Abstract
Study Design
Translational rodent study.
Objectives
To investigate (1) chemokine-mediated mesenchymal stem cell mobilization and homing to the intervertebral disc and (2) using this technique to mitigate intervertebral disc degeneration in a rat model.
Methods
(1) Recruitment of mesenchymal stem cells (MSCs) to intervertebral discs (IVD) was investigated using intradiscal chemokines. Hydrogel containing SDF-1, RANTES, MCP-1, or empty control was injected intradiscally, followed by near-infrared (NIR) imaging to observe MSC localization. (2) A rat IVD degeneration model was induced by annular puncture. Intradiscal RANTES injection and/or systemic AMD3100 injection was performed. Longitudinal imaging and histological analyses including Rutges Score (histologic degeneration) assessed IVD degeneration mitigation post-treatment up to 12-weeks. Statistical analyses included ANOVA and mixed-effects models to evaluate recruitment, retention, and regenerative potential of MSCs.
Results
(1) 24 rats were included in the investigation of MSC recruitment. In vivo NIR signal on 1-day post-intervention was highest with RANTES (P < .05). Ex vivo NIR signal at 14-days post-intervention was highest with RANTES (P < .05). (2) 36 IVD degeneration model rats underwent intradiscal RANTES and/or AMD3100 injection. AMD3100-treated groups showed larger nucleus pulposus (NP) volumes and reduced histologic damage, with lower Total Rutges scores (P = .004). RANTES treatment alone reduced Total Rutges scores (P = .009) and protected against IVD height loss at 6 weeks.
Conclusions
Intradiscal delivery of RANTES/CCL5 promotes a sustained and targeted recruitment of MSCs to the IVD. In a rat model of IVD degeneration, administration of systemic AMD3100 and intradiscal RANTES mitigates IVD degeneration.
Introduction
Chronic low back pain (LBP) is a leading cause of years lived with disability (YLD) globally and can be associated with physically, psychologically, and emotionally disabling symptoms.1-3 The socioeconomic effects, including healthcare costs, missed work, and lost productivity, are additionally significant.4-6 Intervertebral disc (IVD) degeneration is a recognized contributor to LBP. 2 It is associated with pain through multiple interconnected processes – as discs degenerate, they lose height and may herniate, leading to increased mechanical stresses on adjacent structures. 7 This is accompanied by extracellular matrix breakdown, release of inflammatory cytokines, and recruitment of nociceptive nerve fibers, further contributing to discogenic pain. 7 Unfortunately, addressing LBP and IVD degeneration has proven difficult. While nonoperative treatments and appropriately indicated surgical interventions have demonstrated benefit, many patients continue to experience LBP.8-10 Notably, though surgical intervention more reliably relieves radicular pain from nerve compression or pain from spinal canal stenosis, addressing IVD degeneration specifically remains less successful. 8
With the limitations of current treatment options and the irreversible nature of the pathology, the development of biologic treatments to mitigate IVD degeneration is an area of interest. Prior IVD regeneration techniques have included an adequate mechanical environment (eg, matrix or scaffold), growth and differentiation signaling, and multipotent cells.11,12 Specifically, mesenchymal stem cells (MSCs) have shown potential as a cellular source for mitigating IVD degeneration due to their ability to differentiate into IVD-like cells as well as limit damage in IVD.13,14
However, numerous hurdles hinder the use of allogeneic and ex vivo cultured MSCs in clinical pathology, including ethical concerns, immune reactions, and risk of disease transmission.15,16 Additionally, direct implantation of MSCs may have limited efficacy, as a sustained recruitment may be required for IVD repair and persistent changes.17-19 To address this, mobilization and homing of endogenous MSCs to the IVD may offer a strategy to address these challenges. 20
IVD degeneration has been explored preclinically via acute injury models in rats, rabbits, and other animal models via scalpel or needle puncture, using a posterolateral approach.21-25 Although this technique provides reproducibility and simplicity, it does not model the chronicity of IVD degeneration in humans. Our group has recently developed and applied an anterior approach for inducing chronic disc degeneration in a rat lumbar spine model. 26 We subsequently developed and applied a repeat anterior transperitoneal approach to deliver an intervention via intradiscal injection, using the same model. 27 In this study, we utilized these techniques to explore the feasibility of MSC mobilization and homing to the IVD as well as examine the ability of such a technique to mitigate IVD degeneration in situ.
Materials and Methods
Animal Sourcing and Husbandry
All described procedures were conducted under an Institutional Animal Care and Use Committee-approved protocol (IUCAC #AL-17-06, Beaumont Health System). All experiments were carried out using female Lewis rats (retired breeders, aged 5-6 months) acquired from Charles River Laboratories (Wilmington, MA, USA.) Rats were housed in a light/dark facility, fed a standard diet and allowed ad libitum cage activity. At study endpoints, rats were euthanized humanely via CO2 asphyxia.
Recruitment of Mesenchymal Stem Cells (MSCs) to the Intervertebral Disc (IVD)
To assess the capacity for MSCs to be recruited and retained at a target IVD, 24 Lewis rats underwent intradiscal injection of hydrogel loaded with chemokines SDF-1 (CXCL12), RANTES (CCL5), MCP-1 (CCL2), or empty hydrogel (n = 6/group) to the L5/L6 IVD, followed immediately by injection of MSCs labeled with a near-infrared fluorescent (NIR) probe and subsequent NIR imaging to track MSC migration.
