Abstract
Background:
Necrotising soft tissue infection (NSTI) is rare but fatal. Andreasen proposed finger test as an early diagnostic tool to differentiate NSTI from other soft tissue infections. We aim to evaluate the accuracy and reproducibility of the test for the diagnosis of NSTI.
Methods:
Patients who were admitted to our department from 2012 to 2016 with suspicion of NSTI and finger test done were retrospectively reviewed. Finger test was done and interpreted as described by Andreasen. Definitive diagnosis of NSTI was confirmed with surgical and pathological findings.
Results:
Among the 35 patients included in the study, NSTI was confirmed in 10 cases. Finger test had a sensitivity of 100%, a specificity of 80%, positive predictive value of 66.7%, negative predictive value of 100% and an overall accuracy of 85.7%. There was no difference in demographics or comorbidities between NSTI and non-NSTI groups. Surgeons involved had 76.7% agreement and moderate reproducibility (kappa = 0.48) on the diagnostic criteria of finger test.
Conclusion:
A negative finger test was reliable to exclude NSTI and a positive test suggested further surgical exploration. Yet, clinical judgement was still of paramount importance to treat NSTI promptly.
Introduction
Necrotising soft tissue infection (NSTI) is a life-threatening infection involving subcutaneous tissue and muscle fascia. The term ‘necrotising fasciitis’ was introduced by Wilson in 1952 1 and was formally defined by Stamenkovic and Lew by histological patterns of necrosis of the fascia and subcutaneous fat with thrombosis of the microvasculature and sparing of the underlying muscles. 2 Although surgical exploration and pathological examination remained the gold standard for definitive diagnosis, 3 –5 early recognition played an important role so as to provide prompt and appropriate treatment, including both antibiotics and operation. However, differentiating NSTI from other non-NSTIs is difficult, owing to the vague initial presentation and the rarity of the disease, as reported annual incidence in the United States was 4.3 per 100,000 population. After being recognised for more than 50 years, despite technological advancement, mortality could still be as high as 75% from different series. 2,6 –8
Clinicians from all over the world made various attempts to find a way to diagnose NSTI at an earlier stage. Computed tomography (CT) and magnetic resonance imaging (MRI) had promising initial results. Laboratory Risk Indicator for Necrotising Fasciitis (LRINEC) score, developed by Wong et al. 9 to stratify the probability of NSTI from blood markers, was also readily available. But the generalisability of all these modalities in other centres or localities had not been proved.
Andreasen et al. 10 first described the finger test in 2001. Together with rapid frozen-section biopsy, they acted as a quick and accurate guide to early diagnosis of NSTI. However, there had not been any study validating the accuracy and reproducibility of the test. The purpose of the study is, therefore, to evaluate the performance of the finger test for the diagnosis of NSTI and the reproducibility of the test among different surgeons in our centre.
Methods
Patient selection
Patients who were admitted to Alice Ho Miu Ling Nethersole Hospital from 2012 to 2016 with the finger test done for clinically NSTI were included in this retrospective case series.
Data were extracted from computer-generated search from the Medical Records Department for all patients diagnosed of necrotising soft tissue infection or cellulitis in the above time frame. Individual medical records were further revisited for documentations of results of the finger test. Patients who were operated immediately after admission without the finger test done or who died before operation with no definitive pathological diagnosis were excluded. Cases with incomplete documentation of findings of the finger test were excluded as well.
Diagnosis
The finger test was performed in accordance with the decision made by the attending surgeon, in the way originally described by Andreasen in 2001. 10 A 2-cm incision is made in the skin at the suspected diseased area down to the deep fascia. Then, a gentle probing manoeuvre is performed at the level of the deep fascia (Figure 1). With the presence of all three signs – lack of bleeding in subcutaneous tissue, dishwater fluid and tissue dissection with minimal resistance at the level of the deep fascia – the finger test is considered positive and emergency operations would be proceeded either in our hospital or transferred to tertiary centre in non-office hours.
