Abstract
Background:
Tranexamic acid (TXA) has been proved effective in reducing perioperative blood loss in patients undergoing orthopedic operations. However, given the heterogeneity in the diagnoses and spinal operations, there are only limited studies comparing patients with uniform diagnoses and procedures performed.
Methods:
A comparative observational control study was performed on 62 patients with cervical myelopathy. They were divided into control (32) and TXA (30) groups according to whether they had received TXA before and during surgery. Cervical laminoplasty from C3 to C6/7 was performed for all patients using a consistent procedure. Preoperative and postoperative hematological data and perioperative blood loss were compared.
Results:
There were no statistically significant differences between the two groups in terms of age, gender, body mass index, and operating time. The TXA group had significantly less intraoperative (310 ± 215 ml vs. 144 ± 90 ml, p < 0.05) and postoperative blood loss than the control group (150 ± 50 ml vs. 249 ± 94 ml, p < 0.01). The drop of hemoglobin (Hb) level in the TXA group was also significantly lower (0.5 ± 0.42 g/dl vs. 2.1 ± 1.03 g/dl, p < 0.01). No thromboembolic complications found.
Conclusion:
TXA significantly reduced perioperative blood loss and reduced drop of postoperative Hb level in cervical laminoplasty.
Introduction
Cervical laminoplasty is a common procedure for patients with cervical spinal stenosis, caused by ossification of the posterior longitudinal ligament or cervical spondylosis. Compared with other types of spinal decompression or fusion procedures, blood loss during laminoplasty might be less in view shorter operation time and less bone cutting. However, any perioperative bleeding would be significant and lead to severe complications, for example, the formation of epidural hematoma, causing spinal cord compression and devastating neurologic damage. Following major blood loss, patients could develop shock and decrease in tissue perfusion, leading to poor wound healing and wound infection. They may also require allogeneic blood transfusion and could suffer from immunologic reactions or viral transmission. Therefore, we should be meticulous in reducing blood loss, both intraoperatively and postoperatively.
Tranexamic acid (TXA) is a synthetic antifibrinolytic drug that blocks the lysine binding sites of plasminogen, preventing interaction of plasmin with fibrin, thereby preventing dissolution of the fibrin clot. 1 –3 The use of TXA might increase ischemic or thromboembolic complications, such as deep vein thrombosis or pulmonary embolism. 1,2
Perioperative use of TXA has been proved effective in reducing perioperative blood loss in patients undergoing total hip and knee replacement, 4,5 as well as lumbar spinal operations. 6 –9 However, these studies involved patients with different diagnoses, and the surgical procedures were not identical between the treatment group and the control group. To prevent selection bias, uniformity of the diagnosis and the operation should be considered. There was only one study on the effect on cervical laminoplasty. Tsutsumimoto et al. 10 focused on the TXA effects in cervical laminoplasty and reported a reduction in postoperative blood loss only in the TXA group. Therefore, we would like to carry out this retrospective comparative observational control study to evaluate the efficacy and safety of TXA in the reduction of perioperative blood loss in cervical laminoplasty.
Methods
Patients
A retrospective comparative control analysis was performed in patients with multilevel compressive cervical myelopathy, which nonoperative therapy has failed, undergoing cervical laminoplasty of vertebrae C3 to C6/C7. Patients with allergy to TXA, history of thromboembolic disease, chronic renal failure (serum creatinine > 132 µmol/l), liver cirrhosis, history of vascular stent insertion, congenital thrombophilia, epilepsy, all emergency cases, and patients receiving antiplatelet (except Aspirin) or anticoagulant therapy at the time of the operation were excluded. Cases with intraoperative usage of absorbable collagen hemostatic agent with thrombin or other flowable topical hemostats were also excluded.
Between 2014 and 2017, 62 cases fitted the inclusion criteria. The 30 patients who received TXA were categorized as the TXA group. The control group consisted of 32 patients who underwent the procedure without receiving TXA infusion.
Drug dosage
All patients in the TXA group received an intravenous bolus dose of 10 mg/kg bodyweight of TXA after induction and before skin incision, followed by a maintenance dose of 1 mg/kg/h, until wound closure.
Surgical technique
All patients underwent “open-door” cervical laminoplasty with the same technique. Patients were put in a prone position under general anesthesia. The standard posterior midline approach was used for exposure. The junction between the laminae and the facets was opened. Underlying ligamentum flavum was removed. Another gutter in the opposite side was made using a high-speed burr. The preserved ventral cortex would act as a hinge. The laminas were then elevated. Titanium mini-plate and screws were used to maintain the expanded position and prevent the closure of the laminar door. The incision was rinsed, and hemostasis was achieved. A suction drain was inserted before skin closure. The wound was closed with layer-to-layer suture, and a dry sterile dressing was applied.
