chemotherapy administered. This may lead to more effective maintenance therapy, with the potential for shortening duration and reducing the risk of serious side effects.
Introduction
Acute lymphoblastic leukemia (ALL) is the most common cancer in children. There have been substantial improvements in overall survival in the last 50 years, with 5-year survival rates for ALL approaching 80%–90%.1–5 These enhanced treatment outcomes were achieved through effective combination chemotherapy, prophylaxis against central nervous system (CNS) disease, and improvements in postinduction therapy. Despite these important advances, relapsed ALL is the leading cause of death by cancer in children.6–8
Pediatric ALL chemotherapy consists of treatment phases that last 2–3 years from diagnosis. The goal of the prolonged maintenance phase is to prevent disease relapse and includes recurring cycles of oral steroids, 6-mercaptopurine (6MP), and methotrexate, as well as intravenous vincristine and intrathecal methotrexate. Dosing of 6MP and methotrexate is based on body surface area and clinical side effects, including target absolute neutrophil counts (ANCs) greater than 500/µL but less than 1500/µL and platelet counts greater than 50,000/µL. Many patients require ongoing dose adjustments of 6MP and methotrexate in order to avoid myelosuppression, which can cause severe infection and bleeding, as well as to adequately maintain remission and prevent disease relapse.
6MP is methylated by thiopurine methyltransferase (TPMT), which results in its inactivation. 9 TPMT activity is known to vary widely due to common polymorphisms in TPMT, as well as other genes.10–13 Patients with intermediate or deficient activity of TPMT are more likely to experience thiopurine toxicity, whereas those with high TPMT activity are at higher risk of leukemia relapse.14–17 While some patients who have significant myelosuppression during maintenance undergo thiopurine pharmacology testing, assessing all patients requiring 6MP therapy is not yet standard practice. Furthermore, there are multiple germline variants influencing methotrexate pharmacokinetics, such as SLCO1B1 and MTHFR,18,19 which are not routinely tested for and incorporated in the management of pediatric ALL patients. As such, a platform technology that can dynamically and continuously optimize combination therapy outcomes in a patient-specific manner without requiring explicit information pertaining to complex disease mechanisms (e.g., genomics, proteomics, and pharmacokinetics) would be a major advance in ALL therapy.
The consequence of suboptimal combination therapy is a challenge that has confronted nearly every discipline of medicine, ranging from oncology to infectious diseases and cardiovascular medicine, among many others. Previous studies have identified promising approaches toward multidrug optimization, in the context of both drug development and individualizing patient treatment. However, these approaches often utilize drug pairing to predict the efficacy of multidrug combinations, which precludes absolute and dynamic optimization in patients. To overcome this major barrier, we have developed a powerful, clinically validated digital health technology platform based on the field of phenotypic personalized medicine (PPM).20–28 PPM effectively calibrates the patient response to therapy to construct a parabolic map that is unique to each patient. PPM does not require algorithms, predictive modeling, or machine learning, and is capable of dynamically optimizing therapy for the entire duration of care. This is particularly important, since drug synergism and antagonism are dose dependent, and can vary within the same patient over the course of treatment. 29 This study examined the administration of four-drug maintenance therapy regimens (dexamethasone, vincristine, mercaptopurine, and methotrexate) in two patients. The doses of these drugs served as the inputs, while maintaining ANCs and platelet counts within target ranges served as the outputs for optimization. We utilized PPM to construct personalized parabolic response maps for each patient to retrospectively optimize their regimens to successfully eliminate ANC and platelet deviations outside of their respective target ranges. Of note, this study did not involve conventional retrospective analysis or meta-analysis. Instead, PPM-mediated retrospective optimization utilizes patient clinical data coupled with a powerful parabolic correlation process to definitively pinpoint patient-optimized combination therapy administration drug–dose ratios. PPM is not limited by the number of drugs that can be optimized. Therefore, both four-drug and two-drug (6MP and methotrexate) optimization regimens were identified as a demonstration of disease biology and mechanism-independent multidrug optimization. In addition, patient-specific drug response maps were constructed, revealing individualized drug synergism/antagonism that was dose dependent. Finally, analysis of the contribution of drug–drug interaction terms (ITA) and second-order terms (SOT) was conducted in parallel with clinical and PPM-optimized dosing regimens, revealing the potential relevance of maintaining threshold ITA levels balanced with the first-order terms (FOT) and SOT to eliminate target ANC and platelet count deviations. These clinical data-backed readouts provide actionable guidance to maintain patient-specific and dynamically optimized combination therapy regimens for the entire duration of treatment. As such, PPM represents a powerful and broadly applicable digital health technology platform to optimize treatment for a broad array of disorders.
Materials and Methods
Acute Lymphoblastic Leukemia Patient Data
This study was conducted under approved Institutional Review Board (IRB) protocol number IRB#16-000723. Data obtained included drug dosages for dexamethasone, 6MP, vincristine, and methotrexate and corresponding time points (e.g., patient 1, days 28, 57, 85, 113, 148, 176, 211, 242, 270, 302, and 330; patient 2, days 29, 57, 85, 113, 141, 169, 199, 222, 250, 278, and 305). ANCs and platelet counts corresponding to the respective drug dosing dates were also identified for optimization.
Phenotypic Personalized Medicine–Based Optimization
To identify PPM-optimized chemotherapy dosages, a patient-specific second-order polynomial map was constructed using variables, including drug dosages, ANCs, and platelet counts. In addition, individualized second-order effects and ITA were assessed in correspondence with measured and PPM-optimized ANC and platelet values. PPM two- and three-dimensional drug interaction maps were plotted using MATLAB R2014a (MathWorks, Inc., Natick, MA) with a matrix input of drug concentration values correlated to platelet counts or ANCs. 29
Results and Discussion
Clinical Optimization of Combination Therapy Using Mechanism-Independent Digital Health Technology
The implementation of PPM is based on the foundation that a patient’s phenotypic response (output) to drug treatment (input) can be represented by the parabolic response surface. Phenotypic responses can include tumor burden, bacterial/viral load, international normalized ratio (INR), serum toxicity indicators (e.g., myelosuppression and serum alanine aminotransferase), or a combination of these readouts. This parabolic response map is defined by patient-specific constants in a quadratic expression ( Fig. 1 ). 29 This holds true for all patients and all indications. The significance of the parabolic response surface is based on its ability to identify exactly where the best dosing parameters exist at any given time during treatment. PPM allows this response surface to be continually optimized during care as regimen changes occur, or the patient undergoes additional procedures. Furthermore, this phenotypic map implicitly incorporates mechanistic components that drive treatment outcomes, such as disease biology, genetics, proteomics, and pharmacokinetics. Therefore, explicit knowledge pertaining to these elements is not required to continuously mediate optimization.
