Abstract
Study Design
Retrospective single-center review.
Objectives
Assess whether expansile duraplasty was associated with greater motor recovery in patients with acute traumatic spinal cord injury (tSCI) compared with bony decompression alone.
Methods
Retrospective chart review was conducted for patients who underwent surgical stabilization and decompression for tSCI at a level-1 trauma center. Changes in motor scores were calculated and compared between patients who had expansile duraplasty and those who did not.
Results
We studied 96 patients: 11 who underwent expansile duraplasty and 85 who did not. The average pre-operative motor scores for patients with duraplasty was 33.3 ± 22.5 compared to 49.0 ± 29.3 for non-duraplasty (P = 0.11). At hospital discharge the duraplasty group had an average motor score of 43.1 ± 26.3 with an average score of 52.9 ± 29.4 for the non-duraplasty group (P = 0.41). The average motor score at the time of inpatient rehabilitation discharge was 55.3 ± 28.6 for duraplasty and 60.1 ± 30.3 for non-duraplasty (P = 0.37). The change in motor score between baseline and hospital discharge was 9.8 ± 11.8 for duraplasty and 3.9 ± 11.4 for controls (P = 0.088). There was a higher change in motor score from baseline to inpatient rehabilitation discharge in the duraplasty group (20.3 ± 11.6 vs 11.1 ± 13.5 for controls, P = 0.034) even after adjusting for covariates through inverse probability weighting (P = 0.017). The duraplasty group did not have significantly more complications.
Conclusions
Patients who underwent expansile duraplasty at the time of surgical stabilization and decompression for acute tSCI had greater motor improvement between preoperative evaluation and rehabilitation discharge than patients without duraplasty. This technique warrants further study in a multi-center, prospective study.
Introduction
In 2019, there were an estimated 0.9 million new spinal cord injuries (SCI) and a prevalence of nearly 21 million people with SCI worldwide. 1 These injuries can lead to debilitating weakness with long term impacts on quality of life. Following the initial trauma, secondary injury mechanisms including edema, ischemia, and inflammation can lead to considerable extension of the injury, providing a window of opportunity for interventions to preserve function.2-4 Multiple pharmacologic agents have been tested in randomized clinical trials for SCI but none have clearly proven to be advantageous.5-8
In the absence of suitable pharmacotherapy, early surgical decompression and optimization of spinal cord perfusion have emerged as potential targets for intervention. Both animal and human studies have suggested that early decompression of the spinal cord can lead to improved outcomes.9-11 In traumatic brain injury, dural opening is standard of care in decompressive surgeries and is thought to contribute significantly to decompression of the swollen brain following craniectomy.12-14 Data also suggests that the dura may continue to compress the injured spinal cord even following laminectomy.15,16 Expansile duraplasty could provide benefit by extending surgical decompression to the intrathecal space, thereby lowering the intraspinal pressure and increasing spinal cord perfusion.17,18 However, there remains a paucity of data on whether this provides a measurable benefit in terms of patient outcome. We aimed to assess whether expansile duraplasty was associated with greater motor recovery in patients with acute traumatic SCI compared with bony decompression alone.
Material and Methods
The University of Washington Institutional Review Board approved this retrospective study and waived the need for written informed consent. We identified patients who underwent surgical spinal decompression and stabilization for non-penetrating acute traumatic cervical or thoracic spinal cord injury from February 2017 to June 2021 at our academic, level 1 trauma center from a local trauma database. This included patients treated by both orthopedic and neurological surgery attending spine surgeons. Patients were excluded from our analysis if they had evidence of spinal cord transection on imaging, did not discharge to inpatient rehabilitation after their acute hospital stay or if both acute hospital discharge and inpatient rehabilitation discharge motor scores were unavailable.
The decision for duraplasty was at the discretion of the treating surgeon, but was limited to patients with cervical and thoracic acute spinal cord injuries, and was only performed by a single attending surgeon at our institution during this time frame. Intraoperative ultrasound was used to aid in the decision-making. If intraoperative ultrasound showed narrowing of the intrathecal space or cord injury thought to be at risk of significant cord swelling in ensuing days, expansile duraplasty was considered.