Cell Isolation, Processing, and Labeling
One day before surgery, whole bone marrow was harvested from the long bones of naïve donor rats, subjected to red blood cell lysis, and plated. Adherent cell fraction was then collected, labeled with CellVue Burgundy (LI-COR Biosciences, Lincoln, NE, USA), suspended in sterile saline, and loaded for delivery as 1 × 107 cells in 200 µL per 1 mL syringe. This protocol has been shown to result in a concentrated source of MSCs. 28
Preparation of Intradiscal Chemokine-Loaded Hydrogel Injections
Chemokines were dissolved into hydrogel to produce a 100 µL injection volume at their respective Cmax dose (SDF-1: 100 ng; RANTES: 10 ng; MCP-1: 100 ng). Each injection volume was then loaded into a 6.0 mm, 31G insulin needle and placed in a water bath at 37°C until delivery to preserve flowability. Chemokines utilized were carrier-free, recombinant murine proteins (R&D Systems, Minneapolis, MN).
Intradiscal Injection Surgery
Intradiscal injection surgery was performed via a previously described anterior, transperitoneal approach. 27 This approach allowed direct visualization and access to the larger anterior aspect of the lumbar IVDs, facilitating repeatable delivery at the selected anatomic level. Disc level was confirmed via fluoroscopy, and the hydrogel injection was then delivered to the L5/L6 IVD. Immediately post-operative, rats received a tail vein injection of NIR-labeled MSC cells.
Near Infrared Imaging
At 1-, 3-, 7-, and 14-days post-operative, rats underwent NIR imaging of the lumbar spine to track cell migration and retention at L5/L6. Once anesthetized, each rat was laid prone, and the lumbar region was shaven to mitigate fur-associated autofluorescence. The lumbar spine was palpated to locate the L5/L6 IVD, and permanent marker was used to mark the skin cranial, caudal, and lateral to L5/L6 to allow localization on images (Figure 1A). Rats then underwent dorsal planar NIR imaging at 700 nm (Pearl Impulse, LI-COR Biosciences). At 14-days post-operative, rats were euthanized immediately following in vivo imaging. Then, the lumbar spine was dissected and NIR imaging was acquired ex vivo of the L5/L6 IVD. (A) Near-Infrared Imaging of the L5/L6 IVD was Performed in Vivo Longitudinally, as Well as Ex Vivo at the 14-Day Endpoint. Mean Signal was Computed Within a Circular ROI. Representative ROIs Demonstrate Increased Recruitment With RANTES at Both the 1-Day in Vivo Timepoint as Well as the 14-Day Ex Vivo Timepoint, Suggesting Immediate and Sustained Recruitment in These Groups. (B) Numerical Data, Shown as Individual Data Points Overlaid on Box Plots. *Indicates a Significant Difference Between Groups at P < .05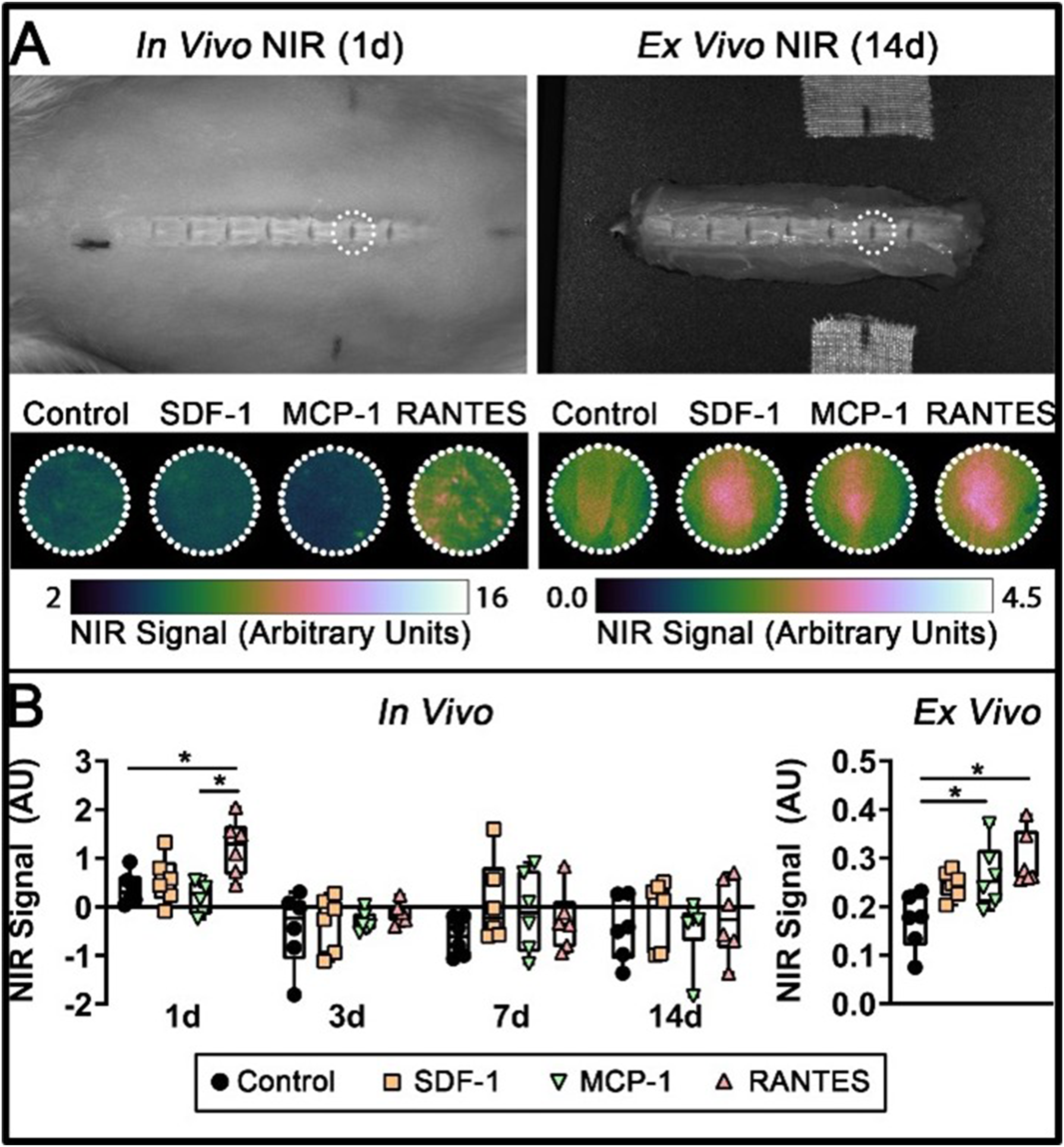
NIR images were analyzed using MATLAB (MathWorks Inc., Natick, MA, USA). For both in vivo and ex vivo images, a 17 mm diameter circular region-of-interest (ROI) was manually placed over the L5/L6 IVD, and mean signal intensity was computed. Background subtraction was performed to skin on in vivo images to limit nonspecific autofluorescence. All statistical analyses were carried out using SPSS (IBM Inc., Armonk, NY, USA). Ex vivo data was computed via univariate ANOVA with chemokine as the sole between-subject factor. In vivo data was computed via mixed ANOVA with time as within-subject factor and chemokine as between-subject factor. F-tests were conducted to assess main effects and interactions, and post hoc tests were performed to assess differences between individual groups and time points, using the Šidák correction for multiple comparisons.