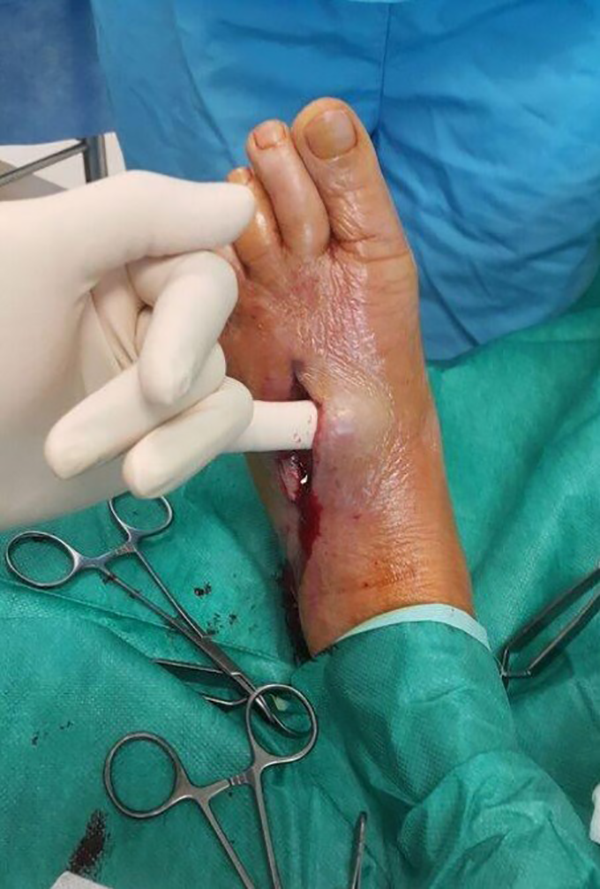
Finger test. Tissue dissection with minimal resistance at the level of fascia upon gentle probing.
Definitive diagnosis of NSTI was determined by a combination of positive operative findings, including necrotic subcutaneous tissue, oedema fascia, thrombosed vessels or watery, thin, often foul-smelling fluid, 11 as described by attending surgeons from surgical exploration, and histopathological tissue examination 2 from intraoperative specimens. Findings included necrosis of fascia and subcutaneous tissue, thrombosis of microvasculature and sparing of underlying muscle. Cases that were treated successfully with antibiotics alone were considered not having NSTI with or without tissue pathology.
Statistical analysis
Demographic and clinical data are analysed, including age, sex, presenting symptoms and signs, duration of symptoms before admission, initial vital signs and laboratory parameters. Results of the finger test and surgeons who performed the finger test, including seniority and their understanding to the test, are also analysed.
Primary outcomes were measured in terms of sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and overall accuracy (defined as the proportion of true results among the total number of cases) of the finger test. Demographic and clinical data mentioned above were also tested – continuous variables were compared using Student’s t-test and categorical data by χ 2 test or Fisher’s exact test, where appropriate. A p value less than 0.05 was considered statistically significant. Significant clinical or laboratory parameters were further categorised with respect to normal limits in local laboratory and analysed in terms of sensitivity, specificity, PPV, NPV and accuracy.
Surgeons who had performed the finger test were invited to complete a questionnaire on the understanding of the finger test, from the findings included in the diagnostic criteria (Q1–3), the interpretation of the findings (Q4–7) and their reliance on the test (Q9). Inter-surgeon reliability was then calculated in terms of percentage agreement and Fleiss’ kappa coefficient on the questionnaire. 12
Comparison
We also attempted to compare the accuracy of the finger test with another readily available and renowned diagnostic tool, LRINEC score, when applied to our cases. The LRINEC score of our patients was calculated retrospectively from admission laboratory parameters. Cut-off of 6 or more 9 was regarded as positive, as originally described by Wong et al. Statistical analysis was performed in terms of sensitivity, specificity, PPV, NPV and accuracy. Receiver operating characteristic (ROC) curve was implemented to evaluate the predictive accuracy of the LRINEC score.
Results
In the study period from 2012 to 2016, 42 patients were admitted to our hospital for suspected NSTI. Four patients were directly sent to the operating theatre because of a full-blown picture of NSTI. They were all confirmed to have the disease intraoperatively and histologically. The remaining 38 patients had the finger test performed. One case had to be transferred to tertiary centre for further management of the condition but succumbed before definitive surgery. Two other cases had incomplete documentation of the finger test and were also confirmed of NSTI after operation. Therefore, 35 patients were included in the study. Mean age of admission is 63.7 (28–102) years. Average duration of symptoms prior to admission was 3.0 (0–14) days. Mortality rate of the entire study population was 8.6% (3 in 35) and 20% in the NSTI group (2 in 10) in particular.
