Abstract
Background
Traumatic spinal cord injury (TSCI) is a debilitating dysfunction of the central nervous system (CNS) particularly affecting the developed world, with 280 to 316 new cases per million within Western Europe annually, 1 and predominantly affecting the male population (78.3%). 2 Most patients are aged between 10 and 40 at the point of injury 3 with varying causes and, depending on the nature of the TSCI, patients can suffer from SCI syndromes 4 encompassing loss of motor and sensory function below the level of the lesion, such as permanent paraplegia (thoracolumbar lesions) or quadriplegia (cervical lesions). Individually, this causes the affected person to suffer health, financial and personal life difficulties, such as the loss of independence, neuropathic pain, incontinence and spasticity. 1
TSCI is often a result of a contusion or compression primary injury, whereby the injured spinal column exerts pressure upon the spinal cord. 5 Other mechanisms of TSCI include the stretching of the cord (distraction), displacement of the vertebrae (dislocation) and complete or partial incisions of the cord (transection), all of which are more difficult to reproduce within animal model studies. 1 Pathophysiological mechanistic events involved in the secondary injury to the spinal cord and prevention of axonal regeneration have been well characterised from rodent studies: cell loss;6,7 excitotoxicity from increased glutamate release;5,8 lipid peroxidation5,9 and changes within the balances of essential ions.5,10 Unlike amphibians and reptiles, 1 the mammalian CNS has a limited capability of naturally regenerating and replacing the damaged cells,1,6 resulting in lifelong neurological dysfunction and chronic disability. Astrogliosis occurs due to extensive proliferation of reactive astrocytes – and migrating ependymal progenitor cells which differentiate into astrocytes1,11,12 that amalgamate with other glial cell types to form a protective glial scar. The glial scar is an important player in the prevention of further secondary injury through the formation of a physical barrier surrounding the lesion site as well as containing the inflammation post-injury.1,5,7,13 Counterintuitively, the glial scar also prohibits TSCI recovery and underlies functional impairment of the spinal cord 14 through the inhibition of axonal regrowth.1,13
Approved treatments currently in the UK for acute TSCI involve surgical intervention, rehabilitation, and physiotherapy. Spinal cord decompression and fusion (stabilisation) is a common surgical procedure, 15 with early decompressive surgery (<24 h after TSCI) being better associated with an improved neurological outcome in human16,17 and canine studies. 18 However, whilst these medical and surgical interventions may allow some spontaneous clinical recovery 15 and are critical for the reduction of secondary injury from spinal swelling and hypoperfusion, they do not address the primary TSCI – which is essential for patients to obtain an optimal recovery and quality of life.
A promising therapeutic strategy for primary TSCI is the use of stem cells (SCs), harnessing their inherent multi- or pluri-potency to reconstitute the damaged or lost oligodendrocytes 7 (to overcome demyelination) and neurons 7 within the spinal cord to regain, or at least improve, normal neurological functions and quality of life. Before SC therapeutics can be introduced within the clinical setting, their superiority in terms of feasibility, safety and relative efficacy must be established in relation to current therapies. Within the last 30 years, there has been a surge in the use of mouse models of contusion and compression injuries. 19 However, major anatomical differences between the murine and human spine,20,21 particularly of the corticospinal tract (CST),21–23 renders a need for cautious interpretation of the results from murine studies, and often the results are not replicated within human efficacy-testing trials. 24 On the other hand, studies in large animals – defined here as canine, porcine and non-human primates (NHPs) – more accurately mimic both the size and anatomical morphology of the human spinal cord and are therefore considered better models for testing SC therapies prior to moving to human clinical trials.
This systematic review analyses the functional outcome (motor and sensory) changes after intraspinal, intrathecal or epidural injections of stem cells into an acutely (<2 weeks) injured spinal cord of large animal models and humans compared to the controls and published over the last 3 decades.
Methods
Protocol
For this systematic review, we followed the PRISMA 2020 Checklist 25 and guidelines adapted for animal, rather than human, participants.
Elibility criteria
Papers were included if the manuscript was published in English between 1990 and February 2020, the SCs were directly injected into the intraspinal, epidural or intrathecal compartments within 2 weeks of a traumatic mechanism of injury, including acute intervertebral disc prolapse. The participants were either large animals – defined as canine, porcine or non-human primate in-vivo models – or human patients. These participants were compared to control groups, who had no/sham or decompressive treatment after induction of a spinal cord injury. The authors must have published data on functional outcomes (motor ± sensory) before and after the injury and subsequent treatments.
The following were excluded from the systematic review analysis:
Reviews, abstract-only and commentaries Articles using only mouse, rat or other small animal models Injury only articles Stem-cell injection-only models without previous spinal cord injury Spinal contusion injury Non-traumatic spinal cord injury Degenerative mechanism of injury, including intervertebral disc disease causing progressive spinal cord compression Time between injury and injection longer than two weeks Use of non-stem cells e.g. Schwann cells, olfactory ensheathing cells Use of genetically modified stem cells Delivery of stem cells using adjuncts e.g. polymer scaffolds No functional outcomes recorded
Search methods and information sources
On 25th February 2020, one author (CH) searched for potentially relevant records in Medline, Embase, Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials and SCOPUS (1990 to February 2020). The search strategies included terms relating to all types of stem cells used to treat traumatic spinal cord injury in large animals and humans (see full Boolean search strategies in Supplementary Table S1).
