Abstract
Study Design
Systematic review and meta-analysis.
Objectives
The objective of this study was to conduct a systematic review and meta-analysis of the literature regarding the therapeutic effect of embryonic and mesenchymal stem cells on the treatment of traumatic spinal cord injury (SCI) in humans. Primary outcome measures were overall American Spinal Injury Association (ASIA) scores, ASIA motor and sensory scores, urinary and bowel function, pain, and adverse events.
Methods
Studies with human patients ages 18-80 years receiving embryonic, induced pluripotent, or mesenchymal stem cells for SCI were included. Study quality was assessed using the Cochrane risk of bias 2 tool and the Newcastle Ottawa scale for randomized and non-randomized studies, respectively. Primary outcomes were overall ASIA grade, ASIA motor scores, ASIA sensory scores, bladder and bowel function, pain, and adverse events.
Results
Thirty total studies with 656 patients were included, with 43.3% of patients experiencing improvement in ASIA grade, 49.4% in motor function, and 73.6% in sensory function. Qualitative analysis of bladder and bowel outcomes suggests overall improved sensation and control. No serious adverse events were reported. The most common side effects were mild and resolved within hours to weeks without requiring additional medical treatment.
Conclusions
Stem cell transplantation for SCI appears to offer moderate improvements in overall ASIA grade, motor, sensory, bladder, and bowel function, accompanied by a relatively mild and transient side effect profile. Further research, particularly high-quality, blinded, randomized controlled trials, is essential to optimize treatment protocols and achieve more consistent and improved clinical outcomes.
Introduction
Spinal cord injury (SCI) results from damage to the spinal cord, causing functional changes. 1 Acute SCI treatment involves hemodynamic management, surgical stabilization, and methylprednisolone administration to stabilize the spine and reduce inflammation.1-5 The use of steroids in acute spinal cord injury (SCI) remains controversial, with early studies suggesting benefit, but more recent meta-analyses showing no improvement in neurologic outcomes and a higher risk of complications. Notably, studies by Liu et al (2019) and Sultan et al (2020) found that high-dose steroids did not enhance motor or sensory recovery and were associated with increased risk of gastrointestinal bleeding, pneumonia, and hyperglycemia.6,7
Subsequent recovery relies heavily on physical therapy. However, SCI recovery is inherently challenging due to the central nervous system’s limited regenerative capacity, including complex neural connections, an inhibitory environment, glial scar formation, and a scarcity of endogenous stem cells. 8 Consequently, cellular transplantation has emerged as a promising therapeutic approach, garnering significant research interest. Transplanted cells can potentially improve outcomes by promoting axonal regeneration and remyelination and by releasing neuroprotective factors that enhance neuronal survival. 9
Stem cells are present in most multicellular organisms and can divide asymmetrically, producing both new stem cells and differentiated cells. 10 They are classified by their differentiation potential as totipotent, pluripotent, multipotent, or unipotent. Totipotent cells can form all cell types, including an entire organism, while pluripotent cells can create cells from all three germ layers but not placental tissues. Multipotent cells differentiate within a single lineage, such as mesenchymal stem cells, and unipotent cells specialize in one type while retaining self-renewal abilities. 11 Stem cells are further categorized by origin as embryonic or adult. Embryonic stem cells (ESCs), derived from pre-implantation embryos, are totipotent or pluripotent, whereas adult stem cells, found post-development, are usually multipotent or unipotent. 11
The historical belief was that stem cells permanently lose their ability to self-renew once differentiated, but this changed in 2006 when Dr Shinya Yamanaka developed a method to reprogram adult somatic cells into induced pluripotent stem cells (iPSCs). By introducing four specific transcription factors, Yamanaka restored their pluripotent properties, enabling the creation of stem cells without using embryos and addressing ethical concerns surrounding embryonic stem cell use.12,13
Mesenchymal stem cells (MSCs) are among the most extensively studied stem cell types for their therapeutic potential in treating traumatic SCI. These multipotent cells can differentiate into all mesoderm-derived cell types, including monocytes, chondrocytes, osteoblasts, adipocytes, and neurocytes. MSCs are traditionally obtained from the umbilical cord blood, the placenta, bone marrow, or adipocytes, each source with their own therapeutic nuances. 8 The use of stem cells for treating traumatic spinal cord injury is promising due to their ease of isolation, maintenance prior to transplantation, and minimal to no immunogenicity after transplantation. The proposed mechanisms by which stem cells exert therapeutic effects include their ability to inhibit the release of inflammatory cytokines, promote angiogenesis, and create an anti-apoptotic environment for surrounding cells. 8
The objective of this study was to conduct a systematic review and meta-analysis of the literature regarding the therapeutic effect of embryonic, induced pluripotent, and mesenchymal stem cells on the treatment of traumatic SCI. We hypothesize that stem cell treatment in patients with SCI will improve clinical outcomes.
Methods
Study Design
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol for this study is registered within the PROSPERO database (ID#: CRD42024568194). The study was exempt from Institutional Review Board approval.
Selection Method and Data Extraction
Using the keywords “mesenchymal stem cell,” “induced pluripotent stem cell,” “embryonic stem cell,” and “spinal cord injury,” a comprehensive literature search was performed in PubMed and Embase via Elsevier for articles published without date limits up until August 2024. Results were restricted to publications in the English language and full-text manuscripts. The literature search was not re-run prior to analyses.
Screening included two stages: title/abstract screening and full-text screening. All imported references were independently screened by two investigators (ZT and DC). Any disagreements on study eligibility during either stage of screening were resolved by a third independent reviewer (TR). All screening was performed utilizing Covidence software (Version 2, Veritas Health Innovation) following importation of references.
Eligibility Criteria
All studies with original data that had human patients with acute or chronic SCI with stem cell treatment were included. Studies were only included if the reported type of stem cells used were embryonic, induced pluripotent, or mesenchymal. Types of studies included were retrospective cohort studies, prospective cohort studies, randomized control trials (RCTs), case-control studies and case series.