Assessment of Mitigation of Intervertebral Disc (IVD) Degeneration
To assess potential therapeutic efficacy of an in situ regenerative medicine (ISRM)-based MSC recruitment technique, IVD degeneration was induced in 36 Lewis rats (n = 6/group) via annular puncture and allowed to progress for 8 weeks, followed by intervention via intradiscal chemokine injection combined with systemic mobilization therapy. Systemic mobilization therapy consisted of once daily subcutaneous injections of the small molecule CXCR4 antagonist, AMD3100 (5 mg/kg). Administration of this drug has been shown to induce rapid egress of MSCs and other cells from the bone marrow via inversion of local CXCL12 gradients in the endothelium of marrow sinusoids.29,30 Based on results of the recruitment characterization, RANTES was selected as the chemokine for further experimentation. Both surgeries were carried out via the transperitoneal approach, as described. 27 The degree of IVD degeneration was then assessed to 12 weeks post-intervention via in vivo micro-computed tomography (µCT), ex vivo contrast-enhanced µCT, and qualitative histologic grading.
Annular Puncture and Intradiscal Injection Surgeries
Annular puncture surgery was performed via the same anterior transperitoneal approach. 27 Upon localization of the L5/L6 IVD, annular puncture was performed via a #11 scalpel, inserted to the first bevel in an “X” pattern, followed by the surgical closure. Rats were allowed ad libitum cage activity for 8 weeks post-operative to allow for the development of established IVD degeneration. After 8 weeks, intradiscal injection surgery was performed, and rats were allowed ad libitum cage activity for an additional 12 weeks.
Micro-Computed Tomography Imaging
To longitudinally assess IVD height and endplate remodeling, in vivo µCT imaging was performed 1 day prior to both annular puncture and intradiscal injection surgeries, as well as at 4, 8, and 12 weeks after intradiscal injection surgery using standard methods. 31 Under isoflurane anesthesia, rats were positioned prone and the L5/L6 IVD and endplates were imaged at 55 kVP, 145 µA, 15.6 µm voxels (VivaCT-80, Scanco Medical AG, Brüttisellen, Switzerland). Immediately after the 12-week imaging timepoint, rats were euthanized and the lumbar spine was dissected. The L5/L6 motion segment was isolated by sectioning through the L4/L5 and L6/S1 IVDs, so that ex vivo contrast-enhanced µCT imaging could be performed.
To quantify glycosaminoglycan content and NP volume within the operative IVD, ex vivo contrast enhanced imaging was performed.32,33 L5/L6 motion segments were incubated for 24 h in a 40% v/v solution of ioxaglate/PBS (Hexabrix 320, Guerbet LLC., Princeton, NJ, USA), patted dry, and imaged at 55 kVP, 145 µA, 20 μm voxel size (µCT-40, Scanco Medical AG). Following imaging, L5/L6 motion segments were fixed for 72 h in 10% neutral buffered formalin and transferred to 70% ethanol for storage.
Image Processing and Analysis
All image processing was performed using MATLAB (Mathworks Inc., Natick, MA, USA). On contrast-enhanced µCT scans, IVD and NP volumes were manually contoured by an expert user (MDN). Attenuation, volumes of the NP, AF, and IVD, as well as the ratio of NP volume to IVD volume (NP Volume Ratio) were calculated.
