Abstract
In medicine, the use of silica nanoparticles (SiO2 NPs) offers new perspectives in biosensor, drug delivery and cancer therapy. However, questions about potential toxic and deleterious effects of SiO2 NPs have also been raised. The aim of this study was to investigate the induction of cytotoxicity, oxidative stress and apoptosis by SiO2 NPs (size 15 nm) in human skin epithelial (A431) and human lung epithelial (A549) cells. SiO2 NPs (concentration range 25–200 µg/ml) induced dose-dependent cytotoxicity in both types of cells, which was demonstrated by cell viability (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide) and lactate dehydrogenase leakage assays. SiO2 NPs were also found to induce oxidative stress in a dose-dependent manner, indicated by depletion of glutathione and induction of reactive oxygen species (ROS) generation and lipid peroxidation. Quantitative real-time polymerase chain reaction analysis showed that following the exposure of cells to SiO2 NPs, the messenger RNA level of apoptotic genes (caspase-3 and caspase-9) were upregulated in a dose-dependent manner. Moreover, activities of caspase-3 and caspase-9 enzymes were also significantly higher in both kinds of cells exposed to SiO2 NPs. This study suggested that SiO2 NPs induce cytotoxicity and apoptosis in A431 and A549 cells, which is likely to be mediated through ROS generation and oxidative stress.
Keywords
Introduction
Nano-biotechnology provides opportunity for the development of new unique materials in the nanometer size range (1–100 nm) that can be applied to many potential applications in biological science. Silica nanoparticles (SiO2 NPs) has been considered an ideal material for biomedical applications and is being widely explored as biosensor, 1 biomarker, 2 cancer therapy, 3 DNA/drug delivery4,5 and enzyme immobilization. 6 Therefore, understanding the toxicity of SiO2 NPs at the cellular level is crucial for rational design of this material for biomedical applications.
Nanoparticles could easily enter into human body via different routes such as inhalation, ingestion and dermal contact. Inhalation and dermal contact are very common exposure routes by which nanoparticles deposit in lung and skin inducing inflammation, fibrosis and cytotoxicity. 7 To date, several studies provided ample evidence that SiO2 NPs distributed in blood, lungs, kidneys, spleen, pancreas or other organs. 8 –10 In vitro studies have shown decreased cell viability and oxidative stress after exposure to SiO2 NPs in human lung epithelial (A549) cells, human endothelial (EAHY926) cells, murine macrophages (RAW 264.7), human embryonic kidney (HEK293) cells, rat neuronal (PC12) cells and human liver (HepG2) cells. 11 –15 However, little is known about the toxic effects of SiO2 NPs on human skin cells that represents the first level of exposure to the nanoparticles.
In this study, we investigated the induction of cytotoxicity, oxidative stress and apoptosis by SiO2 NPs in human skin epithelial (A431) cells. To avoid cell type-specific response, we also used human lung epithelial (A549) cells for SiO2 NPs toxicity studies. To achieve this objective, we determined the cytotoxicity by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT; cell viability) and lactate dehydrogenase (LDH) leakage assays in A431 and A549 cells exposed to different concentrations of SiO2 NPs. We further measured the oxidative stress markers such as reactive oxygen species (ROS), glutathione (GSH) and lipid peroxidation (LPO) in response to SiO2 NPs exposure. Quantitative real-time polymerase chain reaction (PCR) was employed to see the effects of SiO2 NPs on the messenger RNA (mRNA) level of apoptosis genes (caspase-3 and caspase-9). Moreover, activities of caspase-3 and caspase-9 enzymes were also examined in both the types of cells against SiO2 NPs exposure. Both A431, derived from human epidermoid carcinoma, and A549 cells, derived from human lung carcinoma, have been widely utilized in toxicological studies. 16 –20
Materials and methods
Silica nanoparticles
Silica (SiO2) nanopowder (product no. 4850MR) was purchased from Nanostructured and Amorphous Materials, Inc. (Houston, Texas, USA). Physicochemical characterization of SiO2 NPs utilized in the present study was reported in our previous publication. 15
Chemicals
Fetal bovine serum (FBS), Dulbecco’s modified Eagle’s medium (DMEM), Hank’s balanced salt solution (HBSS), penicillin–streptomycin and trypsin were bought from Invitrogen Co. (Carlsbad, California, USA). GSH, MTT, 5,5-dithio-bis-(2-nitrobenzoic acid) (DTNB), 2,7-dichlorofluorescin diacetate (DCFH-DA), thiobarbituric acid (TBA) and trichloroacetic acid (TCA) were purchased from Sigma-Aldrich (St Louis, Missouri, USA). LDH, caspase-3 and caspase-9 enzymes assay kits were obtained from Bio-Vision Inc. (Milpitas, California, USA). All other chemicals used in this study were of the highest purity available from commercial sources.
