Abstract
Study Design
Systematic Review.
Objectives
To evaluate the impact of proton pump inhibitors (PPIs) and histamine-2 receptor antagonists (H2 blockers) on spinal fusion outcomes.
Methods
A systematic search of PubMed (MEDLINE) was conducted for studies published between January 2000 and July 2025 using Title/Abstract terms for PPIs, H2 blockers, spine, and fusion. Eligible studies were English-language articles assessing the relationship between acid suppression and spinal fusion outcomes. Two reviewers independently screened studies and extracted data.
Results
Of 798 records screened, five studies met inclusion criteria: three retrospective cohort studies, one animal study, and one prospective study evaluating H2 blocker use. Four of the five studies evaluated PPIs. All three clinical studies observed higher pseudarthrosis rates among postoperative PPI users undergoing spinal fusion. The animal model found no significant fusion impairment. The H2 blocker study showed reduced inflammatory markers but did not assess osseous union.
Conclusions
Current evidence suggests an association between PPI use and impaired spinal fusion, particularly in the cervical and lumbar spine. In contrast, H2 blockers appear to have a more neutral skeletal profile, though no studies have directly evaluated their impact on spinal fusion or pseudarthrosis rates. Most available data on PPIs are derived from retrospective studies or animal models, with substantial heterogeneity in surgical level, fusion construct, PPI type, and follow-up duration. Future research should prioritize prospective, randomized studies or large registry-based analyses to clarify the causal relationship between acid suppression therapy and spinal fusion.
Introduction
Spinal fusion is a widely performed surgical procedure to address degenerative disc disease, instability, deformity, and trauma. While advances in surgical techniques and instrumentation have improved outcomes, pseudarthrosis, defined as failure to achieve solid bony union, remains a significant complication. Reported rates of pseudarthrosis range from 5% to 24% depending on spinal level, patient risk factors, and fusion construct. 1 Pseudarthrosis can lead to persistent pain, neurological symptoms, and the need for revision surgery, representing both clinical and economic burdens.2,3
Acid-suppressive medications, particularly proton pump inhibitors (PPIs) and histamine-2 receptor antagonists, also known as H2 blockers, are frequently prescribed in the perioperative setting. They are used for gastrointestinal protection in patients receiving NSAIDs, anticoagulants, or corticosteroids. However, growing evidence suggests that PPIs may have unintended effects on bone metabolism and healing. 4 Chronic PPI use has been associated with increased risk of osteoporotic fractures in large population-based studies.3,4 Mechanistically, this may result from impaired calcium absorption, decreased bone mineral density, and altered osteoblast activity.4-9 In a prospective cohort of midlife women, PPI initiation was linked to accelerated hip bone mineral density (BMD) loss, whereas H2 blocker use was not. 8 More recent data suggest that PPIs may also disrupt calcium and phosphate homeostasis and promote secondary hyperparathyroidism, further contributing to bone fragility. 9 PPIs have been linked to impaired healing in animal fracture models and delayed union in orthopedic trauma patients.10-12 In contrast, H2 blockers appear to have a more neutral skeletal profile, though their effect on fusion biology remains poorly understood.8,9
Given the widespread use of PPIs and H2 blockers in spine surgery patients, and the high clinical importance of achieving spinal fusion, understanding whether these medications influence spinal fusion outcomes is essential. Although several studies have examined this question in isolation, to date no systematic review has comprehensively evaluated the impact of PPI or H2 blocker use on spinal fusion healing. The objective of this review is to systematically identify and synthesize PubMed-indexed clinical and preclinical studies that directly evaluate the effects of PPIs and H2 blockers on spinal fusion outcomes.
Methods
Literature Search Strategy
A systematic literature search was conducted using the PubMed (MEDLINE) database to identify studies published from January 1, 2000, through July 2025. The search strategy included combinations of the following terms, restricted to the Title/Abstract fields: “proton pump inhibitor,” “proton pump inhibitors,” “PPI,” “H2 blocker,” “H2 blockers,” “H2 receptor antagonist,” “H2 receptor antagonists,” “spine,” “spinal,” “spine surgery,” “spinal surgery,” “cervical,” “lumbar,” and “fusion.” Only English-language studies indexed in PubMed were eligible. Both original studies and review articles were considered if they provided relevant data on the relationship between acid suppression therapy and spinal fusion outcomes. All inclusion criteria and search parameters were prospectively defined and followed without deviation.