For longitudinal data, the L5/L6 IVD and its cranial and caudal endplates were segmented from in vivo images using a semi-automatic registration algorithm. Briefly, IVD and cranial/caudal endplate volumes were manually contoured from baseline scans by expert users (MDN and SEH, respectively), and rigidly registered onto all subsequent timepoints. Any errors were addressed via manual contouring. Endplates were characterized in terms of bone volume fraction (BV/TV), bone mineral density (BMD), tissue mineral density (TMD), and endplate thickness (Th). Using a custom MATLAB algorithm, IVD heights were computed longitudinally for the whole IVD, as well as the anterior aspect, NP, and posterior aspect. All scans were rigidly registered onto the baseline scan. Three sagittal slices were chosen, corresponding to the sagittal midline and ±10% of the midline. On each slice, five height lines were drawn corresponding to anterior, mid-anterior, middle, mid-posterior, and posterior locations (Figure 4A). The endpoints of each line segment were automatically determined using the pre-segmented endplate volumes, and the length of each line was averaged over the three slices. Whole IVD height was computed as the average of all five lengths, while anterior, NP, and posterior heights were computed from anterior/mid-anterior, mid-anterior/middle/mid-posterior, and mid-posterior/posterior lines, respectively (Figure 4A). All longitudinal data was normalized to the baseline scan to account for anatomical variability.
Histology
To characterize tissue- and cell-level changes in the IVD and endplates, L5/L6 motion segments were processed for decalcified histology. Specimens were bisected down the sagittal midline and serial 5 µm sections were cut, mounted, and stained with Toluidine Blue. Damage grading was performed via the Rutges score, a histological grading system for the severity of disc degeneration on a scale of 0-12 (higher score indicating more severe degeneration), by the senior author (KCB). 34 NP area was also computed for each slice, based on manual contouring performed in QuPath. 35
Statistical Analysis
For endpoint data (contrast-enhanced µCT, histology), initial review of the dataset showed a subpopulation of IVDs completely lacking a discernable NP on both µCT and histology. Further review determined that the initial injury resulted in nucleotomy in these rats, inconsistent with our intended injury model and thus were excluded from statistical analyses. Data were computed via univariate ANOVA with injection/sham, RANTES, and AMD3100 as between-subject factors. F-tests were conducted to assess main effects and interactions, and post hoc tests were performed to assess differences at individual time points, using the Šidák correction for multiple comparisons.