All patients received standard of care treatment, including spinal decompression and stabilization. In the duraplasty group, dura was opened after completing laminectomies and performing ultrasound evaluation. A 3.5 cm midline linear durotomy was made. A 1.5 cm × 3.5 cm acellular, expansile patch was then sutured to the surrounding dura with running 6-0 polypropylene (Figure 1). Valsalva maneuver was used to verify no cerebrospinal fluid (CSF) leak along the suture line; if necessary, additional reinforcing sutures were placed. After confirming no CSF leak, the suture line was covered with a polyethylene glycol ester / trilysine amine hydrogel sealant for further protection against CSF egress. Hemostasis was achieved and multilayer soft tissue closure was performed in the same manner as in patients without duraplasty, according to the preferences of the attending surgeon. Surgical Technique for Expansile Duraplasty (A) Left: After Performing Laminectomies, the Dura Over the Injured Spinal Cord is Carefully Opened With a Midline, Linear Incision (B) Right: An Acellular, Expansile Patch is Sutured to the Surrounding Dura to Expand the Intrathecal Space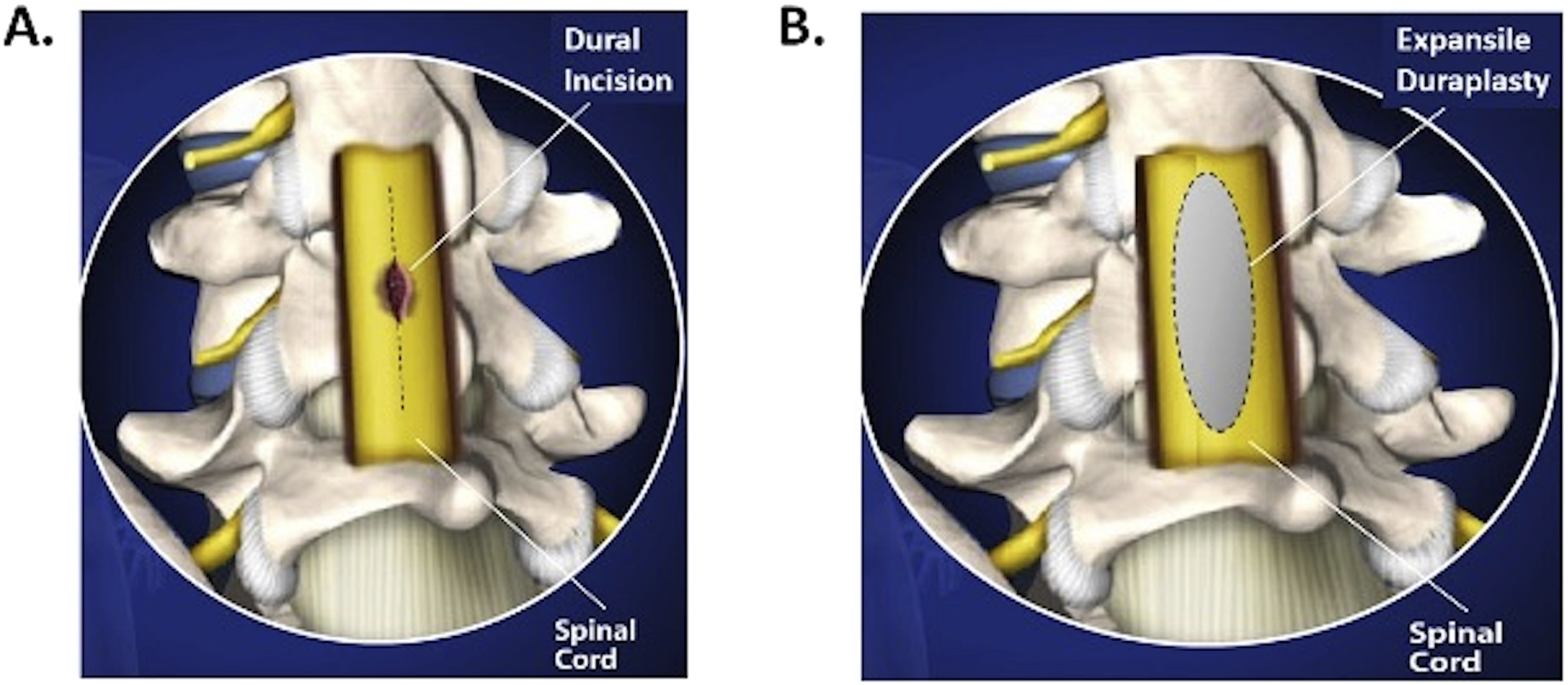
Data was collected from the electronic medical record retrospectively. Patient information collected included age at the time of surgery, sex, level of injury, steroid administration, American Society of Anesthesiologists (ASA) class, time from injury to surgery, and intensive care unit (ICU), hospital, and inpatient rehabilitation length of stay. We collected information on the surgical case length and estimated blood loss. We additionally collected information on complications, including deep vein thrombosis (DVT), pulmonary embolism (PE), urinary tract infection (UTI), pneumonia, return to the operating room for wound revision, and CSF infection. The American Spinal Injury Association (ASIA) International Standards for Neurological Classification of SCI (ISNCSCI) exam was performed preoperatively and at the time of acute hospital and inpatient rehabilitation discharge. 19 The primary outcome was change in total ISNCSCI motor score between preoperative evaluation, acute hospital discharge, and discharge from inpatient rehabilitation.
