Abstract
Introduction
Management of significant bone defect has been challenging to orthopedic surgeons. Iliac crest bone harvesting has been the gold standard for autogenous bone graft 1 until the Reamer-Irrigator-Aspirator (RIA) system (Synthes, Inc., West Chester PA) provides a viable alternative since 2005. 2 There were several studies that revealed the superiority of RIA as compared to iliac bone harvesting in terms of donor site pain, nerve injury, operative time, and volume of bone graft harvested.3–7 Meanwhile a systemic review and meta-analysis conducted by Laubach et al. demonstrated a low prevalence of complications rate associated with RIA. 8 While the usage, efficacy, and safety of RIA system seem promising, it has been superseded by a redesigned RIA2 since 2020. 9 To date, there are only handful reports on this new system and its related complications.9,10 There was only one case report on device failure—breakage of reamer head with metallic debris retention in RIA2. 10
Therefore, we conducted a review of the current literature on the applications and complications of the RIA and RIA2 systems. A case with a similar forementioned complication in RIA2 system is also illustrated in our study. Analysis for the corresponding device failure and prevention of such complication is presented.
Illustrative case
A 33-year-old overweight (BMI 29.9) man had right tibial infected non-union fracture resulting from a Gustilo 3B right tibial shaft open fracture treated with nailing 1 year ago. After debridement and hardware removal, there was a 12 cm tibial bone defect. Membrane induction by antibiotic loaded cement spacer and fracture fixation by external fixator were performed. Second stage of Masquelet procedure was performed 11 weeks later. The right tibial shaft was fixed with a tibial nail, and the bone gap was filled with autologous bone grafting.
Autograft was obtained from the femoral canal by RIA-2 system. Autograft harvesting procedure was performed strictly following RIA-2 standard protocol 11 with careful selection of entry point, central guidewire placement, slow advancement of reamer, and diligent use of fluoroscopy avoiding eccentric reaming. Before reaming, the size of medullary canal was estimated by the method suggested in the surgical technique manual. The diameter tabs of radiological ruler were placed perpendicularly over isthmus on the limb side closest to the fluoroscopy receiver in both anteroposterior and mediolateral views. 11 A disposable, single-use12.5 mm RIA2 reamer head was chosen, as the isthmus diameter was estimated to be 13 mm. A power tool with 3.5–6Nm torque and 700–900 RPMs in drill setting was used. Continuous reaming was performed at full speed, applying gentle, and controlled pressure with slow advancement. During the antegrade reaming, the reamer head reached femoral condyle but became stuck on the way up at the isthmus. Consequently, the reamer was restarted for retrograde reaming. It was noted that the reamer head was dislodged from the shaft when the shaft was pulled out. The dislodged reamer head was retrieved by pulling the ball-tipped reamer guide rod out. Further fluoroscopic imagining revealed multiple radiopaque fragments inside the femoral canal (Figure 1A). The retained metal fragments were removed using a regular flexible intramedullary reamer, tooth curette, and long forceps under fluoroscopic guidance (Figures 1B, 2A and 2B). Around 20 mL autogenous bone graft was harvested from the femur intramedullary canal after a single pass of the 12.5 mm RIA2 reamer.