Study selection process and data collection process
The searches were conducted, and 344 duplicates were removed by one author (CH). Two authors (CL, DK) screened the titles and abstracts of 356 records retrieved from the searches. One author (CL) screened the retrieved full-text articles and collected the data using a standardised data-collection form as well as an Excel spreadsheet with desired data collection fields. The following data were collected:
Injury model Spinal level of injury Length of time from injury to injection Delivery method Stem cell type Stem cell dosage Number of animals studied (in intervention and control groups) Motor improvement* Olby score Deep pain perception (DPP) MRI Findings Histological Findings Somatosensory evoked potentials (SSEPs) Follow-up period
* Motor improvement was categorised into full recovery, partial recovery, and nil recovery. Full recovery was defined as back to baseline assessment score, Olby score 14 (see Appendix, Table A1), Revised Tarlov (RT) scale 9, Modified Tarlov (MT) scale 5 (see Appendix, Table A1), Basso, Beattie, Bresnahan score (BBB) 21 (see Appendix, Table A2). Partial recovery was defined as an improvement in scores but not back to original, recovery of ambulation (including limited), mild ataxia, Olby score 10–13, RT scale 6–8, MT scale 4, BBB 14–20. Nil recovery was defined as no improvement in baseline scores, cannot walk without support though may stand unassisted, Olby score 0–9, RT scale 1–5, MT scale 0–3, BBB 0–13.
Study outcomes were grouped into early (<8 weeks), intermediate (8 weeks-6 months) and late (>6 months) to provide categories for the meta-analysis. Studies were not included in the meta-analysis if the functional outcome data was averaged across the treatment groups, rather than providing individual scores, or if the data was incompletely reported.
Risk of bias
Bias was assessed by one reviewer (CL) using the Cochrane ‘Risk of bias’ assessment across 6 evidence-based domains. The ‘Blinding of outcome assessment (detection bias): Self-reported outcomes’ was not included because the studies all involved animal (rather than human) participants. Risk of bias was assessed at the study level. We have reported the risk of bias in Supplementary Figures S1 and S2.