We divided patients into those who underwent expansile duraplasty at the time of surgery vs those who did not. Differences between groups were compared with Wilcoxon rank sum test for continuous variables and Fisher’s exact test for categorical variables. If a patient was missing an outcome measure, they were excluded from that particular analysis. Longitudinal analysis of delta motor scores was performed using a linear mixed model (LMM) with random intercept and slope. Analysis of variance (ANOVA) of the LMM was performed with Satterthwaite’s method for degrees of freedom approximation. Given the study’s observational nature and the potential for confounding, we also adjusted the LMM analysis by the inverse probability weights of the propensity score. The propensity score was calculated with logistic regression with duraplasty or not as the response and age at surgery, sex, level of injury (cervical vs thoracic), pre-operative motor score, time to surgery (hours), time from surgery to hospital discharge (days), and time to rehabilitation discharge (days) as predictors. Stabilized weights were calculated from the propensity score and used as regression weights in the LMM analysis, effectively reducing the covariate imbalance between treatment groups. A P value < 0.05 was considered significant. Statistical analysis was performed using R (v 4.1.0). Due to the consideration that patients who had a traumatic durotomy from their spinal fracture might not benefit from duraplasty as much as those without, we performed a post-hoc analysis with additional exclusion of patients with CSF egress noted during their operation. Most of these cases were clearly traumatic durotomies that occurred at the time of injury. In this post hoc analysis, we additionally excluded cases in which there was CSF egress noted in the operative report, when it was unclear whether this was due to a traumatic CSF leak or an incidental durotomy during surgery.
Results
One hundred forty-seven acute traumatic spinal cord injury patients from our local trauma database who had undergone surgery were screened for this study. Five patients were excluded for having lumbar level injuries and 6 for having penetrating mechanisms or imaging evidence of spinal cord transection. Twenty-four patients were excluded as they did not discharge to inpatient rehabilitation and another 16 were excluded due to unavailable documentation of motor scores at both acute hospital discharge and rehabilitation discharge.
Patient Demographics and Clinical Variables
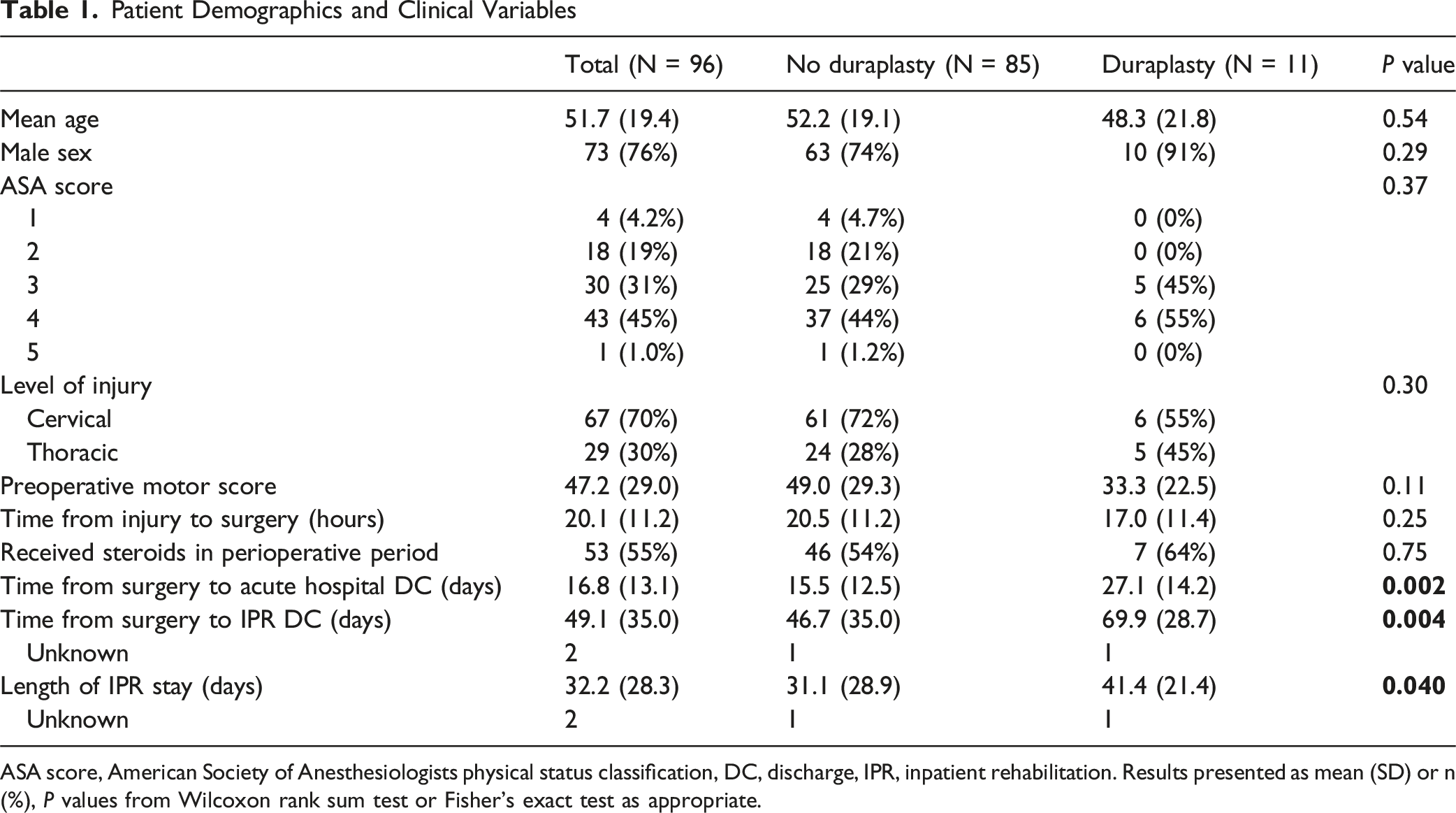
ASA score, American Society of Anesthesiologists physical status classification, DC, discharge, IPR, inpatient rehabilitation. Results presented as mean (SD) or n (%), P values from Wilcoxon rank sum test or Fisher’s exact test as appropriate.
Patient Outcomes
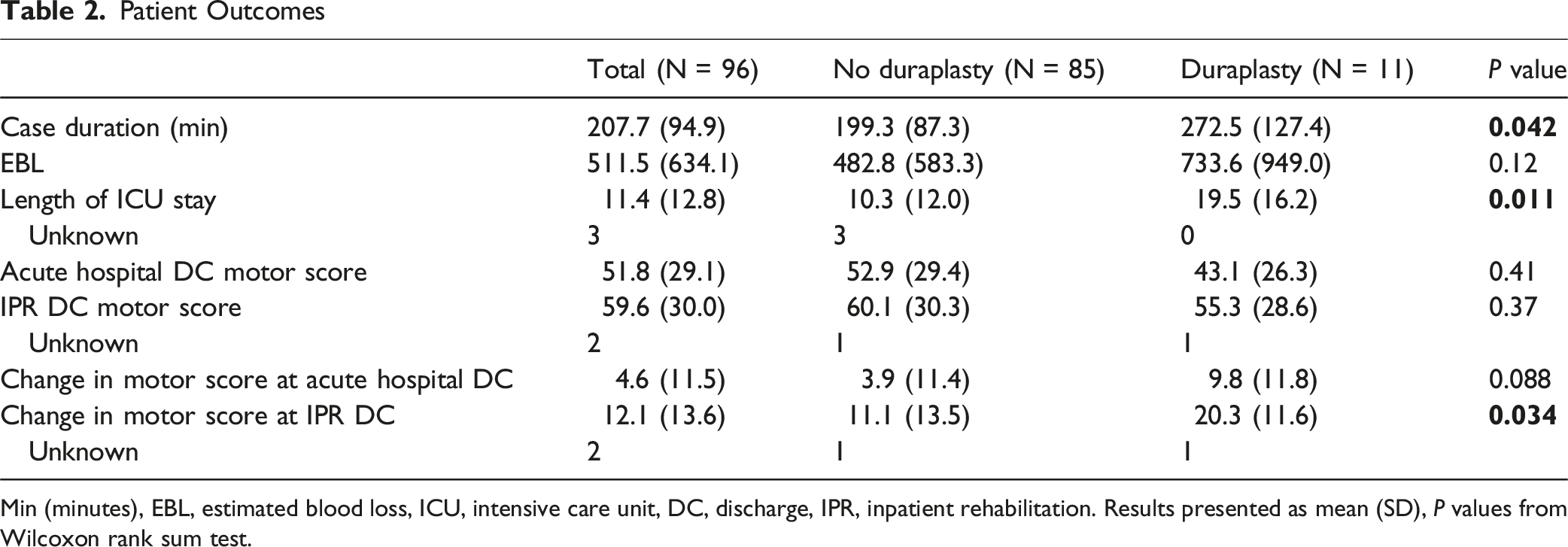
Min (minutes), EBL, estimated blood loss, ICU, intensive care unit, DC, discharge, IPR, inpatient rehabilitation. Results presented as mean (SD), P values from Wilcoxon rank sum test.