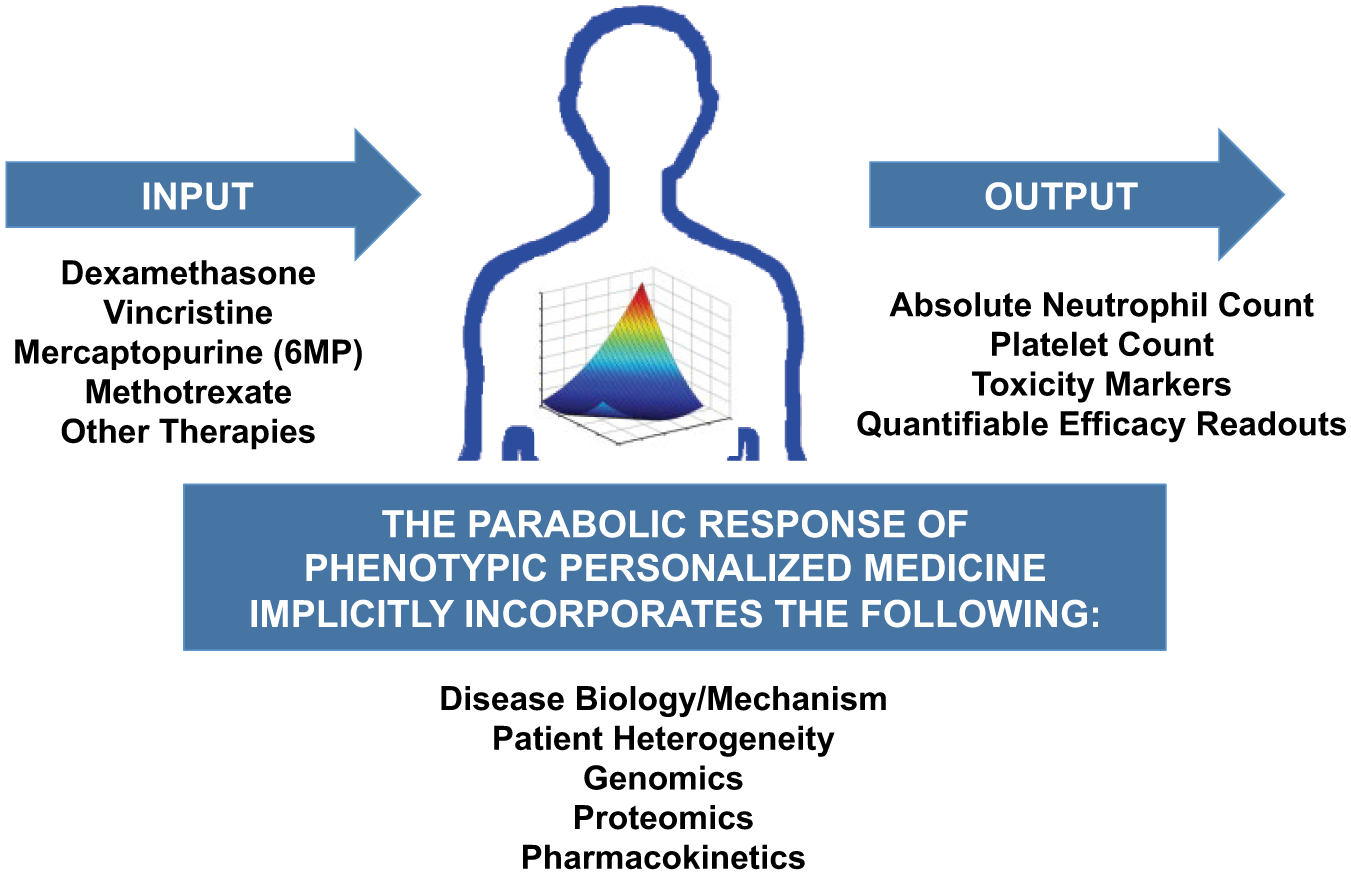
Parabolic medicine implicitly incorporates mechanistic information such as genomics, proteomics, pharmacokinetics, and other components of disease biology. Explicit information pertaining to these factors is not necessary to optimize personalized combination therapy regimens using PPM. The primary components of PPM are the inputs, comprised of the therapies, and the outputs, which are quantifiable indicators of treatment efficacy and safety.
Dynamic Optimization of Personalized Combination Therapy: Four Drugs
Conventional maintenance therapy involves the modulation of 6MP and methotrexate in order to maintain ANCs and platelet levels within target levels. Titration is often used as the primary means of adjusting the doses of these two therapies. However, clinical titration has shown that the proper dosing of even one drug can be challenging. 29 Therefore, due to barriers that prevent rational dosing of multiple drugs, let alone optimized multidrug administration, a technology platform such as PPM may be particularly important for improving response rates in oncologic indications.
Since PPM is not limited by the number of drugs in a regimen, we conducted a four-drug optimization assessment for two patients. For patient 1, clinical chemotherapy administration resulted in substantial deviation from the ANC target range on day 85, and deviations on days 242 and 302, and platelet levels that remained within the target range. In contrast, PPM-optimized administration of dexamethasone, 6MP, vincristine, and dexamethasone eliminated the ANC deviations while also maintaining normal platelet levels ( Fig. 2 ). In comparing the clinical and PPM-optimized administration of each drug, PPM optimization resulted in substantially lower dexamethasone administration than clinical dosing. In fact, PPM-optimized dexamethasone and 6MP dosages were approximately fourfold lower than the clinically administered dosages. Initial vincristine dosing was higher for the PPM-optimized regimen than for clinical dosing. However, PPM optimization identified substantially lower dosages of vincristine than clinical administration starting on day 211 (with the exception of day 270) through the end of treatment that eliminated ANC deviations. PPM suggested that 6MP dosing resulted in sharply lower drug dosages than clinical administration starting on day 148, while PPM-guided methotrexate dosing was sharply lower than clinical dosing parameters on day 302.
