Abstract
The aim of this study was to identify an improved delivery scheme for intracellular delivery and anticancer therapy. Novel gold nanorods (GNRs) bioconjugates were prepared by using GNRs as nonviral vectors conjugated with thiol-modified antisense oligodeoxynucleotides (ASODNs). We then investigated the possibility of using GNRs as carriers for nucleus delivery of ASODNs genes by using confocal microscopy and cell transmission electron microscope imaging. Our results demonstrate that the GNRs-ASODNs entered into the cell nucleus and increased cell apoptosis. Therefore, the GNRs could hold great promise for biological delivery applications and gene therapy.
Introduction
The applications of nanomaterials as drugs and gene carriers in biological systems have recently attracted great interest.1,2 In comparison to conventional delivery methods, the nanoparticles are so small and safe that they can penetrate the cell wall and deliver drugs or biomolecules into living systems, usually for a therapeutic purpose.3–5
Various transfection agents have been used to deliver genes into cells, including viral carriers, cationic lipids, 6 and carbon nanotubes, but each system has particular limitations. For example, viral carriers are a most effective but rather dangerous method because of the risk of recombination, leading to the generation of viruses capable of replication, and cationic compounds are usually toxic.
Gold nanorods (GNRs) with unique electronic and mechanical properties show great promise for a range of applications in optical sensing,7,8 biomedical imaging,9,10 photothermal therapy,11–13 and cancer diagnostic marker14,15 and biosensing applications.16,17 They especially have a high affinity for biomolecules and have been used as nonviral vectors for DNA delivery. 18 In addition, chemical modification of the surface of GNRs can be easily achieved.
Telomerase activity has been found in ~85% to 90% of all human tumors but not in adjacent normal cells,19–21 and the tumor cells can gain eternal life due to the telomerase activation that maintains a stable telomere length. This makes telomerase a target not only for cancer diagnosis but also for the development of novel gene therapeutic agents. Among nucleic acid–based methods for controlling gene expression, antisense (AS) therapies are potentially powerful candidates for clinical treatments of various ailments, including cancer.22,23 However, their development into viable therapeutic systems has faced challenges with regard to stable transfection and low efficacy.
In this work, we prepared GNRs directly conjugated with thiol-modified antisense oligodeoxynucleotides (ASODNs). Cellular uptake and delivery efficiency of GNRs bioconjugates into HeLa cells had been observed through a combination of confocal microscopy and flow cytometry. We further monitored cellular apoptosis induced by the ASODNs transfected with GNRs. This study clearly demonstrates the potential of these metallic nanostructures for use as novel vectors in gene therapeutic applications.
Experimental
Materials
Cetyltrimethylammonium bromide (CTAB), hydrogen tetrachloroaurate(III) trihydrate (HAuCl4.3H2O), silver nitrate (AgNO3), L-ascorbic acid, and sodium borohydride (NaBH4) were purchased from Sinopharm Chemical Reagent Co., Ltd. The antisense oligodeoxynucleotides (ASODNs) sequence modified with thiol group on the 3′ end (3′SH-(C3H6)-A10-GGGCGCTACGGCGCGCGA-G-G5′), and FAM fluorescence-labeled 5′ end (3′ SH-(C3H6)-A10-GGGCGCT- ACGGCGCGCGA-G-G-FAM5′) were purchased from Shanghai Sangon Biological Engineering Technology & Services Co., Ltd. All reagents used were available commercially and were of the high-purity grade.