Risk of Bias Assessment
The Cochrane Risk of Bias Version 2 (ROB2) tool was used to evaluate RCTs. ROB2 evaluates RTCs based on 5 domains: randomization process, effect of assignment to intervention, missing outcome data, outcome measurement, and selection of reported results. Each domain poses a series of questions that once answered are implemented into an algorithm yielding a low, moderate, or high risk of bias for each domain and ultimately the overall study. 14
The Newcastle-Ottawa Scale (NOS) was used to assess the quality of nonrandomized studies. The NOS evaluates studies on strength of selection, comparability, and exposure or outcomes depending on study type. Each study may receive a maximum of 9 “stars” to indicate the highest quality study. 15 The total number of “stars” was converted to a low risk (7-9 stars), somewhat concerning risk (4-6 stars), and high risk (0-3 stars) of bias for comparability with the ROB2 for the RCTs.
Data Synthesis and Statistical Analyses
The data were extracted and tabulated in an excel spreadsheet. Data extracted included authors, year published, journal of publication, study design, level of evidence, number of patients, average duration of follow up, type and route of therapy, American Spinal Injury Association Impairment (ASIA) motor and sensory scores before intervention and on follow-up, study quality, and description of complications (if any). Since included studies were primarily single arm cohorts, a traditional meta-analysis involving calculating and pooling effect sizes could not be performed. Hence, weighted means for continuous variables and weighted odds ratios for categorical variables were calculated to describe the sample of studies. The primary categorical outcome variables were improvement in overall ASIA scores, and improvement in ASIA sensory and motor scores. The pooled incidence of improvement was calculated with 95% confidence intervals (CIs) using a random effects model with double arcsine transformation (Freeman-Tukey) for sensory and motor symptoms separately. If there was no improvement in any patient, then a continuity correction was performed by adding 0.5 to the numerator and denominator. Analyses were not controlled for study quality. All other variables were described qualitatively. One of our planned sub-analyses was to compare the incidence of improvement between MSCs and human ESCs. However, only 3 out of 30 included articles used ESCs and they were all from the same author. Their rate of improvement was similar to the pooled rate of improvement, so this analysis was not performed. A P-value of < .05 was considered statistically significant and all analysis were performed using JBI Sumari (Adelaide, Australia). 16
Results
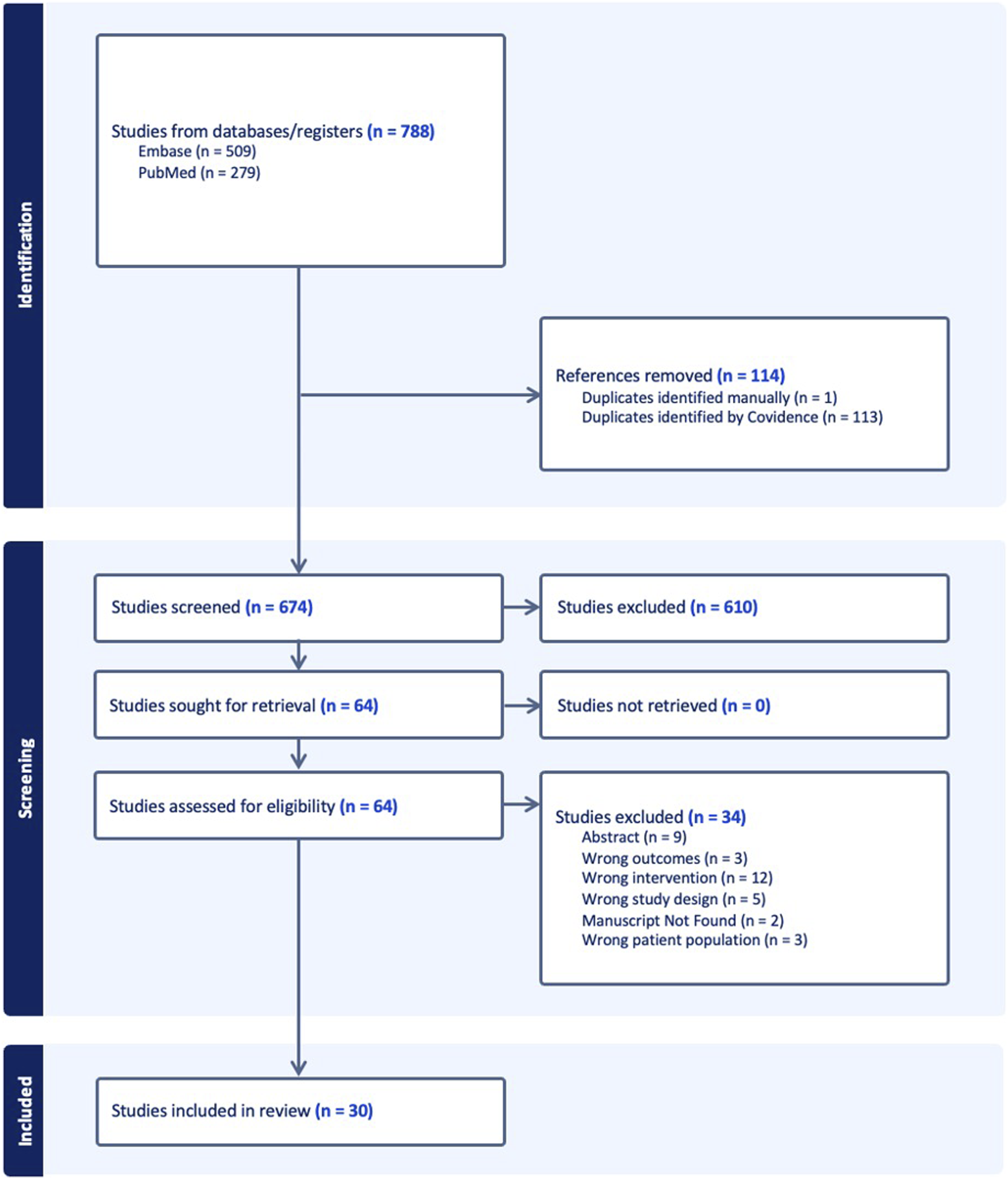
The initial search included 788 studies total: PubMed (279) and Embase (509). A total of 114 duplicates were removed, leaving 674 unique studies to screen. An evaluation of titles/abstract followed by full-text assessment for eligibility resulted in the removal of 644 studies. The remaining 30 studies were included in the analysis. Figure 1 displays the PRISMA search strategy utilized in this study. PRISMA study inclusion flow chart (adopted from covidence software).
