Abstract
Background:
In the field of cartilage repair, cell-based therapy, employing the use of mesenchymal stem cells and chondrocytes maybe a propitious treatment option. Once transferred, success of transplanted cells is determined by their immunogenicity making assessment imperative. Another contender in the field, may be articular chondroprogenitors (CPs) shown to possess chondrogenic potential and reduced expression of hypertrophy markers. Our aim was to assess immunogenic properties of CPs (non-diseased and osteoarthritic (OA)) and compare them with chondrocytes since it may be a good alternative for cell-based therapy and data regarding its immunogenic profile is limited.
Methods:
Human chondrocytes and CPs from the same cartilage source were isolated. Passage 0 cells characterized by fluorescence-activated cell sorting against human surface antigen were human leucocyte antigen class I (HLA-A2 and HLA-B7), HLA-DR and its costimulatory molecules (CD80 and CD86) and macrophage/monocyte marker (CD14).
Results and conclusion:
Our observations indicated that CPs isolated from human non-diseased and OA cartilage showed similar immunogenic properties to chondrocytes isolated from the same source with no significant difference in expression. High-to-moderate expression of MHC class I (HLA-A2 and HLA-B7) and moderate-to-low expression of MHC class II (HLA-DR and its co-stimulatory molecules CD80 and CD86) were observed. This is the first report to shed insight on the immunogenic properties of human cartilage-derived progenitors, a potential contender in the field of regenerative therapy for formation of genuine hyaline cartilage.
Introduction
Current techniques for treatment of cartilage diseases include cell-based therapies, which employ the use of cells like mesenchymal stem cells (MSCs) and chondrocytes. 1 Studies using these cell types demonstrate the formation of fibrocartilage or a combination of fibrohyaline cartilage in place of pure hyaline cartilage when transplanted, thus necessitating further investigation of a cell type with higher chondrogenic potential. Articular chondroprogenitors (CPs) derived using fibronectin adhesion assay have been classified as MSCs conforming to the minimal criteria of International Society of Cellular Therapy. 2,3 In addition to being native to the cartilage, CPs have been shown to possess inherent chondrogenic potential and reduced hypertrophy. 4 –6 However, reports pertaining to characterization of its biological profile are under par and ongoing studies are rapidly adding important data to the phenotypic repository of CPs.
Cellular immunogenicity plays an important role in determining the success of the transplant; therefore, assessment of immunogenic behavior is imperative. Literature suggests that MSCs are weakly immunogenic as they express low levels of major histocompatibility complex class I (MHC I) and costimulatory molecules, whereas chondrocytes have been reported to express high levels of MHC class I and low levels of costimulatory molecules. 7 –10 Creation of cellular immunogenic profile is crucial as allogeneic immune response posttransplant is dependent on the level of MHC class I/II/costimulatory molecule expression.
The aim of our study was to assess immunogenic properties of CPs and compare them with chondrocytes, since this cell type maybe a good alternative for cell-based therapy and data regarding its immunogenic profile are limited. We also compared cells derived from non-diseased (N) and osteoarthritic (OA) knee joints as we wanted to assess the effect of tissue inflammation and degeneration on immunogenicity of the cell types under question.
Methods
The study was approved by the Institutional Review Board and performed in accordance to the guidelines laid down by the Ethics Committee (study clearance no: IRB min no: 10183). The samples were collected after obtaining written informed consent. Human articular cartilage was obtained from three N and three OA joints. Mean age of donors was 22 ± 4 years (N) and 63 ± 7 years (OA), respectively. OA joints were acquired from patients requiring knee replacement surgery, diagnosed with primary degenerative osteoarthritis based on history, blood parameters, and radiological grading (Kellgren–Lawrence grade 4). Patients with post-traumatic, systemic, infective, inflammatory arthritis, and conditions secondary to metabolic diseases were excluded. Post-trauma patients requiring above knee amputation as a part of their treatment provided N joint. Patients presenting with inflammation, infection (e.g. osteomyelitis), and tumors of the knee were excluded. Cells were isolated from nonweight-bearing areas with preserved full-depth cartilage and cultured to passage 0 (p0).
Isolation and culture of chondrocytes
Shavings were harvested and subjected to overnight enzymatic digestion using Dulbecco’s modified Eagle’s medium (DMEM-F12-Himedia, India) containing 0.15% collagenase type II (Worthington, Washington, US) under standard culture conditions.