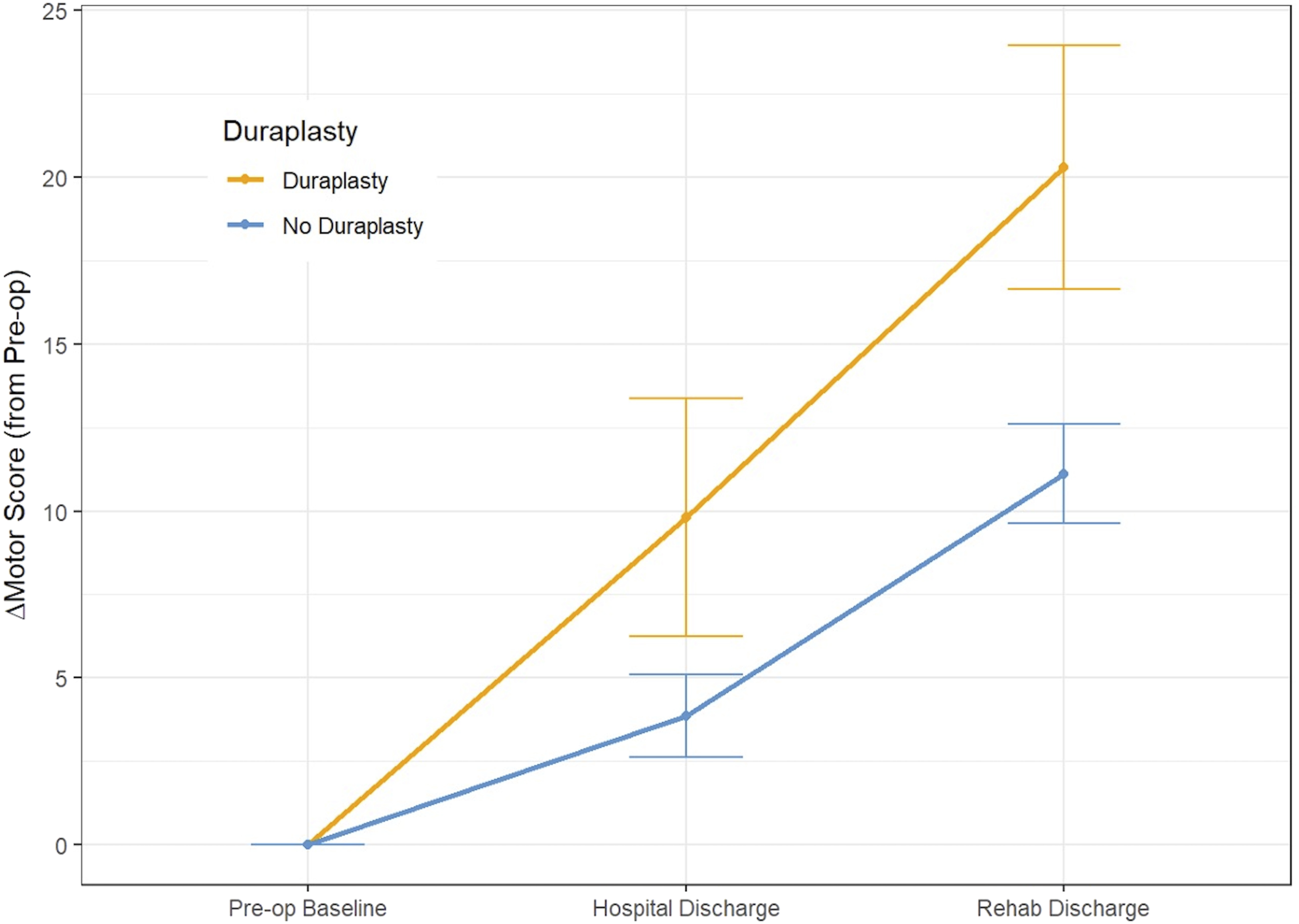
Delta Motor Scores Over Two Recovery Periods (Preoperative to Hospital Discharge and to Inpatient Rehabilitation Discharge)
Subgroup analysis of cervical SCI showed 13.1 ± 13.1 points improvement at the time of rehabilitation discharge for non-duraplasty patients and 18.4 ± 10.9 points for duraplasty patients. Thoracic SCI patients had 5.8 ± 13.5 points improvement at the time of rehabilitation discharge for non-duraplasty patients and 22.2 ± 13.1 points of improvement for duraplasty patients.