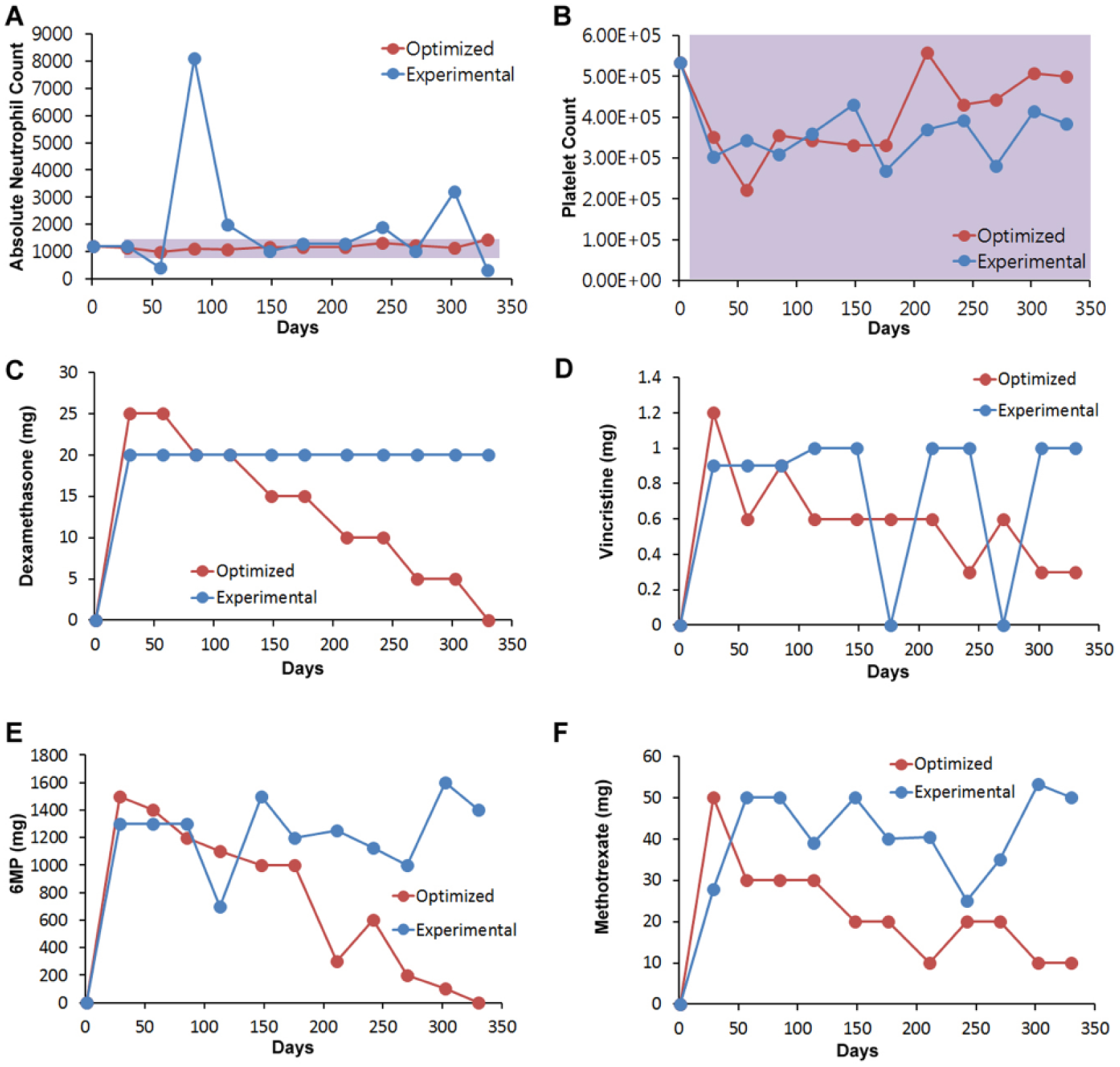
PPM-optimized and clinically administered four-drug regimens. (
In patient 2, clinical chemotherapy resulted in a substantial deviation from the ANC target range that spanned from day 141 to 169, while platelet levels remained within the target range during the course of treatment. To eliminate this ANC deviation, PPM optimization resulted in a steady decrease in dexamethasone dosing over time compared with clinical administration that alternated between dosage increases and decreases. PPM-suggested vincristine dosages alternated between being higher and lower than clinically prescribed dosages. However, PPM-optimized 6MP and methotrexate dosages were notably lower than the clinically administered dosages for virtually the entire duration of care ( Fig. 3 ).
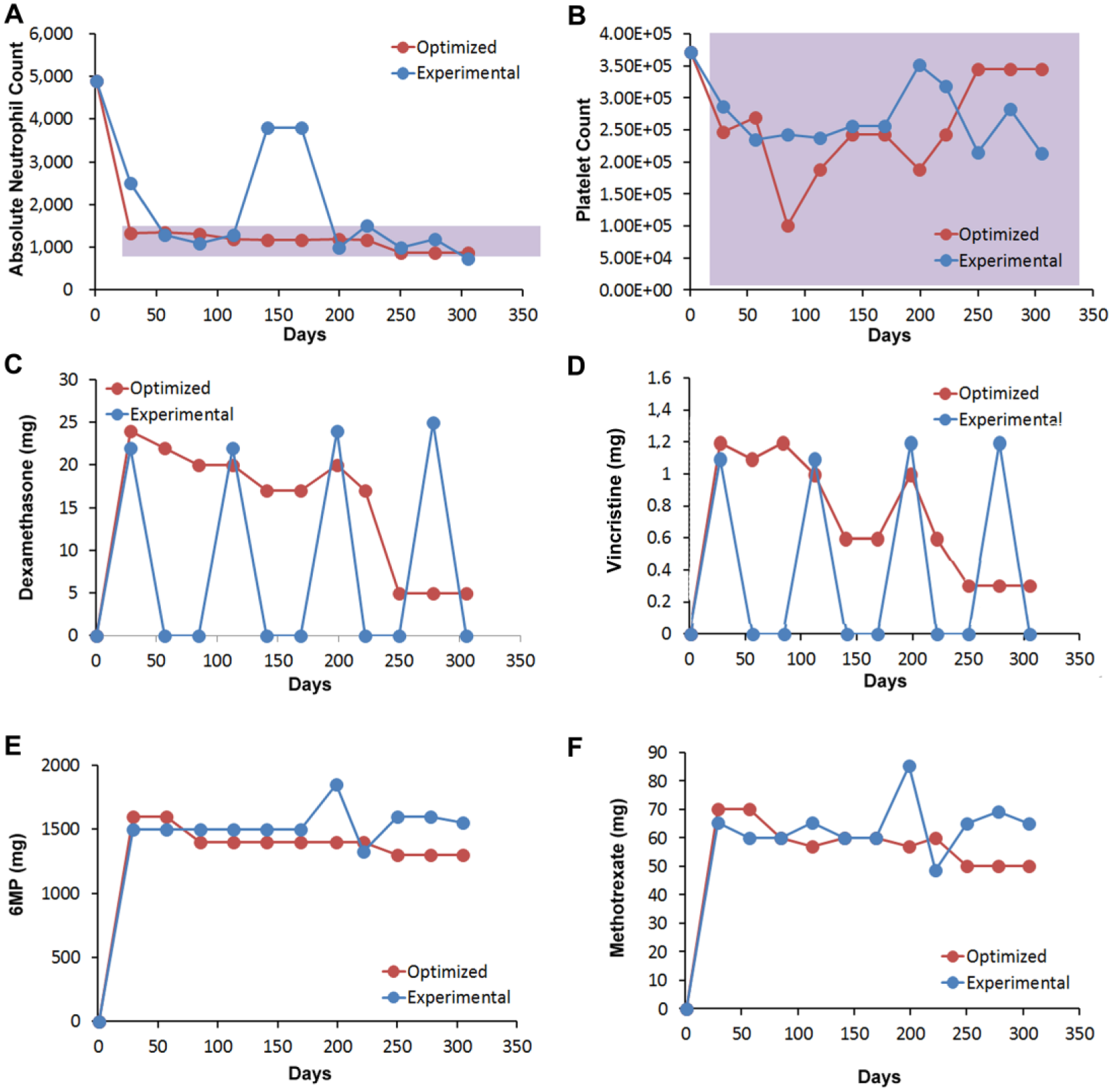
(
While four-drug modulation is currently not utilized for ALL maintenance therapy, this optimization study demonstrated that PPM is uniquely capable of dynamically personalizing the administration of all four drugs to optimize dosing to prevent ANC deviations for the entire duration of care.
Dynamic Optimization of Personalized Combination Therapy: Two Drugs
A two-drug PPM optimization study to modulate 6MP and methotrexate was also performed ( Fig. 4 ). In patient 1, the ANC fluctuated outside of the target range while the platelet levels remained within range during care ( Fig. 4A , B ). In patient 1, the elimination of ANC deviations was achieved by 6MP doses, which, with the exception of days 113 and 242, where PPM recommended a higher dose, as well as a comparable dose at day 270, were notably lower than the clinically administered dosages ( Fig. 4C ). PPM-determined methotrexate dosages were also notable lower for a vast majority of the treatment period ( Fig. 4D ).