Study Selection
All records identified through the search were imported into Covidence, (Veritas Health Innovation, Melbourne, Australia) a systematic review management platform, for screening. Two independent reviewers screened titles and abstracts to identify studies addressing spinal surgery in the context of acid suppression therapy (ie, PPIs or H2 blockers). Studies were excluded if they did not investigate the relationship between acid suppression and spinal fusion. Full texts of potentially eligible studies were reviewed independently by the same two reviewers. Discrepancies were resolved by consensus.
Data Extraction and Synthesis
Data from each included study were extracted into a standardized spreadsheet. Extracted variables included study design, population characteristics, surgical procedure, acid suppression therapy type, and spinal fusion–related outcomes. Given the heterogeneity in study design and outcome reporting, a meta-analysis was not performed. Instead, findings were synthesized qualitatively in accordance with systematic review methodology.
Risk of Bias Assessment
The risk of bias for each included study was independently assessed using the Cochrane Risk of Bias 2 (RoB 2) tool.
13
Five domains were evaluated: (1) bias arising from the randomization process, (2) bias due to deviations from intended interventions, (3) bias due to missing outcome data, (4) bias in measurement of the outcome, and (5) bias in selection of the reported result. Each domain was judged as low risk, some concerns, or high risk of bias, with an overall risk of bias judgment generated accordingly. Assessments were performed in duplicate, with disagreements resolved by discussion. Results of the risk of bias assessment are presented in Figure 1. Risk-of-Bias (RoB 2) Assessment Across the Five RoB 2 Domains for Included Studies
Results
Study Characteristics
Summary of Studies Evaluating Proton Pump Inhibitors (PPIs) and Histamine-2 Receptor Antagonists (H2 Blockers) in Spinal Fusion
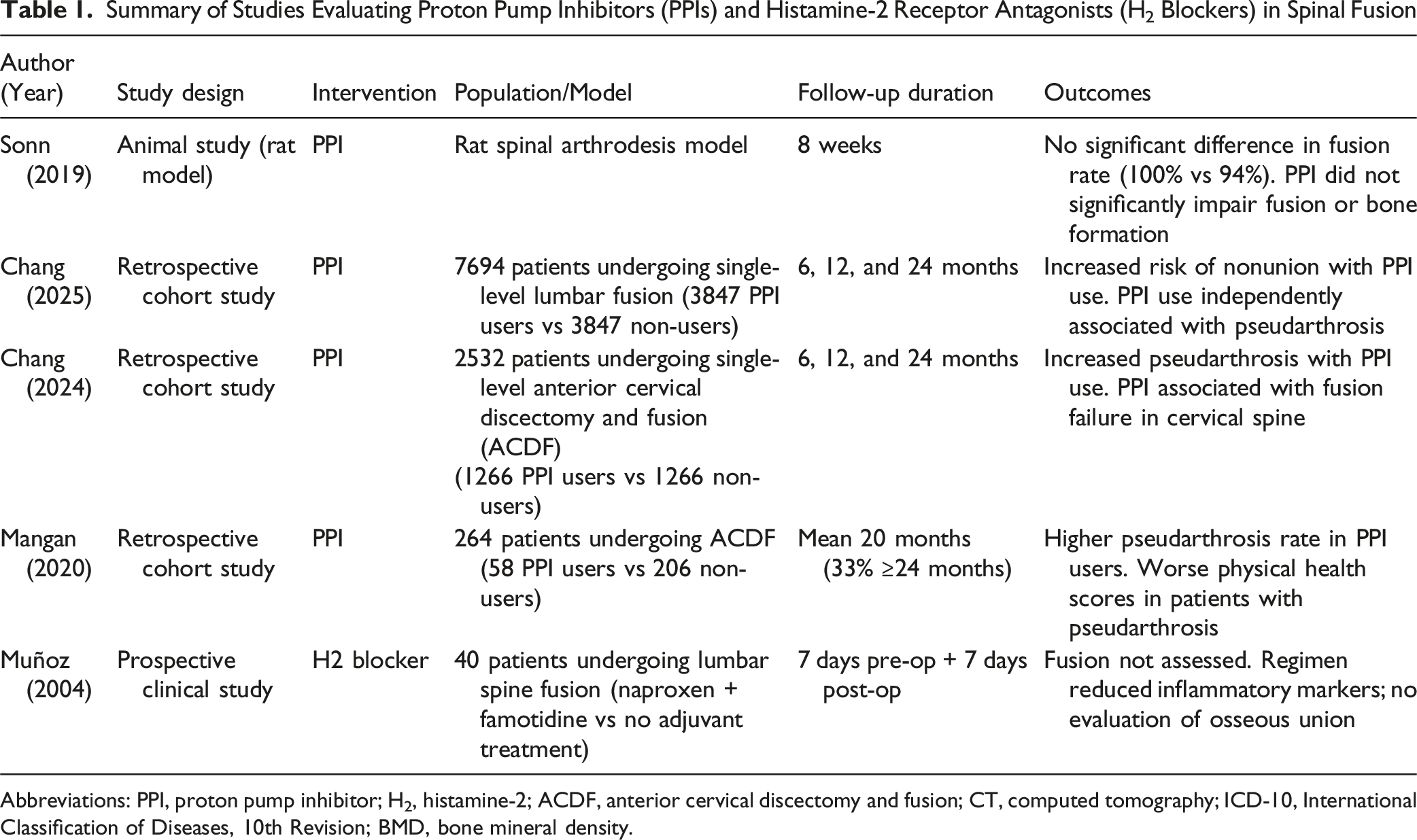
Abbreviations: PPI, proton pump inhibitor; H2, histamine-2; ACDF, anterior cervical discectomy and fusion; CT, computed tomography; ICD-10, International Classification of Diseases, 10th Revision; BMD, bone mineral density.
