Abstract
The aim of the present study was to determine the relation between the simultaneous fasting plasma glucose level and HbA1c in a large population of patients presenting to the hospital, based on various measurement methods available for HbA1c. HbA1c levels of 162,210 patients presenting to various hospitals and laboratories were measured based on seven different systems, and at the same time, eAG levels were calculated based on HbA1c levels. The correlation coefficients (
Keywords
Introduction
The A1c-derived average glucose (eAG) study recommended translating HbA1c into estimated eAG equivalents for monitoring glycemic control. 1 However, there is an ongoing disagreement regarding the underlying assumption that HbA1c levels depend exclusively on long-term previous blood glucose concentration.2-6 Numerous studies have shown that biological variation in HbA1c is influenced by factors other than blood glucose concentration,7-10 suggesting that eAG might be a systematically biased estimate of self-monitored mean blood glucose.
An American Diabetes Association (ADA), European Association for the Study of Diabetes (EASD), and International Diabetes Federation (IDF) sponsored study, which was designed with an aim to provide a better mathematical relationship between HbA1c and eAG, included 507 subjects with type 1 and type 2 diabetes, and without diabetes from 10 international centers: 11 “eAG was calculated by combining weighted results from at least 2 days of continuous glucose monitoring performed four times, with seven-point daily self-monitoring of capillary glucose performed at least 3 days per week. The relationship between eAG and HbA1c based on linear regression analysis was: eAG (mg/dL) = (28.7*HbA1c) – 46.7.” 1 Given et al., however, suggested that mean plasma glucose levels corrected for measurement error were more appropriate than the linear regression model between HbA1c and eAG levels, and that the relevant correlation values could increase from 0.71 to approximately 0.88. 12
The studies on eAG have proved to be highly complex and difficult. In fact, HbA1c concentrations were analyzed based on four different methods in the present study. Given that there are more than a hundred methods currently available for measuring HbA1c, it is vital that they have been standardized to report the same (or at least a very similar) result for a single blood sample. The present study had two basic aims. The first was to obtain the rather difficult eAG calculation procedure by retrospective use of hospital data, and the second was to determine, based on multicentric data mining, whether the same was affected by the methodological differences in the seven most frequently used HbA1c measurement approaches.
Materials and Methods
Study subjects were recruited from seven centers in Turkey, according to the consensus protocol. A total of 162,210 patients with type 1 or type 2 diabetes, and without diabetes, between the ages of 20 and 70 years, were enrolled in the study. The data were obtained from patients with fasting glucose and HbA1c measured simultaneously during 12–24 months. This was because of the fact that the present study included multiple measurements of the same patients (median 2, min 1, max 6). HbA1c values were analyzed using Bio-Rad Variant II Turbo (Bio-Rad Laboratories, Hercules, CA), Tosoh G8 (Tosoh Corporation, Yamagata, Japan), ADAMS A1c HA-8180 (ARKRAY, Inc., Kyoto, Japan), Trinity (Primus) Boronate Affinity HPLC (Trinity Biotech Plc, Bray, Ireland), Chromsystems HPLC (Chromsystems Instruments & Chemicals GmbH, Gräfelfing, Germany), Tina-quant II immunoassay on Hitachi (Roche Diagnostics Corporation, Penzberg, Germany), and Abbott Architect immunoassay (Seradyn ReAGents, Abbott Diagnostic, Lake Forest, IL). Fasting plasma glucose measurements were performed based on hexokinase methods by using AU2700/5400 (Beckman Coulter, Inc., Indianapolis, IN), Cobas Integra and c8000 (Roche Diagnostics Corporation), and Architect c8000 (Abbott Diagnostic). The laboratory assays were approved by the National Glycohemoglobin Study Program and had intra- and interassay coefficients of variation lower than 2.5% for lower and higher values. In all the laboratories, glucose measurements employed the Centers for Disease Control (CDC) hexokinase reference method for glucose, and intra- and interassay coefficient of variation (CV) values were found to be lower than 2%.