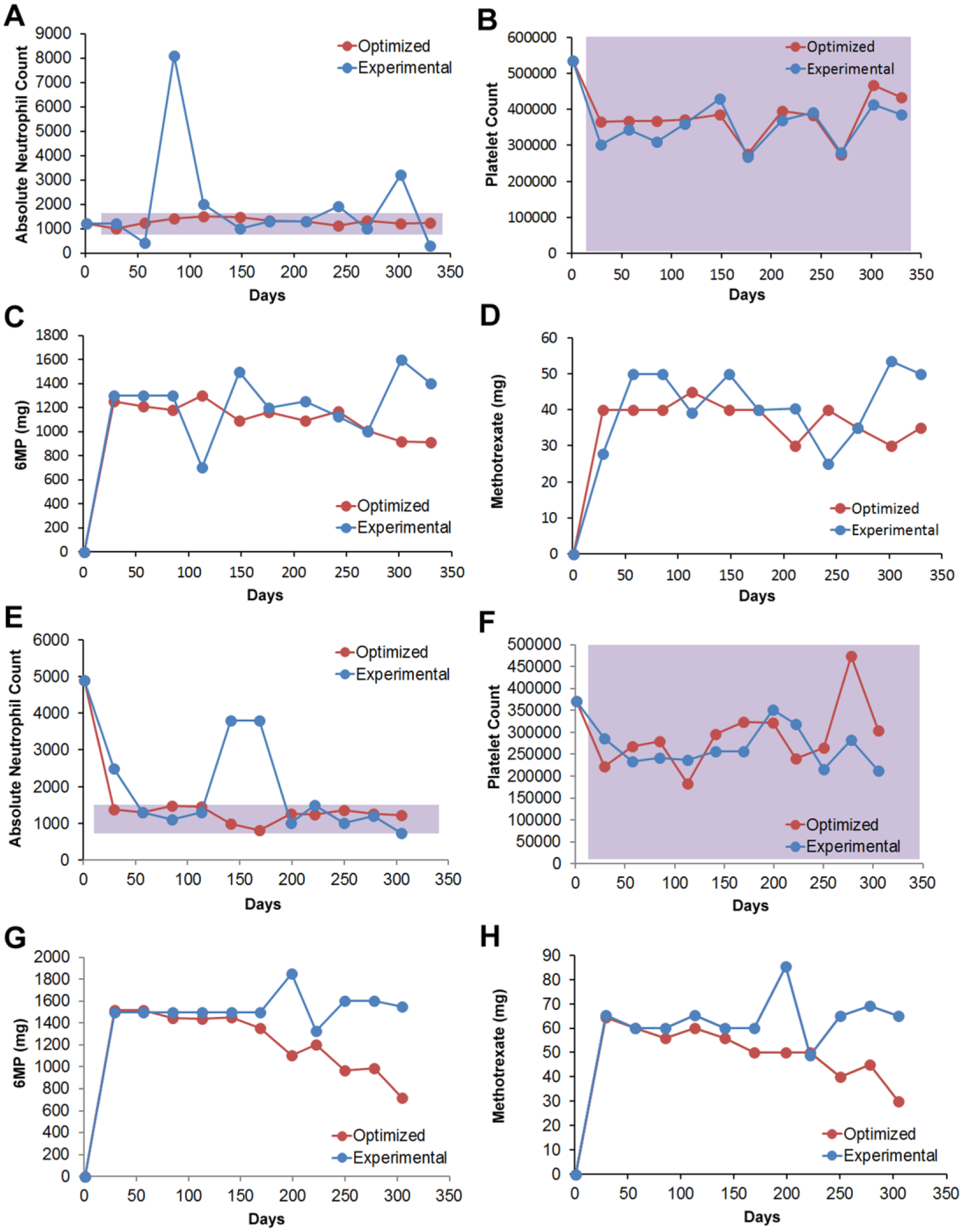
PPM-optimized and clinically administered two-drug regimens. (
Patient 2 also had ANC fluctuations outside of the target range, while the platelet levels remained within the target range ( Fig. 4E , F ). In this patient, the disparity in dosing recommendations was more apparent between the PPM-determined and clinically administered protocols. For both 6MP and methotrexate, the PPM-optimized dosages were lower than the clinically administered dose for virtually the entire duration of care ( Fig. 4G , H ). In some cases, the PPM-determined dose was approximately 40% lower than the clinical dose.
Drug Interaction Mapping
Drug interaction mapping for both two- and four-drug modulation was conducted to visually correlate drug dosing with target ANC levels. The substantial variations in drug interactions and resulting outputs between the patients are clear indicators of the need to personalize and continuously optimize treatment. For both patients, ANCs were correlated with 6MP–methotrexate, 6MP–dexamethasone, and methotrexate–dexamethasone interactions. Since the maps are based off of clinical data, they are not predictive and provide a deterministic strategy to assess optimal drug–dose ratios. Drug interaction mapping correlated to ANC using four-drug modulation was performed for both patients 1 and 2 for the aforementioned interactions. These studies identified the range of drug–dose ratios along the phenotypic landscape that enabled the patient to stay within the target ANC range (greater than 500/µL but less than 1500/µL, Fig. 5A–F , blue region).