Preparation of GNRs
GNRs were prepared in high yields in the presence of cyltrimethylammonium bromide (CTAB) and silver nitrate using the seeded-growth conditions described by Sau and Murphy. 24 First, the gold seeds were synthesized by adding 0.250 mL of HAuCl4 (0.01 M), 9.15 mL CTAB (0.10 M). The solutions were gently mixed by the inversion. Then, 0.600 mL ice-cold NaBH4 (0.01 M) solution was added all at once, followed by rapid inversion mixing for 2 min, and the resulting seed solution turned to a dark brownish yellow color. Second, the growth of the GNRs solution was synthesized as follows: 28 mL of 0.1 M CTAB, 1.2 mL of HAuCl4 (1 mM), and 0.18 mL of silver nitrate (0.01 M), and after gentle mixing of the solution, 0.2 mL of ascorbic acid (0.10 M) was added, with continuous stirring, and 0.15 mL of the seed solution was finally added into the mixture and left overnight. The solution was then centrifuged at 14,000 rpm for 10 min to remove the excessive CTAB. Finally, the nanorod sample was characterized by CARY 100 Scan UV–vis spectrophotometer and the transmission electron microscope (TEM; JEOL 2100).
Preparation of GNRs-ASODNs Conjugates
For preparing GNRs bioconjugates ( Fig. 1 ), we used 100 µL of 0.01 mM thiol-modified ASODNs (or ASODNs-FAM) added to 500 µL 2 mM GNRs. The resulting mixture was shaken gently for 24 h to complete the antisense oligodeoxynucleotides functionalization process, and then the particles were centrifuged (10,000 rpm, 10 min) and the supernatant was decanted to remove unbound SH-ASODNs. Finally the GNRs-ASODNs conjugate precipitate was resuspended in phosphate-buffered saline (PBS; pH = 7.4).The Zeta potential of GNRs and GNRs-ASODNs were performed on a Zeta Sizer Nano ZS (Malvern Zetasizer 3000HS and He/Ne laser at 632.8 nm at scattering angles of 90 at 25 °C).
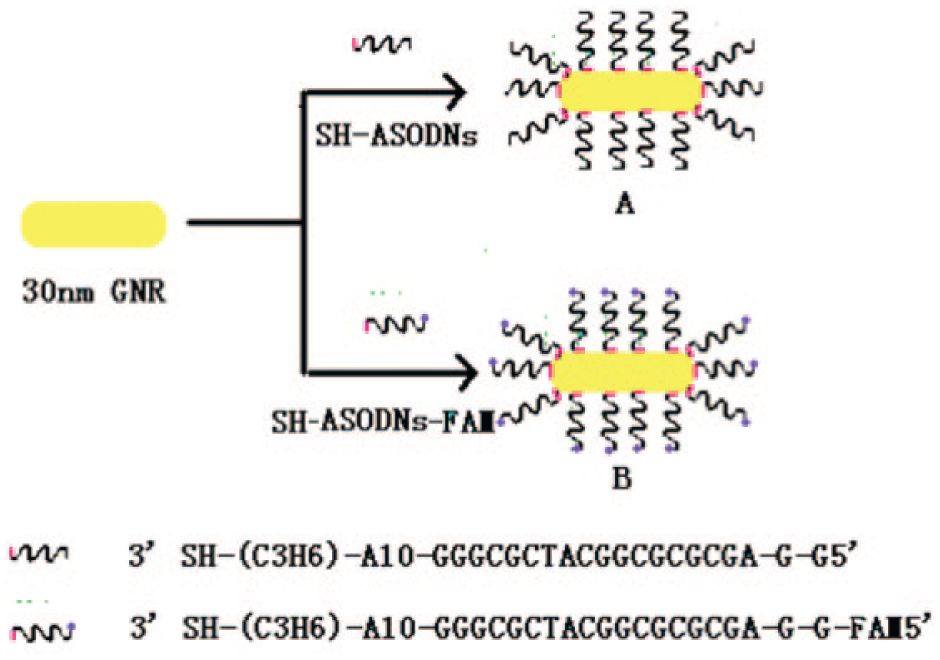
Schematic illustration of gold nanorod (30 nm) functionalized with SH-ASODNs or SH-ASODNs-FAM to produce antisense particles A and B, respectively.