Demographics, clinical presentation and laboratory results are presented in Tables 1 and 2. It is noticeable that patients in the NSTI group showed significantly low sodium level (135 mmol dL−1 as lower limit of normal in local laboratory) and high creatinine level (110 µmol L−1 as upper limit of normal in local laboratory), compared with the non-NSTI group. Further analysis (Table 3), however, revealed that the overall accuracy was less than 80% (74.3% and 62.9%, respectively) when either sodium level or creatinine level was used to separate NSTI patients from other soft tissue infections, which was suboptimal as either a screening or diagnostic tool. Presence of bullae or soft tissue crepitation, which are known to be more commonly seen in NSTI patients, was not particularly prevalent (p = 0.381) in NSTI patients in this series though.
Demographics and clinical presentation.a
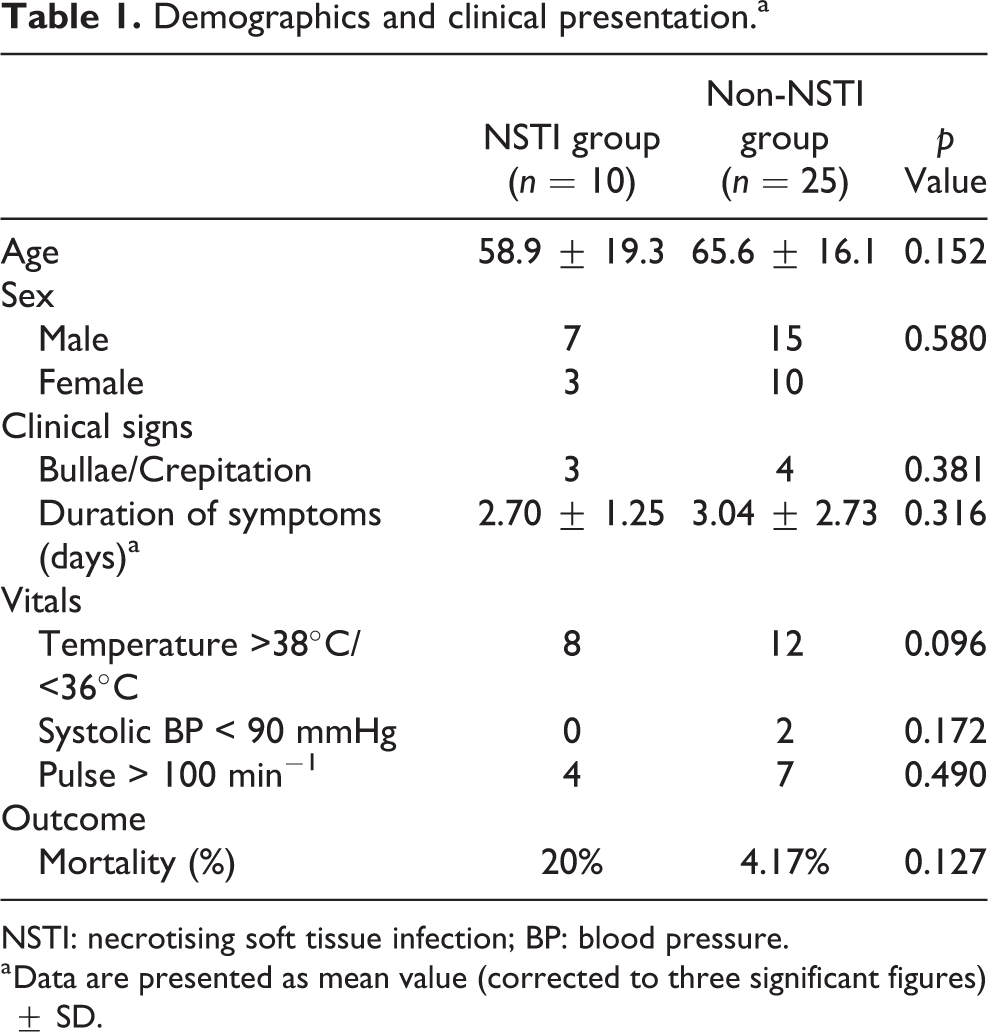
NSTI: necrotising soft tissue infection; BP: blood pressure.
a Data are presented as mean value (corrected to three significant figures) ± SD.
Laboratory results.