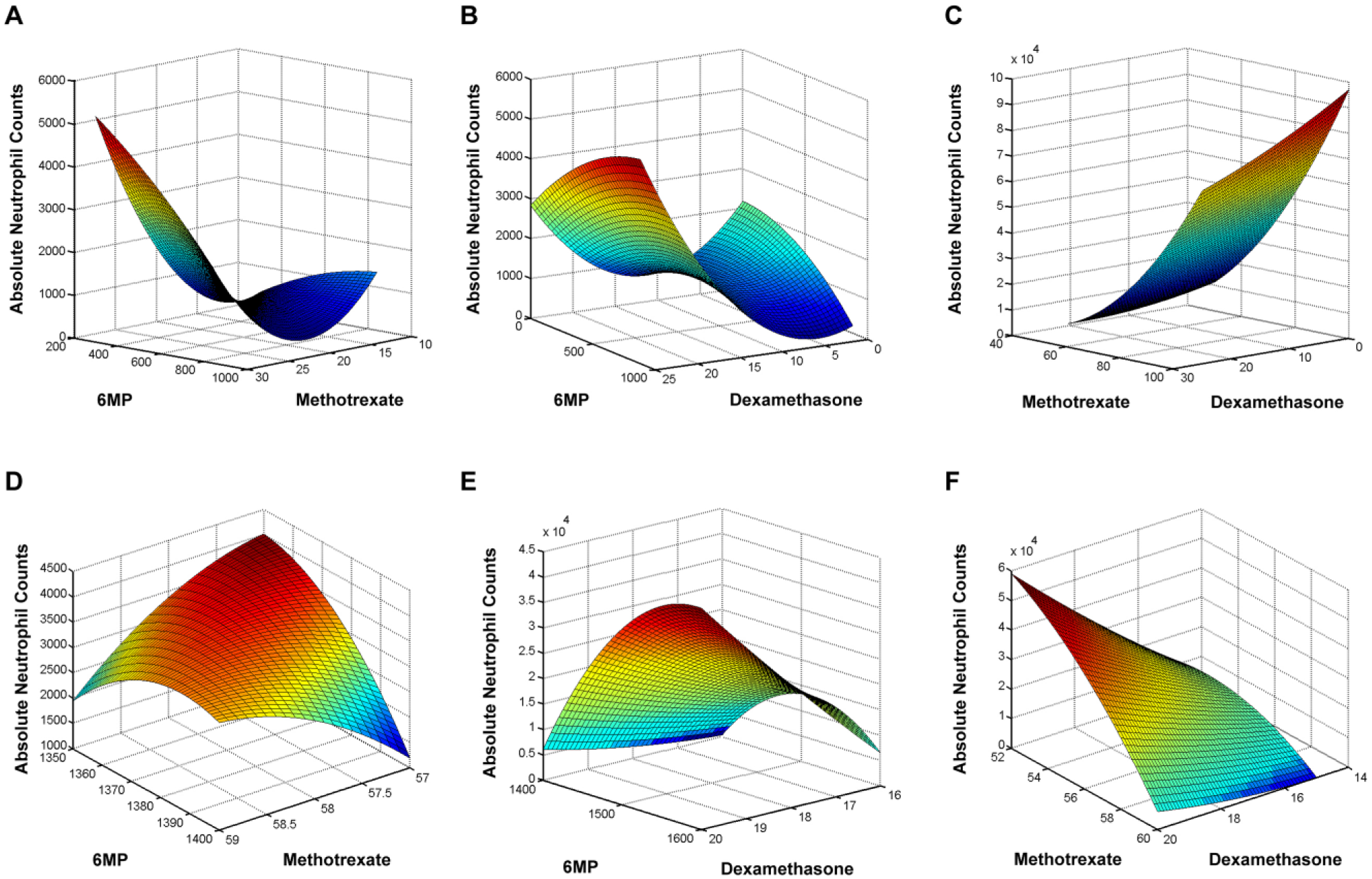
Phenotypic mapping to correlate 6MP–methotrexate, 6MP–dexamethasone, and methotrexate–dexamethasone interactions with ANCs using four-drug modulation. (
For two-drug modulation in both patients, 6MP and methotrexate were correlated with ANC output, with the maps clearly showing a clear difference in drug interaction behavior between patients 1 and 2 ( Fig. 6A , B ). This was a demonstration of the importance of personalizing care in ALL. For patient 1, a broad range of 6MP doses was capable of maintaining the patient within the ANC target range. A narrower range of methotrexate doses resided within the ANC target ( Fig. 6A ). The methotrexate dosing spectrum exhibited a clear dose-dependent fluctuation from the ANC target range at the lower and higher dosing domains, further demonstrating the importance of pinpointing the right patient-specific doses. For patient 2, a narrow range of doses for both 6MP and methotrexate were required to maintain the ANC target range ( Fig. 6B ). This was also a clear indicator for the need to optimize personalized care, since titration outside of this range would have led to substantial deviations from the ANC target range.
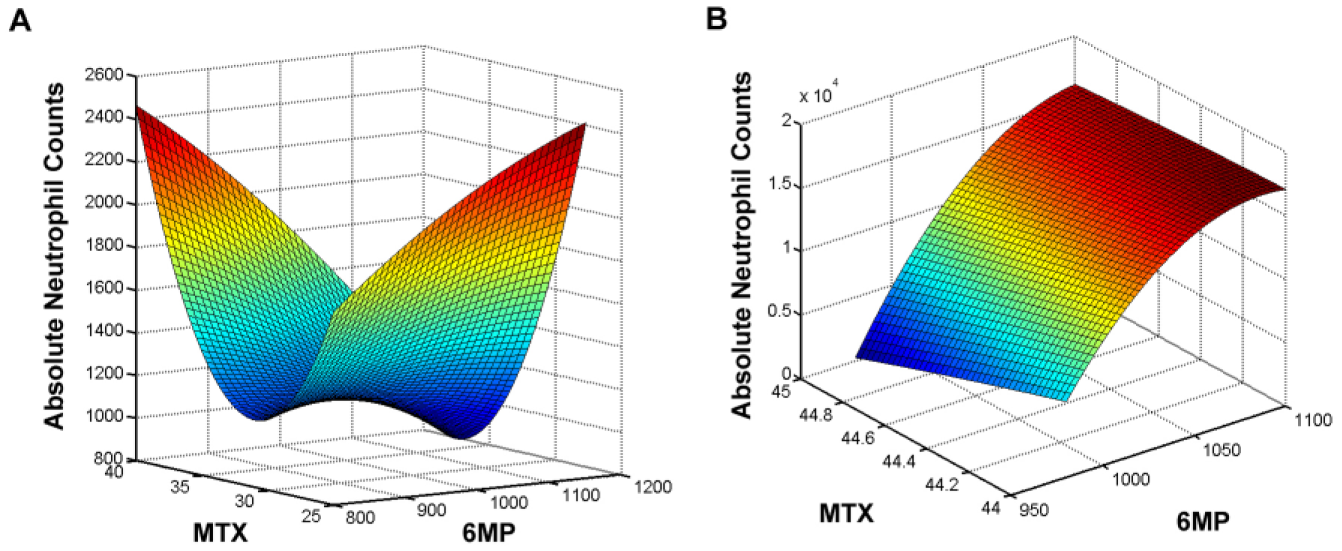
Phenotypic mapping to correlate 6MP–methotrexate interactions with ANCs. (A) Patient 1 mapping. (B) Patient 2 mapping.
Drug Interaction Analysis
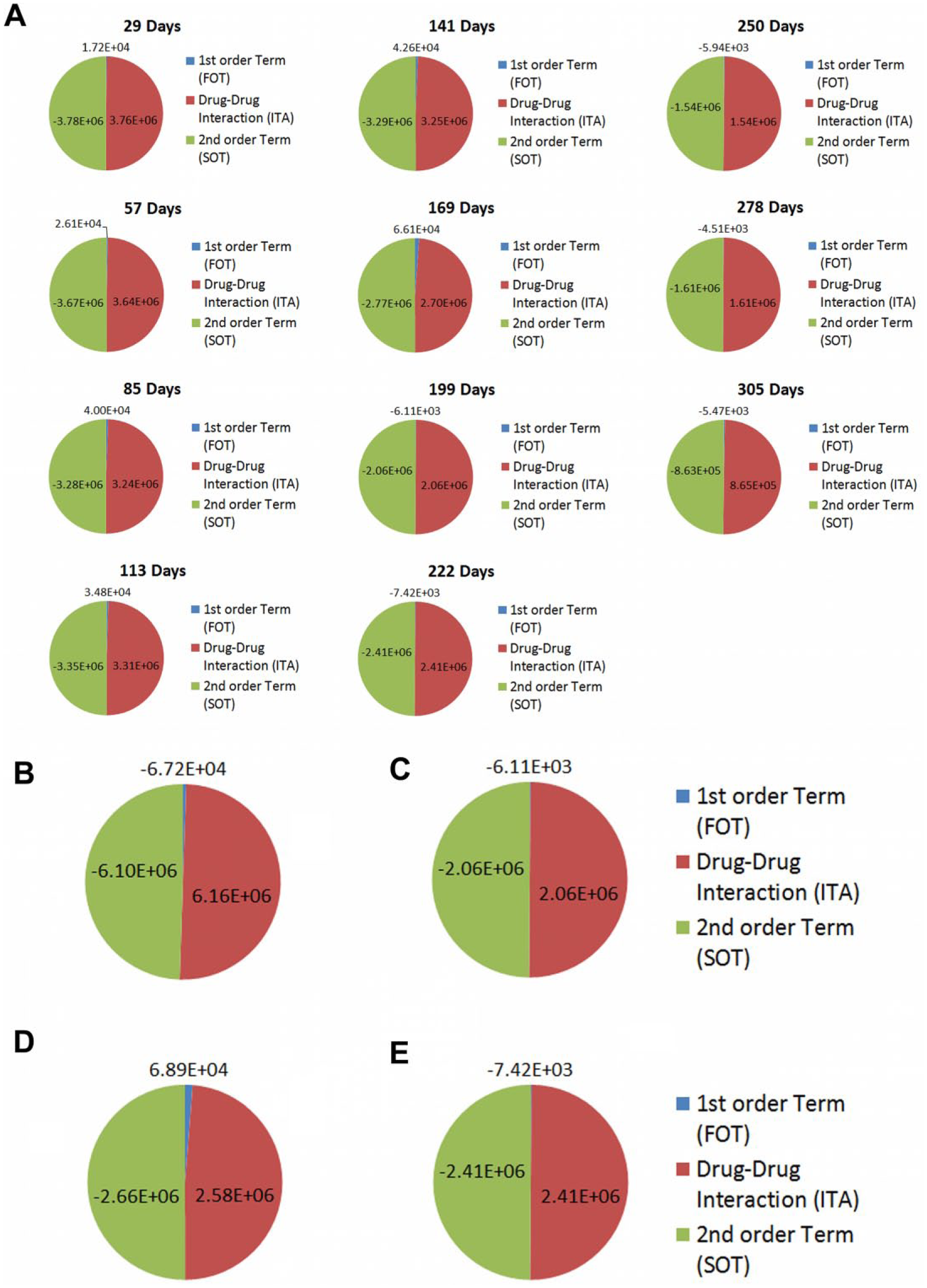