Data evaluation
In both groups, patients demographic data (age at surgery, gender, comorbidities) were analyzed. Duration of operation was defined as the time from the initial incision to the completion of wound closure. Intraoperative blood loss was estimated by the volume of blood collected by suction and weighing surgical gauzes. The postoperative blood loss was measured by the total amount of drainage output collected in the bottle. The bottles were reviewed daily after the operation, and the drain would be removed if the daily output was less than 100 ml. Preoperative and postoperative day 1 hematological data were obtained in both groups, and the hemoglobin (Hb) level was compared.
Statistical analysis
Continuous variable data were presented as mean and standard deviation. Student’s t-test was used to calculate the statistical differences. Categorical variable data were compared using a χ 2 test or Fisher exact test. A value of p < 0.05 was considered statistically significant.
Results
There was no statistical difference in demographic values (age, gender, numbers of overweight, hypertension, diabetes) between the two groups. The number of levels operated on was similar (Table 1).
Demographical data.
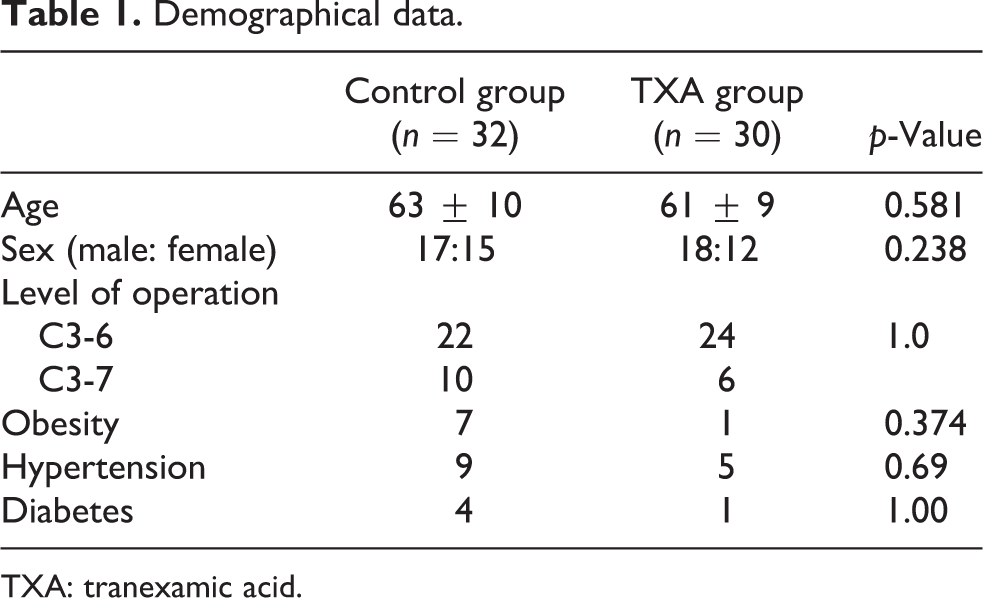
TXA: tranexamic acid.
There was no significant difference in the operation time between the control group (177 ± 39 min) and the TXA group (159 ± 30 min, p = 0.698). The intraoperative blood loss in the TXA group (144 ± 90 ml) was significantly less than that in the control group (310 ± 215, p = 0.04). The TXA group had significantly less postoperative blood loss than the control group (150 ± 50 ml vs. 249 ± 94 ml, p < 0.01). The drop of Hb level in the TXA group was significantly lower than those of the control group (0.5 ± 0.42 g/dl vs. 2.1 ± 1.03 g/dl, p < 0.01) (Table 2).
Results.
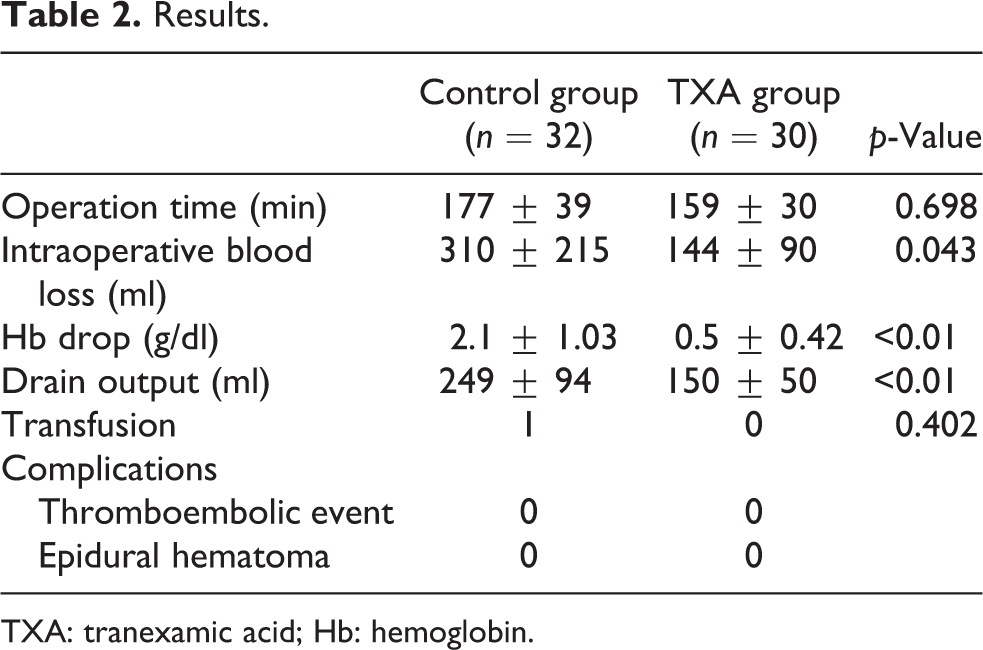
TXA: tranexamic acid; Hb: hemoglobin.