Data analysis
We summarised the functional outcome data of included stem cell studies into tables. Due to the varying methods of measuring functional motor outcomes in each study, motor recovery at the end point was categorised into nil, partial or full recovery. Full recovery was defined as back to baseline assessment score, Olby score 14 (see Appendix, Table A1), Revised Tarlov (RT) scale 9, Modified Tarlov (MT) scale 5 (see Appendix, Table A1), Basso, Beattie, Bresnahan score (BBB) 21 (see Appendix, Table A2). Partial recovery was defined as an improvement in scores but not back to original, recovery of ambulation (including limited), mild ataxia, Olby score 10–13, RT scale 6–8, MT scale 4, BBB 14–20. Nil recovery was defined as no improvement in baseline scores, cannot walk without support though may stand unassisted, Olby score 0–9, RT scale 1–5, MT scale 0–3, BBB 0–13.
The data were extracted as available from the published papers and the outcomes grouped into 3 follow-up periods, Early (<8 weeks), Intermediate (8 weeks–6 months) and Late (>6 months). Forest plots were produced using RevMan 5, 26 summarising the Odds ratios, with 95% confidence intervals (CI) and using the Mantel-Haenszel (M-H) method for random effects summaries.
Results
Study selection
In total, 700 records were retrieved from the searches. Of these, 30 were identified for full-text screening (Figure 1). The inclusion criteria were met by 9 studies (Table 1). Excluded papers are presented in Supplementary Table S2.
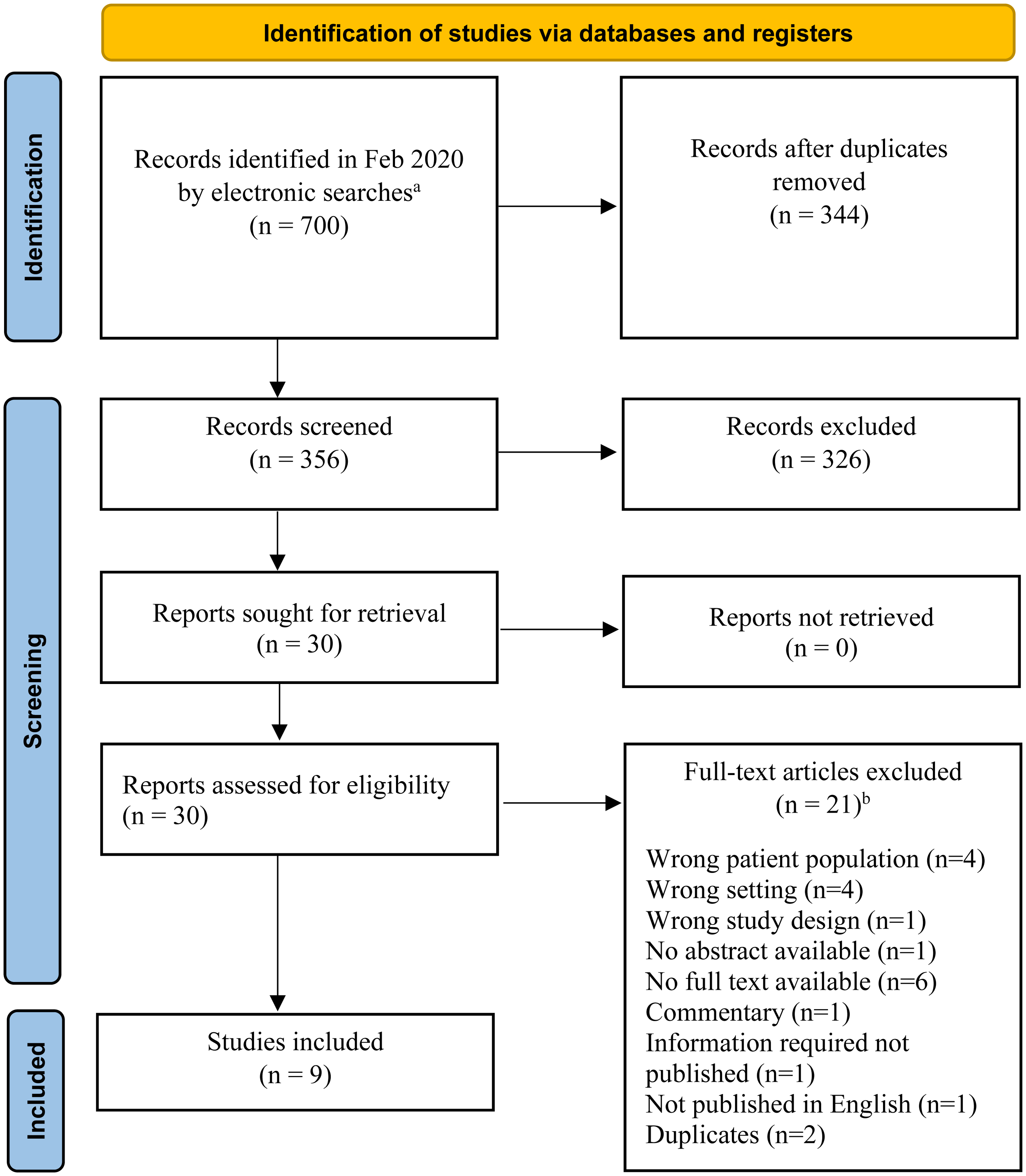
Flow diagram of identification, screening, and inclusion of studies. aRecords identified from Ovid Medline, Ovid Embase, Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials, SCOPUS. bExcluded papers are presented in Supplementary Table S2.
Characteristics of included studies using stem cell therapeutics with controls in large animal and human TSCI.

MRI: magnetic resonance imaging; SCI: spinal cord injury; UCB: umbilical cord blood; PBS: phosphate-buffered saline; ASC: adipose derived stem cells.
Motor improvement was categorised into full, partial, and nil recovery based on determined criteria.
Study characteristics and results of individual studies