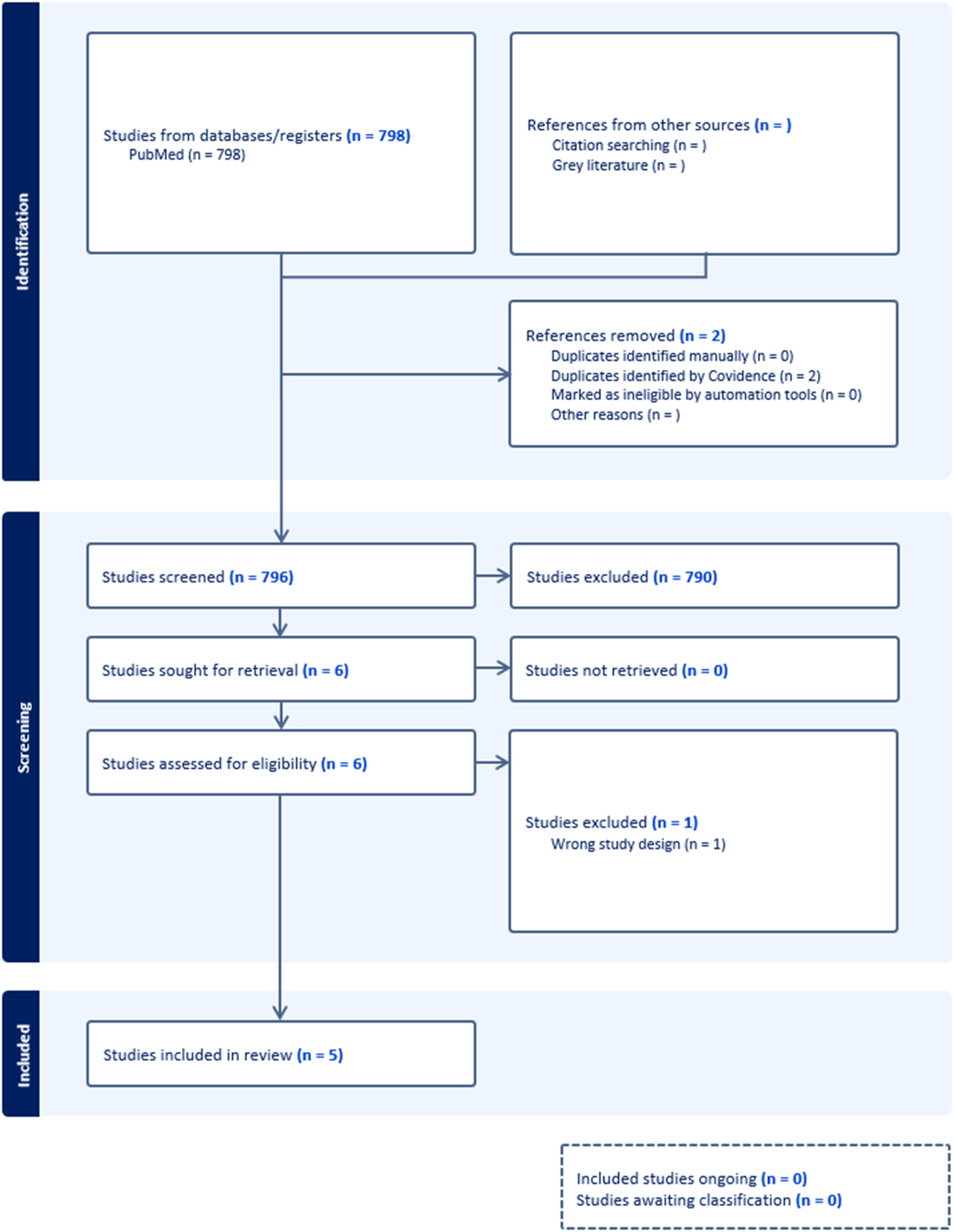
PRISMA Flow Diagram Summarizing the Study Selection Process
The PPI studies consisted of one animal study using a rat spinal arthrodesis model and three retrospective clinical cohort studies. The cohort studies examined outcomes following single-level lumbar or cervical spinal fusion, with matched comparisons between PPI users and non-users. Sample sizes for these clinical studies ranged from 264 to 7694 patients. The animal study evaluated the effects of PPI exposure on fusion and bone formation.