Cellular uptaking of GNRs-ASODNs conjugates by confocal microscopy
HeLa cells were incubated with GNRs-ASODNs-FAM (100 µM GNRs) solution separately for 1, 3, and 24 h at 37 °C in 5% CO2 atmosphere; after that, the cells were thoroughly washed, resuspended in RPMI (Roswell Park Memorial Institute) 1640 medium, then the cellular internalization of FAM fluorescence-labeled GNRs-ASODNs were obtained with a Carl Zeiss LSM 5 PASCAL confocal laser scanning microscope. An argon laser for FAM excitation at 488 nm was used for imaging. A 515 nm band-pass filter for the FAM emission signal was placed in the front of detectors. An oil immersion objective was used for the cellular fluorescence imaging.
Subcellular localization of GNRs-ASODNs by TEM
The subcellular localization of GNRs was observed on a TEM (HT7700, 100 kV, Hitachi, Japan). Briefly, Hela cells of 1 × 106 were seeded into a 100 mm dish and left overnight to allow cell attachment. GNRs-ASODNs were added to the dish at a GNR concentration of 10 nM. After incubation for 3 and 24 h, the cells were washed separately with PBS buffer solution for several times and scraped gently from the dish. The collected cells were centrifuged into a small pellet and fixed in 3% glutaraldehyde solution for 1 h at room temperature. The resulting pellets were dehydrated gradually by alcohol and embedded in Epon. Ultrathin sections were cut and placed on a copper meshwork.
Cell Viability Assay
Hela cells were seeded at 5 × 103 cells per well in a 96-well plate, preincubated for 24 h, then incubated with 20 µL GNRs (100 nM), GNRs-ASODNs (100 nM GNRs, 150 nM ASODNs), and naked ASODNs (150 nM) for 24 h at 37 °C in 5% CO2 atmosphere. The medium was then replaced with 100 µL of 0.5 mg/mL MTT, and after 3 h, the MTT solution was replaced with 150 µL of DMSO solution. The absorbance at 570 nm of each well was measured by a microplate reader (Infinite M200; Tecan, Durham, NC). The absorbance at 630 nm was also measured as a reference. Untreated cells in medium were used as control. All standard deviations were calculated from three replicates.
Cellular Apoptosis Assay
HeLa cells were incubated in 2 µL GNRs (100 µM), GNRs-ASODNs (100 µM GNRs, 150 nM ASODNs), and naked ASODNs (150 nM) for 24 h at 37 °C in 5% CO2 atmosphere, respectively. Then the cells were washed and supplemented with fresh RPMI 1640 medium and collected for analysis. For the cell cytometry, the cells were observed immediately after the initial incubation, and the level of cell death was compared with control cells by PI staining.
Results and Discussion
Figure 2A shows two absorption bands of GNRs from the UV-Vis optical absorption spectral, one at a shorter wavelength of ~519 nm, which is due to the transverse oscillation of electrons, and the other at a long wavelength of ~815 nm, due to the longitudinal oscillation of the electrons, which depends on the aspect ratio of the GNRs. The resonances are due to a collective oscillation of the free electrons through the metal, which depends on the boundary conditions. 25 Figure 2B shows that from the TEM image of GNRs, which is 40 ± 5 nm, most of GNRs are more uniform and stable in water solution and so can be used as good gene carriers.