The one-sample Kolmogorov–Smirnov test was used to evaluate the distribution of the variables. eAG was calculated based on each HbA1c level. Thereafter, the relation between eAG and plasma glucose was determined by correlation, regression analysis, and the concordance correlation coefficient (CCC). The correlation between HbA1c and serum glucose levels was examined using Pearson correlation tests. Microsoft Excel and MedCalc (MedCalc Software, Inc., Mariakerke, Belgium) software were used for analyses.
Results
Regression equations and coefficients, correlation and confidence intervals values regarding HbA1c, and fasting plasma glucose levels were presented for each method in
Figure 1
. The highest correlation between fasting plasma glucose and HbA1c was observed in Trinity Premier HPLC (
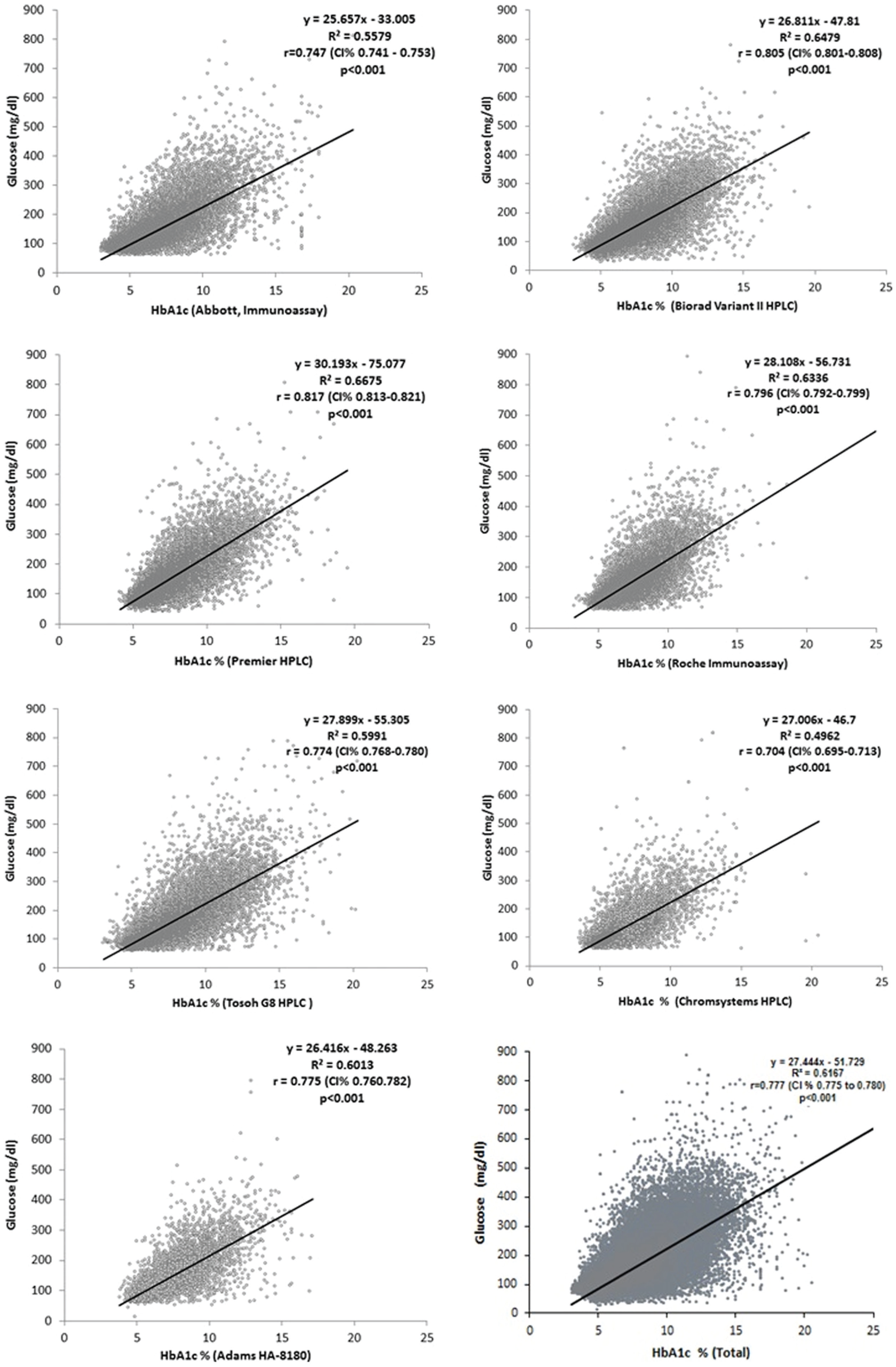
Regression equations and coefficients between HbA1c and plasma glucose levels.