50
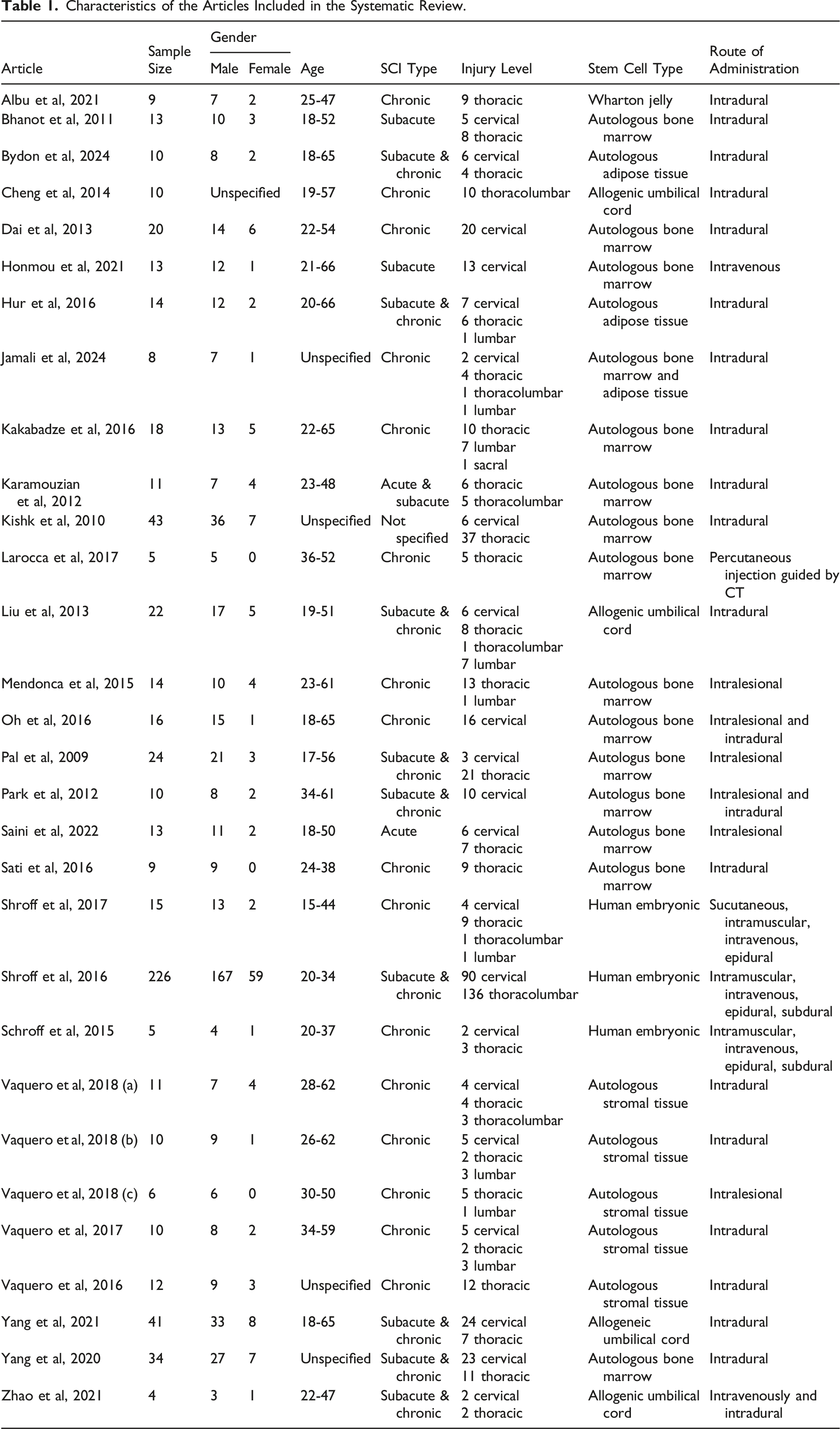
Characteristics of the Articles Included in the Systematic Review.
Twenty-two studies with 578 patients reported pre-treatment and post-treatment overall ASIA scores. Before stem cell therapy, 408 (70.6%) patients were ASIA A, 82 (14.2%) ASIA B, 77 (13.3%) ASIA C, and 11 (1.9%) ASIA D. Figure 2A presents the pooled incidence of improvement which was 0.433 (0.384, 0.483), meaning 43.3% of patients experienced improvement in their overall ASIA score. In the 18 studies that reported the average change in ASIA grades, 13 reported an average increase of at least 1 grade, 2 studies reported average increases less than 1 grade and 3 studies reported no change in ASIA grade. No studies