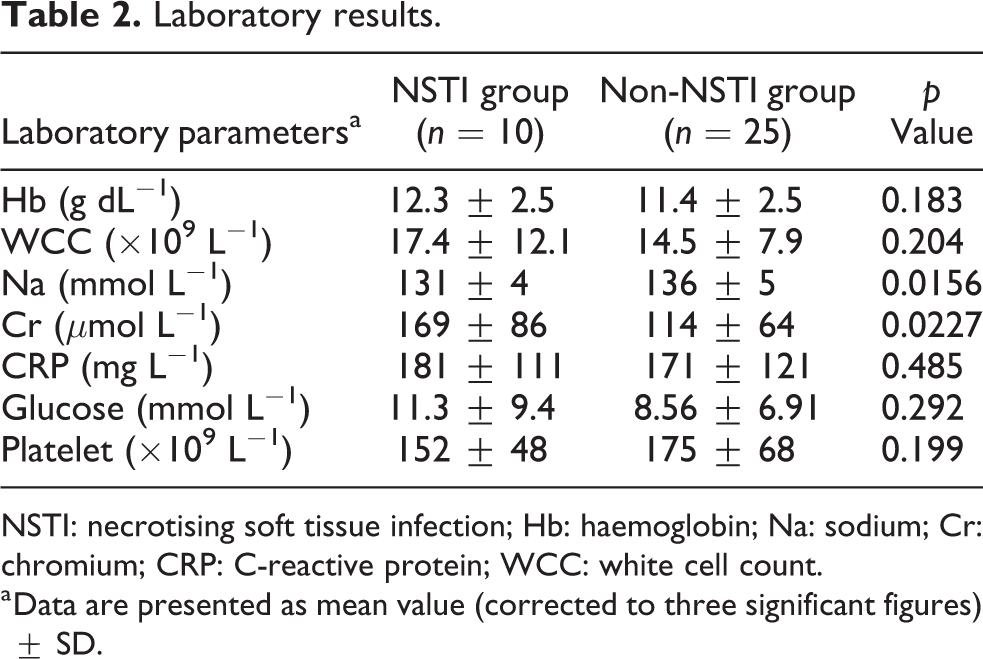
NSTI: necrotising soft tissue infection; Hb: haemoglobin; Na: sodium; Cr: chromium; CRP: C-reactive protein; WCC: white cell count.
a Data are presented as mean value (corrected to three significant figures) ± SD.
Statistical analysis of clinical parameters in evaluation of NSTI.
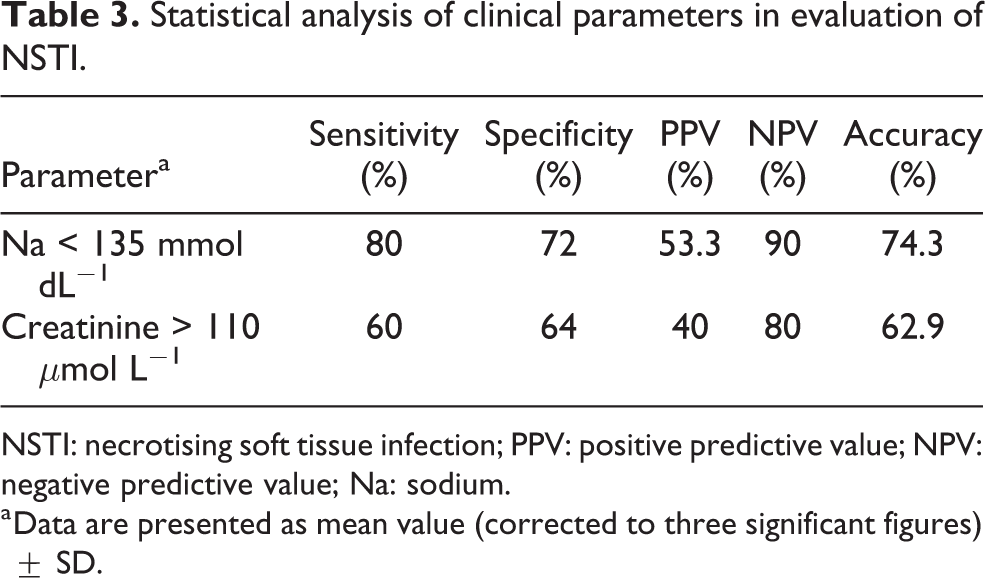
NSTI: necrotising soft tissue infection; PPV: positive predictive value; NPV: negative predictive value; Na: sodium.
a Data are presented as mean value (corrected to three significant figures) ± SD.