The characteristics and summary data for each study are presented in Table 1. All studies involved canine participants, and no porcine, non-human primate or human studies met the inclusion criteria. Three studies used natural injury models27,29,35 and 5 used experimentally-induced TSCI using a balloon catheter.28,30–34 The spinal levels of injury ranged from T3 to L5. Most studies injected the SCs after 7 days,27–32,34 most commonly intraspinal injections.29–34 Other studies injected the SCs intrathecally28,35 or into the epidural space. 27 Allogenic SCs27–34 were used more than autologous SCs28,35 at a dose range between 1 × 106 to 2.3 × 109 cells. The study sizes were relatively small, with the largest number of included cases (including controls) being 82. 35 Olby score was the most common functional motor outcome measured.28,32–34 Three studies used non-standardised measures of motor and/or deep pain perception outcomes.27,29,35
Results of each meta-analysis done, including confidence intervals and measures of consistency, are presented in Figures 2–4. For Figures 2 and 3, three studies28,32,34 were not included in the meta-analysis for motor improvement as they reported mean Olby scores, rather than individual participant scores. In Figure 4, one study 30 was not included in the meta-analysis, as DPP data in the stem cell subgroups were only partially reported.
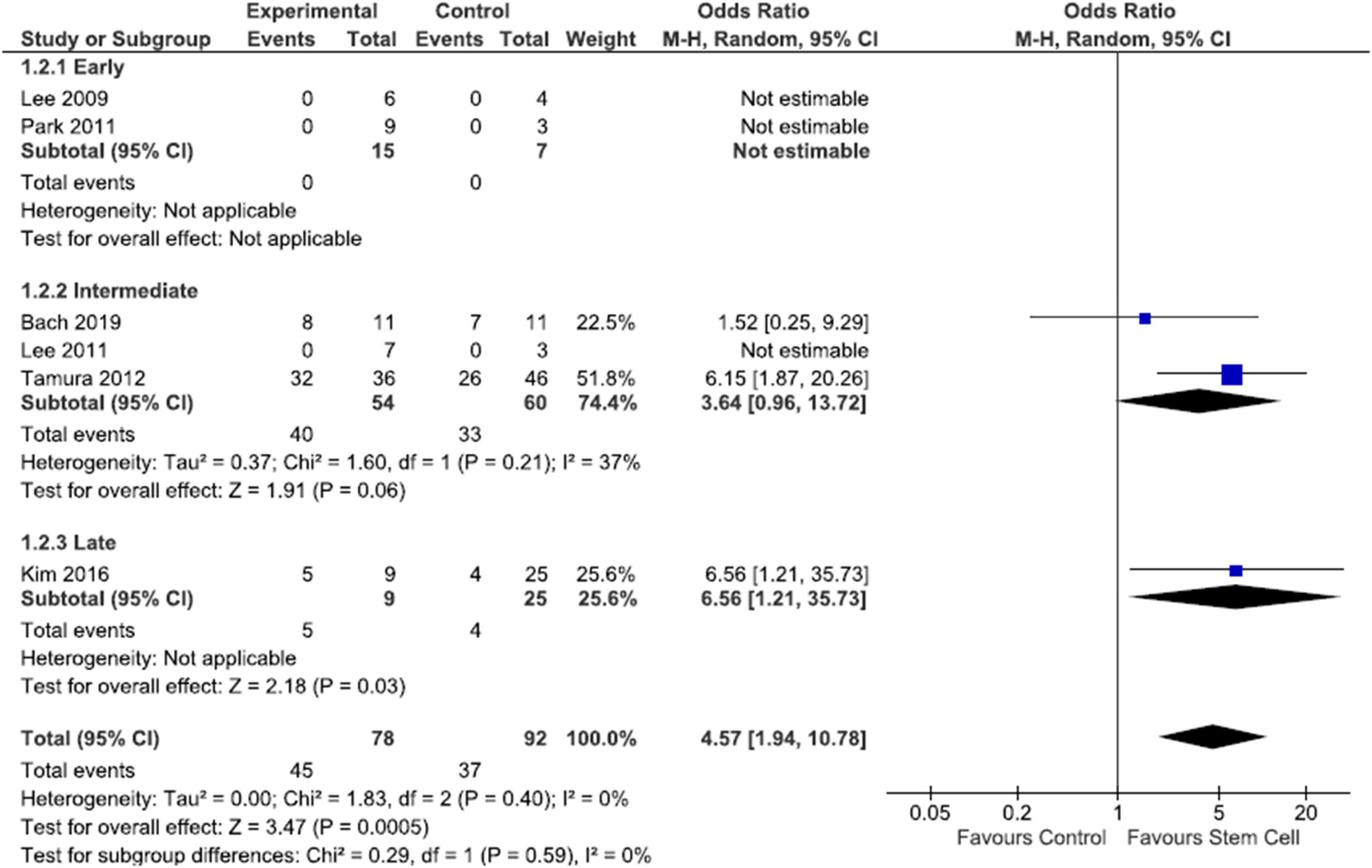
Forest plot of full motor recovery data. Study outcomes were categorised into early (<8 weeks), intermediate (8 weeks-6 months) and late (>6 months). Full recovery was defined as back to baseline assessment score, Olby score 14 (see Appendix, Table A1), Revised Tarlov (RT) scale 9, Modified Tarlov (MT) scale 5 (see Appendix, Table A1), Basso, Beattie, Bresnahan score (BBB) 21 (see Appendix, Table A2).
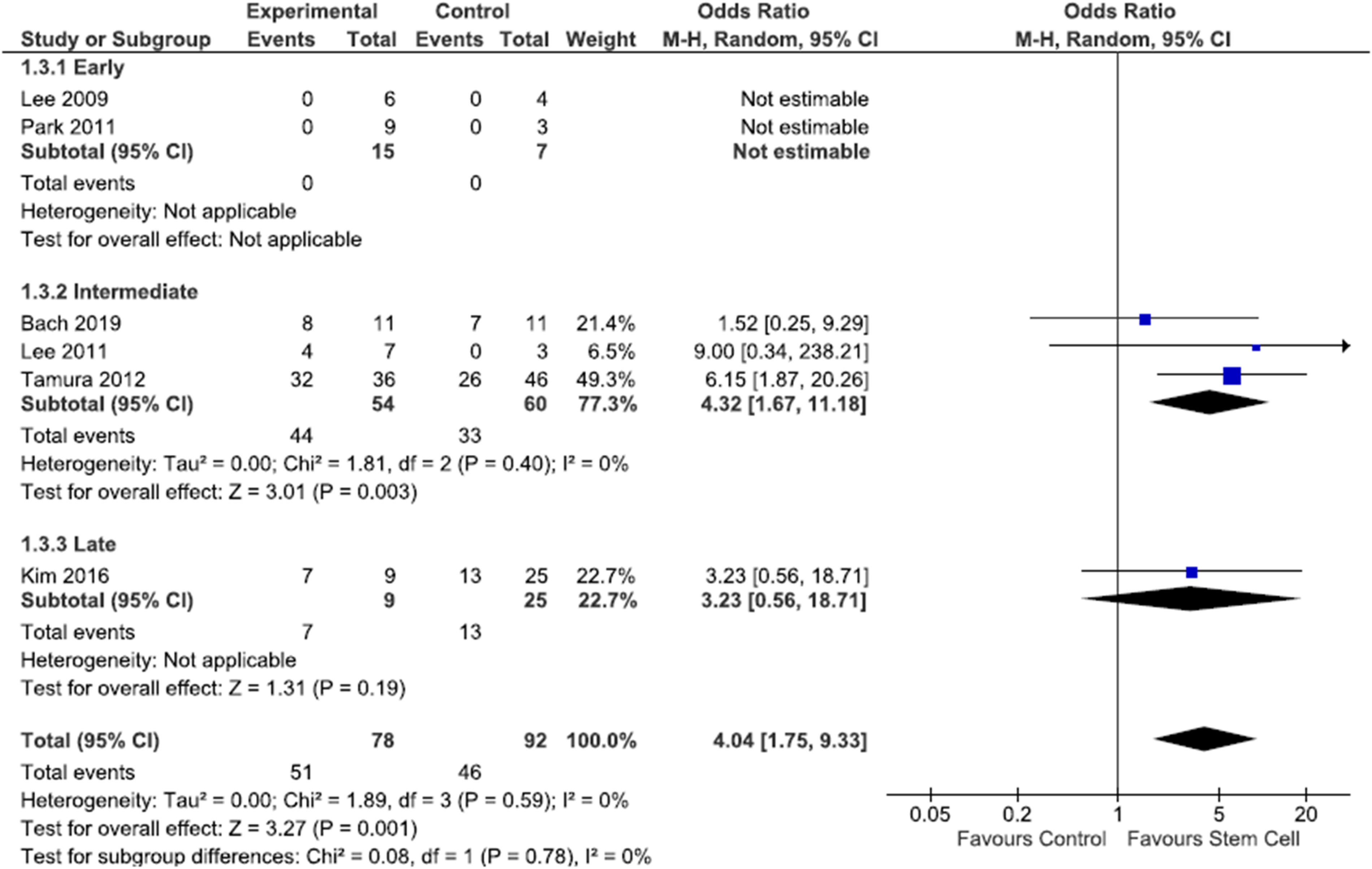
Forest plot of partial motor recovery data. Study outcomes were categorised into early (<8 weeks), intermediate (8 weeks-6 months) and late (>6 months). Partial recovery was defined as an improvement in scores but not back to original, recovery of ambulation (including limited), mild ataxia, Olby score 10–13, RT scale 6–8, MT scale 4, BBB 14–20.