reported a worse post-treatment ASIA grade (Table 2). Improvement in overall ASIA score for stem cell therapy (a), and controls (b). Clinical Outcomes of the Articles Included in the Systematic Review.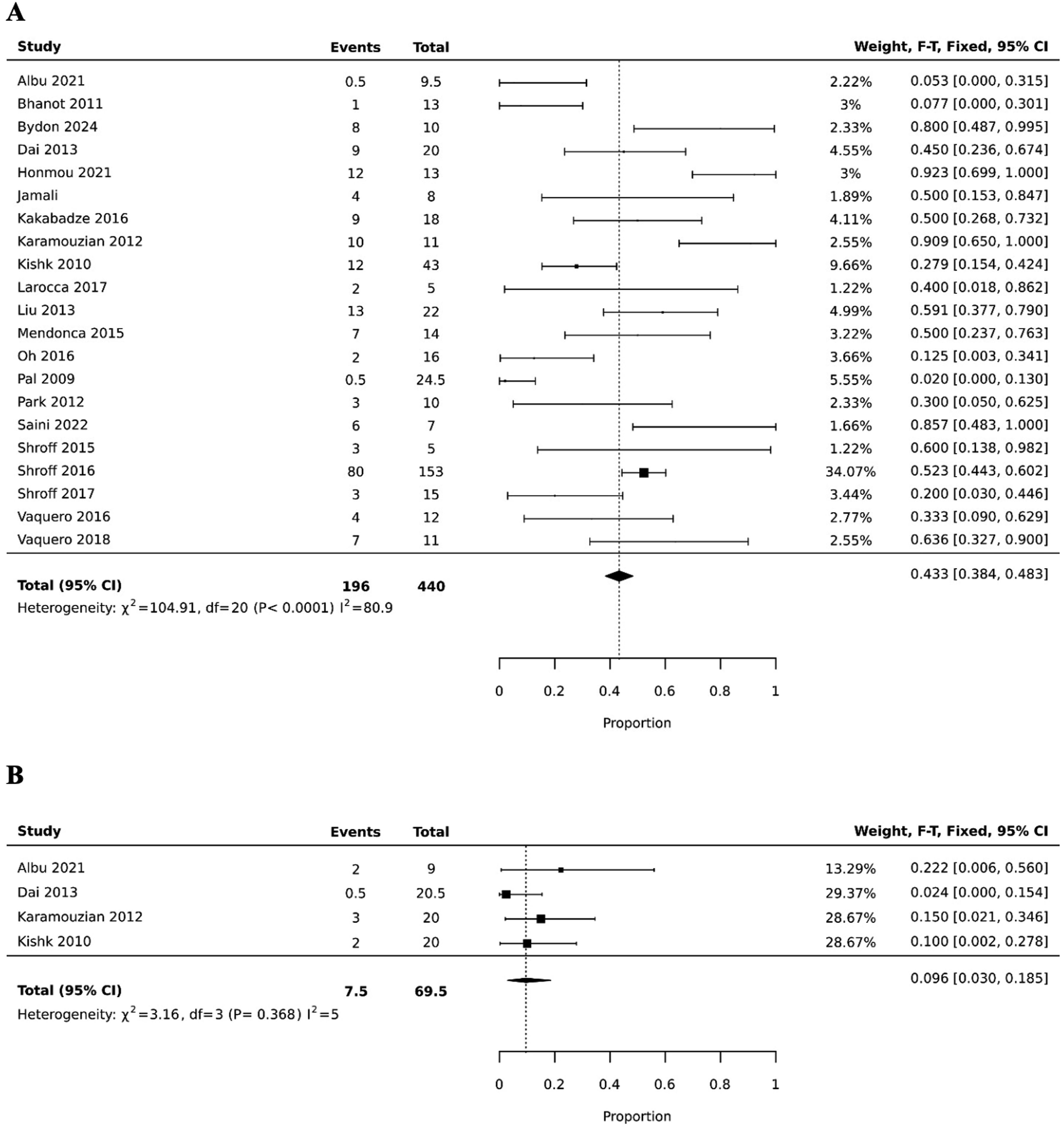
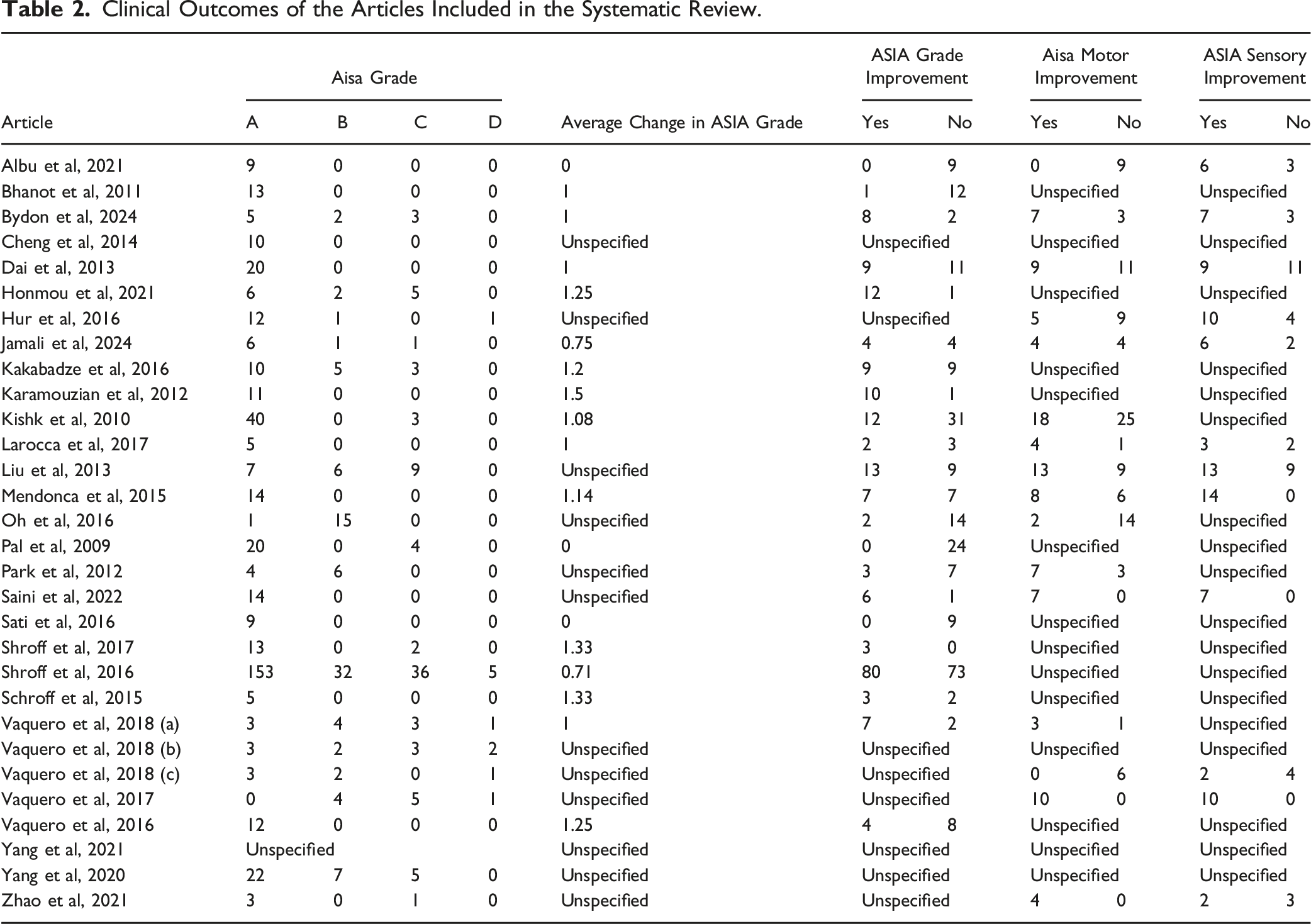
Four studies reported ASIA grade improvement data for their control populations, comprising a total of 68 patients. Sixty-seven patients were classified as ASIA A and 1 patient as ASIA B at baseline; no patients were categorized as ASIA C or D. The pooled incidence of ASIA grade improvement among control patients was 9.6% (0.030, 0.185) (Figure 2B). This rate was significantly lower than that observed in the stem cell therapy group (P < .001).