The finger test was positive in 15 patients. All proceeded with surgical exploration and 10 were diagnosed with NSTI in operative and histological terms. One patient had operative suspicion of NSTI but histopathology did not reveal any tissue necrosis or microvascular thrombosis. Four patients had alternative diagnoses including severe cellulitis, tenosynovitis or deep ulcers after surgical exploration.
Among test-negative cases, two patients required surgical exploration despite negative findings in the finger test because of clinical deterioration; neither of them showed NSTI operatively or histologically. They were treated successfully with antibiotics after single surgical exploration. The remaining test-negative cases were successfully treated by antibiotics and hence not regarded as having NSTI.
Statistical analysis of the finger test is presented in Table 4. It demonstrated a sensitivity of 100% and a specificity of 80%. PPV was 66.7%, and NPV was 100%. Overall accuracy was 85.7%. LRINEC score was also retrospectively calculated from available laboratory results in all the cases in our series. Among all 35 cases, the LRINEC score was unreliable in two cases due to unavailable admission C-reactive protein (CRP) result. Analysis was therefore carried out in the remaining 33 cases. At a cut-off of ≥6, LRINEC score had a sensitivity of 80%, specificity of 56.5%, PPV of 44.4%, NPV of 86.7% and overall accuracy of 63.6% when applied to the cases in the series. The area under ROC curve for accuracy of LRINEC score was 0.698 (±0.097) (Figure 2).
Statistical analysis of finger test.
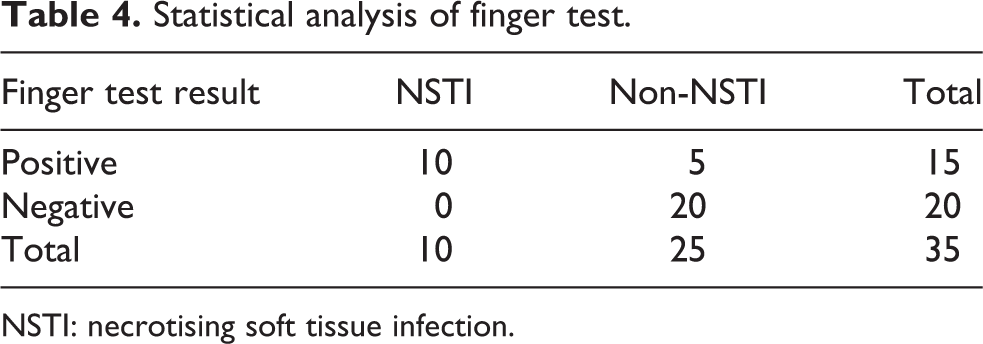
NSTI: necrotising soft tissue infection.
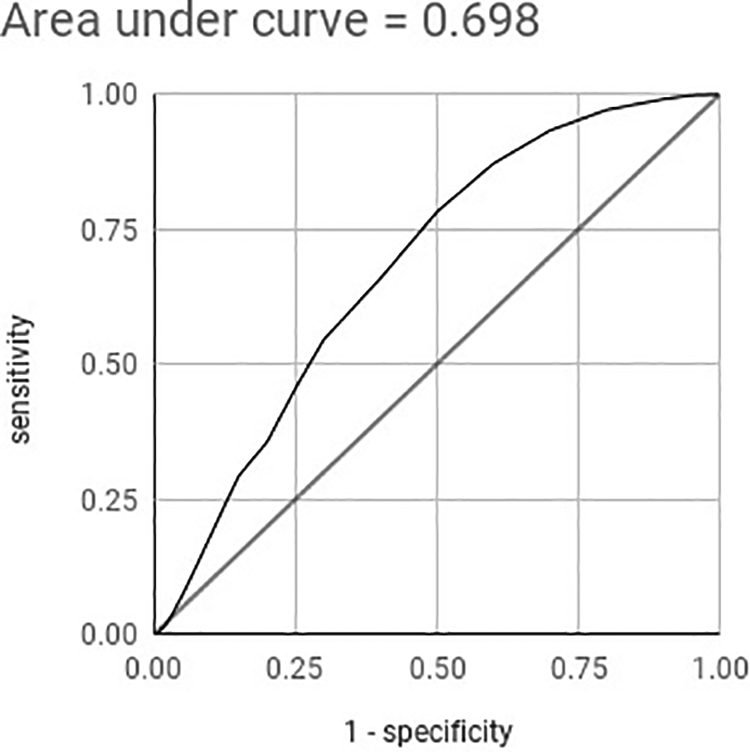
Area under ROC curve for the accuracy of LRINEC score. ROC: receiver operating characteristic; LRINEC: laboratory risk indicator for necrotising fasciitis.