11
Isolated cells were seeded at a concentration of 10,000 cells/cm2 and expanded to p0 with DMEM/F12 containing 10% fetal bovine serum (FBS, Gibco, Waltham, US), ascorbic acid 62 μg/mL,
Isolation and culture of CPs
Cartilage shavings were washed and subjected to overnight sequential enzymatic digestion with pronase (Roche, Switzerland) and collagenase type II to obtain chondrocytes. These were subjected to differential adhesion on fibronectin (10 µg/mL containing 1 mM calcium chloride and 1 mM magnesium chloride) precoated plates for 20 min at a concentration of 2000 cells/9.6 cm2. Postincubation, excess media, and nonadherent cells were removed and replaced with standard growth media (DMEM-F12-Glutamax plus 10% FBS, ascorbic acid 62 μg/mL, penicillin–streptomycin 100 IU/mL, and amphotericin-B 2 μg/mL). Adherent cells were maintained at standard culture conditions for 10 days to obtain colonies of >32 cells, known as clones. Isolation of clones and replating at a ratio of 1 clone per 5 cm2 was performed. Further expansion of enriched polyclonal CPs to p0 was done as per protocol described by Nelson et al. 12 For expansion, the above mentioned basal medium containing transforming growth factor β2 1 ng/mL (human-recombinant, BioVision, San Francisco, US) and fibroblastic growth factor 2 5 ng/mL (human-recombinant, BioVision) was used to refresh the medium once every 3 days and cells were harvested at subconfluence (Figure 1).
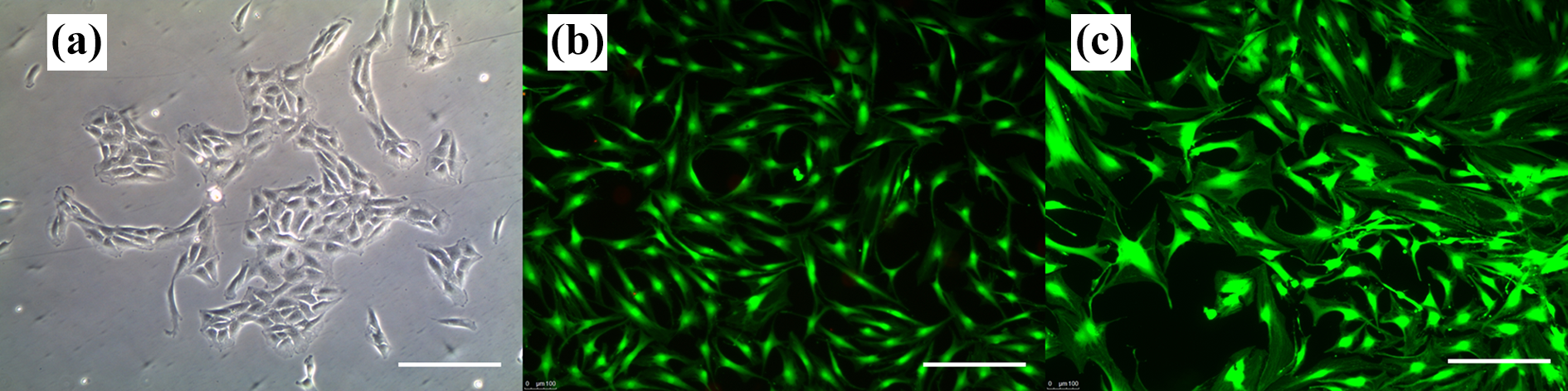
Representative clonally derived human articular chondroprogenitors (fibronectin differential adhesion). (a) A clone at day 12 forming a compact cluster (×10). Live dead assay using calcein AM/ethidium homodimer of (b) CPs and (c) chondrocytes at subconfluence (×10). Scale bar denotes 100 μm. CP: chondroprogenitor.
Phenotyping: Fluorescence-activated cell sorting and statistical analysis
Cell surface marker expression of chondrocytes and CPs from p0 was characterized by fluorescence-activated cell sorting (FACS). The studied antibodies against human surface antigen were human leucocyte antigen class I (HLA-A2-PE), HLA class I: NK modulator (HLA-B7-PE), HLA-DR: T-cell receptor (HLA-DR V500), B7-1 costimulatory molecule (CD80-PE), B7-2 costimulatory molecule (CD 86-PE), and macrophage/monocyte marker (CD14-FITC). The staining method followed instructions provided in the manual received with individual antibodies and data was acquired using BD FACS Celesta flow cytometers. Gating was applied using BD FACS Diva v 5.0.2 software; unstained and isotype controls (for nonspecific staining) were run for the specific CD markers. Results of FACS were reported as mean ± standard error. Statistical analysis was done using SPSS 17.0 software. Mann–Whitney test was used to compare surface marker expression between different cell groups (chondrocytes vs. CP) and cell-source (N vs. OA) at p0. A P value of <0.05 was considered as significant.