The single H2 blocker study was a prospective clinical trial involving 40 patients undergoing lumbar spine surgery. Patients received perioperative naproxen combined with famotidine or no adjuvant treatment.
Quality Assessment
Risk of bias assessment is summarized in Figure 1. All five studies received an overall judgment of high risk of bias according to the RoB 2 tool. However, several individual domains were rated as low risk or raised only some concerns, particularly for missing outcome data and selective reporting. The overall ratings primarily reflect the nonrandomized study designs, limited adjustment for confounding, and methodological constraints inherent to retrospective analyses.
Proton Pump Inhibitors
Initial animal studies yielded mixed results. Sonn et al. 14 employed a rat spinal arthrodesis model and reported no significant differences in fusion rates, bone formation, or biomechanical integrity between the control and PPI-treated groups (100% vs 94%). Three clinical cohort studies examined spinal fusion outcomes in patients receiving PPIs. Chang et al. 15 conducted a retrospective cohort study evaluating lumbar fusion outcomes and, after propensity score matching to adjust for age, sex, race, and comorbidities, found that postoperative PPI use was independently associated with increased risk of pseudarthrosis following single-level lumbar fusion. A subsequent propensity-matched study 16 by the same research group found similar associations between PPI use and pseudarthrosis following anterior cervical discectomy and fusion (ACDF). These findings were supported by Mangan et al., 17 who also identified PPI use as a significant predictor of clinically diagnosed pseudarthrosis following ACDF. In their cohort, patients who developed pseudarthrosis experienced less improvement in physical health–related quality of life scores (PCS-12) compared to those who achieved successful fusion.
H2 Receptor Antagonists
Evidence regarding H2 blockers in the context of spinal fusion is limited. The only study meeting inclusion criteria was conducted by Muñoz et al., 18 who examined the perioperative use of naproxen combined with famotidine in lumbar spine surgery. The regimen was well tolerated and associated with reductions in inflammatory markers both clinically and at the laboratory level. However, because fusion outcomes were not assessed, the effect of H2 blockers on spinal arthrodesis could not be determined.
Discussion
This systematic review highlights emerging evidence that PPIs, while commonly used for gastrointestinal protection in the perioperative period, may adversely affect spinal fusion outcomes. Although prior studies focused on fracture risk and disrupted skeletal metabolism, more recent investigations raise concern that these effects may also impair spinal fusion. However, the current body of evidence remains limited in both scope and quality, and a definitive causal relationship between PPI use and impaired fusion cannot yet be established.
The biological plausibility of PPI-related impairment in spinal fusion is supported by both experimental and clinical observations. By inhibiting gastric acid secretion, PPIs can reduce calcium absorption, impair osteoblast function, and lower bone mineral density, all essential for successful bone healing.4,7-9 These biologic effects are consistent with population-level data showing higher fracture risk among chronic PPI users. A meta-analysis by Liu et al 19 further supports these concerns. This study demonstrated that PPI use is associated with significantly increased risks of any-site fractures (HR: 1.30; 95% CI: 1.16-1.45), hip fractures (HR: 1.22; 95% CI: 1.15-1.31), spine fractures (HR: 1.49; 95% CI: 1.31-1.68), and osteoporosis (HR: 1.23; 95% CI: 1.06-1.42). Notably, these associations were observed even in the absence of consistent evidence for bone mineral density loss, suggesting that PPIs may impair skeletal integrity through additional mechanisms. Animal studies also lend support to these mechanisms, with pantoprazole delaying fracture healing in mice, and omeprazole compromising implant osseointegration in rat tibiae.10-12 While one spinal arthrodesis model showed no difference in fusion rates between treated and control animals (P = 0.47), 14 this may reflect limited follow-up, underpowered design, or variability in drug exposure.