Cell culture and nanoparticles exposure
Both A431 and A549 cells were obtained from American Type Culture Collection (ATCC) (Manassas, Virginia, USA). Both types of cells were used between passages 10 and 20. Cells were cultured in DMEM supplemented with 10% FBS and 100 U/ml penicillin–streptomycin at 5% CO2 and 37°C. At 85% confluence, cells were harvested using 0.25% trypsin and were subcultured into 75 cm2 flasks, six-well plates or 96-well plates according to selection of experiments. Cells were allowed to attach the surface for 24 h prior to nanoparticles treatment. SiO2 NPs were suspended in cell culture medium and diluted to appropriate concentrations (25–200 µg/ml). The dilutions of SiO2 NPs were then sonicated using a sonicator bath at room temperature for 10 min at 40 W to avoid SiO2 NPs agglomeration prior to cells exposure. Selection of SiO2 NPs dosage (25–200 µg/ml) and exposure time (72 h) was based on a preliminary dose–response study (data not shown).
Cell viability assay
Viability of A431 and A549 cells after exposure to SiO2 NPs was assessed by MTT assay as described by Ahamed et al.
18
In brief, 10,000 cells/well were seeded in 96-well plates and exposed to different concentrations (25–200 μg/ml) of SiO2 NPs for 72 h. At the end of exposure, culture medium was removed from each well to avoid interference of SiO2 NPs and replaced with new medium containing MTT solution in an amount equal to 10% of culture volume and incubated for 3 h at 37°C until a purple colored formazan product developed. The resulting formazan product was dissolved in acidified isopropanol. Furthermore, 96-well plate was centrifuged at 2300
LDH leakage assay
LDH is an enzyme widely present in cytosol, which converts lactate to pyruvate. When plasma membrane integrity is disrupted, LDH leaks into culture media and its extracellular level is elevated.
21
LDH assay was carried out by the LDH-cytotoxicity assay kit (Bio-Vision) according to the manufacturer’s protocol. In brief, 10,000 cells/well were seeded in 96-well plates and exposed to different concentrations (25–200 µg/ml) of SiO2 NPs for 72 h. At the end of exposure, 96-well plate was centrifuged at 2300
Determination of ROS
ROS level was determined in A431 and A549 cells exposed to different concentrations (25–200 μg/ml) of SiO2 NPs for 72 h. The production of intracellular ROS was measured using DCFH-DA.
22
The DCFH-DA passively enters the cell, where it reacts with ROS to form the highly fluorescent compound dichlorofluorescein. Briefly, 10 mM of DCFH-DA stock solution (in methanol) was diluted in culture medium without serum or other additive to yield a 100 µM working solution. At the end of exposure, cells were washed twice with HBSS. Then cells were incubated in 1 ml working solution of DCFH-DA at 37°C for 30 min. Cells were lysed in alkaline solution and centrifuged at 2300
Preparation of crude cell extract
For the measurement of LPO, GSH and caspase enzymes, A431 and A549 cells were cultured in 75 cm2 culture flask and exposed to different concentrations (25–200 µg/ml) of SiO2 NPs for 72 h. After the treatment, cells were harvested in ice-cold phosphate buffer saline by scraping and washed with phosphate buffer saline at 4°C. The cell pellets were then lysed in cell lysis buffer (1× 20 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1 mM Na2EDTA, 1% Triton and 2.5 mM sodium pyrophosphate) as described in our previous publication.