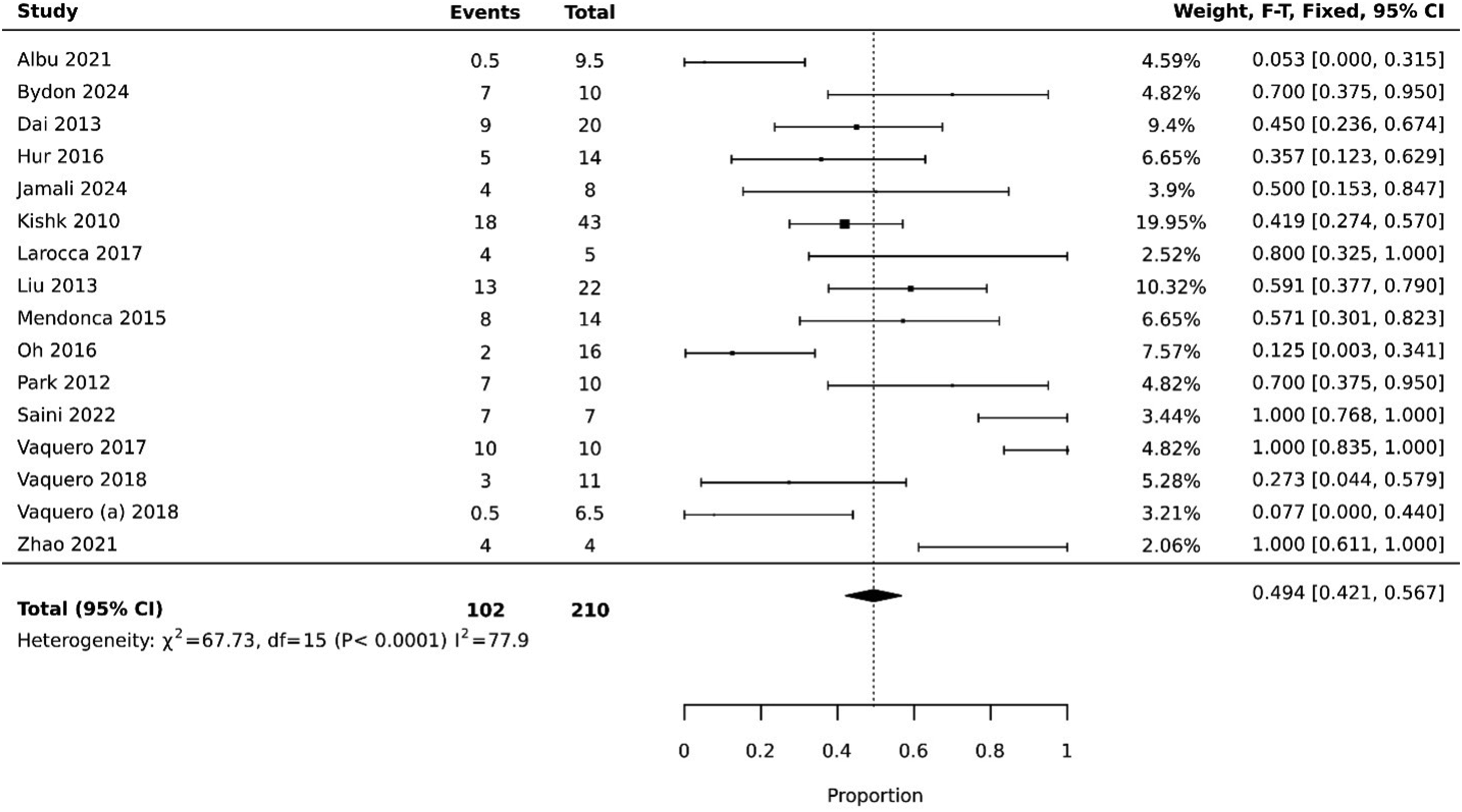
Twelve studies with 129 patients reported ASIA sensory outcomes, and Figure 3 presents the pooled incidence of improvement. The pooled incidence was 0.736 (0.648, 0.816), which means that 73.6% of patients reported an improvement in sensory symptoms on follow-up. Sixteen studies with 209 patients reported ASIA motor outcomes, and Figure 4 presents the pooled incidence of improvement. The pooled incidence of improvement was 0.494 (0.421, 0.567), meaning 49.4% of patients reported an improvement in motor symptoms on follow-up. Improvement in ASIA sensory symptoms. Improvement in ASIA motor scores.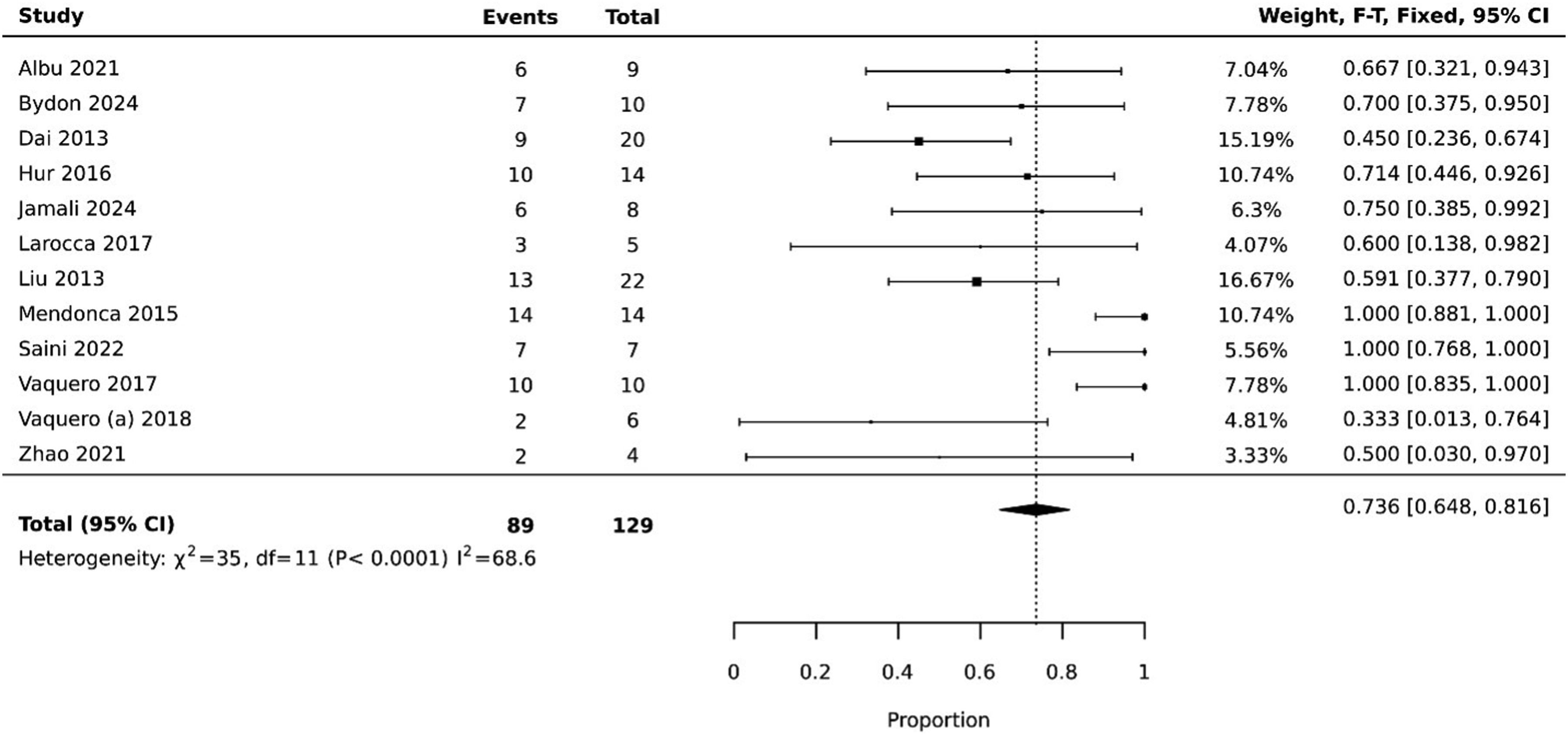
Changes in Bladder and Bowel Function, Improvement in Pain, and Adverse Events in the Articles Included in the Systematic Review.