Average surgical case duration was longer in the duraplasty group (272.5 ± 127.4 min vs 199.3 ± 87.3 min for the nonduraplasty group, P = 0.042) and ICU length of stay was longer (19.5 ± 16.2 days vs 10.3 ± 12.0 days for nonduraplasty, P = 0.011). The difference in estimated blood loss between the groups was not statistically significant. See Table 2. Patients who underwent expansile duraplasty had longer post-surgery hospital stays than those without duraplasty (27.1 ± 14.2 days for duraplasty vs 15.5 ± 12.5 days for non-duraplasty, P = 0.002). There was additionally a significant difference between groups in how long patients spent in inpatient rehabilitation (41.4 ± 21.4 days for duraplasty vs 31.1 ± 28.9 days for non-duraplasty, P = 0.040). This was also reflected in longer total days from surgery to inpatient rehabilitation discharge (69.9 ± 28.7 days for duraplasty vs 46.7 ± 35.0 days for non-duraplasty, P = 0.004). See Table 1.
Surgical and Medical Complications

OR, operating room, CSF, cerebrospinal fluid, DVT, deep vein thrombosis, PE, pulmonary embolus, UTI, urinary tract infection. Results presented as n (%), P values from Fisher’s exact test.
Motor Outcomes excluding Patients With a Durotomy
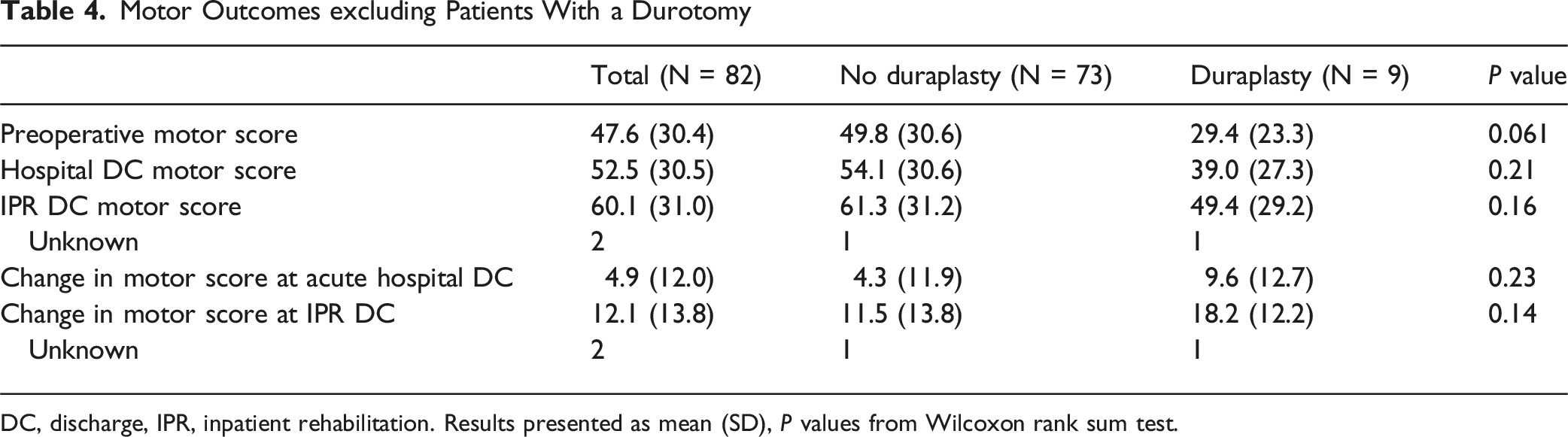
DC, discharge, IPR, inpatient rehabilitation. Results presented as mean (SD), P values from Wilcoxon rank sum test.
Discussion
Our study found that patients who underwent expansile duraplasty had a significantly greater increase in motor score from baseline to inpatient rehabilitation discharge than patients who did not undergo duraplasty. This suggests that duraplasty may be an effective therapy to increase rehabilitation and recovery potential in acute spinal cord injury.
Prior studies indicate that the initial, instantaneous, mechanical injury to the spinal cord is followed by a secondary injury phase of necrotic and apoptotic death of surrounding cells, which expands the size of the cord insult.2,3,20 This secondary injury is mediated by mechanisms such as edema, ischemia, glutamate cytotoxicity, free radical production, nitric oxide production, membrane disruption, and inflammation.2-4 Rodent studies have shown near complete loss of spinal cord perfusion at the injury epicenter and decreased perfusion of the surrounding penumbra. 21 Other preclinical studies suggest that there is a window of opportunity after SCI during which therapeutic intervention may improve outcomes.2,4
Traumatic brain injury literature demonstrates that dural opening provides a significant reduction in ICP and increase in cerebral perfusion pressure compared to bony decompression alone.12-14 It goes with reason that the same might be true for the spinal cord following spine trauma.
