Abstract
Introduction
Periprosthetic joint infection (PJI) after total knee arthroplasty (TKA) is an uncommon yet devastating complication with severe morbidity and mortality. Different factors have been suggested to be associated with PJI and it is paramount for clinicians to be aware of them in order to minimize the risks of our patients developing this condition.
Out of these risk factors, postoperative hyperglycemia and glucose variability have been reported to be modifiable ones.1–3 Another possible modifiable risk factor includes the use of steroid perioperatively. Although steroid injection is increasingly used to improve pain control and enhance recovery, the risk of hyperglycemia and PJI remains a concern.4–6 Moreover, glycemic markers, such as hemoglobin A1c (HbA1c) and fructosamine, correlate with postoperative hyperglycemia and PJI, where the threshold for optimization, have been proposed with conflicting evidence.7–11
There have been previous studies investigating the effects of these risk factors on perioperative hyperglycemia in patients undergoing TKA.12,13 However, most of these studies rely on traditional intermittent self-monitoring of blood glucose level,14,15 which has its limitations in reflecting the glucose profile after TKA. Recently, continuous glucose monitoring (CGM) technology has become a common alternative method for monitoring patients’ blood glucose levels. It enables continuous and instantaneous glucose level monitoring. It can also provide valuable information such as time in hyperglycemia, average blood glucose level, and glucose variability, which traditional glucose measurement methods cannot offer. Despite its more prevalent use and aforementioned advantages, the use of CGM in literature in investigating the risk factors of hyperglycemia after TKA are scarce.
Our study aims to investigate the risk factors for postoperative hyperglycemia and glucose variability in patients undergoing unilateral primary TKA, by looking at their average blood glucose level, the amount of hyperglycemic time, and the fluctuation of glucose level with the use of CGM technology.
Methods
We performed a prospective cohort study on patients undergoing unilateral primary TKA from 2020 to 2022. Patients’ demographic factors were collected. Diabetic status was based on medical history and HbA1c level. According to the American Diabetes Association, HbA1c ≥ 6.5% and ≥5.7% were considered diabetic and prediabetic, respectively. 16 HbA1c and fructosamine levels were checked one month before surgery. Patients received either 16 mg or no dexamethasone as decided by the anesthetist prior to their surgery. Potential risk factors for hyperglycemia were analyzed, including body mass index (BMI), diabetic status, perioperative dexamethasone injection, HbA1c, and fructosamine level. Patients who were having poorly controlled Diabetes mellitus requiring regular insulin injections, patients who were immunocompromised or with contraindications of using steroids (e.g., hepatitis carrier) were not given dexamethasone after anesthetists’ assessment. Patients who were scheduled for one-staged bilateral TKA or refused to use CGM were excluded from this study. Patients who were having HbA1c > 7.5% were postponed for surgery and were not recruited in our study as well. Patients who were newly diagnosed for diabetes mellitus but with HbA1c ≤7.5% were not excluded from surgery or from using dexamethasone. Since the use of fructosamine level is still at an investigative stage as a correlation measurement for perioperative hyperglycemia, we did not use fructosamine as a parameter for the decision of the use of dexamethasone or postponing surgery.
The intravenous fluids and calorie intake during the in-hospital stay of the patients in this study were standardized according to their body weight. We divided the patients into three different groups according to their weights (49 kg or below, 49–59 kg, and 60 kg or above) and limited their calories intake to 1200 kcal, 1500 kcal, and 1800 kcal, respectively, which is about 25–30 kcal per kilogram of their body weight. Normal saline was the only type of intravenous fluid given to our patients in this study in order to standardize their intravenous fluid intake.
We used CGM to monitor the glucose level from before the operation to at least postoperative day 4. Outcome measures include the percentage of time in hyperglycemia (percentage of time with blood glucose level >10.0 mmol/L), increased average glucose level (>10.0 mmol/L), and increased glucose variability (percent coefficient of variation >36%) during the monitoring period. The CGM system we have used in this study is the FreeStyle Libre System (Abbott, IL, USA), which is a CGM system that has been frequently used in other clinical studies on blood glucose monitoring.17–19 A CGM sensor was applied to each of the patients’ upper arm on the day of admission until at least postoperative day 4. The blood glucose level and the blood glucose variability of these patients were continuously recorded by the sensors during this period.
According to a sliding scale, insulin was injected whenever the patient's blood glucose level was ≥10.1 mmol/L due to ethical considerations to prevent the development of dangerously high hyperglycemia.
Statistical analysis
IBM SPSS Statistics Version 28.0.1.0 was used for statistical analysis for this study. Unpaired Student t-test and correlation and multivariate regression analysis were performed between the risk factors and the outcome measures. Multicollinearity between the predictor variables was checked. Continuous variables are presented in means and standard deviations (SDs), while qualitative variables are presented in percentages. P-value ≤0.05 was considered statistically significant.
Results
A total of 83 patients were analyzed in our study; 82% of the patients were females. The mean age and BMI were 69.1 years old (SD 6.6) and 28.8 kg/m2 (SD 3.8), respectively. The average of HbA1c and fructosamine level was 6.0% (SD 0.5) and 249.3 mmol/L (SD 27), respectively. Diabetic, prediabetic, and nondiabetic patients were composed of 26.5%, 47.0%, and 26.5%, respectively; 65.1% received 16 mg dexamethasone perioperatively. Patients’ demographics and their baseline characteristics are shown in Table 1.
Patients’ demographics and baseline characteristics.