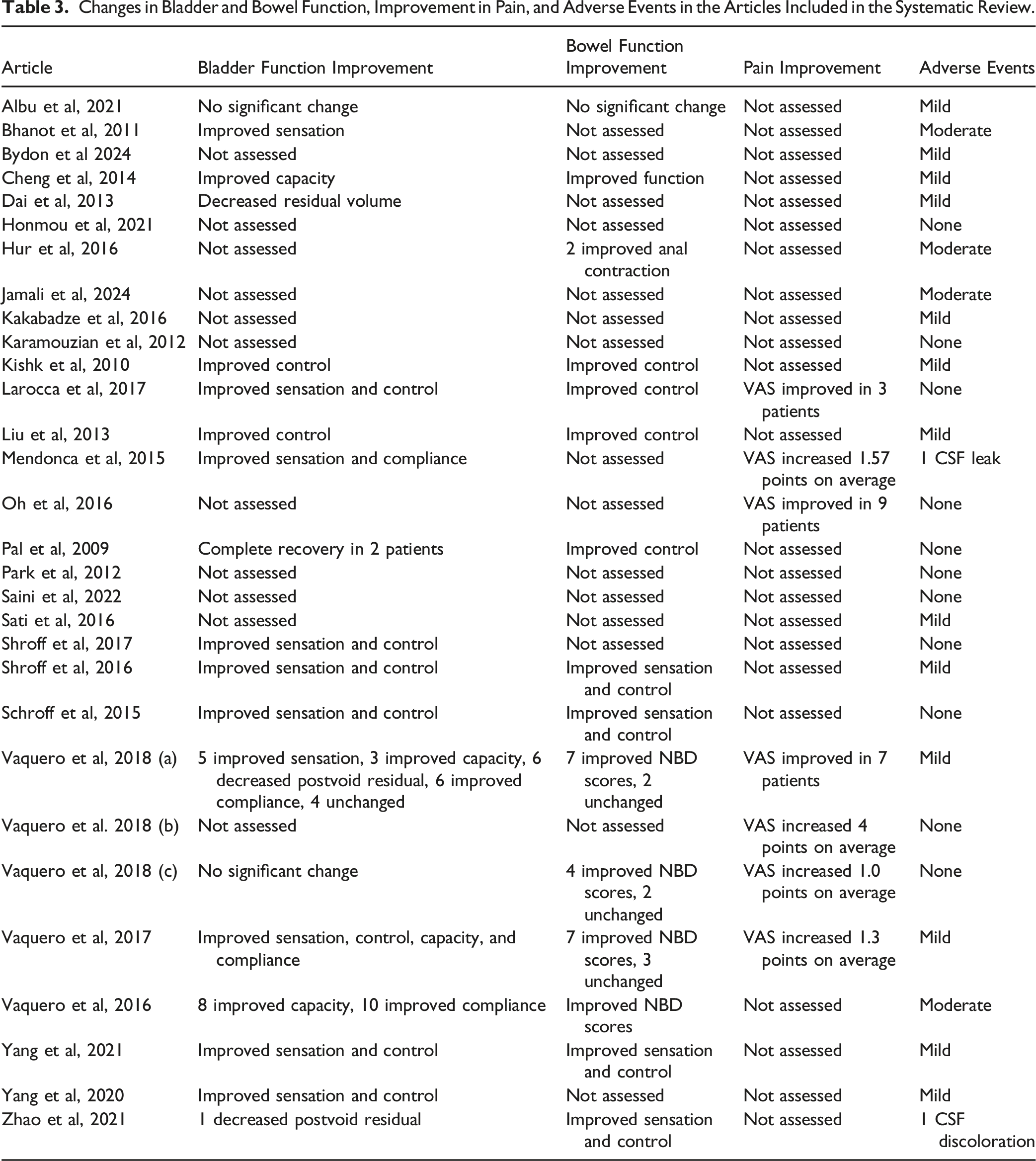
Fifteen studies reported outcomes regarding bowel function. Nine studies reported improvements in overall bowel sensation and control following treatment of SCI with stem cell therapy.17,18,20,21,24,26,28,30,32 Vaquero et al 33 reported significant improvements in neurogenic bowel dysfunction (NBD) scores. In a series of 3 independent trials by Vaquero et al, they reported improved NBD scores in 7 of 11 patients, 22 4 of 6 patients, 35 and 7 of 10 patients. 36 Hur et al 37 reported improved anal contraction in 2 of 14 patients. Albu et al 34 reported no significant changes in bowel function between the control and stem cell therapy groups (Table 3).