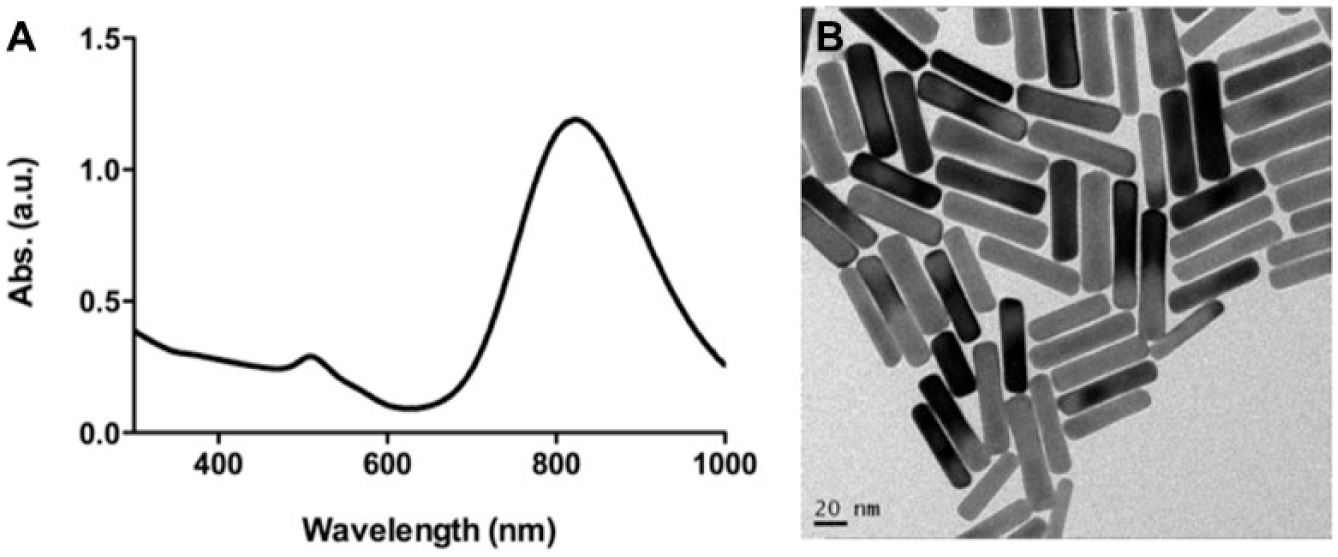
(
GNRs with aspect ratio of ~4 were attached to thiol-modified ASODNs by interactions of Au-S to prepare GNRs-ASODNs (or GNRs-ASODNs-FAM) conjugates. After the conjugation reaction, the resulting conjugation showed very good water solubility without precipitation or aggregation in water-based solutions. The conjugates were kept in a 4 °C refrigerator following the experiment. We applied Zeta potential to characterize the ASONDs linked on GNRs. The average Zeta potential of the as-prepared GNRs is 50.7 ± 2.3 mV; after being bonded with ASONDs, the average Zeta potential of GNRs-ASONDs is 25.2 ± 1.1 mV. The Zeta results show that the surface charges of GNRs decrease about 25 mV, and this means that GNRs have been modified successfully with ASODNs because of the negatively charged ASONDs (
To investigate the intracellular ability of GNRs-ASODNs conjugates, we carried out incubating HeLa cells for 1, 3, and 24 h with the functionalized fluorescence-labeled GNRs-ASODNs-FAM. The confocal microscopic image in Figure 3A shows that the GNRs-ASODNs-FAM have not entered into the cell after 1 h of culture, and the growing status of cells is normal. After 3 h ( Fig. 3B ), the GNRs-ASODNs-FAM conjugates entered into the cell cytoplasm from the observation of the FAM fluorescent signal (green) within cells and the fact that the Hela cells were also growing well. However, when incubated for 24 h with the GNRs-ASODNs-FAM, observation of cell morphology and the anchorage-dependent rate indicated that most of the cells died ( Fig. 3C ). The localization of intense fluorescence of FAM-labeled GNRs-ASODNs was mainly in the cell nucleus, which suggests that ASODNs were delivered into the cell nucleus and targeted the mRNA of telomerase, inhibiting the activity of telomerase and resulting in cell apoptosis.