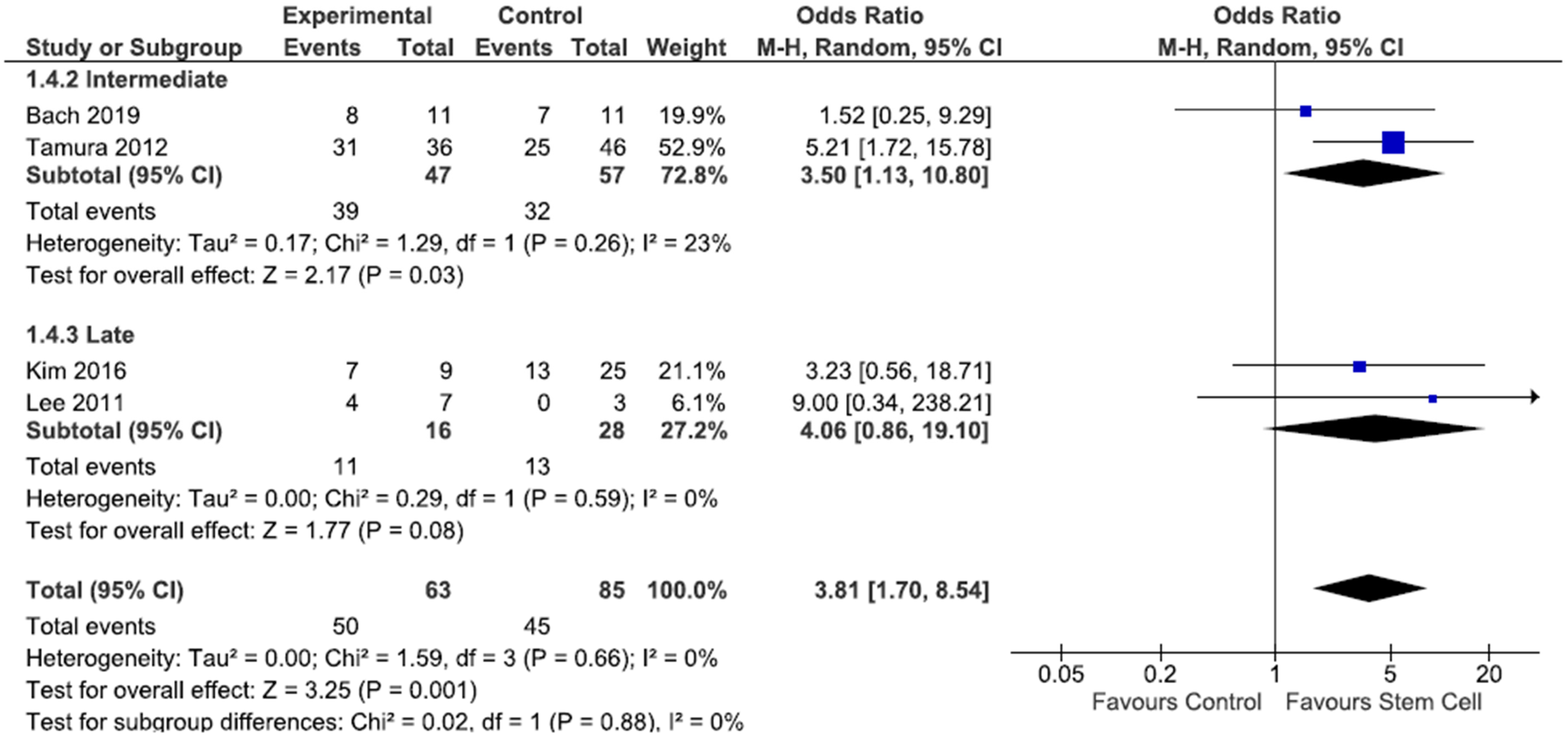
Forest plot of deep pain perception data. Study outcomes were categorised into early (<8 weeks), intermediate (8 weeks-6 months) and late (>6 months). Events are defined as the recovery of deep pain perception as reported by the authors of the papers.
Risk of bias
The risk of bias for each study is presented as a summary (Supplementary Figure S1) and a graph (Supplementary Figure S2).