Seven studies reported changes in pain using the Visual Analog Scale (VAS) after stem cell therapy. Improvement in VAS score was reported in 3 of 5 patients by Larocca et al, 26 9 of 16 patients by Oh et al, 38 and 7 of 8 patients by Vaquero et al. 22 In studies that reported changes in VAS scores, on average scores improved by 4 points, 39 1.57 points, 31 1.3 points, 36 and 1 point 35 (Table 3).
No studies reported serious adverse events such as death, surgical site infection, intracranial infection, permanent neurologic dysfunction, respiratory distress, tumors, or immune reactions. Eleven studies reported no adverse events. Nineteen studies reported minor or moderate adverse events. The only serious adverse event was reported by Mendonca et al 31 where one patient sustained a cerebrospinal fluid (CSF) leak and was managed non-surgically without further sequelae. Zhao et al 30 reported one patient with clear-yellow colored CSF upon administration of the second stem cell dose, however biochemical testing was normal (Table 3).
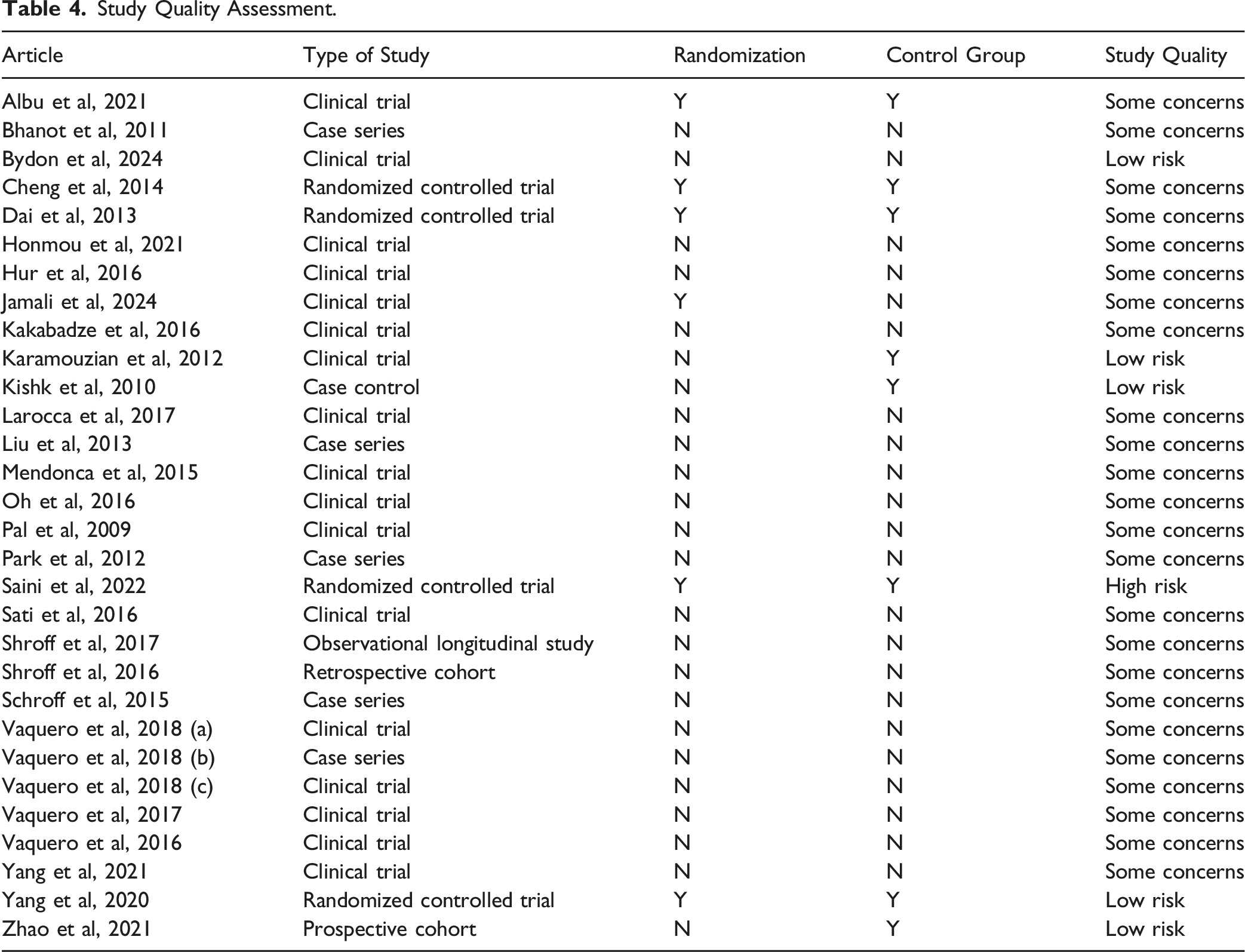
Study Quality Assessment.
Discussion
Spinal cord injury, with a reported global incidence of 8.0 to 57.8 cases per million, poses substantial economic, social, and functional burdens on individuals and their communities.40,41 These burdens stem from the need for intensive acute care and long-term management of complications such as bladder/bowel dysfunction, chronic pain, and frequent hospital readmissions. The estimated lifetime cost per individual with SCI can reach $1.5 to $5.4 million, underscoring the critical need for safe and effective treatments to alleviate these lifelong hardships.42,43
Stem cell therapy has been studied in animal populations for the past few decades with the aim of developing a treatment that restores neurological function. A comprehensive meta-analysis by Antonic et al, in 2013, encompassing 156 publications on animal models, reported a 25% improvement in overall motor and sensory function following allogeneic stem cell treatment. 44 This promising preclinical evidence has fueled growing interest in translating stem cell therapies to human SCI patients, motivating the present systematic review of the clinical literature.