Quantitative drug interaction analysis was conducted to assess the contribution of FOT, ITA, and SOT toward patient treatment outcomes. More specifically, this analysis assesses the extent to which the phenotypic outcome is mediated by FOT, where one drug molecule is interacting with the target; SOT, where two of the same drug molecules are interacting with the target; or ITA, where two different drug molecules are interacting with the target. When substantial deviations from target ranges occur in the clinic, drug interaction analysis assesses which terms are playing a role in these deviations. As such, the application of PPM to optimize drug–dose ratios/therapeutic inputs and resulting treatment outcomes pinpoints the patient-specific FOT, ITA, and SOT profiles, providing key insight into the contributing factors that are regulating maximal efficacy and safety on an individualized basis. Patient 1 interaction analysis was plotted for clinician-guided therapy ( Fig. 7 ) and PPM-optimized therapy ( Fig. 8A ). When ANC levels deviated from the target range on days 57 and 85, a noticeable reduction was observed in the ITA, demonstrating the importance of balancing the FOT, ITA, and SOT that maintain target ANC levels. This is further illustrated when a direct comparison of the FOT, ITA, and SOT are made between the aforementioned clinician-guided and PPM-optimized regimens on day 242, where another deviation from the target ANC range was observed during clinician-guided care ( Fig. 8B , C ).
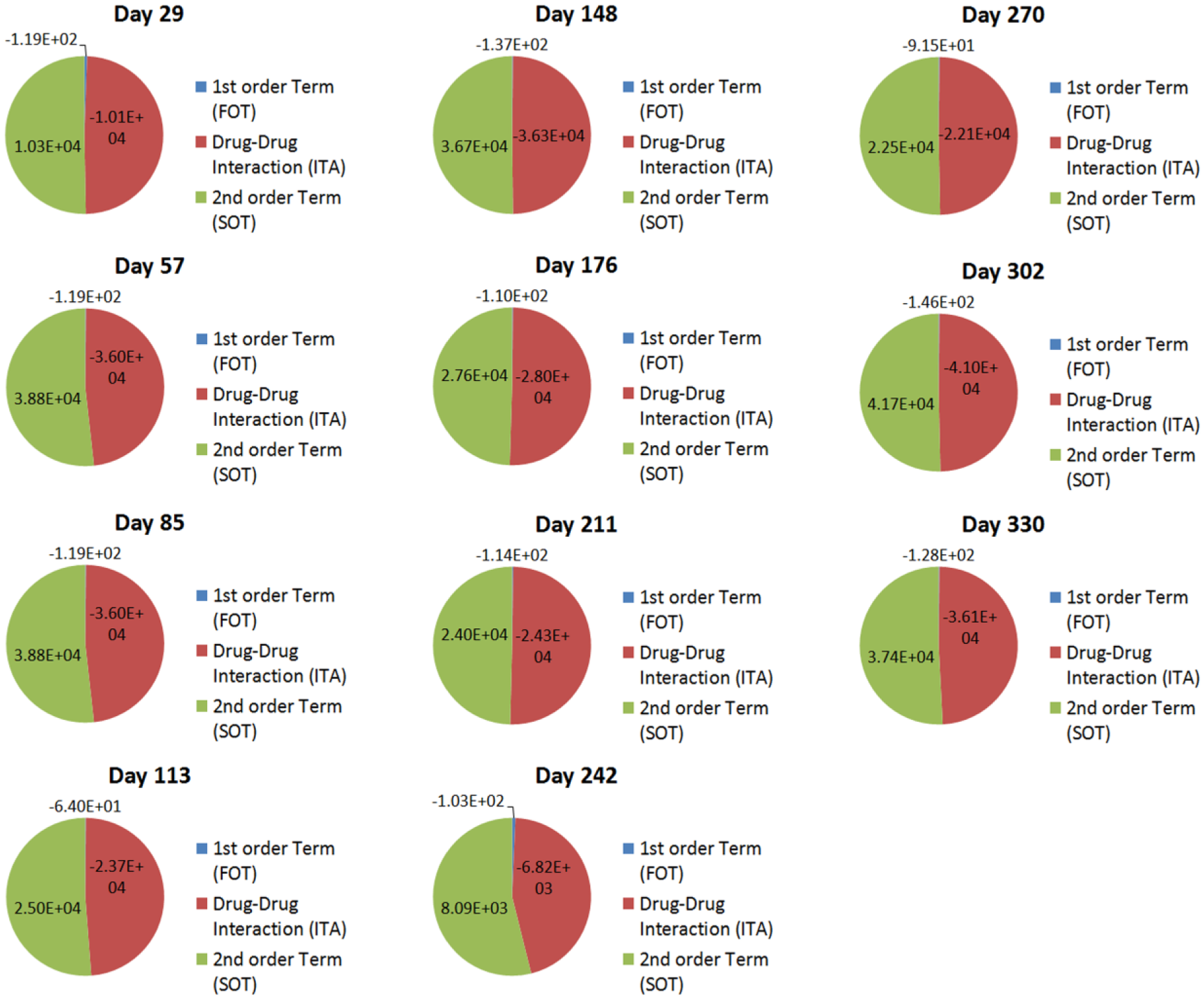
ITA, FOT, and SOT contributions toward clinically observed treatment outcomes for patient 1.