Synthesis of results
From Figure 2, the overall meta-analysis showed homogeneity across the studies included (Chi2 = 1.83, P = 0.40, I2 = 0%). There was a statistically significant favour of stem cells from combined early, intermediate, and late full motor recovery data across included studies. Odds ratio, heterogeneity and overall effect could not be assessed at the early (<8 weeks) timepoint after SC injection. Only at the late (>6 month) timepoint after SC injection was there a statistically significant finding that SC injections for TSCI achieve full motor recovery compared to control.
From Figure 3, the overall meta-analysis showed homogeneity across the studies finding a partial motor recovery after spinal injection of stem cells (Chi2 = 1.89, P = 0.59, I2 = 0%). There was a statistically significant favour of stem cells from combined early, intermediate, and late partial motor recovery data across included studies. Odds ratio, heterogeneity and overall effect could not be assessed at the early (<8 weeks) timepoint after SC injection. At the intermediate (8 week-6 month) timepoint after SC injection, there was a statistically significant finding that SC injections for TSCI achieve partial motor recovery compared to control, but not at the late (>6 month) timepoint after SC injection.
From Figure 4, the overall meta-analysis showed homogeneity across the studies included (Chi2 = 1.59, P = 0.66, I2 = 0%). There was a statistically significant favour of stem cells from combined intermediate and late deep pain perception data across included studies. There were no studies reporting deep pain perception data at the early (<8 week) timepoint after SC injection. At the intermediate (8 week-6 month) timepoint after SC injection, there was a statistically significant finding that SC injections for TSCI restore deep pain perception compared to control, but not at the late (>6 month) timepoint after SC injection. Four studies28,32–34 did not report on DPP outcomes and one study 30 was excluded as the numbers were unreported for some groups.
Discussion
Summary of evidence
Full and partial motor recovery
We have insufficient data to make conclusions about the early effects of SC injection for TSCI and achieving full or partial motor recovery. There is promising evidence of a statistically significant benefit of SC injection in the intermediate (partial motor recovery only) and late (full motor recovery only) timepoints after SC injection, suggesting that at least some motor recovery can be achieved within the 8-week to 6-month time frame.
For full motor recovery, the 3 studies that were possible to show meta-analysis data for were all conducted with canines with natural injuries,27,29,35 which more accurately resembles the spectrum of TSCI seen in human patients. These same 3 studies used differing injection methods: epidural, 27 intraspinal 29 and intrathecal. 35 Bach et al. 27 and Kim et al. 29 used allogenic adipose-derived mesenchymal stem cells, which are relatively easy to harvest and use and therefore may have a higher chance of translation into human clinical medicine.
For partial motor recovery, data from the study conducted by Lee et al. 31 could be included in the meta-analysis. Due to the small sample size (10 dogs) and the non-significant favour of stem cells, we cannot draw meaningful conclusions from this single study data. However, this data fed into the overall effect of stem cells at the intermediate timepoint post-injection, which significantly favoured the use of stem cells to achieve a partial motor recovery within the 8-week to 6-month time frame.
Overall, this gives a promising clinical indication that SC injection, in whichever modality, is more likely to achieve partial motor recovery within 8 weeks to 6 months post-injection compared to controls and more likely to achieve full motor recovery >6 months post-injection compared to controls.
Deep pain perception
There were no studies recording the early effects of SC injection for TSCI and restoring deep pain perception. There is promising evidence of a statistically significant benefit of SC injection within the intermediate timepoint after SC injection, suggesting that participants are more likely to restore deep pain perception than controls within the 8-week to 6-month time frame. The included studies for meta-analysis27,29,31,35 used differing injection methods and stem cell types – forbidding us to draw meaningful conclusions on the best SC type and injection protocol to recommend in further progression studies and trials.
Progression of SC therapy for TSCI
There are several factors as yet to be optimised before SC therapy can become clinical protocol.
Human and large animal studies currently use a range of delivery methods of SCs: direct intraspinal injection;29–34 intrathecal28,35 and epidural 27 administration. Our data does not lend itself to recommending a particular injection modality. Intraspinal injection targets the lesion directly, but may have little SC migration past the site of injection, 36 risk of infection and iatrogenic secondary damage. 37 Lamanna and colleagues (2017) 38 developed an MRI-guided intraspinal injection apparatus in their proof-of-principle study in mini pigs. This boasts the advantages of reduced invasiveness, lowered risk of iatrogenic injury (by eliminating the need for surgical laminectomy), direct lesion targeting and increased accuracy over standard injection conventions. However, as with most intraspinal injections, there is the risk of leakage of cerebrospinal fluid (causing headaches), spinal cord displacement during injection, vasculature haemorrhage and limited injection sites due to anatomical barriers, such as vertebrae. 38 A desirable alternative is the minimally invasive intravenous injection. 39 Despite this, it is conducted with the assumption that SCs will migrate via the broken blood-spinal cord barrier to the lesion 40 within around 7 days post-injury. 37 Whilst intrathecal and epidural injections are also less invasive than intraspinal injections, they are comparatively less precise and less popular within SC research. 20
Choosing the best type of SCs for optimal TSCI recovery is also an important factor to refine. From Table 1, it appears that allogenic umbilical cord blood is a favourable SC source within canine model experiments at a dosage of 1 × 106 cells. Ideally, autologous SCs are a preferred source of SCs for injection due to their lack of immunogenicity and the ability to source directly from the patient at hand.1,41 Unfortunately, autologous cells require more prior preparation. Furthermore, their therapeutic potency is considered to be lower than foetal SCs. 42 Potency is highest when the source is from foetal or umbilical cord tissue – but at what ethical cost?. 42 Moreover, there is a patient safety risk of teratoma formation with embryonic 5 and pluripotent 43 SCs. Foetal and other allogeneic SCs also risk immunological rejection, which requires lifelong immunosuppressive therapy to achieve success.