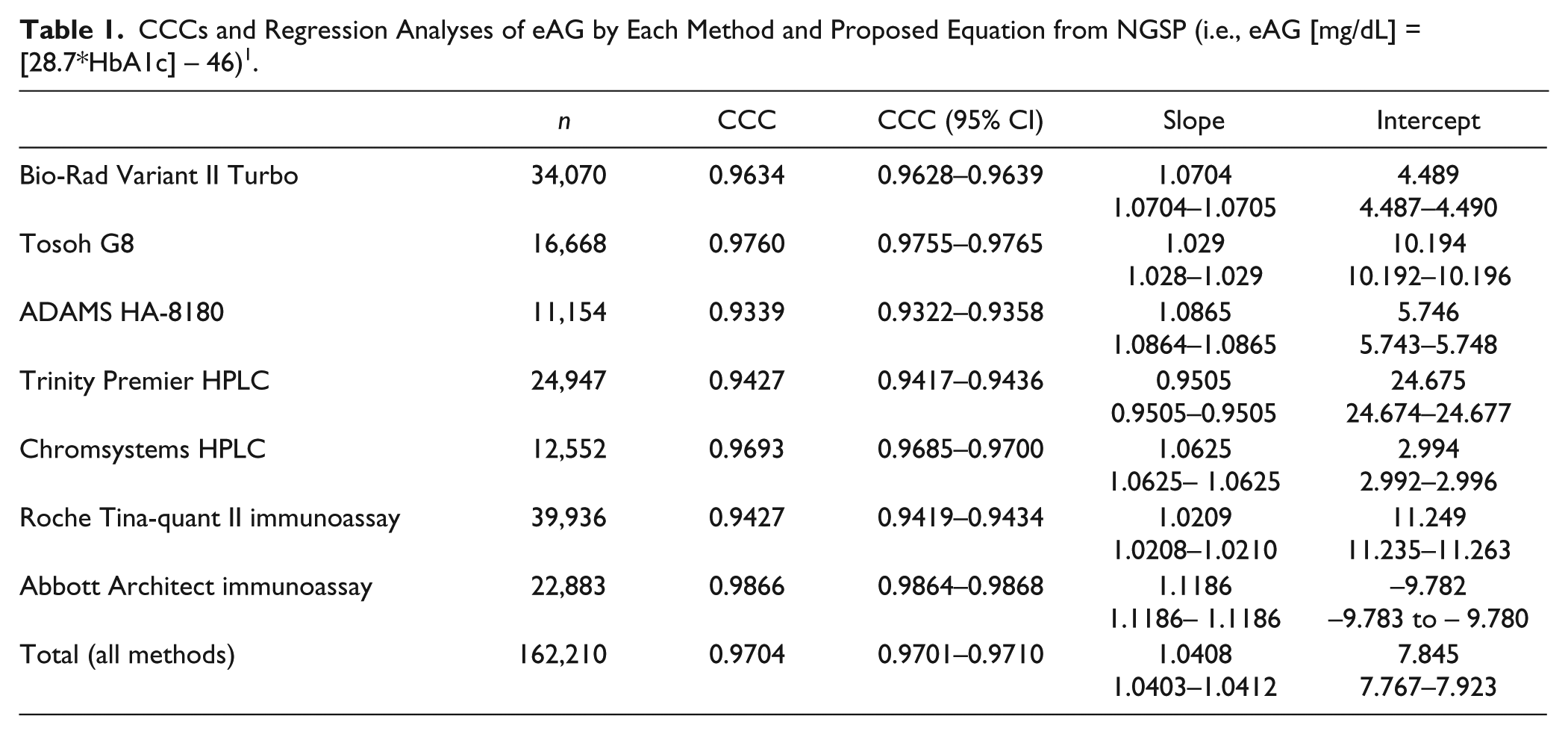
CCCs and Regression Analyses of eAG by Each Method and Proposed Equation from NGSP (i.e., eAG [mg/dL] = [28.7*HbA1c] – 46) 1 .