A total of eight surgeons were involved in the series. Seniority ranged from 5-year resident trainee to 15 years after fellowship. Six of them replied to the questionnaire (Table 5). The overall percentage agreement was 76.7%, and moderate reproducibility (kappa = 0.48) was observed on the diagnostic criteria and interpretation of the finger test.
Questionnaire on the understanding of the finger test.

NSTI: necrotising soft tissue infection.
Discussion
NSTI is difficult to be diagnosed in the early stage. Initial presentation could be indistinguishable from cellulitis. 13 However, NSTI is rapidly progressive and becomes life-threatening within the first few hours of presentation. 2,6,7 Kobayashi et al. 8 suggested a delay of surgical treatment more than 12 h would result in higher rate of septic shock, acute renal failure, increased number of surgical debridement required and also the overall mortality rate.
NSTI is known to be a clinical diagnosis in conventional teaching. A triad of disproportionate pain, swelling of the involved limb and high fever should have raised the suspicion of NSTI and urgent treatment is required. Surgical exploration with proceeding debridement is the gold standard for diagnosis and treatment of NSTI. 3,5,11,14 However, a similar clinical picture could also be seen in patients with cellulitis, which could be treated by antibiotics alone. Signs more directing to NSTI, including multiple haemorrhagic bullae, soft tissue crepitations, skin necrosis, septic shock, are considered in the more advanced stage of NSTI in which treatments could already be late. Wong et al. 13 first proposed a clinical staging system of NSTI based on skin changes and physical signs. Stage 1 was the initial stage, presenting with signs of local erythema, swelling and tenderness beyond margin of skin erythema. Stage 2 was the stage of critical skin ischaemia, with clear blister or bullae formation and skin fluctuance. Stage 3 was more advanced. Signs including haemorrhagic bullae, skin necrosis and crepitus, which were more pathognomonic of NSTI, appeared at this stage. But stage 3 also signified the condition of high mortality rate. This clinical staging gives clues for the attending physician on the evolving disease, but treatment could still be delayed in the rapidly progressing NSTI. The finger test, therefore, aimed at picking up patients who are still in stage 1 of the disease, which was hardly different from other non-life-threatening soft tissue infections.
In our series, bullae formation or presence of soft tissue crepitations was only present in 30% among NSTI patients and did not significantly differ from the non-NSTI group. This echoes with the series of Elliott et al. 14 which reported only 20–30% of NSTI patients presented with bullae or crepitations. It revealed that presence of bullae or crepitations are alarming features but not reliable diagnostic criteria for NSTI. We also demonstrated that admission vital signs including body temperature (p = 0.096), blood pressure (p = 0.172) or pulse rate (p = 0.490) are unreliable to separate patients with NSTI from patients with other soft tissue infections.
On the other hand, the finger test is a simple procedure, which can be performed in bedside with preliminary results readily available. Different studies had also proposed the use of the finger test as diagnostic adjunct to NSTI. Stamenkovic and Lew 2 initiated the use of frozen-section soft-tissue biopsy as a rapid and accurate diagnostic tool to pick up NSTI even in the early stage of the disease. However, it was not readily available out-of-office hours in our centre and interpretation of biopsy specimens takes time and experience from the pathologist. Andreasen et al. 10 therefore proposed the use of the finger test, which was a quick and simple procedure for attending surgeons. Goldstein et al. 3 from the United States, Goh et al. 15 from Singapore, Morgan 16 from the United Kingdom and Cheung et al. 5 from Hong Kong all recommended the ‘finger test’ in their reviews, revealing the finger test as a renowned tool for diagnosis of NSTI. Goh et al. even suggested the ‘finger test’ as the best way to diagnose NSTI. Nonetheless, there had not been any study attempted to validate the accuracy and reproducibility of the finger test.