Timing of the injection of SCs is vital for achieving their expected efficacy. Injection of SCs within the acute stages of TSCI encourages healing of the lesion and restoration of signalling. Yet, the harsh microenvironment severely diminishes the effectiveness of SCs. 42 Late injection faces the physical barrier of the glial scar, particularly within chronic patients. 42 We decided to focus on stem cell therapy administered within the acute phase (<2 weeks) of the timing of the TSCI, as this would be the realistic time for administration within humans. It is likely that SC therapy may involve injections within both the acute and chronic phases – potentially with different types of SCs. 44 Most studies within this review injected SCs 7 days after the TSCI.27–32,34
Elucidating the optimal parameters for TSCI treatment, probably in small animal models,19,21 will optimise the functional gain in large animal studies and future patients. The multifactorial nature of TSCI pathophysiology and heterogeneity between patients will undoubtedly require the optimal SC therapeutic protocol to harness multiple synergistic interventions22,42,45,46 (Figure 5):
Fast, directed referral to specialist sites with experienced orthopaedic and neurosurgeons Early decompressive surgery16,17 – reducing swelling and hypoperfusion Aggressive management of mean arterial pressure and inflammatory state – preventing further secondary injury Stem cell therapy (including necessary scaffolds,22,47 neurotrophic/growth factors
48
etc.) in both the acute and chronic phases – repairing the primary injury through remyelination,1,49 cell replacement
49
and remodelling of the pathophysiological microenvironment46,50 Anti-astrogliosis therapy51,52 – preventing the formation of the glial scar Immunosuppressant therapy (if applicable) Rehabilitation
21
and physiotherapy programmes
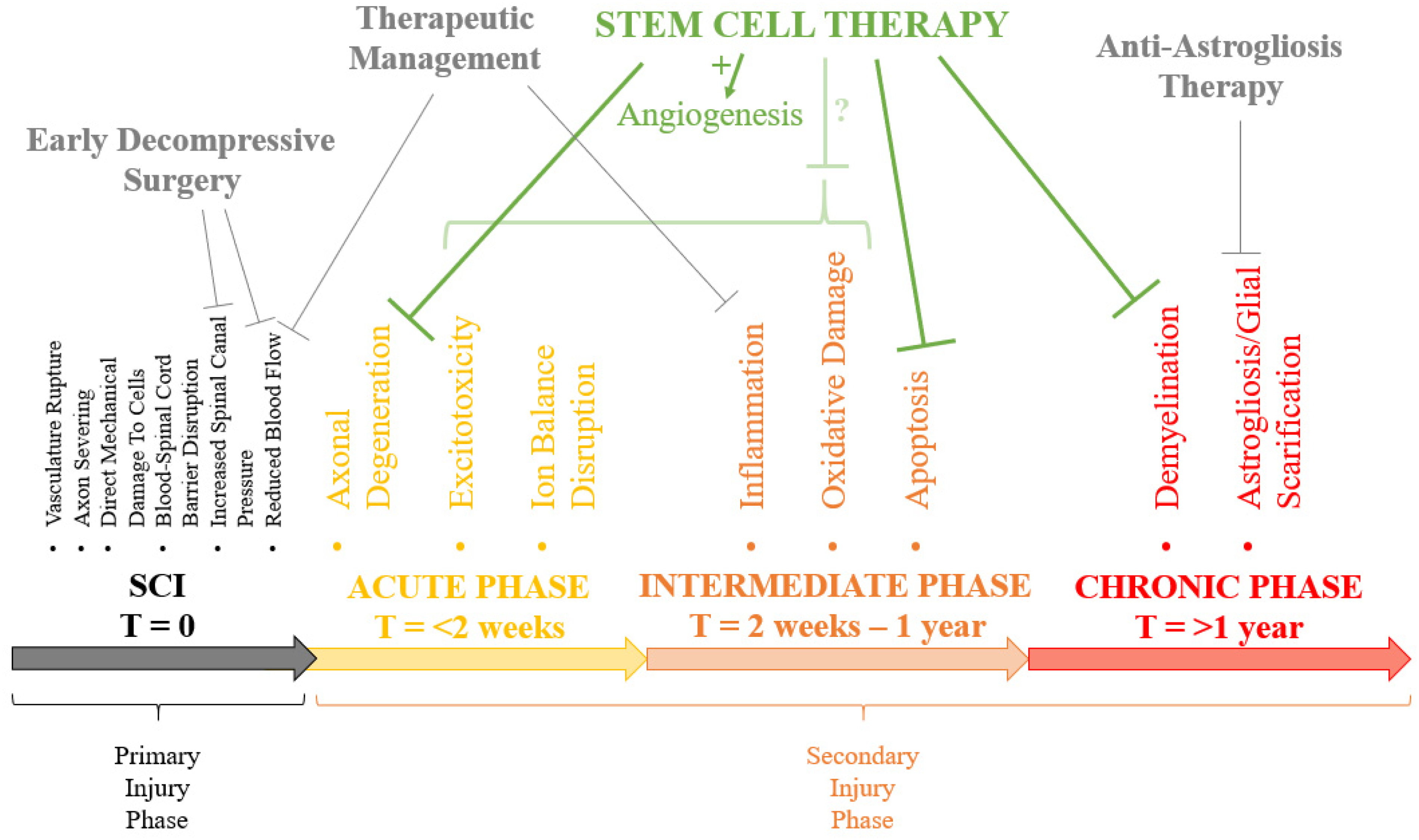
Unfortunately, our searches did not draw out any human studies to include within this systematic review. The exclusion criteria also removed all porcine and NHP studies. From separate online searches, there are a number of trials currently being conducted or published assessing the feasibility and safety of stem cell therapies for TSCI in humans (see Appendix, Table A3). Recruitment numbers in both published and unpublished studies are consistently low, and consequently, we require larger studies over longer time periods to elucidate the actual therapeutic benefits of SC injection. High levels of mild adverse events of SC therapy are observed, 37 including spinal cord oedema, 53 headaches, 6 pain3,6 and non-inflammatory fever. 3 There are also largely unavoidable but powerful modulators of outcome measures that are uncontrolled within clinical trials, including age, mechanism of injury and hormone states. 45 Therefore, it is difficult to predict a time frame before SC therapeutics potentially become routine TSCI treatment.