15
Following centrifugation (15,000
LPO assay
LPO was estimated by measuring the formation of malondialdehyde (MDA) using the method of Ohkawa et al.
23
MDA is one of the end products of membrane LPO. Briefly, a mixture of 0.1 ml cell extract and 1.9 ml of 0.1 M sodium phosphate buffer (pH 7.4) was incubated at 37°C for 1 h. After incubation, the mixture was precipitated with 5% TCA and centrifuged (2300
GSH assay
GSH level was quantified using Ellman’s method.
24
Briefly, a mixture of 0.1 ml of cell extract and 0.9 ml of 5% TCA was centrifuged (2300
Total RNA isolation and quantitative real-time PCR analysis
Both A431 and A549 cells were cultured in six-well plates and exposed to different concentrations (25–200 μg/ml) of SiO2 NPs for 72 h. At the end of exposure, total RNA was extracted using Qiagen RNeasy mini Kit (Valencia, California, USA) according to the manufacturer’s instructions. Concentration of the extracted RNA was determined using Nanodrop 8000 spectrophotometer (Thermo-Scientific, Wilmington, Delaware, USA), and the integrity of RNA was visualized on a 1% agarose gel using a gel documentation system (Universal Hood II, BioRad, Hercules, California, USA). The first strand of complementary DNA (cDNA) was synthesized from 1 μg of total RNA by reverse transcriptase using M-MLV (Promega, Madison, Wisconsin, USA) and oligo (dT) primers (Promega) according to the manufacturer’s protocol. Quantitative real-time PCR was performed by QuantiTect SYBR Green PCR kit (Qiagen) using an ABI PRISM 7900HT Sequence Detection System (Applied Biosystems, Foster City, California, USA). Overall, 2 ml of template cDNA was added to the final volume of 20 μL of reaction mixture. Real-time PCR cycle parameters included 10 min at 95°C followed by 40 cycles involving denaturation at 95°C for 15 s, annealing at 60°C for 20 s and elongation at 72°C for 20 s. The sequences of the specific sets of primer for caspase-3, caspase-9 and β-actin used in this study are given in our previous publication. 18 Expressions of selected genes were normalized to the β-actin gene, which was used as an internal housekeeping control. All the real-time PCR experiments were performed in triplicate, and data were expressed as the mean of at least three independent experiments.
Caspase-3 and caspase-9 enzymes assay
Activities of caspase-3 and caspase-9 enzymes were measured using Bio-Vision assay kit. Crude cell extract was prepared as described above. This assay is based on the principle that activated caspases in apoptotic cells cleave the synthetic substrates to release free chromophore
Protein estimation
Protein content was estimated using a bicinchoninic acid protein assay kit (Pierce Biotechnology, Inc., Rockford, Illinois, USA).
Statistical analysis
Data presented in this study are mean ± SD of three identical experiments made in three replicate. Statistical significance was determined by one-way analysis of variance followed by Dunnett’s multiple comparison test. Significance was ascribed at
Results
SiO2 NP-induced cytotoxicity
Cytotoxicity of SiO2 NPs in A431 and A549 cells was evaluated by MTT and LDH assays. MTT results showed that cell viability was reduced by SiO2 NPs and degree of reduction was dose-dependent. Viability of A431 cell was decreased to 89%, 77%, 60% and 49%, while A549 cell viability reduction was 87%, 74%, 58% and 45% at the concentrations of 25, 50, 100 and 200 µg/ml, respectively (

SiO2 NPs induced cytotoxicity in A431 and A549 cells. Cells were exposed to different concentrations (25–200 µg/ml) of SiO2 NPs for 72 h. At the end of exposure, cytotoxicity was determined as described in the Materials and Methods section. (a) MTT assay and (b) LDH assay. Data represented are mean ± SD of three identical experiments made in three replicate. *Significant difference compared with the controls (
We further observed that SiO2 NPs-induced a dose-dependent LDH leakage in both the types of cells. LDH leakage in A431 cells was increased to 125%, 157%, 189% and 208%, while in A549 cells LDH leakage was increased to 129%, 164%, 194% and 223% for the concentrations of 25, 50, 100 and 200 µg/ml, respectively (

Correlation between cell viability and LDH leakage in A431 and A549 cells after 72 h exposure to 25, 50, 100 and 200 µg/ml of SiO2 NPs. LDH: lactate dehydrogenase; SiO2 NPs: silica nanoparticles.