There is a growing body of preclinical evidence in favor of expansile duraplasty. A study by Camlar et al found a decrease in the number of apoptotic neurons in the spinal cords of rats who received duraplasty within 6-8 h of SCI compared to those without duraplasty, suggesting a positive impact of duraplasty on a cellular level. The same was not true for rats who underwent duraplasty at 24 h after injury, highlighting the importance of early decompression. 22 Another study in rats found that rats treated with durotomy demonstrated more white matter sparing, less axon degradation, and a decreased inflammatory response compared to those with laminectomy alone. In addition, they showed improved recovery of neurologic function, as measured by the Basso-Beattie-Bresnahan (BBB) scores, in rats treated with durotomy. 23
Smith et al compared outcomes in rats who underwent cervical SCI without durotomy, with durotomy alone, vs with durotomy plus duraplasty. 24 On immunohistochemical analysis spinal cords from the rats who underwent durotomy had the greatest inflammatory response. The inflammatory response seen in the duraplasty group was less than that seen in either the durotomy-only group or the group without durotomy. The duraplasty group had significantly smaller lesional volumes than either the durotomy-only or the injury-only groups. They also found that duraplasty after spinal trauma was associated with improved grip strength at multiple time points compared to the control injury group. Interestingly the durotomy-only group consistently showed worse function at multiple time points. 24 This study highlights the potential importance of dural repair after expansion, beyond just the importance of avoiding CSF leak.
Khaing et al examined the effects of relieving intrathecal pressure (ITP) by opening the dural and pial membranes in a rodent model of acute traumatic SCI. 25 Prior to treatment, ITP significantly increased after SCI. Following dural decompression, ITP decreased. Durotomy significantly increased sparing of gray matter in the spinal cord. Additionally, dural decompression led to improved functional recovery, evaluated using BBB and Ladder walk analysis. 25 Combined, these studies suggest that elevated ITP following acute SCI negatively impacts functional recovery in rodents. Therefore, intrathecal decompression may be an effective way to promote functional recovery in humans.
There are several human studies that support the role of intrathecal decompression to promote functional recovery after SCI. One study demonstrated that laminectomy, in comparison to anterior decompression without laminectomy, in cervical SCI increased the rate of complete spinal cord decompression. However, even with both anterior and posterior decompressions, not all patients had full radiographic decompression with visible CSF surrounding the injured spinal cord. 15 Another study looked at MRIs of SCI patients and found that in 25% of those with spinal cord compression, the dura appeared to be contributing. 16
Squair et al. found that spinal cord perfusion pressure (SCPP) optimization in the first five days after injury positively correlated with motor score improvement in SCI patients at 6 months. 26 A study looking at intraspinal pressures in SCI patients found that intraspinal pressure was higher at the site of the injury than at a CSF site below the injury or extradurally and that intraspinal pressures were >20 mmHg even in patients with laminectomy, suggesting ongoing compression due to dura. 18 In two patients in which SCPP was monitored, motor scores positively correlated with higher SCPP on serial exams. This study also demonstrated increased spial cord blood flow with increased SCPP. 18 This group later went on to demonstrate that patients who underwent laminectomy with duraplasty had lower intraspinal pressures and higher SCPP postoperatively than patients who underwent laminectomy alone. 17
One potential concern about duraplasty is the possibility of persistent CSF leak. This could lead to postoperative complications such as infection, delayed wound healing, and/or symptomatic intracranial hypotension. Meticulous closure of the dural repair as well as overlying soft tissue layers reduce these risks. 27 Providing reassurance that such potential risks can be overcome is a study of pediatric patients with Chiari malformation in which there was no increase in infectious complications between groups treated with bone only decompression and bony decompression plus duraplasty. 28 In our study, no expansile duraplasty patients had to return to the operating room for wound complications, in comparison to 5.9% of non-duraplasty patients. No patient in either group had positive CSF cultures during their hospitalization. This suggests that performing expansile duraplasty does not increase the risk of wound breakdown or infection.
Rates of DVT and PE did not differ significantly between the groups. The rates of UTI and pneumonia were relatively high in both groups compared to other complications. The high rates likely reflect the high rates of these complications in patients with aSCI. 29 Five out of 11 patients with duraplasty (45%) had UTIs and pneumonia, which is higher than the rates seen for the nonduraplasty group, but the difference was not statistically significant. The higher rate is likely an artifact of the small sample size as these complications do not have a clear link to duraplasty. These complication rates should be further examined in larger studies. As expected given the additional procedure, the average case duration in the expansile duraplasty group was longer than in the non-duraplasty group.