Univariate analysis between each risk factor and outcomes found that HbA1c, fructosamine, and dexamethasone injection are statistically significant as a risk factor on the percentage of time in hyperglycemia (p < 0.05) and average blood glucose level (p < 0.05). Also, it has been shown that dexamethasone injection is a statistically significant risk factor for glucose variability in our patients (p < 0.05). The mean values, SDs, and p-values of the outcome measurements are shown at Tables 2–6.
Means, standard deviations, and p-values (BMI ≥30 kg/m2 vs patients with BMI <30 kg/m2).

Means, standard deviations, and p-values (HbA1c ≥ 5.7% vs HbA1c < 5.7%).
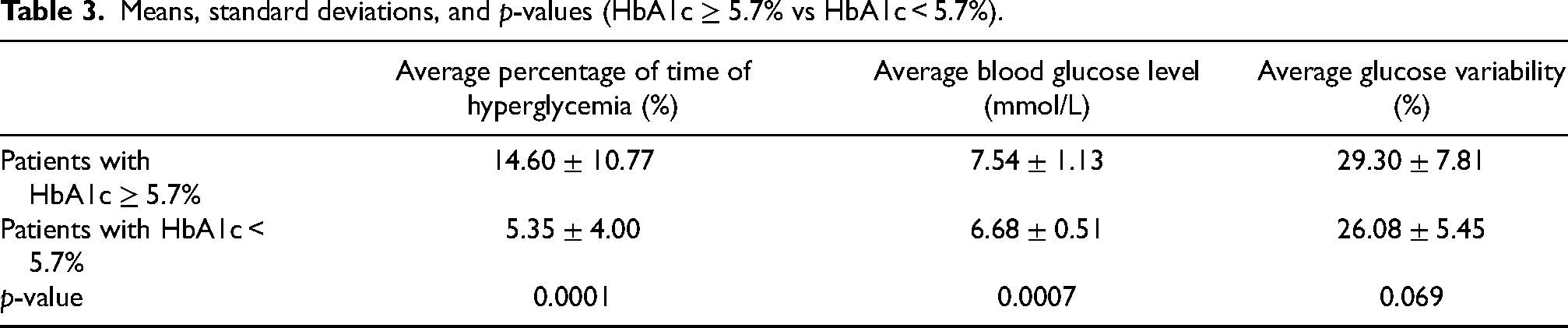
Means, standard deviations, and p-values (Fructosamine ≥ 285 µmol/L vs Fructosamine < 285 µmol/L).

Means, standard deviations, and p-values (Known history of diabetes vs Without known history of diabetes).

Means, standard deviations, and p-values (received dexamethasone vs did not receive dexamethasone).

Multivariate regression analysis found that HbA1c, fructosamine, and dexamethasone injection are the significant risk factors for the percentage of time in hyperglycemia (R2 = 0.374, p < 0.001) and average blood glucose level (R2 = 0.493, p < 0.001).
For the analysis for the time in hyperglycemia, HbA1c had the highest standardized coefficient (0.469, p < 0.001), followed by fructosamine (0.277, p = .01) and dexamethasone injections (0.194, p = 0.02) (Table 7).
Multivariate regression analysis for time of hyperglycemia (percentage of time of blood glucose level >10 mmol/L).

For the analysis of high average blood glucose level, once again HbA1c had the highest standardized coefficient (0.397, p < 0.001), followed by fructosamine (0.320, p = 0.003) and dexamethasone injections (0.261, p = 0.002) (Table 8).
Multivariate regression analysis for high average blood glucose level (average blood glucose level >10 mmol/L).

For the analysis for glucose variability, although dexamethasone usage was significant in multivariate regression analysis on predicting glucose variability (standardized coefficient 0.230, p = 0.044) (Table 9), the effect size of the model is very weak (R2 = 0.05, p < 0.05). All independent variables did not show any multicollinearity with tolerance >0.25 and variance inflation factor <4.
Multivariate regression analysis for glucose variability (percentage of coefficient of variation >36%).