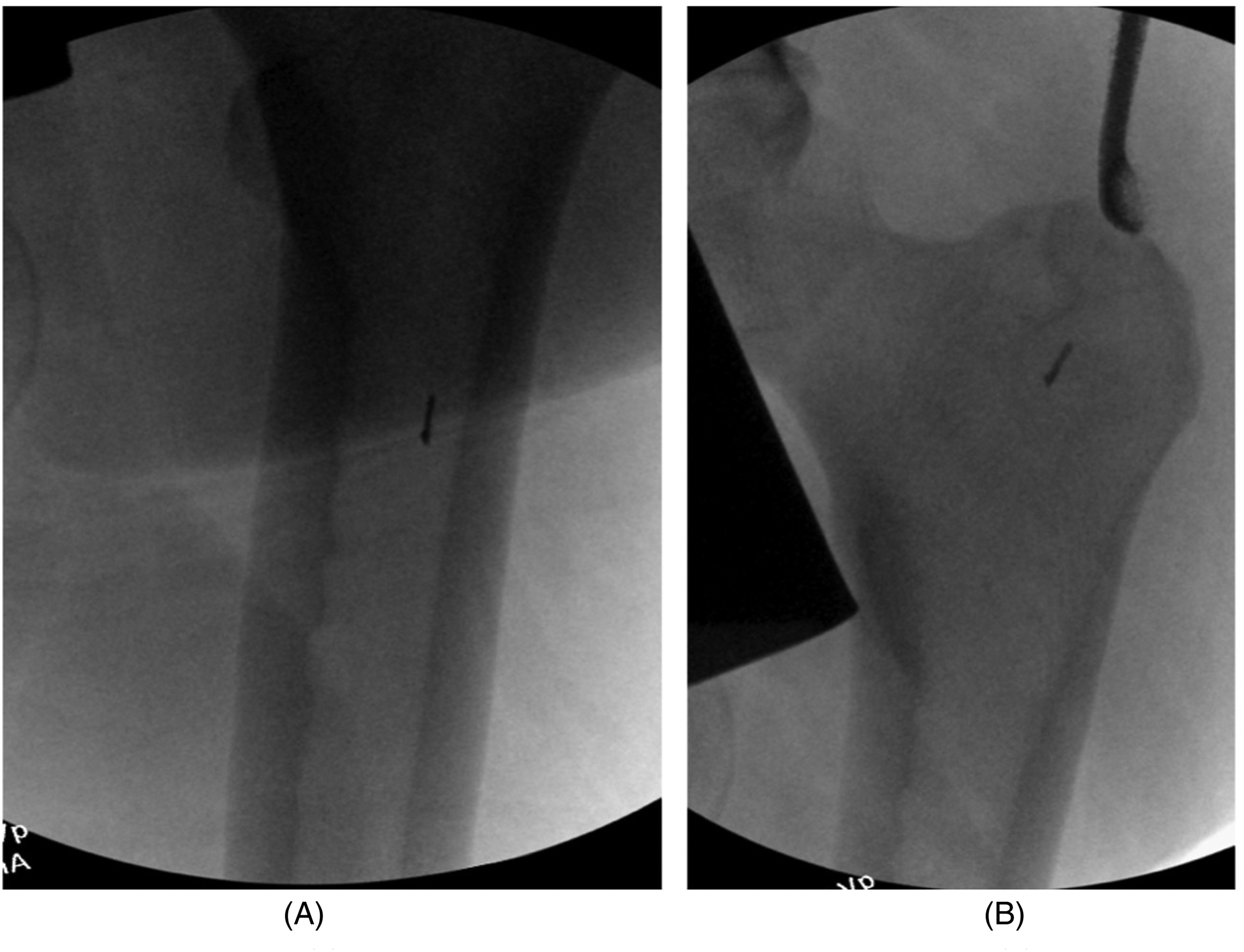
A (Left)—fluoroscopic image showing one of the three retained metal debris. B (Right)—Fluoroscopic image showing removal of fragment using a tooth curette.

(A) Broken reamer head together with three retrieved fragments (a fourth piece could not be found). (B) Close up view of Reamer-Irrigator-Aspirator 2 (RIA2) reamer head with broken prongs. (C) Close up view of intact RIA2 reamer head.