Due to our consideration that a traumatic durotomy occurring at the time of injury may partially decompress the intradural space and thus impact functional outcomes, we performed a post-hoc analysis, excluding all patients with unintended CSF egress encountered at the time of surgery. In this analysis, there was no significant difference in motor score change, although the expansile duraplasty group did still trend in the direction of having greater improvement. A possible explanation is that traumatic durotomy is not sufficient for decompressing the intrathecal space and that even patients with traumatic durotomy may still benefit from having an expansile patch sutured in. Our post-hoc analysis also had decreased power to detect a difference due to the smaller n = 9 duraplasty patients after exclusion.
Limitations
Patients who underwent expansile duraplasty had longer acute hospital and inpatient rehabilitation stays than patients who did not undergo duraplasty. This time difference is a potential confounder and warrants further study in a prospective randomized trial. Additionally, the expansile duraplasty group had lower baseline motor scores than the non-duraplasty group, although not statistically significant. By the time of hospital discharge, there was not yet a significant difference in motor improvement between groups, although the trend towards improvement with duraplasty had begun. To fully realize any potential for motor recovery, patients in this study needed to complete SCI rehabiliation. Whereas the average baseline values differ by 15.7 points, the average values at rehabilitation discharge differ by only 4.8 points, demonstrating that the duraplasty group was able to reach a similar motor outcome despite starting worse, again suggesting an increased recovery potential. The longer times to hospital and rehabilitation discharge may be due to the lower initial motor scores, indicative of more severe initial injury, in the duraplasty group. The lower baseline motor scores in the duraplasty group could have biased the results against duraplasty, yet these patients still achieved greater motor score increase. Alternatively, one could assert that the lower baseline scores in the duraplasty group leave room for a greater amount of improvement as there may be a ceiling effect that impacts the non-duraplasty group. However, the average preoperative motor score in the non-duraplasty group was only 49, which would seem to leave room for the 20 point improvement seen in the duraplasty group by the time of rehabilitation discharge. These limitations, related to the retrospective study design, should be further studied via a prospective randomized trial.
Additional limitations include that our patient population comes from a single academic level one trauma center and all duraplasty surgeries were performed by a single attending surgeon. Due to the exploratory nature of this study, the sample size is small. Full sensory scores were not consistently available preoperatively. Therefore ISNCSCI motor scores were used for analysis. Recent studies have shown that ISNCSCI motor score may be a better metric than AIS conversion for assessing SCI recovery. 30 Postoperative MRI was not routinely available to allow for quantitative analysis of postoperative decompression and should be assessed in future studies. Despite these limitations, we demonstrate some important findings. First, duraplasty is feasible and safe in acute traumatic spinal cord injury patients, consistent with findings in other international groups.17,18 Second, duraplasty has the potential to increase motor recovery in these patients. Our findings offer the first validation of this technique within the United States and a basis on which to perform larger, multicenter, prospective studies on the role of expansile duraplasty in acute traumatic spinal cord injury. Two such prospective studies, Duroplasty for Injured Cervical Spinal Cord with Uncontrolled Swelling (DISCUS) and Duraplasty for Acute Traumatic Spinal Cord Injury (DATSCI), have since commenced and should provide important data in the coming years.
Conclusions
In this contemporaneous, single-center, retrospective study, SCI patients undergoing expansile duraplasty had significantly greater improvement in ISNCSCI motor scores at rehabilitation discharge compared to non-duraplasty patients. These data present an independent and the first United States validation of preliminary data from international centers in favor of expansile duraplasty. Further prospective, randomized studies are warranted to assess whether this adjunctive surgical technique is efficacious in treating acute traumatic SCI.
Footnotes
Ethical Considerations
The University of Washington Institutional Review Board approved this study, number 07742.
Consent to Participate
Consent to participate was waived by the IRB due to the retrospective nature of this study.
Author Contributions
M.G. collected data, interpreted results, drafted and critically revised the manuscript. E.R. collected data and drafted the manuscript. A.T.E. drafted the manuscript, performed data analysis and interpretation of results. R.S. performed study conception and design, study supervision, interpreted results, and critically revised the manuscript. All authors approved the final version of the manuscript and are accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RS is a consultant for Spiderwort, Propio, and Globus. MG, ER, and ATE have no competing interests.
Data Availability Statement
Data analyzed for this study are available from the corresponding author upon reasonable request.