Discussion
Postoperative hyperglycemia is a risk factor for complications in orthopedics and other types of surgeries.1,2,20–22 The surgical stress after major surgeries triggers a catabolic response and the release of hormones, such as cortisol and glucagon, which in turn leads to hyperglycemia and increase insulin resistance. This transient hyperglycemia creates an optimal environment for bacterial growth while impairing our immune system's ability to combat infection.23,24 This explains the clinical finding of increased PJI and wound complications after arthroplasty with postoperative hyperglycemia.2,10 Therefore, we should identify, screen, and optimize the risk factors for postoperative hyperglycemia before joint replacement surgeries.
Our study found that patients’ preoperative HbA1c level was the most robust predictor for the time in hyperglycemia and high average blood glucose, followed by fructosamine and steroid injections. On the contrary, the patient's medical history of diabetic status was not a significant risk factor after multivariate regression analysis. Our findings are supported by other studies demonstrating hyperglycemia is more critical than diabetic status in predicting postoperative complications.20,25 Also, postoperative hyperglycemia can develop in diabetic and nondiabetics. 26 Hence, screening and optimizing the glycemic markers, especially HbA1c, is essential in all patients before arthroplasty to help minimize their risks of developing infection. Due to the relatively small sample size of our study, we were not able to conclude a cutoff value of HbA1c as a contraindication to TKA. However, despite excluding patients with HbA1c level >7.5%, in our study HbA1c still appeared to be a predictor of hyperglycemia. From this result, it appears that a cutoff HbA1c level lower than 7.5% may be beneficial to guide us in assessing the hyperglycemic risk of patients undergoing TKA; however, a study with a larger sample size will be required for further confirmation. Even though steroid injection is one of the risk factors for hyperglycemia, its standardized coefficient is the lowest out of the three risk factors. This may suggest that the correlation between the use of it and perioperative hyperglycemia may not be as strong as conventionally presumed. Thus, consideration of a more lenient use of this medication should be explored in the future especially when many studies have shown that steroids are effective in enhancing recovery which is particularly useful in fast-track arthroplasty.27,28 Therefore, the risk and benefits of its use should be weighed and discussed for individual patients.
Shohat et al. reviewed point-of-care capillary and serum blood glucose levels in arthroplasty patients and reported that glucose variability in the postoperative period is associated with an increased rate of PJI. 3 The traditional methods of glucose measurements might not capture the peaks and troughs of the glucose level, thus, underestimating its variability. On the other hand, CGM can accurately reflect the variation in glucose over time. Unfortunately, the risk factors analyzed in this study only had a weak effect size in predicting glucose variability. This indicates glucose variability is more volatile and more predictors are needed to explain its fluctuation.
Although we standardized the intravenous fluid and diet, controlling extra intake and snacks between meals is challenging. Moreover, insulin injections are given to hyperglycemic patients per protocol as it is unethical to leave them untreated, thus underestimating the actual duration of hyperglycemia. In addition, patients with complex medical comorbidities and a higher risk of postoperative glucose disturbance will likely opt not for steroid injection by the anesthetist, which will further confound our results. Nevertheless, although these limiting factors can potentially affect our results, we believe that they also reflected real-life clinical scenarios where similar treatments would have been provided accordingly. As patients with HbA1c > 7.5% were not recruited in our study, it means our study was mainly focused on patients who were supposed to have relatively good control of their diabetes, and our results may not be directly applicable for patients with poorly controlled diabetes. However, since the variation of glucose control is still commonly observed in patients with relatively well-controlled diabetes, we believe our results are still useful in providing insight into perioperative hyperglycemia risk of this group of patients.
Despite the limitations, our study does shed some light on the risk factors for postoperative hyperglycemia after TKA and we hope it can provide evidence for clinicians to consider when managing this group of patients. However, the sample size of our study is relatively small to see the effect of controlling the risk factors identified in the current study, including HbA1c, fructosamine, and steroid injections. Also, in our study, we were not able to draw any conclusion on a target threshold for these risk factors for patient optimization, and this is once again likely due to the limited size of our study. In view of these limitations, a larger scale of study is welcomed to further examine the relationships between these risk factors and postoperative hyperglycemia. Furthermore, in our study, we have only been looking at patients undergoing unilateral primary TKA. Nowadays bilateral one-staged TKAs or revision surgery are becoming more common, so our results may not be directly applicable to those patients. Future studies on the risk factors for postoperative hyperglycemia for these patients will be useful.
Conclusion
Our study found that the most potent risk factor for hyperglycemic time and increased average blood glucose is HbA1c, followed by fructosamine and dexamethasone injections. With our results, we suggest that universal screening of these glycemic markers, that is, HbA1c and fructosamine, is beneficial for patients undergoing primary TKA, even in nondiabetic patients. However, the factors analyzed in our study cannot adequately explain the difference in glucose variability in our patients, and further study in this regard is welcomed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