For longitudinal µCT data, an outlier analysis was conducted, and several datapoints were determined to result from motion artifact on in vivo µCT scans; subsequently, these data were removed from further analyses. Data were then analyzed via a mixed effects model, with time as a within-subject factor; injection/sham, RANTES, and AMD3100 treatments as between-subject factors; pre-treatment value of each measurement included as a covariate. Data were significantly non-spherical, and several alternative covariance structures were assessed, with a heterogeneous autoregressive covariance structure determined to be most appropriate for the dataset and used for all variables. This analysis facilitated sensitive assessment of longitudinal trends by accounting for within-subject and between-subject variance, allowed rats with excluded timepoints (due to motion artifact) to be incorporated into the analysis, and accounted for non-spherical covariance. Again, F-tests were conducted to assess main effects and interactions, and post hoc tests were performed to assess differences at individual time points, using the Šidák correction for multiple comparisons.
Results
Recruitment of Mesenchymal Stem Cells (MSCs) to the Intervertebral Disc (IVD)
In vivo NIR signals at 1-day post-operative indicated measurable recruitment of marrow-derived cells at the L5/L6 IVD. At this timepoint, RANTES showed the highest mean NIR signal of any group and significantly higher signal compared to the Control (hydrogel only) group (Figure 1). At subsequent timepoints, in vivo NIR signal dropped to undetectable levels, suggesting that recruitment was highest immediately post-injection and gradually decreased over time. Ex vivo imaging enabled increased sensitivity compared to in vivo imaging, which demonstrated measurable NIR signal in all groups at 14 days post-injection, indicating that a fraction of MSCs were retained up to this timepoint. Further, ex vivo NIR signal of both RANTES and MCP-1 were significantly higher compared to Control, and RANTES had the highest mean signal of any group. Thus, RANTES was the chosen chemokine for assessing therapeutic efficacy.
Assessment of Mitigation of Intervertebral Disc (IVD) Degeneration
AMD3100 Protected Against NP Loss and Histologic IVD Damage
Injured IVDs were characterized morphologically, biochemically, and histologically via longitudinal in vivo µCT, endpoint ex vivo contrast-enhanced µCT, and histology, respectively. All IVDs included in the analysis exhibited a distinct NP, discernable on both contrast-enhanced µCT and histology (Figure 2). Mean Total Rutges histologic damage scores ranged from 4 to 9 (out of a possible 12), indicating moderate IVD degeneration across groups. Compared to Sham and Hydrogel Only, all treatment groups had qualitatively increased cellularity, with de-centralized cell clusters noted