Discussion
Even though HbA1c stands for the mean glycemic exposure for almost the past 3 months, it does not provide information about the daily alterations in glucose levels. Self-monitoring of blood glucose represents an important adjunct to HbA1c, because it allows us to distinguish fasting, preprandial, and postprandial hyperglycemia; to detect glycemic excursions; to identify hypoglycemia; and to provide immediate feedback to patients about the effect of food choices, activity, and medication on glycemic control. Many studies have shown that HbA1c serves as an index of eAG over the preceding weeks to months.
We had two different aims in this study: (1) Is it possible that eAG can be calculated with patient HbA1c results applying a data mining procedure? (2) Are there any differences among eAG calculations by HbA1c methods?
In the Diabetes Control and Complications Trial (DCCT)
13
study of patients with diabetes mellitus, 3-monthly HbA1c determinations were the principal measure of right glycemic control; study subjects also performed quarterly 24 h, 7-point capillary blood glucose profiles. Premeal, 90 min postmeal, and bedtime blood specimens were obtained from the subjects in the home setting. In an analysis of the DCCT glucose profile data,
5
mean HbA1c and eAG were calculated for each study subject. Rohlfing et al. found a linear relationship between HbA1c and eAG (eAG [mg/dL] = [35.6*HbA1c] – 77.3), with a correlation coefficient (
There were certain limitations in the present study, which was based on the historical patient system data, although calculation of eAG, as an easier method that is less affected by methodological differences, was easier to employ. Whether the eAG formulation has been affected by the methodological differences involved in the study data should be supported by studies based on serial capillary glucose measurements. Nevertheless, such studies are costly and difficult, and it should be considered that it is rather impractical to apply a range of Hb1Ac measurement methods on the samples collected from the same patient. The second limitation is associated with the incomplete specific demographic and clinical data (clinical state, drug use, etc.) of the patients. However, the same is also valid for the previous studies, and such data are limited in the hospital systems.
A study by Bozkaya et al. included 3891 samples evaluated only by Chromsystems kit and Agilent HPLC systems, and found a correlation of 0.757, while our study showed that the foregoing systems had the lowest correlation (
Statistical differences were found in eAG, depending on the HbA1c measurement methodology, based on a comparison between the CCCs and eAG levels as calculated in line with the given methodology and with the eAG formulation recommended by NGSP (95% confidence interval [CI]). It was seen upon review of the data regarding the CCC calculation involving the joint use of bias and correlation data that although it complied well with the accordingly developed formulation, compared to the various Hb1Ac measurement methods, there were statistical differences between the methods. Based on a comparison between CCCs and regression models, which was introduced for the first time in literature, our findings suggested that the CCC HbA1c measurement method might affect the eAG assessment.
We propose that each laboratory can establish a more suitable equation to calculate eAG based on its own data. In addition, it should be noted that more effective formulations can be readily developed by collecting wider data sets thanks to the use of data mining techniques, and the same will contribute more comprehensively in the assessment of patients.
The International Federation of Clinical Chemistry (IFCC) established a working group on HbA1c standardization in 1995. This group ended in 2010, having fulfilled its mission. The Laboratory Network is now overseen by the IFCC Committee on Traceability in Laboratory Medicine. The clinical and educational responsibilities of the group will be assumed by a new “integrated project” designed by the IFCC.17-20 Despite the progress and development of the standardization studies for measurement of HbA1c, there are still differences regarding the relation between serum blood glucose and HbA1c by the respective results of the analyzers.
It is indispensable that the said discrepancies among HbA1c measurement systems affect the calculation of eAG, which is especially demonstrated in HbA1c measurement methods involving preliminary processes. Clinicians and laboratory specialists should be careful in assessing diabetes patients in consideration of the above differences. The present study showed that use of simultaneously measured serum glucose and HbA1c levels could help with facilitating eAG calculation, which would minimalize the methodological differences inherent in the available HbA1c measurement methods and allow a more reliable comparison of eAG results.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