Results
Both chondrocytes and CPs derived from N and OA cartilage demonstrated high expression of HLA-A2 without any significant difference in the levels between them. When expression of HLA-B7 and its costimulatory molecules (CD80 and CD86) was compared, both cell types showed moderate-to-low expression without any significant difference between them. No significant difference was observed between the two cell populations when HLA-DR and CD14 expression was compared with low levels seen in both groups (Figures 2 and 3).
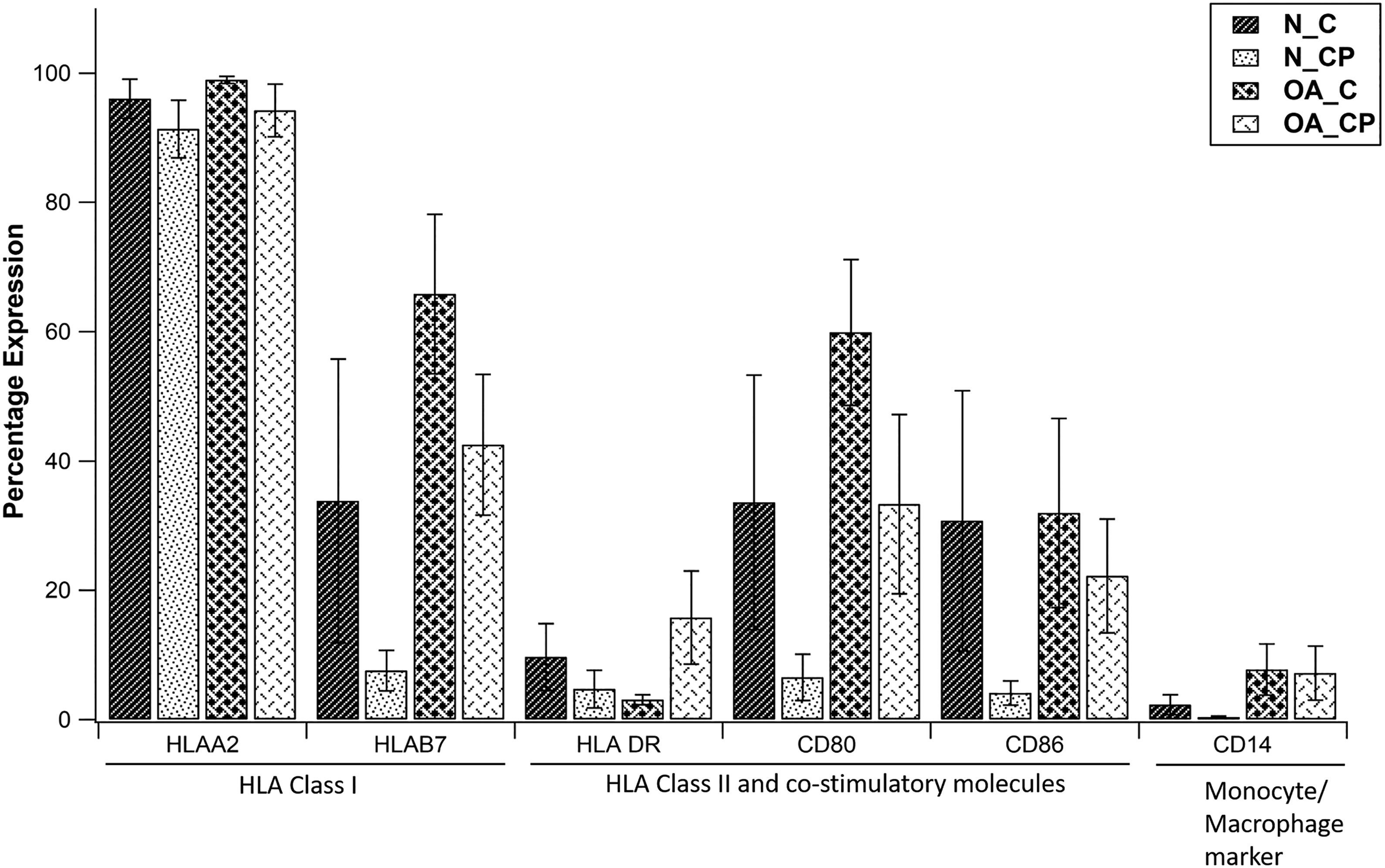
Comparison of percentage expression of HLA-A2, HLAB7, HLA-DR, CD80, CD86, and CD14 across different cell groups at passage 0.