SiO2 NP-induced oxidative stress
The potential of SiO2 NPs to induce oxidative stress was examined by measuring the ROS, LPO and GSH levels in A431 and A549 cells. We found that SiO2 NPs induced intracellular production of ROS in dose-dependent manner (
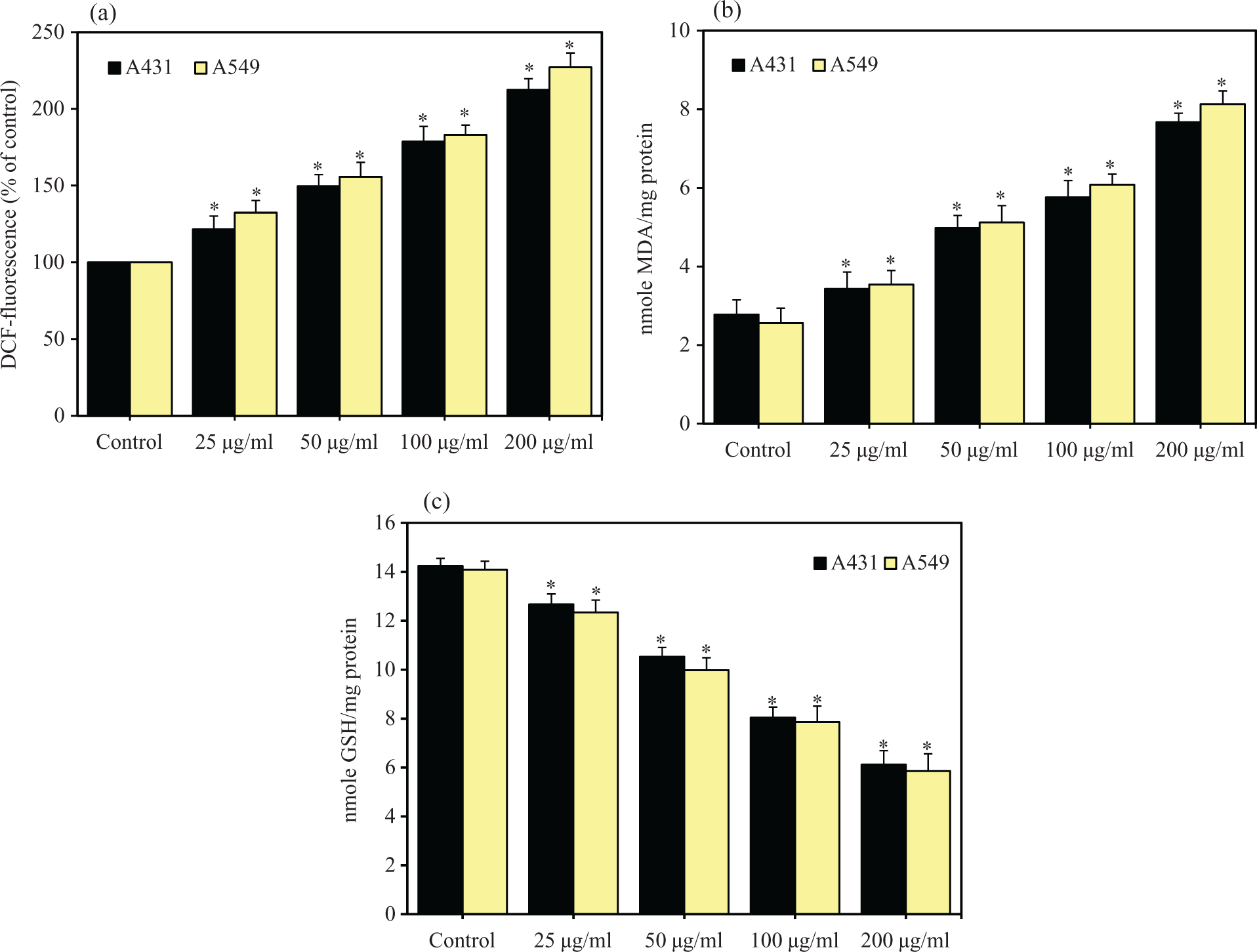
SiO2 NPs induced oxidative stress in A431 and A549 cells. Cells were exposed to different concentrations (25–200 µg/ml) of SiO2 NPs for 72 h. At the end of exposure, oxidative stress parameters were determined as described in the Materials and Methods section. (a) ROS, (b) LPO and (c) GSH. Data represented are mean ± SD of three identical experiments made in three replicate. *Significant difference compared with the controls (

Correlation between ROS level and GSH level in A431 and A549 cells after 72 h exposure to 25, 50, 100 and 200 µg/ml of SiO2 NPs. GSH: glutathione; ROS: reactive oxygen species; SiO2 NPs: silica nanoparticles.

Correlation between cell viability and ROS level in A431 and A549 cells after 72 h exposure to 25, 50, 100 and 200 µg/ml of SiO2 NPs. ROS: reactive oxygen species; SiO2 NPs: silica nanoparticles.
SiO2 NP-induced apoptosis