Clinically, several retrospective cohort studies have reported an association between postoperative PPI use and elevated pseudarthrosis rates in both cervical and lumbar fusion patients.15-17 Both Chang et al studies, however, defined pseudarthrosis using ICD-10 coding (M96.0) rather than postoperative CT or dynamic imaging. This limitation may have underestimated the true incidence by excluding asymptomatic cases. Nevertheless, reduced improvement in physical health scores among patients with pseudarthrosis underscores the possible patient-centered consequences of routine perioperative PPI use. 17 Together, these findings highlight the need for cautious prescribing and further prospective investigation.
In contrast to emerging studies on the potential impact of PPIs on spinal fusion, the effects of H2 blockers on spinal fusion remain poorly defined. To date, no study has directly assessed the impact of H2 blocker use on spinal fusion or pseudarthrosis rates. While some observational studies have demonstrated no association between H2 blocker use and bone mineral density loss, 8 an umbrella review by Meng et al 20 evaluating 46 meta-analyses of H2 blocker–associated outcomes reported a modestly increased risk of hip fracture. However, no significant associations were found for spine, vertebral, or osteoporotic fractures, and the certainty of evidence was rated low to very low across most outcomes. Although the study by Muñoz et al 18 provides some reassurance regarding the perioperative safety of famotidine, it did not evaluate osseous union. Accordingly, any conclusions regarding the skeletal safety of H2 blockers in the context of spinal fusion remain speculative and should be interpreted with caution.
Importantly, the quality of available evidence is limited. Most clinical studies15-17 are retrospective and therefore susceptible to confounding by indication. Advanced age, smoking, diabetes, osteoporosis, and chronic corticosteroid or NSAID use are well-established risk factors for pseudarthrosis.15,16,21,22 These comorbidities were more common among PPI users in the unmatched cohorts of Chang et al15,16; although propensity matching reduced imbalances, residual bias remains possible. Consequently, PPI use may function as a surrogate marker of higher-risk patients rather than a direct causal factor in impaired fusion. In addition, heterogeneity in PPI dose, duration, surgical level, fusion construct, and follow-up further limits comparability. The umbrella review by Alanazi et al 4 underscores these constraints and highlights the need for rigorous prospective studies.
The consistent association between PPI use and impaired bone healing across both preclinical and clinical domains raises important questions about their routine use in patients undergoing spinal fusion. While evidence remains limited and largely retrospective, the biological plausibility and emerging clinical signals suggest that caution may be warranted. At the same time, PPIs remain essential for gastrointestinal prophylaxis in high-risk patients such as those receiving chronic NSAIDs or anticoagulants. Broad avoidance is therefore not appropriate; rather, acid suppression should be tailored to individual risk profiles, balancing skeletal risks with gastrointestinal benefits until higher-quality prospective data are available. Collectively, these findings define key priorities for future research, establishing an agenda for prospective and randomized studies to better characterize the relationship between acid suppression therapy and spinal fusion outcomes. Future investigations should also aim to standardize definitions of pseudarthrosis, report PPI dose and duration, and distinguish perioperative from chronic use.
Conclusion
This systematic review is limited by the quality and design of the available studies. All included studies were judged to be at high risk of bias, as most clinical evidence derives from retrospective cohorts, which are inherently susceptible to confounding by indication, incomplete adjustment for comorbidities, and variations in concurrent medication use. No randomized controlled trials were identified. In addition, substantial heterogeneity was present across studies. Some examined cervical fusions while others investigated lumbar procedures, and follow-up periods ranged from several months to more than two years. Definitions of PPI exposure also varied, and no study provided details on the specific drug, dose, or treatment regimen, limiting interpretation of potential dose–response or drug-specific effects. Details on surgical technique, graft selection, and instrumentation were generally not reported, limiting assessment of these potential confounders. These limitations underscore the need for prospective, high-quality studies to clarify the relationship between acid suppression therapy and spinal fusion.
Current evidence suggests an association between PPI use and impaired spinal fusion, particularly in the cervical and lumbar spine. In contrast, H2 blockers appear to have a more neutral skeletal profile, though no studies have directly evaluated their impact on spinal fusion or pseudarthrosis rates. Most available data on PPIs are derived from retrospective studies or animal models, with substantial heterogeneity in surgical level, fusion construct, PPI type, and follow-up duration. Future research should prioritize prospective, randomized studies, or at minimum large registry-based analyses with imaging-confirmed fusion outcomes, to clarify the causal relationship between acid suppression therapy and spinal fusion.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