Literature review and discussion
The RIA system is a versatile tool, not only for clearing up the medullary canal in the case of infection, reducing the incidence of fat embolism and thermal necrosis during regular reaming for intramedullary fixation but also for harvesting autogenous bone graft. 12 The development of RIA-2 further increases intraoperative flexibility with its new features. 11 The prevalence of complications associated with older RIA system varied from 0.7% to 11.9%. 8 In Laubach et al.'s meta-analysis, they suggested that the complications rate varied significantly depending on specific application of RIA system. When RIA system was used for intramedullary reaming before femoral nail fixation, the complication rate could be up to 11.9%. However, this could be attributed to confounding factors in the index injury, which could lead to a higher prevalence of systemic and local infection, as well as cardiopulmonary complications. 13 Meanwhile, the overall prevalence of complications in other application is significantly lower, including 1.4% in autologous bone graft harvest and 0.7% in intramedullary canal clearance. 8 Among those complications, cortex perforation is the most commonly reported, followed by cardiopulmonary complications, wound infection, low energy fracture, and RIA device failure.5,6,8,14–16 There were 6 out of 1834 cases reporting device failure with breakage of the reamer head and metallic debris retention in RIA system from 2007 to 20225,6,8,14–16 (Table 1). Yet, there was not much information on the causes and precautions on this complication.
Literatures on RIA reamer head breakage with patients’ demographics and features.

For the new RIA2 system, Chloros et al. was the only group that reported on retained metal debris from broken reamer head at autograft harvest in a 33-year-old female patient. 10 Reamer-Irrigator-Aspirator 2 surgical technique manual has suggested on proper guide wire entry angle less than 10° from canal axis as a precaution to avoid reamer head connection damage which could lead to retained metal fragments. 11 Chloros et al. further speculated that such device failure could be due to improper assembly between reamer head and shaft, suboptimal surgeon technique, or device malfunction. They had also proposed troubleshoots for metal fragments retrieval. In our patient, we have encountered similar device problem as Chloros et al. did at femoral isthmus reaming. Despite proper entry site, careful estimation of isthmus size under fluoroscopy, starting RIA2 reamer head size 0.5 mm smaller than estimated isthmus diameter, proper device assembly, judicious surgical technique, and diligence use of fluoroscopy, this hardware problem still occurred.
From the mechanical and technical aspect, the breakage of the reamer head prongs could be due to the excessive torque acting at the reamer head-shaft junction when reamer head got jammed in a small femoral intramedullary canal. Initial torque and friction of restarting reaming head in a tight canal not only can lead to reamer head fragmentation or reamer shaft breakage, 14 more catastrophic complication such as iatrogenic fracture can also occur.16–18 Therefore, in addition to the forementioned precautions, sequential reaming starting from a smaller reamer head, vigilant reaming at isthmus, and continuous reaming in intramedullary canal might help to mitigate the risk of these complications. There have been studies speculating about the inaccuracy of using fluoroscopy for estimating medullary size.19,20 Marchand et al. determined the size of RIA reamer by commencing sequential standard flexible reaming until diaphyseal chatter was reached. They then adopted RIA reamer head that was 2 mm larger for the subsequent reaming. They proposed that obtaining better estimation of the RIA head size using standard reamers was one of the modified techniques for reducing intraoperative blood loss. This could also reduce the risk of head incarceration due to medullary size overestimation by direct measurement from fluoroscopy.
In our case, the device complication could be attributed to technical factors. Following the surgical manual, we estimated the reamer head size by fluoroscopy and performed one-step reaming using the disposable RIA2 reamer head 0.5 mm smaller than the estimated diameter. Additionally, restarting the jammed reamer head in the narrow isthmus could also result in excessive torque, leading to reamer head fragmentation. Considering this first-time experience with RIA2 system, there are various technical bailouts described in the current literatures that can be employed in the future uses of RIA2 system.