It is well known that ROS generation leads to apoptotic cell death. We utilized quantitative real-time PCR to analyze the mRNA levels of two apoptotic genes (caspase-3 and caspase-9) in A431 and A549 cells exposed SiO2 NPs at concentrations of 25, 50, 100 and 200 µg/ml for 72 h. Results showed that mRNA levels of caspase-3 and caspase-9 genes was upregulated in dose-dependent manner in both the types of cells due to SiO2 NPs exposure (

SiO2 NPs upregulated the mRNA levels of caspase-3 and caspase-9 genes in A431 and A549 cells. Cells were treated with different concentrations (25–200 µg/ml) of SiO2 NPs for 72 h. Quantitative RT-PCR was performed by QuantiTect SYBR Green PCR kit using ABI PRISM 7900HT Sequence Detection System. The β-actin was used as the internal control to normalize the data. SiO2 NPs-induced alterations in mRNA expression levels are expressed in relative quantity compared with those for the respective unexposed control cells. Data represented are mean ± SD of three identical experiments made in three replicate. *Statistically significant difference compared with the controls (
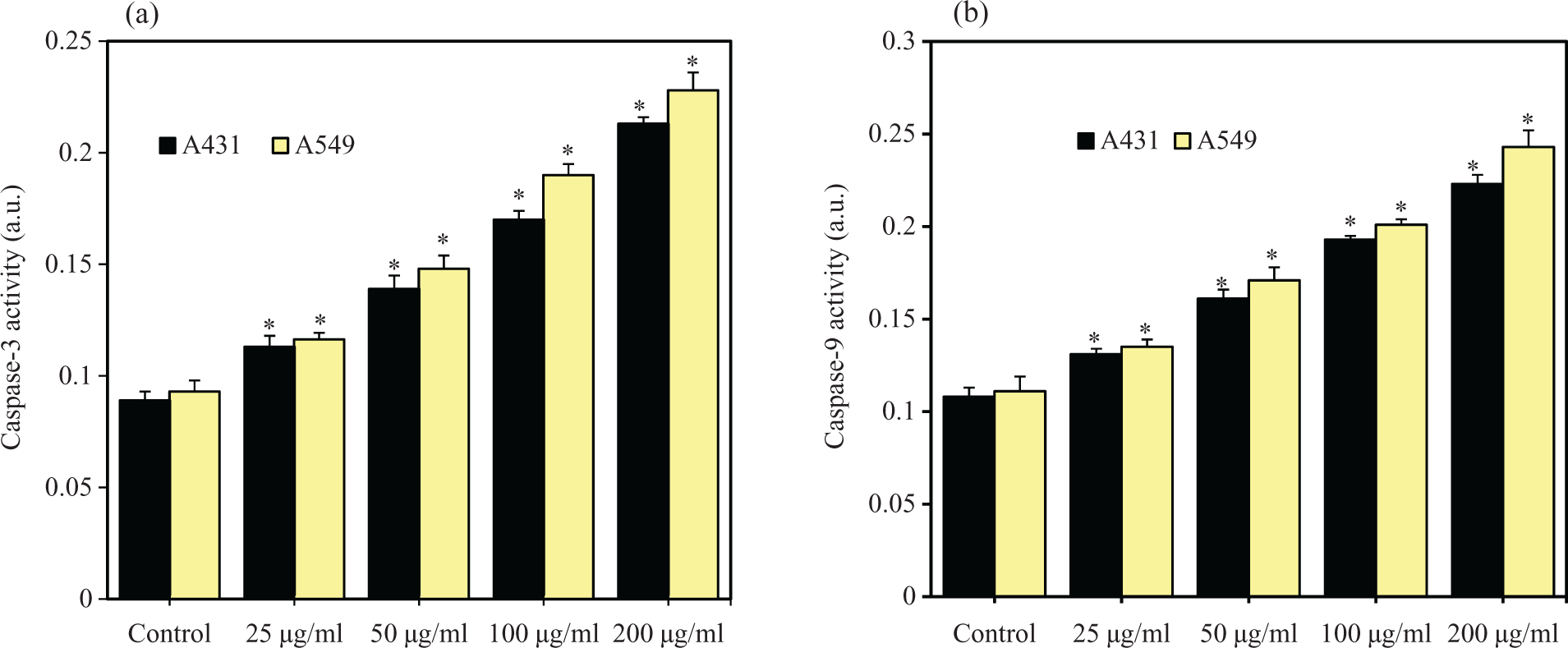
SiO2 NPs induced the activities of caspase-3 and caspase-9 enzymes in A431 and A549 cells. Cells were treated with different concentrations (25–200 µg/ml) of SiO2 NPs for 72 h. At the end of exposure, activities of caspase-3 and caspase-9 enzymes were determined using BioVision assay kit as described in the materials and methods section. (a) Caspase-3 and (b) caspase-9. Data represented are mean ± SD of three identical experiments made in three replicate. *Statistically significant difference compared with the controls (
Discussion
Physicochemical characterization of nanoparticles is an important aspect of nanotoxicity research. Characterization of present SiO2 NPs was reported in our previous article. 15 Briefly, SiO2 NPs were highly pure, amorphous, smooth surface and spherical with an average diameter of 15 nm (transmission electron microscopy (TEM) size). The average hydrodynamic diameter of SiO2 NPs in complete cell culture medium as determined by dynamic light scattering was around 96 nm. The higher size of SiO2 NPs in aqueous suspension compared with TEM size might be due to the tendency of particles to agglomerate in aqueous state. 15
SiO2 NPs are being considered a potential candidate for biomedical applications because of their unique physicochemical properties. However, recent studies reported that SiO2 NPs have a potential to induce toxicological effects to human health. 10,26 –29 These studies suggested that hazards have not been defined yet and more safety research is necessary. Therefore, we investigated the toxic response of SiO2 NPs in human skin epithelial (A431) and lung epithelial (A549) cells. We found that SiO2 NPs have a potential to induce cytotoxicity, oxidative stress and apoptosis in both the types of human cells.