Only one patient in the control group received an allogeneic blood transfusion, with his Hb level dropped from 10.1 to 6.8 postoperatively. There were no significant complications, for example, epidural hematoma formation, deep vein thrombosis, pulmonary embolism, allergic reaction, renal failure, or other cardiopulmonary complications.
Discussion
When patients undergo surgery, there is a temporary increase of fibrinolysis, 2 and it has been suggested to be a contributing factor to blood loss during spinal surgery. 11 TXA acts as a competitive antagonist of the lysine-binding sites of plasminogen, plasmin, and tissue plasminogen activator. The reversible blockade impedes fibrinolysis and blood-clot degradation. 12 Since fibrinolysis is activated during and immediately after the operation, TXA should be given before the operation.
Dose regimens of TXA vary widely in the literature. Pharmacokinetic evidence suggests the use of 10–15 mg/kg loading dose, followed by an infusion dose of 1 mg/kg/h or repeated bolus dosing. 13
Elwatidy et al. 9 used a high single dose of TXA (2 g for adults or 30 mg/kg for children) and found that it reduced total blood loss by 49%. Incidence of blood transfusion was also reduced. Yang et al. 14 conducted a meta-analysis of nine randomized controlled trials and observed a dose-independent beneficial effect of TXA.
Tsutsumimoto et al. 10 reported on the effect of TXA in cervical laminoplasty. They reported that a single dose of TXA (15 mg/kg) before skin incision reduced postoperative blood loss. In their study, there was no significant difference between the TXA group and the control group in terms of intraoperative blood loss.
In this study, we evaluated the effects of TXA with a dose of 10 mg/kg, followed by a maintenance dose of 1 mg/kg/h. The TXA group lost significantly less blood both intraoperatively and postoperatively than that of the control group.
The half-life of TXA is around 80 min in patients with normal renal function. Both single bolus dose and continuous infusion of TXA have shown similar effects in different studies. 9,11,14 However, the optimal dose and treatment duration need to be determined by further studies.
The primary concern surrounding the use of TXA and other antifibrinolytics is the potential for an increased risk of thrombotic events, such as deep vein thrombosis or pulmonary embolism. None of our patients suffered from these complications. Many other studies also reported no increased thromboembolic event with TXA usage in spinal or other orthopedic operations. 2 –6,9,10 A typical misconception is that these drugs are procoagulants and promote the formation of blood clot. The drugs do not alter blood clotting but reduce the dissolution of blood clots instead. The clots have formed will, therefore, remain or enlarge. However, spontaneous formulation of clots should not occur. Benoni et al. 15 suggested that TXA was not associated with thromboembolic events because the effects of TXA are more pronounced in the operative wounds than in the peripheral venous blood. Although topical application of TXA theoretically could reduce systemic thrombotic complications and has been widely used in arthroplasty, its efficacy and safety for spinal operation are unknown.
Although the efficacy of TXA in lumbar spinal operations has been established by numerous studies, 6 –9,16 the patient population was usually heterogeneous, and the patients underwent various types of procedures. All of these determinants could influence perioperative blood loss. The strength of our study was the uniformity in the diagnosis and type of procedure. This uniform cohort is prudent to evaluate the effect of TXA.
The weakness of this study is the small sample size and the retrospective data collection. Checking the Hb level on postoperative day 1 might also fail to reflect the actual Hb drop due to the hemodilution by the intravenous fluid given intraoperatively and postoperatively. Another limitation is that the operations were performed by different surgeons. However, the significance of this may be minimal, given that cervical laminoplasty was a fairly consistent procedure, and all operations were performed at the same institution by the same team.
In summary, intraoperative blood loss, postoperative blood loss, and Hb level drop in the TXA group were significantly lower than that in the control group. There was no significant thromboembolic side effect caused by the TXA. Our regimen of prophylactic TXA may be beneficial for limiting excessive blood loss in cervical laminoplasty and other cervical spinal operations. A prospective randomized controlled study is required for superior evidence.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