From the patient aspect, strong cortical bone in a young patient or the one with high BMI may be more at risk of reamer head breakages. There was a report of a case in which retained metallic debris was found at the forearm graft recipient site in a young patient who had autologous bone graft harvested by RIA system. 5 Another middle-age overweight man with BMI 29 had retained metal fragments during reaming. Hence, smaller starting reamer head, extra reaming time with incremental reamer advancement would be more favorable in patients with high bone resistance. Nevertheless, the patient must be forewarned of these possible complications. In our case, the overweight young patient is likely to have dense bone quality. It is important to consider that the estimation of reamer head size by fluoroscopy might be inaccurate, especially that the exact estimation of thigh thickness is more unreliable. This estimation inaccuracy could lead to the selection of a slightly oversized reamer. Combined with the patient's dense bone quality, the risk of reamer breakage could have been compounded. Therefore, it is crucial to consider patient's specific characteristics, such as bone quality and age, when selecting the appropriate reamer head size and reaming technique.
From implant aspect, both RIA and RIA2 reamer heads have four prongs for engagement with the reamer shaft. The size of the connecting part with four prongs for shaft engagement does not differ between different reamer diameters within the same system. Comparing RIA and RIA2 reamer head as shown in Figure 3, prongs of new RIA2 reamer head are thinner and narrower. Diameter of the connecting end is also smaller in RIA2. Hence, the reamer head prongs, being the weakest link in the whole chain, are the most vulnerable to break off at peak torque in a jammed reamer head.

Side-by-side comparison of 12 mm Reamer-Irrigator-Aspirator (RIA) reamer head (left) and 10 mm RIA2 reamer head (right).
Retained metal debris in the donor femoral canal is indeed a rare complication of the RIA and RIA2 systems, and retrieval of the debris can be challenging. Various techniques have been proposed in different studies for the removal of retained broken reamer heads and metal fragments including closed and open techniques.10,21–25 Use of ball tipped reamer guide rod is the most important safeguard to enable retrieval of the reamer head in case the head-shaft junction breaks. Chloros et al. utilized different size of curettes, graspers, and nail extraction hook under fluoroscopic guidance to remove the fragmented metal prongs after reamer head was removed using ball-tipped guide rod. 10 Other groups have employed Kocher clamps or endoscopic graspers for the retrieval of the small debris.24,25 In cases where closed retrieval of the reamer head was unsuccessful or when ball-tipped guide rod was unavailable, use of a cortical window was reported.22,23 In our case, we successfully retrieved the reamer head and metal debris by closed technique with ball-tipped guide rod, regular flexible intramedullary reamer, tooth curette, and long forceps under fluoroscopic guidance.
It is important to note that retained metal fragments rarely cause severe local or systemic harm. Therefore, the decision to pursue their removal should be carefully considered. In some cases, it may not be worthwhile to expend significant effort and prolong operative time to remove them. Despite taking all necessary precautions, this hardware complication could still occur. It is essential to ensure that patients are well-informed and acknowledged the possibility of retained debris during the consent process. By considering the potential risks, benefits, individual patient's condition, and available resources in the surgical setting, the most appropriate decision regarding the management of retained reamer head and metal fragments should be made by an experienced surgeon.
By incorporating considerations from technical, patient, and implants factors, we can further improve the safety and effectiveness of the use of RIA and RIA2 system.
Conclusion
Broken reamer head with retained metallic debris is a rare complication in RIA and RIA2 system. Apart from following the manufacturer's recommendations in the surgical manual, we recommend starting with sequential reaming utilizing a regular reamer to better select an appropriate RIA head. After all, both RIA and RIA2 are still very reliable, safe, and effective tools for autologous bone graft harvest.
Footnotes
Authors’ note
Contributing author: Lau Janice Chi Kay, Shen Wan Yiu.
Availability of data and materials
Data were not generated or analyzed in this study. There was no data sharing.
Research Ethnic and consent
The current study has obtained the approval from Hong Kong Hospital Authority Central Institutional Review Board on clinical research ethnics. Formal written consent was obtained from the patient who was involved in this study with the participant information sheet provided. Permission was obtained from the patient for the de-identified images, demographics, and details of this case for the purpose of publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