Our MTT data showed that SiO2 NPs-induced reduction in the viability of A431 and A549 cells in dose-dependent manner. LDH leakage from cells is another evidence for the penetration of nanoparticles into the cells and cell membrane damage. 14,30 It has been well documented that LDH levels in the cell medium elevated after the cells exposed to nanoparticles. 18,19,31 –33 We also observed that LDH leakage was higher in a dose-dependent manner in both the types of cells treated with SiO2 NPs. A significant reverse correlation between MTT and LDH was also observed. These results are in agreement with the results from other investigators showing a dose-dependent cytotoxicity induced by SiO2 NPs in human embryonic kidney cells, human neuronal cells and human liver cells. 13 –15
Concomitant cellular oxidative stress was manifested by elevated oxidant levels and depleted antioxidant levels. In the present study, the oxidant (ROS and LPO) levels were higher, while the antioxidant (GSH) level was lower in A431 and A549 cells due to SiO2 NPs exposure. Furthermore, negative linear correlation between the ROS and GSH indicated that free oxygen radical species were generated by exposure to SiO2 NPs, which reduced intracellular antioxidant GSH levels. There was also a strong inverse correlation between decreased cell viability and increased ROS level. The inverse correlation between the decreased cell viability and the increased ROS level suggested that cell death could be the primary cause of ROS generation. Consequently, higher production of ROS leads to increased membrane LPO with a concomitant decrease in antioxidants such as GSH to scavenge these free oxygen radicals appears to be the underlying mechanisms of SiO2 NPs toxicity.
ROS production and oxidative stress have received much attention in nanoparticles-induced apoptosis. 18,34 –36 ROS has been suggested to be signaling molecule for the initiation and execution of the apoptotic death program. 37 ,38 We analyzed the mRNA expression of two apoptotic genes (caspase-3 and caspase-9) in response to SiO2 NPs in A431 and A549 cells, because apoptosis is controlled through these pathways. Our quantitative real-time PCR results showed that SiO2 NPs upregulated mRNA level of these genes. Furthermore, activities of caspase-3 and caspase-9 enzymes were also higher in SiO2 NPs-treated cells. Taken together, ROS induces permeabilization of the outer mitochondrial membrane, which releases soluble proteins from the intermembrane space into the cytosol, where they promote caspase activation. 39,40 The best studied of these proteins is cytochrome c, which binds to apoptosis protease activating factor-1 (Apaf-1) and leads to the assembly of an apoptosome complex. This apoptosome binds to procaspase-9 and causes its auto-activation through a conformational change. Once initiated, caspase-9 goes on to activate caspase-3 (effector caspase), which cleaves substrates at aspartate residues and activation of this proteolytic activity appears to be an event in nanoparticle-induced apoptosis. 18,41 Apoptotic response of SiO2 NPs in A431 and A549 cells are supported by our previous investigation, where SiO2 NPs induced apoptosis in human liver cells (HepG2). 15
In conclusion, we have shown that SiO2 NPs produce significant cytotoxicity to A431 and A549 cells in dose-dependent manner in the concentration range of 25–200 µg/ml. This nanoparticle was also found to induce oxidative stress by inducing oxidants (ROS and LPO) and depleting antioxidant (GSH). Furthermore, mRNA levels and activities of casapase-3 and capase-9 enzymes were upregulated by SiO2 NPs. It is also worth mentioning that A549 cells seem to be marginally more susceptible to SiO2 NPs exposure than A431 cells. Taken together, this study suggesting that SiO2 NPs may induce cytotoxicity and apoptosis in A431 and A549 cells, which is likely to be mediated through ROS generation and oxidative stress. Further studies are underway to examine the molecular mechanism of toxicity induced by SiO2 NPs.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by King Abdulaziz City for Science and Technology under the National Plan for Science and Technology (Grant No.: 10-NAN1201-02).