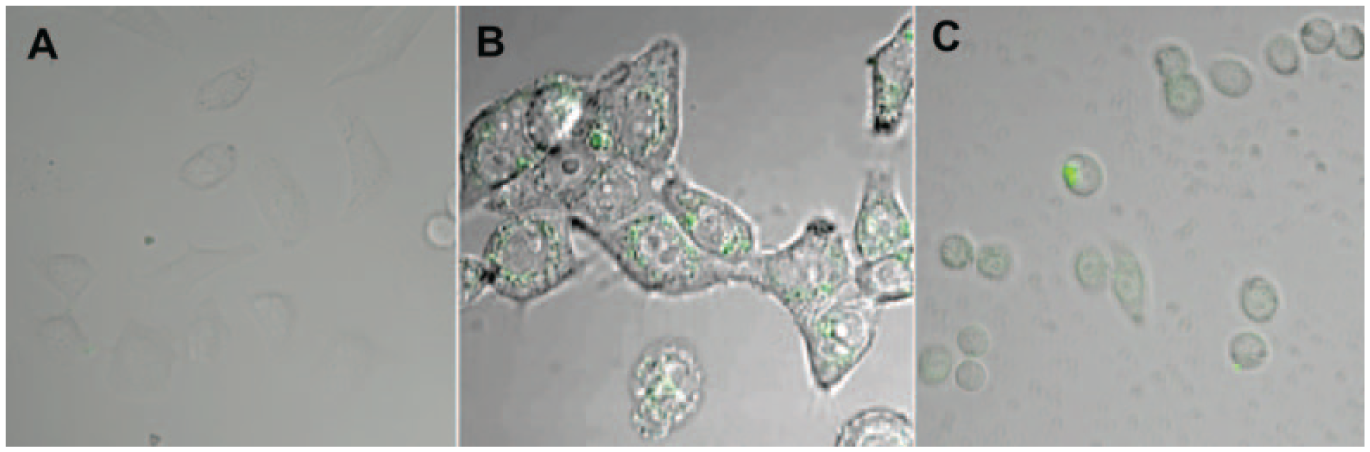
Experiments aimed at understanding the transfection stability of gold nanorods–antisense oligodeoxynucleotides (GNRs-ASODNs). Confocal fluorescence images of HeLa cells incubated for (
To further observe and understand the subcellular localization of GNRs-ASODNs, TEM was further conducted. After 3 h of incubation, the GNRs-ASODNs entered into the cell cytoplasm ( Fig. 4A ). When the incubation time was over, more GNRs were found inside the cells. Figure 4C displays a TEM image showing that the GNRs-ASODNs entered into the cell nucleus after 24 h of culture and induced cell death. This suggests that after transport inside the cell cytosol by the GNRs carriers, ASODNs were entirely delivered into cell nuclei to order to inhibit the target mRNA of telomerase and to increase cell apoptosis.
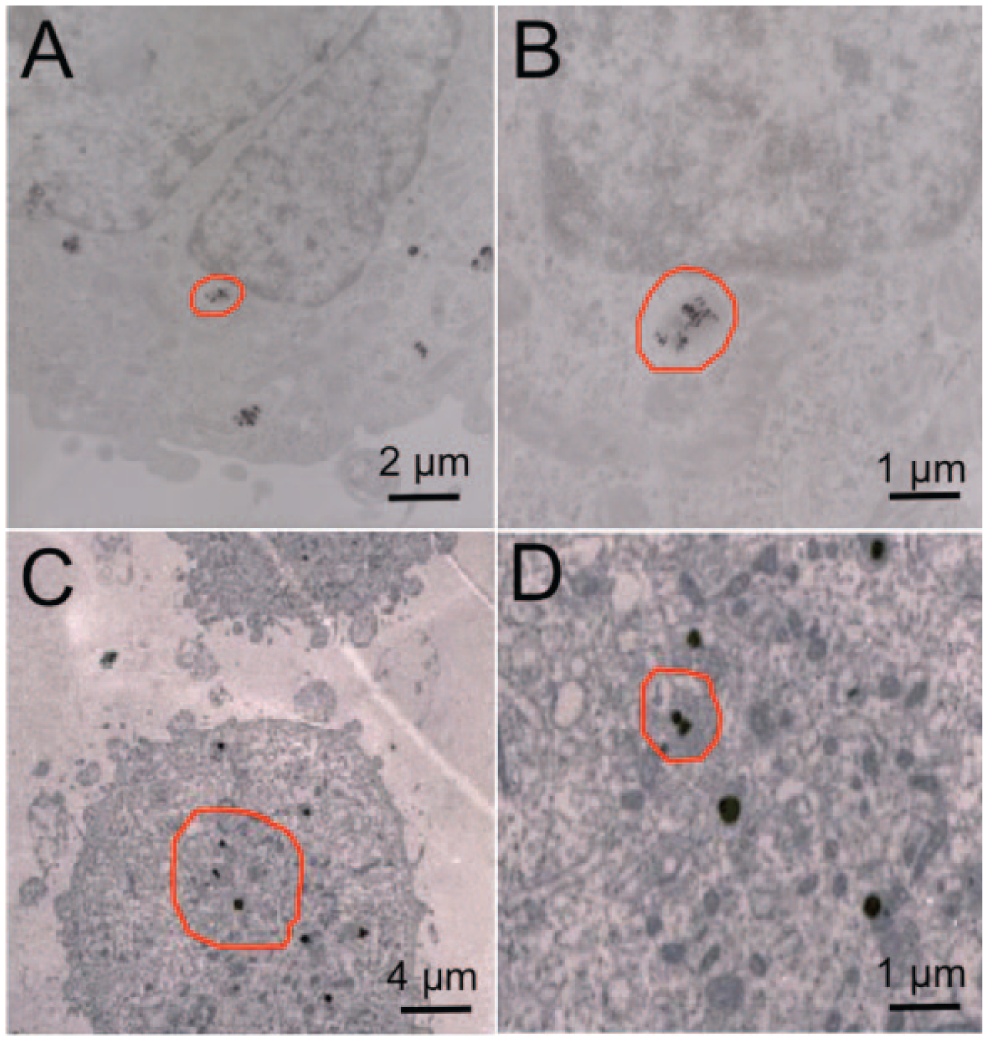
Transmission electron microscope images of Hela cells after (