Strengths
Our systematic review is strengthened by the identification of current advancements in the use of stem cells in treating acute traumatic spinal cord injury in large animals and humans from 4 relevant databases. Secondly, detailed information on the functional outcomes after injection of stem cells into the injured area of the spinal cord was collected so that the best methods could be assessed for use within human trials and the clinical setting. Finally, canine studies are an improvement on the use of mice in terms of size and anatomical similarity, particularly because the canine major CST is located within the lateral column of the spinal cord, which more accurately compares to the lateral funiculus CST and voluntary motor function within humans and primates than the dorsal column CST in rodents. 22
Limitations
Our systematic review is limited by the screening of the full text, data collection and bias assessment all being performed by only one of the authors and therefore may have introduced some omissions and errors to the data.
Secondly, the heterogeneity of currently published canine SC studies hampers their comparability, especially as functional outcome measurements use combinations of: Olby score 54 (Table A1), (revised/modified) Tarlov scale 55 (Table A1), modified Basso, Beattie and Bresnahan scores 56 (Table A2) and various other measurements. These scores were used in 6 studies28,30–34 out of 9 reviewed.27–35
Thirdly, the numbers of canines used within each study (range 10 to 82) are relatively small with varying sources of SCs, hence we require larger, standardised studies for greater statistical power and to allow direct comparison.
Fourthly, studies using ‘natural’ cord injuries,27,29,35 as opposed to experimentally-induced trauma, are considered to be better models for the heterogeneity of human TSCI. 57 Limitations lie within the use of experimental models, as they rely on reproducibility and homogeneity across the test animals 24 and as a result, do not reflect heterogenous human cases recruited in clinical trials. 42 Despite this, it is unlikely that recruitment of veterinary patients with ‘natural’ cord injuries will be sufficient for statistical power, considering exclusion criteria. Furthermore, the presence of the owner-canine bond can result in dissatisfaction if SC therapy is not administered (if placed within the control group) and often must be overcome by ‘promising’ later SC therapy. 57
Finally, the studies included generally had unclear or a high risk of bias at the study level (Supplementary Figures S1 and S2), which warrants cautious interpretation of the results and further studies on the superiority of stem cells with surgical decompression compared to surgical decompression alone. At the review level, our systematic review did not include (after full-text exclusions) any non-human primate or human studies for stem cell transplantation in acute TSCI. Primate model studies are more clinically relevant for translation into human clinical trials, due to the similar size and anatomy of the spinal cord.19,58 Manual dexterity, autonomic function (e.g. urodynamics) and bipedalism in macaque monkeys are also very similar to humans. 21 Primate studies also allow better predictions of human side effects from SC therapy, such as neuropathic pain from the inappropriate targeting of growing axons. 45 Without the option of large numbers of veterinary macaque TSCI patients, experimental studies mostly rely on a drop weight dorsal contusion TSCI model. Despite being easy to reproduce and potentially a better representation of adult contusion TSCI, 42 the variable recovery of contusion injuries makes it almost impossible to know whether the injury is self-healing or helped by the introduction of stem cells into the injury. Therefore, we excluded contusion injuries from the systematic review, unfortunately encompassing all primate studies initially retrieved from the search.
Conclusions
Human TSCI has a tremendously poor prognosis and current therapies do not yet offer a complete recovery. We have reviewed whether SCs could play a beneficial role in TSCI therapeutics. We have found that SCs do offer early improvement for motor function and deep pain perception within canine models, when injected within 2 weeks of injury. Further studies are required in porcine and NHP models as well as further published data from current clinical trial data in humans to further refine protocols in the clinical setting.
Supplemental Material
sj-docx-1-otr-10.1177_22104917221087401 - Supplemental material for A systematic review of large animal and human studies of stem cell therapeutics for acute adult traumatic spinal cord injury
Supplemental material, sj-docx-1-otr-10.1177_22104917221087401 for A systematic review of large animal and human studies of stem cell therapeutics for acute adult traumatic spinal cord injury by Charlotte Laycock, David Kieser, Connor Fitz-Gerald, Sherry Soltani and Chris Frampton in Journal of Orthopaedics, Trauma and Rehabilitation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