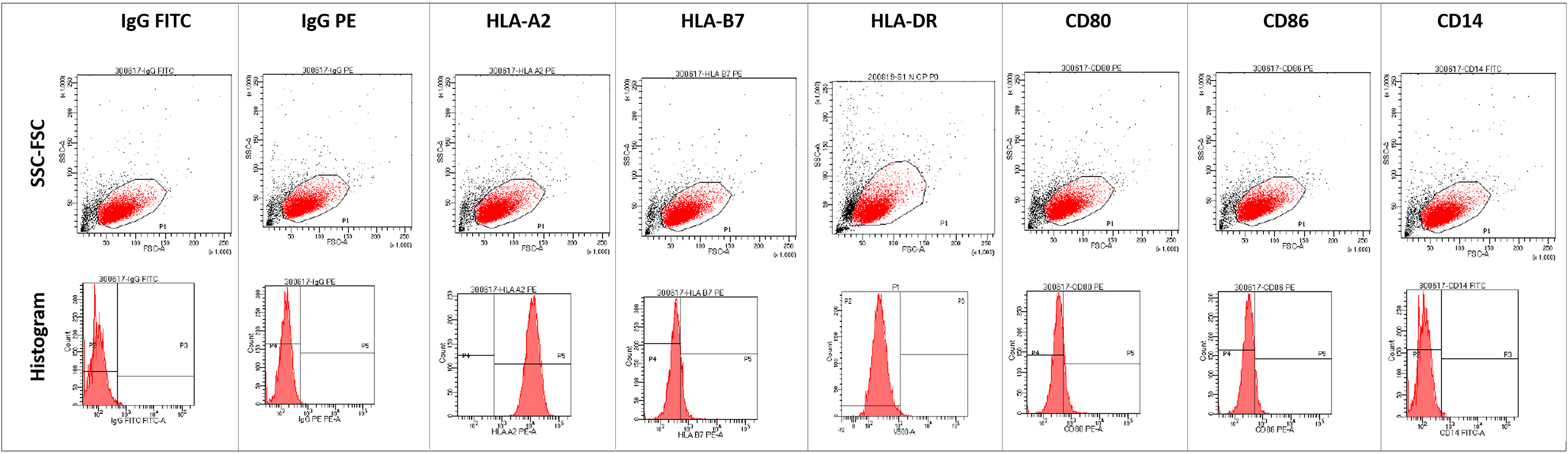
Representative FACS graphs for the corresponding antibodies. Upper panel: Forward scatter-size scatter plot, lower panel: histogram. Data are expressed as mean ± standard error of the mean (Mann–Whitney
Discussion and conclusion
This is the first in-vitro study to shed insight on the immunogenic properties of human cartilage-derived progenitors, a potential contender in the field of regenerative therapy for formation of genuine hyaline cartilage. It has been reported that chondrocytes constitutively display high levels of MHC class I molecules (HLA-A, HLA-B, and HLA-C). 8 From our observations, CPs express high levels of class I molecules which is not comparable to the reported low levels expressed by MSCs. 9 In the present study, since it was difficult to age match patients due to paucity of nondiseased joints, we were unable to assess the effect of age on immunogenic expression, although our results did not reflect any difference when the cell groups were compared. Moreover, alteration in the immune response could also have occurred due to trauma sustained in patients from whom the non-diseased joints were harvested. This possible variation in response could not be assessed and values obtained from cells isolated from these joints were considered as baseline. Although it should be marked that the level of expression observed in the cell groups reflects the in-vitro behavior of the cellular phenotype and not its inherent ability to elicit an immune response when transplanted, thereby warranting a detailed analysis prior to implantation.
Footnotes
Acknowledgements
The authors would like to acknowledge the Centre for Stem Cell Research (A unit of InStem, Bengaluru), Christian Medical College, Vellore, for infrastructural support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by AO Trauma Asia Pacific (AOTAP 16-19) of the AO foundation.