(
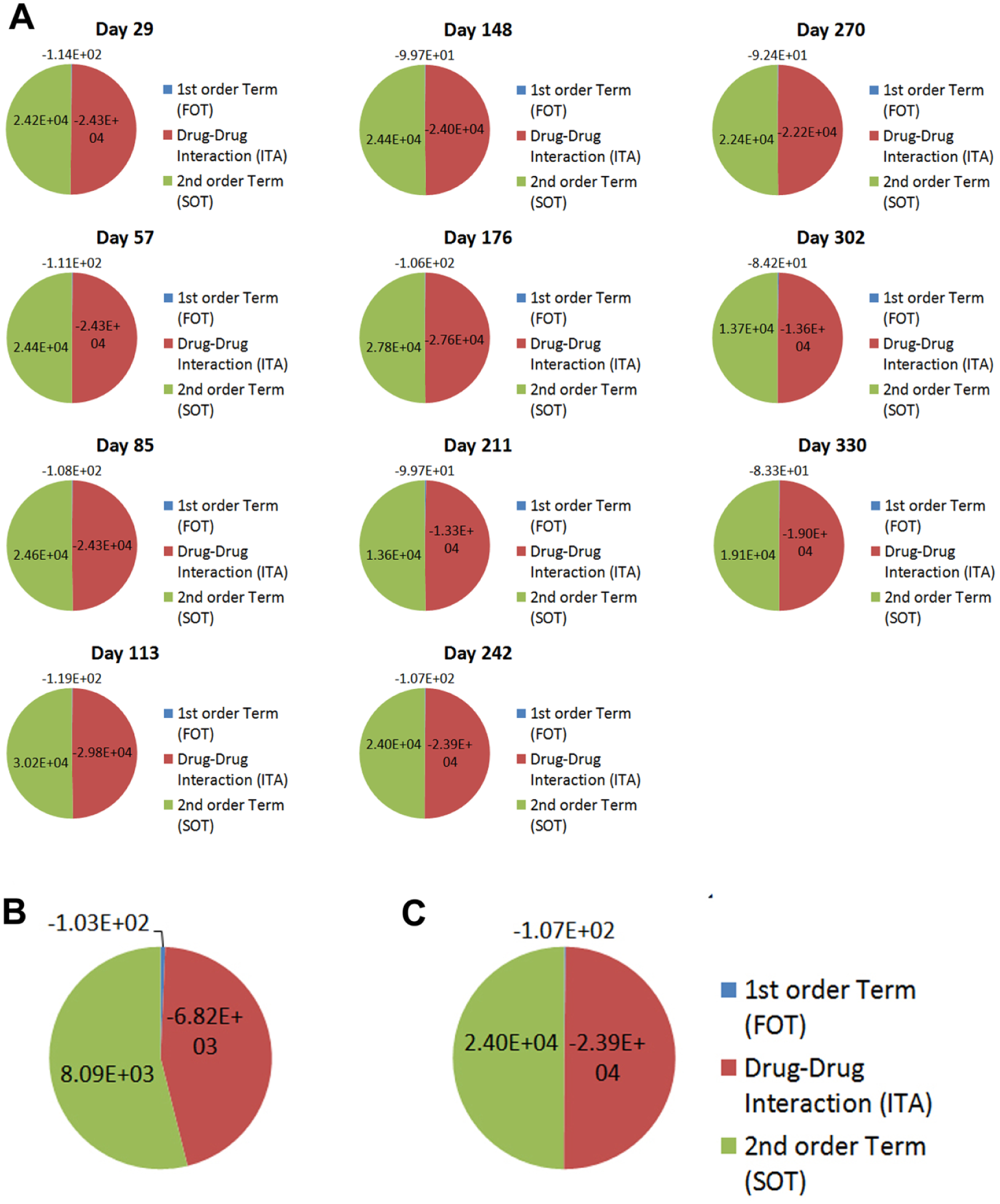
For patient 2, a reduction in ITA from days 141 to 169 was observed that correlated with a substantial deviation from the target ANC range under clinician-guided therapy ( Fig. 9 ). PPM-guided interaction mapping resulted in modest shifts in the FOT, ITA, and SOT values ( Fig. 10A ). This further indicates the importance of balancing FOT, ITA, and SOT values in order to maintain target ANC levels. Further examination shows that modest shifts between the FOT, ITA, and SOT are observed between the clinician-guided and PPM-optimized regimens on days 199 ( Fig. 10B , C ) and 222 ( Fig. 10D , E ). Specifically, a reduction in FOT toward the maintenance of threshold ITA levels is observed for days 199 and 222. Importantly, the modulation of FOT, ITA, and SOT were revealed to be patient specific, demonstrating the need for continuous and dynamic optimization of combination therapy administration for the entire duration of care in ALL. Future prospective studies coupled with PPM-guided optimization will likely result in more apparent shifts in the FOT, ITA, and SOT that are coupled with multidrug optimization. This will provide patient-specific insight into the directed/clinical modulation of these terms to prevent deviations from ANC target ranges, potentially resulting in lower drug dosages required to maintain efficacy, with reduced acute and long-term side effects.
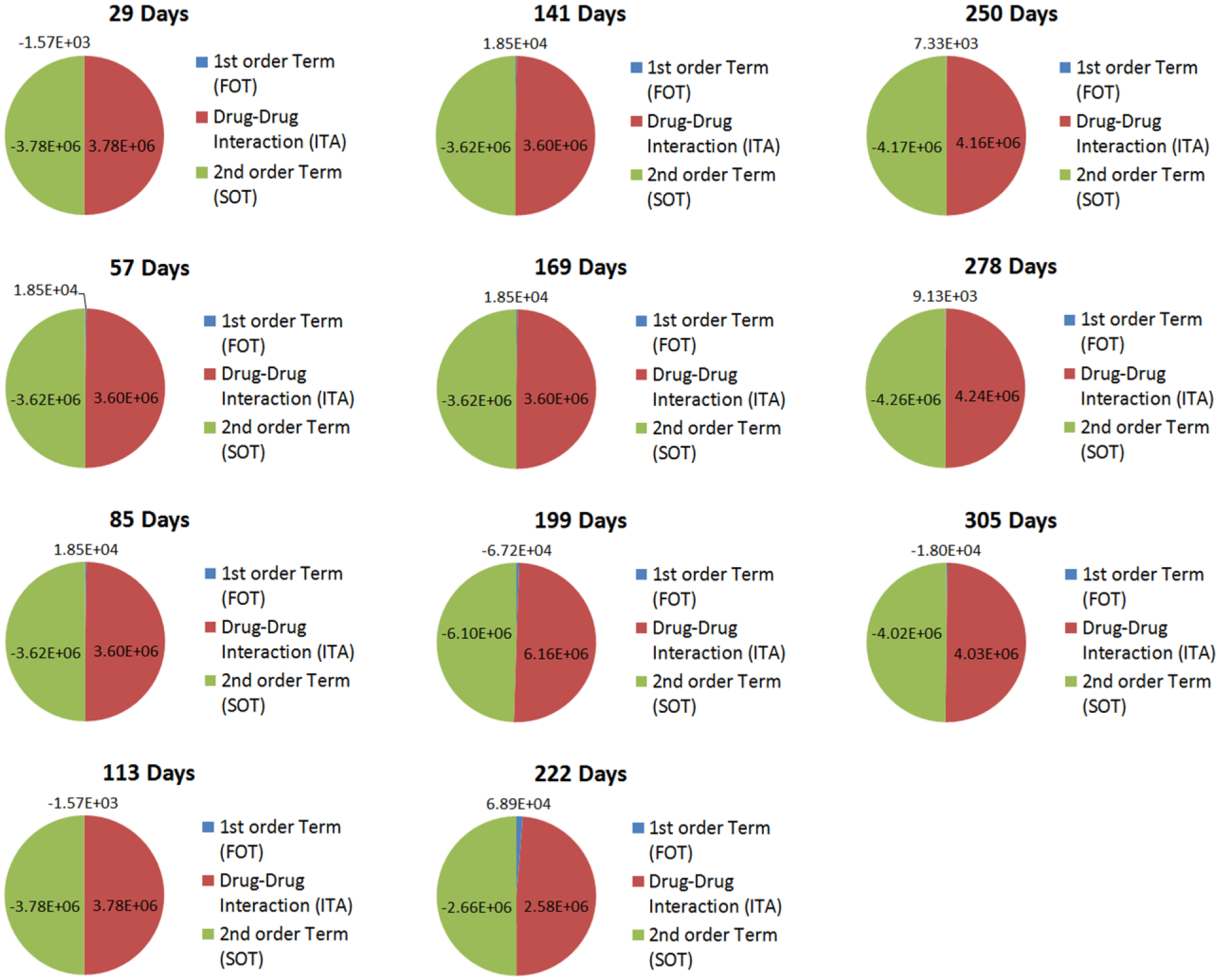
ITA, FOT, and SOT contributions toward clinically observed treatment outcomes for patient 2.