throughout the NP (Figure 4). Hydrogel injection increased Total Score compared to Sham (P < .001), indicating an independent deleterious effect associated with the injection surgery (Figure 3). IVD height loss following injury was most evident in the anterior IVD, consistent with the location of the injury (Figure 4). Generally, IVD heights decreased following injury, exhibited a slight recovery at 6 weeks post-intervention, and then decreased again at 12 weeks post-intervention. (A) Representative Sagittal Toluidine Blue-Stained Histology and Axial µCT Slices. NP Area and Volume (Dotted White Line on Histology and µCT, Respectively) was Higher in Groups Treated With AMD3100, While IVD and AF Attenuation (Inversely Correlated to GAG Content) Were Higher in Groups Treated With RANTES. (B) Numerical Data, Shown as Individual Data Points Overlaid on Box Plots. Main Effects of AMD3100 (“a:”) and RANTES (“R:”), Computed via ANOVA F-Test, are Given in the Top Right Corner of Each Graph. # Indicates a Significant Difference Between Groups at P < .05 (A) Representative Sagittal Toluidine Blue-Stained Histology of the IVD and Endplates. Treatment With AMD3100 Reduced Overall Rutges IVD Damage Score, as Evidenced by Increased NP Cellularity (Green Arrows) With a More Distinct NP/AF Boundary (Yellow Arrow) Compared to the Hydrogel Only Group. RANTES Reduced Total Endplate Damage Score, Evidenced by Reduced Osteophyte Formation (Cyan Arrow) and Retention of a More Distinct Cartilage Endplate (Red Arrow) Compared to the Hydrogel Only Group. (B) Numerical Histological Scores and Subscores, Presented as Individual Data Points Overlaid on Box Plots. Main Effects of AMD3100 (“a:”) and RANTES (“R:”), Computed via ANOVA F-Test, are Given for Total Scores in the Top Right of the Graph (A) IVD and Anterior, NP, and Posterior Zone Heights Were Derived From 5 Height Lines Placed Across 3 Sagittal Slices per IVD. (B) Representative Zonal Sagittal µCT IVD Heights Demonstrate Increased IVD Height in the RANTES-Only Treatment Group at 6 weeks Post-intervention. (C) Longitudinal Numerical Data of Bony Endplate Thickness, Volume, and Densitometry, Normalized to Baseline Values, are Presented as Summary Plots Showing Estimated Marginal Mean ±95% Confidence Interval for Each Treatment Combination, Along With ANOVA Interaction Plots Showing Interactions Between RANTES (Red), AMD3100 (Blue), and Time Point. * and # Represent Significant Differences With Respect to RANTES and AMD3100, Respectively, at P < .05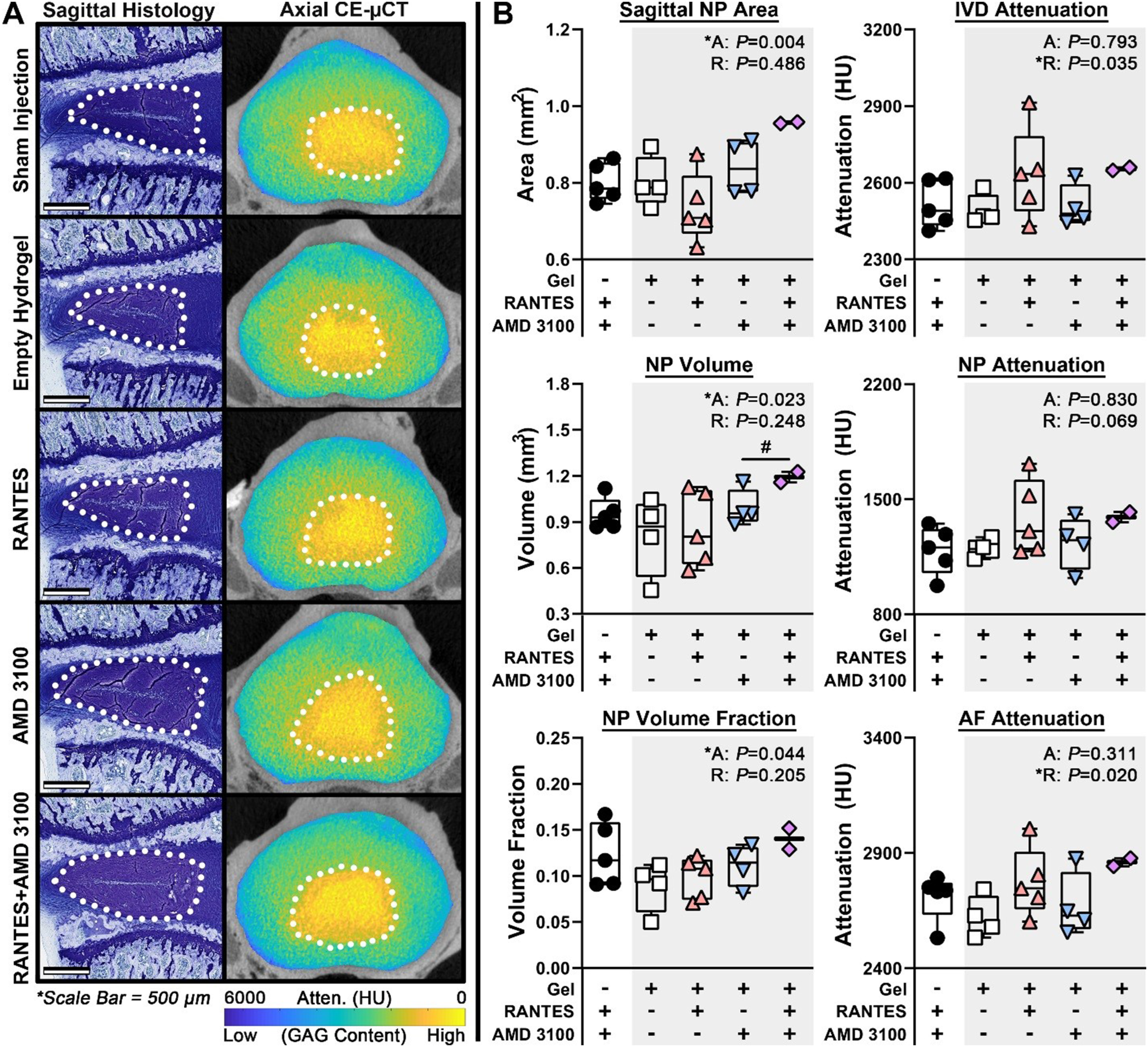
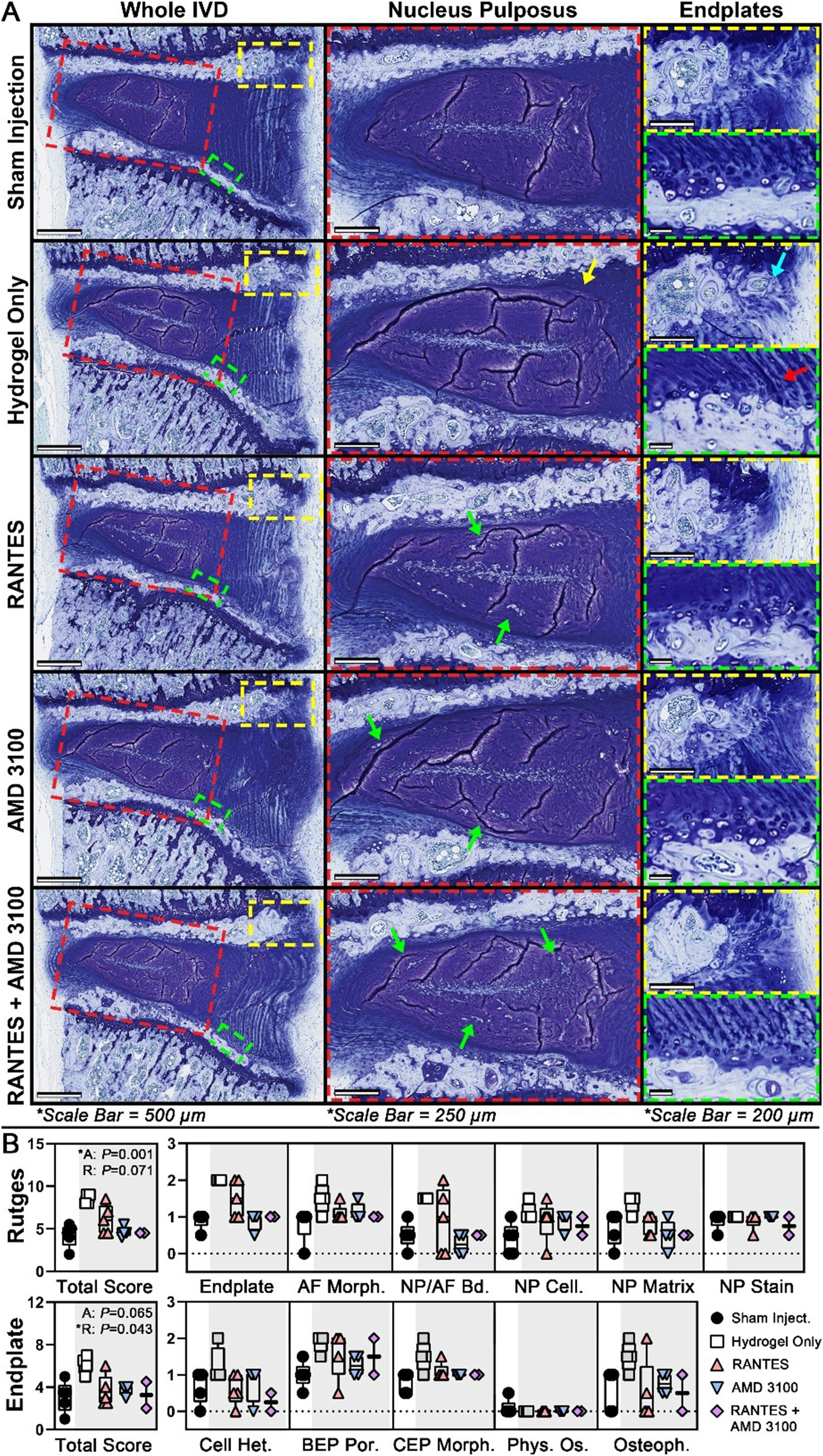
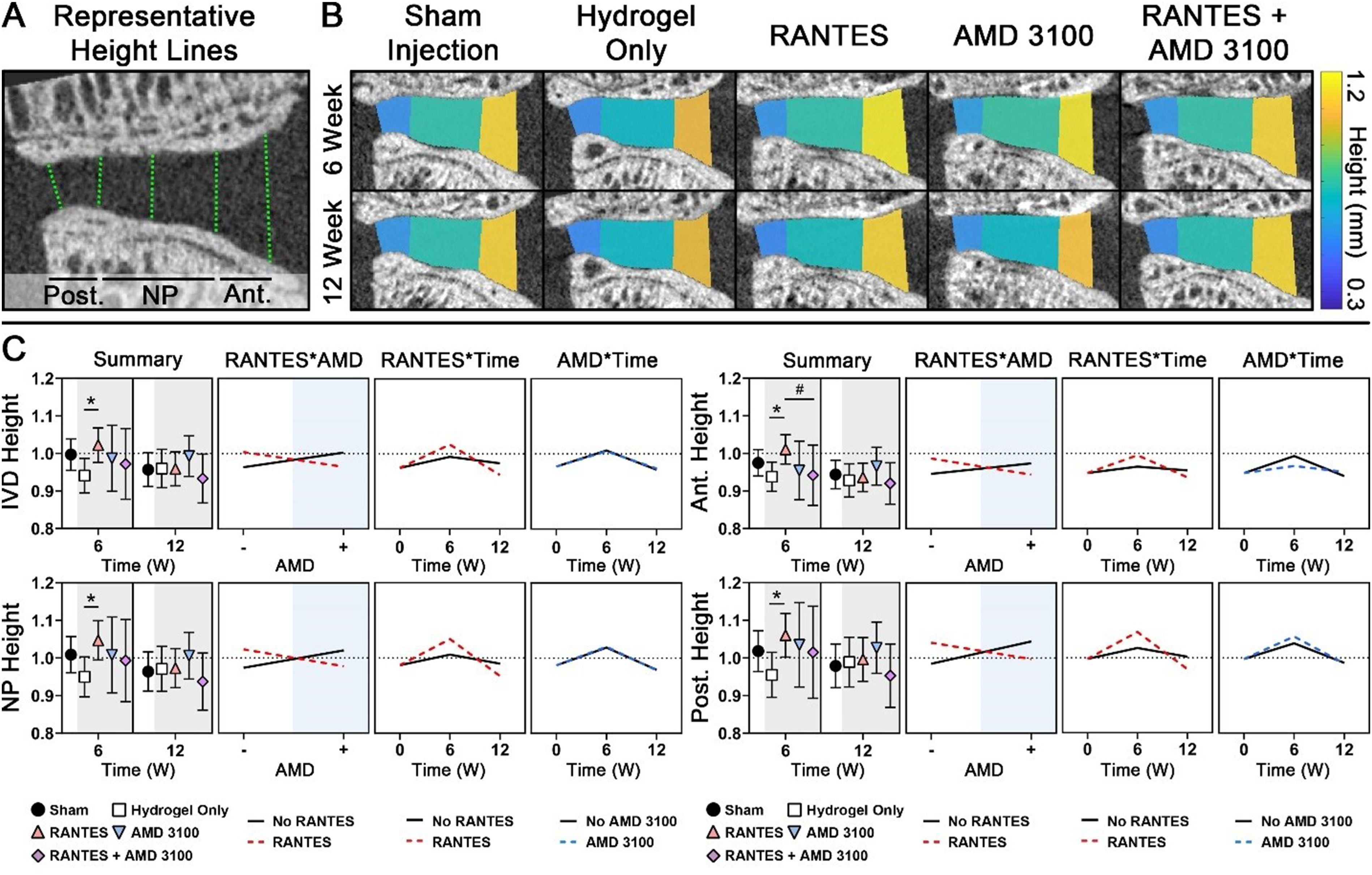