Our findings suggest that a negative finger test could successfully exclude patients from suffering NSTI and a positive test identified the disease with a likelihood of 66.7% after surgical exploration. The relatively low PPV could be explained by the low overall incidence of NSTI. Meanwhile, sensitivity of 100%, specificity of 80% and accuracy of 85.7% suggest that the finger test being reliable enough to establish the need for further surgical exploration and debridement in suspected cases. Among the five false-positive cases, tenosynovitis and infected ulcer are confirmed in two cases which still required operative treatment. One case was suspicious of NSTI with suggestive operative findings, though tissue necrosis or microvascular thrombosis was not found in the surgical specimens eventually. The patient died of multiorgan failure the next day after surgical debridement, which was done within 24 h of admission. The two other false-positive cases were found to have cellulitis on exploration and infection resolved with intravenous antibiotics later. Finger test, therefore, is able to accurately determine surgical candidates and prevents unnecessary surgical exploration and their associated complications.
Various diagnostic modalities had also been investigated worldwide in an attempt to diagnose NSTI in its early stage. Imaging is one of the modalities. Wysoki et al. 17 and Zacharias et al. 18 suggested CT with contrast. Wysoki et al. retrospectively looked into 20 patients with pathologically confirmed NSTI and found asymmetrical fascial thickening and fat stranding in 80% of patients, gas tracking in 55% and abscesses in 35%. Zacharias et al. modified the study and performed contrast CT in suspected cases as well and showed a sensitivity of 100%, specificity of 81%, positive predictive rate of 76% and negative predictive rate of 100% in a total of 67 cases included. Schmid et al. 19 proposed the use of MRI with gadolinium contrast with sensitivity of 100% and specificity of 86% in their series of 17 cases. But there are a few limitations of imaging studies. In many institutions, CT or MRI scans may not be readily available around the clock. The use of iodinated contrast in CT scans could impose extra risk or even irreversible damage on septic patients who might have evolving acute renal failure despite protective medication. Routine MRI screening in all suspected cases could also impose financial problems in small-scale institutions. 9 Further case reports from Arslan et al. 20 pointed out unreliable MRI findings to diagnose NSTI. In this aspect, the finger test is readily available and could be done as a bedside procedure with scalpel and local anaesthetics, which are common apparatuses. It also allows direct visualization of soft tissue which provides the surgeon more accurate information. Suggestive cases could then be immediately sent for surgical exploration by extending the wound from the finger test.
The use of laboratory parameters is another area of diagnostic interest. Wall et al. 21 compared 21 NSTI patients with matched non-NSTI controls in 2000, suggesting admission white cell count > 14 × 109 L−1, serum sodium < 135 mmol L−1 and blood urea nitrogen > 15 mg dL−1 as objective criteria to proceed further surgical intervention for suspected NSTI. Wong et al. 9 further derived LRINEC score, comprising admission haemoglobin level, white cell count, serum creatinine, sodium and glucose level and CRP level, with a cut-off of 6 giving PPV of 92% and NPV of 96% from 89 NSTI cases and 225 control cases. Area under ROC curve was 0.980. However, we are unable to reproduce the same result. When applied to our cases retrospectively, at cut-off ≥6, LRINEC score only gives PPV of 44.4% and NPV of 86.7%. Overall accuracy was 63.6%. Area under ROC curve was 0.698, which was inferior to the original paper. In addition, Wong et al. suggested the finger test as the next step of management after risk stratification by LRINEC score in the original paper. This affirms the applicability and recognisability of the finger test.
Nonetheless, there are still some limitations of the finger test. One main challenge is the interpretation of findings. We had a percentage agreement of 76.7% in our centre from junior trainees to senior fellows, and a moderate inter-observer reproducibility (kappa = 0.48), implying that the interpretation of the finger test is standardised in our centre, but it was limited to a single-centre study. Similar interpretations and study results might not be able to be reproduced in other centres. The interpretation of findings of the test is subjectable to the clinical judgement of the attending surgeon.
Conclusion
To our knowledge, this is the first report analysing accuracy and reproducibility of the finger test since its introduction in 2001. A negative test reliably excluded NSTI and a positive test suggested further surgical exploration. But we should always bear in mind that the finger test is a bedside adjunct to aid diagnosing NSTI and is not mutually exclusive to other diagnostic tools. Clinical judgement is still of paramount importance concerning the use of different diagnostic tools and surgical exploration should not be delayed if clinical suspicion is high.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