(
Impact of Parabolic Medicine on Optimized Drug Development and Personalized Therapy
The clinical management of combination therapy is challenging, given the infinite dosing space in which patient-specific drug administration parameters reside. This further confounds the identification of these parameters since patient physiology changes over time as new drug regimens are introduced to address comorbidities, or additional procedures are administered (e.g., bone marrow transplants). These factors have made the optimization of patient-specific combination therapy, at a specific time point, or during the entire course of care, virtually impossible.
The inability, until now, to pinpoint drug–dose ratios in combination therapy is a problem that spans both drug development and individualized care. 30 In the context of drug development, this is due to the fact that optimized dosing parameters can also exist on a universal, or population-based, scale. This makes population-optimized combination therapy possible. However, current drug development strategies preclude the design of population-optimized therapy. More specifically, current combination therapy strategies use high-throughput screening to identify lead compounds. These compounds are then evaluated further to assess efficacy and safety. Designing combination therapies or monotherapies subsequently involves dose escalation toward maximum tolerated dose (MTD)–based additivity. In addition, drug synergy–based prediction serves as a conventional strategy to further enhance treatment outcomes. However, the study reported here, as well as others based on PPM, demonstrates that pinpointing drug dose can have a profound impact on drug synergy and antagonism, and that dosing outside of optimal ranges can render combinations that were predicted to be synergistic ineffective. At the same time, less effective or ineffective combinations can be rendered maximally efficacious through drug–dose optimization via PPM. Therefore, dose escalation, prediction-based design, conventional drug screening, or the use of disease mechanism as the sole means of determining which drugs to use in a combination therapy implicitly precludes the optimization of drug–dose ratios and identification of the drugs that comprise the best therapeutic formulations. These approaches serve as major drivers of the exorbitant costs and high failure rates of drug development. Therefore, PPM demonstrates that drug–dose optimization profoundly impacts drug selection, and that both must be optimized simultaneously to realize maximal therapeutic efficacy and safety in the clinic.
As this study and others demonstrate, PPM overcomes these aforementioned challenges, eliminating the use of brute force to assess both the dosing and drug selection space. By substantially reducing bias during the initial drug development process, we can interrogate the entire drug–dose and selection space to rapidly optimize both attributes that will mediate maximal efficacy and safety. To overcome the burden of suboptimal dosing and combinatorial drug selection, PPM reoptimizes at each stage of development, where in vitro optimization refines the list of lead drug combination candidates. Drug–dose ratios and drug selection are reoptimized at the preclinical stages, and as this study and a recent prospective clinical show, inpatient optimization can be realized as well. 29 In lieu of brute-force screening, parabolic medicine substantially accelerates these processes. Population and personalized optimization collectively provide powerful information for drug–dose ratios that enable dramatic increases in efficacy and safety. This can be achieved using PPM-determined, novel fixed-dose combination therapy products for population-wide administration, as well as dynamically individualized combination therapy within a single patient for the entire duration of care. The implicit validation of these combinations during optimization and reoptimization has already made major strides toward improving clinical outcomes over conventional standards with dramatically accelerated developmental timelines.24–26,29,30
In the context of personalizing patient regimens, conventional approaches have included dosing algorithms, pharmacogenomics, pharmacokinetic modeling, and other strategies.31,32 However, due to substantial changes in patient physiology that are inevitably encountered during treatment from regimen changes (e.g., changes in drug dose and formulation from intravenous to oral administration) and other procedures (e.g., bone marrow transplant), dynamically optimized care that is not enabled by these aforementioned modalities is required. To overcome this challenge, PPM correlates patient response to therapy in a parabolic fashion that pinpoints, at all times, the best possible combination therapy regimen. PPM patient-specific dosing is particularly effective at co-optimization of combination chemotherapy and simultaneous immunosuppression following bone marrow transplantation, as it is not limited by the number of drug inputs. This ability to calibrate optimized and patient-specific regimens makes PPM broadly applicable toward oncologic, infectious disease, cardiovascular, wound healing, neurological, and a broad spectrum of other indications.
Conclusion
Maintenance therapy for ALL utilizes dexamethasone, vincristine, 6MP, and methotrexate in an effort to maintain treatment efficacy, as well as target ANCs and platelet levels. Using conventional titration, ANC levels often fluctuate outside of the target range, and can result in treatment complications. Furthermore, eliminating these fluctuations can often be achieved using drug dosages that are substantially different from those given in conventional clinical practice. This opens the doors to possibly reducing acute or long-term side effects due to the duration of maintenance therapy. Toward this objective, this study used PPM to retrospectively optimize combination therapy regimens by modulating all four drugs, or 6MP and methotrexate (clinical practice). While this was achieved using clinical data that had already been collected, prospective PPM can achieve profoundly improved patient outcomes based on the acquisition of a modest level of increased serum analysis compared with conventional clinical protocols. Therefore, when the prospective clinical procedure is readily adapted to PPM-based treatment optimization, obtaining the right data will redefine the way that patients are treated, forging a path toward substantially improving durable response rates.
Footnotes
Acknowledgements
The authors gratefully acknowledge the patients, families, and clinical staff at the Mattel Children’s Hospital at Ronald Reagan UCLA Medical Center, as well as the Division of Pediatric Hematology and Oncology.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.-K. Lee, T. Kee, C.-M. Ho, and D. Ho are inventors on multiple patents pertaining to PPM.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: V.Y.C. gratefully acknowledges support by the UCLA Children’s Discovery and Innovation Institute (CDI) Today’s and Tomorrow’s Children Fund (TTCF) Award. D.H. gratefully acknowledges support from the National Science Foundation CAREER Award (CMMI-1350197), Center for Scalable and Integrated NanoManufacturing (DMI-0327077), CMMI-0856492, DMR-1343991, OISE-1444100, V Foundation for Cancer Research Scholars Award, Wallace H. Coulter Foundation Translational Research Award, National Cancer Institute grant U54CA151880 (the content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health), Society for Laboratory Automation and Screening Endowed Fellowship, Beckman Coulter Life Sciences, and the American Academy of Implant Dentistry Research Foundation under grant 20150460.