Groups receiving AMD3100 had significantly larger NPs (P = .004) compared to groups without AMD3100 treatment, as measured by 3D NP Volume and Volume Fraction as well as 2D histologic NP Area (Figure 2), with the group receiving both AMD3100 and RANTES having the highest mean NP size of any group, suggesting a possible synergistic effect. Histologically, groups receiving AMD3100 exhibited significantly lower Total Rutges score (P = .001, Figure 3), as well as lower damage subscores in the Endplate (P < .004), NP/AF Boundary (P = .039), and NP Organization (P = .020) categories. Despite these protective effects, AMD3100 had no quantitative effect on IVD height at the 6- or 12-week time points (Figure 4). However, groups treated with RANTES had higher whole IVD and AF attenuation, indicating lower GAG concentration (Figure 2). When given alone, RANTES also significantly improved Total Rutges score (P = .009), along with Endplate (P = .027) and NP Organization (P = .025) subscores, compared to Hydrogel Only (Figure 3). RANTES, when given alone, also protected against IVD height loss across all zones at 6 weeks post-intervention (Figure 4), but this same effect was not seen at 12 weeks post-intervention.
RANTES Protected Against Endplate Thickening and Histologic Endplate Damage
Cranial and caudal endplate volume, thickness, and densitometry were characterized via longitudinal in vivo µCT and endpoint histology. Mean total Endplate histological damage scores ranged from approximately 3 to 6 (out of a possible 10), suggesting mild to moderate changes to the endplate consistent with those found in the IVD (Figure 3). As in the IVD, hydrogel injection increased total score compared to Sham (P = .003), but showed little quantitative change on µCT. Compared to baseline, both endplates exhibited increased thickness, volume, and bone density at all post-injury timepoints (Figure 4).
Groups receiving RANTES exhibited significantly lower histologic Total Endplate damage scores (Figure 3), and specifically lower damage subscores in the Cell Heterogeneity (P = .036), Cartilage Endplate Morphology (P = .001), and Osteophyte (P = .042) categories. Groups receiving RANTES also showed significantly decreased caudal endplate thickness across both time points (P = .016), and with AMD3100 treatment (P = .008) (Figure 5). RANTES groups also demonstrated significantly lower caudal endplate BV and TV, as well as lower cranial endplate BV/TV at 6 weeks post-intervention though not 12 weeks. AMD3100, when given alone, was associated with lower histologic Total Endplate score (P = .017) and Cartilage Endplate Morphology subscore (P = .015), as well as significantly increased cranial and caudal endplate thickness at 6 weeks post-intervention. Co-treatment with RANTES, however, mitigated the increases in endplate thickness in AMD3100-treated groups. (A) Representative Sagittal µCT of the L5/L6 IVD and Endplates (Top) and Endplate Thickness Maps (Bottom) Demonstrate Decreased Cranial Endplate BV/TV (Yellow Arrow) and