This systematic review and meta-analysis examined 30 studies published between 2009 and 2024, encompassing 656 patients who received stem cell therapy for SCI. Mesenchymal stem cells were overwhelmingly the most common cell type used (27 studies), likely due to their ease of acquisition from sources like bone marrow, adipose tissue, and placenta, often under local anesthesia. Their straightforward culturing, storage, and injectability further contribute to their popularity. Embryonic stem cells (3 studies) were less frequently used, likely due to the ethical and moral considerations surrounding their derivation from human embryos, which restricts research. 45 The absence of studies utilizing iPSCs likely reflects the relative novelty of this technology, the technical challenges associated with differentiation, and potential concerns regarding tumorigenicity and immunogenicity.46,47
This meta-analysis revealed that 43.3% of SCI patients receiving stem cell therapy experienced an improvement in their overall ASIA impairment scale grade, with 72% of studies reporting an average improvement of at least one grade. This overall ASIA improvement was significantly higher than the 9.6% of SCI patients who improved after receiving a placebo. Furthermore, 73.6% of patients demonstrated improved ASIA sensory scores, and 49.4% showed improvements in ASIA motor scores. These findings align with previous meta-analyses. Mototo-Meijide et al reported ASIA grade improvements ranging from 7% to 100% across studies, with most demonstrating sensory improvements and, to a lesser extent, motor improvements. 8 Similarly, Chen et al found significant improvements in ASIA grade, sensory, and motor scores in MSC-treated patients compared to those receiving physical therapy alone. 48 Shang et al observed that 48.9% of patients improved by at least one ASIA grade following stem cell treatment, noting “slight improvement” in sensory and motor function. 49 Collectively, these data suggest that stem cell therapy may offer clinically meaningful motor and sensory recovery for a substantial proportion of SCI patients.
Bladder and bowel function improvements were assessed across the included studies using a variety of metrics, ranging from patient-reported changes in sensation and control to objective measures like bladder compliance, detrusor pressure, and residual volume. The heterogeneity in reporting data precluded meta-analysis of these outcomes. Of the 19 studies with data on changes in bladder function, 17 reported overall improvements in bladder function. Similarly, 14 of 15 studies with bowel function data reported improvements (Table 3). While Shang et al observed improvements in nearly half of their patients, they also noted that these benefits may not have met patient expectations, highlighting the need for standardized, objective metrics for assessing bladder and bowel function in future studies. 49
The safety profile of stem cell therapy for SCI appears favorable. Of the 30 studies included in this review, 11 reported no adverse events, and 19 described minor to moderate transient events that resolved without long-term consequences. These events, typically resolving within 24 h to 2 weeks without intervention, included headache, nausea/vomiting, dizziness, fever, myalgias, radiculopathy, paresthesias, and urinary tract infections. One CSF leak, reported by Mendonca et al, was successfully managed non-operatively, 31 and Zhao et al reported a case of benign CSF discoloration. 30 This generally benign safety profile is consistent with other published data. Chen et al, 48 in their 18-study systematic review, found that only 13 studies reported adverse events, with no severe complications or tumor development. Their most common findings (fever, headache, back pain, and paresthesias) mirrored those observed in the present review. Similarly, Shang et al 49 reported a low incidence of mild, self-limiting adverse events (28 in 2439 patients). The low frequency and mild nature of these events, particularly with MSCs, may be attributable to the autologous nature of the cells used in most studies, minimizing the risks of immunogenicity and rejection.
Study quality was assessed using the Cochrane ROB2 tool for RCTs and the NOS for non-randomized studies. ROB2 evaluates five domains: randomization process, deviations from intended interventions, missing outcome data, measurement of the outcomes, and selection of the reported results, classifying studies as having low, moderate, or high risk of bias. 14 NOS assesses patient selection, comparability, and outcome, awarding up to nine “stars.” 15 To maintain consistency, NOS scores were translated into risk categories: 0-3 stars (high risk), 4-6 stars (moderate risk), and 7-9 stars (low risk). The included studies comprised 17 clinical trials, 5 case series, 4 RCTs, one case-control study, one retrospective cohort, one prospective cohort, and one longitudinal study. Only 6 (20%) studies employed randomization, and 8 (26.7%) included a control group. Overall, five studies were rated as having low risk of bias, 24 as moderate risk, and one as high risk (Table 3). This moderate study quality is a recurring issue in stem cell therapy research for SCI. Shang et al reported that only 27.4% of their included studies used blinding protocols, 49 while Chen et al found that 55.5% of their studies used randomization and only 22.2% used blinding of participants, personnel, and outcome assessors. 48 This lack of randomization and blinding may contribute to an overestimation of stem cell therapy’s effectiveness.
This systematic review and meta-analysis, along with others, suggests that stem cell therapy for SCI may lead to modest but clinically meaningful functional improvements with a favorable safety profile. However, significant heterogeneity exists across studies regarding stem cell type, inclusion/exclusion criteria, dosage, and administration route. This variability underscores the need for future research to establish a standardized, optimized treatment protocol, ideally evaluated in high-quality, double-blind, RCTs. Such trials should also investigate factors potentially influencing treatment efficacy, including SCI location, severity, and timing of intervention. In this evolving field of regenerative medicine, analyzing homogeneous data across multiple studies is crucial for drawing definitive conclusions about treatment efficacy.
This study has several limitations. The inclusion of predominantly non-randomized studies with moderate methodological quality and a lack of detailed data limits the strength of our conclusions. The ASIA impairment scale, while widely used, relies on physician interpretation of clinical exams, potentially introducing misclassification bias. Considerable heterogeneity across studies in factors such as stem cell dosage and timing, follow-up duration, SCI level and severity, and administration route further complicates analysis. This heterogeneity, combined with the limited number of controlled studies, precluded more in-depth meta-analyses and restricts the scope of our findings. Despite these limitations, this study provides valuable insights by systematically reviewing the latest research on the most promising and extensively studied stem cell lines, offering a comprehensive update to the existing literature.
Conclusion
Stem cell transplantation for SCI appears to offer moderate improvements in overall ASIA grade, motor, sensory, bladder, and bowel function, accompanied by a relatively mild and transient side effect profile. This review found that 43.3% of patients experienced improvement in ASIA grade, 49.4% in motor function, and 73.6% in sensory function. While these findings suggest MSC transplantation may be a viable therapeutic option for SCI, further research, particularly high-quality, blinded, RCTs, is essential to optimize treatment protocols and achieve more consistent and improved clinical outcomes.
Footnotes
Author Contributions
ZT, DEC, TJR, and CL developed the methodology of this protocol. ZT, DEC, and TJR collected the data. ZT, DEC, TJR, and CL drafted the manuscript. MNH provided all statistical analyses and calculations. ZT, DEC, TJR, MNH, DK, BH, MS, EV, and CL edited and approved the final draft of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