To evaluate the efficiency of ASODNs on cell apoptosis, we quantified the cell apoptosis efficiency by flow cytometry after incubating HeLa cells for 24 h with GNRs, GNRs-ASODNs, and naked ASODNs, respectively. HeLa cells treated with GNRs-ASODNs exhibit much higher cellular apoptosis ( Fig. 5 ), which can reach 35.78% higher than those treated with GNRs (6.95%) or naked ASODNs (3.73%). To further prove the cell apoptosis efficiency by GNRs-ASODNs, MTT assays were used, and the result showed that GNRs-ASODNs inhibit Hela cell viability by about 35% at 100 µM GNRs-ASODNs after 24 h. The GNRs and the ASODNs inhibit the cell viability only by about 15% and 10% separately ( Fig. 6 ), which means that the GNRs-ASODNs increase cellular apoptosis compared with GNRs and ASODNs.
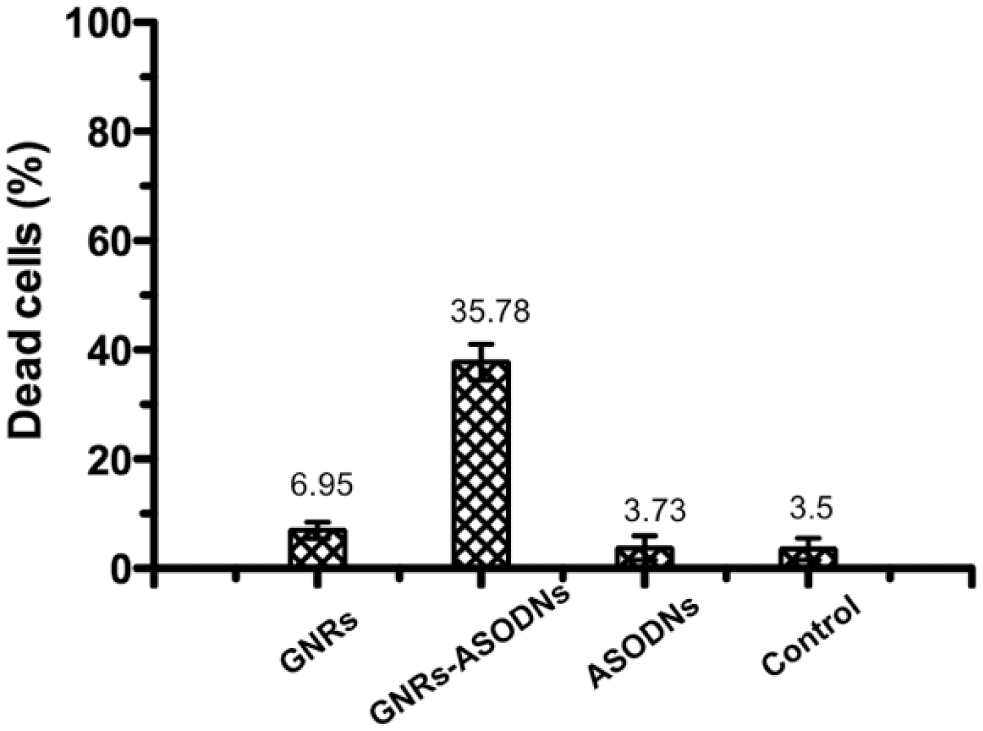
Flow cytometric analysis of the cells in the presence of (
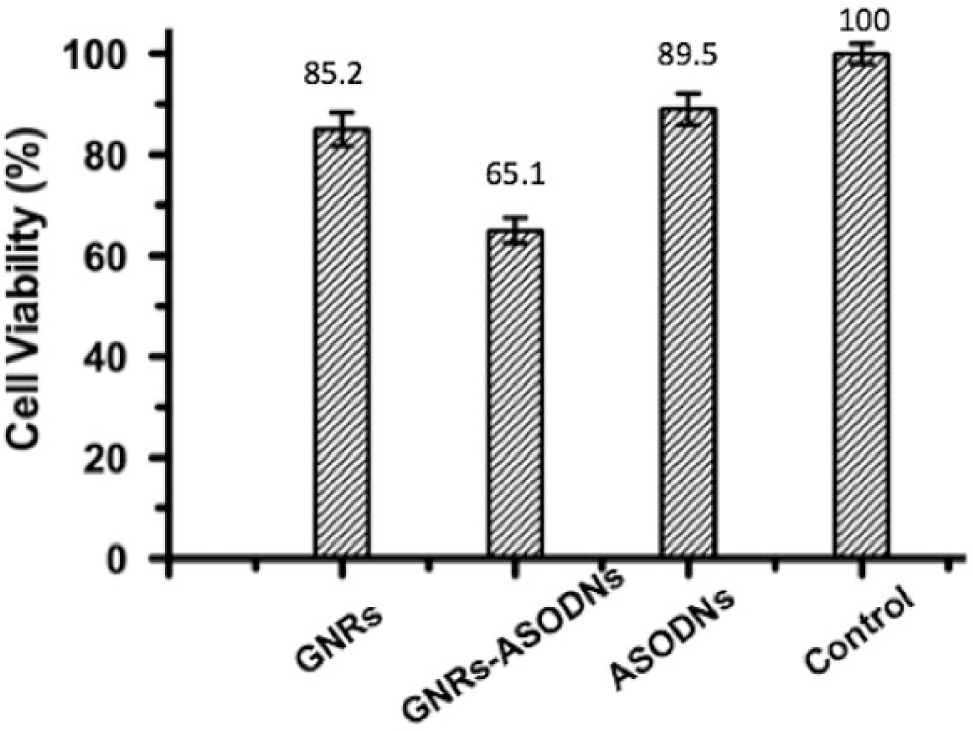
Cell viability by MTT assays after incubating cells for 24 h with (
We prepared the nanomaterial conjugate GNRs-ASODNs. By using the fluorescent FAM to label ASODNs, efficient intracellular transporting and strong cell nucleus localization of ASODNs by GNRs carriers could clearly be seen. Furthermore, we used TEM to further observe the subcellular localization of GNRs-ASODNs, and the results show that GNRs are good carriers for delivering ASODNs to the cell nucleus. Flow cytometry and the MTT assay demonstrate that ASODNs bound to GNRs display an effective anticancer activity. Therefore, GNRs could hold great promise for biological delivery applications and gene therapy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (81301293, 51375444), National Science and Technology Major Projects for “Major New Drugs Innovation and Development” (2014ZX09507008), and a project funded by the China Postdoctoral Science Foundation (2014M561475).