Increased Caudal Endplate Volume (Green Arrow) in RANTES-Treated Groups, Particularly Compared to AMD3100 Along (Red Arrow). (B) Longitudinal Numerical Data of Bony Endplate Thickness, Volume, and Densitometry, Normalized to Baseline Values, are Presented as Summary Plots Showing Estimated Marginal Mean ±95% Confidence Interval for Each Treatment Combination, Along With ANOVA Interaction Plots Showing Interactions Between RANTES (Red), AMD3100 (Blue), and Time Point. * and # Represent Significant Differences With Respect to RANTES and AMD3100, Respectively, at P < .05. Treatment With RANTES was Associated With Increased Caudal Endplate Volume at 6 and 12 weeks Post-Intervention, as Well as Increased Caudal Endplate Volume and Cranial Endplate BV/TV at 6 Weeks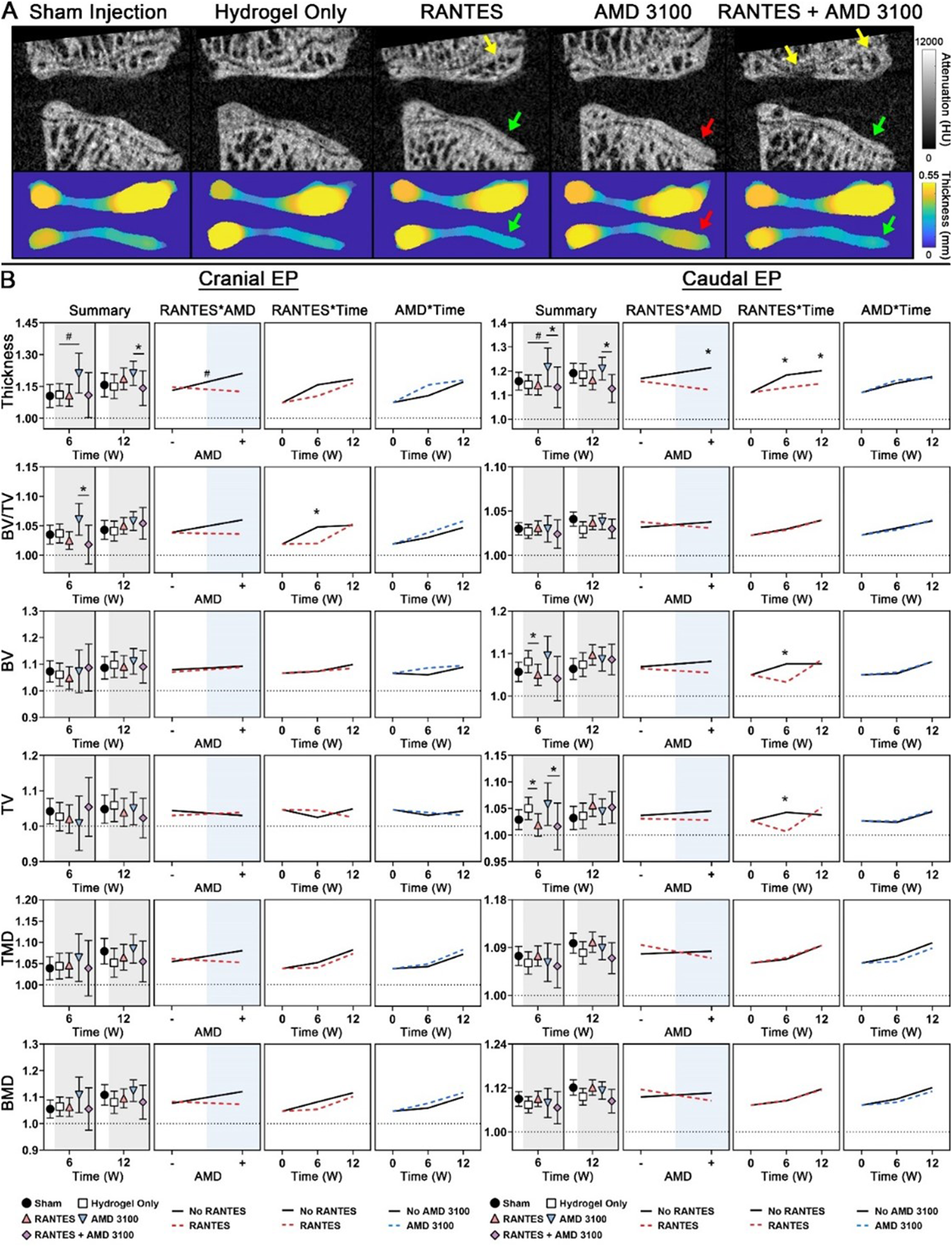
Discussion
Low back pain is among the most prevalent medical conditions with significant physical, mental and economic burdens; while its etiology is multifactorial, intervertebral disc degeneration is recognized as a major contributor.1,7 Currently, degenerative disc disease is considered a progressive and irreversible condition. Therefore, treatments to mitigate IVD degeneration have garnered significant interest. Optimal structural, biomolecular, and cellular approaches must be established prior to clinical success of a biologic treatment of IVD degeneration.36-38 Although the exact mechanisms of repair and interaction between cell and microenvironment remain unclear, MSCs have shown promise as a cellular source for IVD regenerative approaches, given their ability to differentiate into IVD-like cells.13,14,39-41 Given the challenges associated with autologous or allogenic MSCs, endogenous MSC mobilization and homing to the IVD presents a potential solution. In our study of an endogenous MSC-based treatment, we observed that intradiscal delivery of RANTES with a hydrogel was able to promote sustained targeted recruitment of MSCs to the IVD in a preclinical rat model. Additionally, we found that the use of systemic CXCR4 antagonist AMD3100, in combination of intradiscal delivery of RANTES, mitigated IVD degeneration.
In the setting of IVD degeneration, direct implantation of MSCs may be limited in efficacy, as a sustained recruitment may be required for lasting IVD change.17-19 Chemotactic homing of endogenous MSCs may provide an attractive alternative to direct implantation. MSCs have shown the ability to migrate to sites of injury throughout the body via a number of chemotactic factors.42,43 However, the IVD presents a challenging environment for MSCs to reach and exert their effects, including poor vascularization. 44 The ability of MSCs to migrate to the disc space has been examined by several groups. In a rabbit model, Tam et al 45 showed that intravenously injected MSCs have little ability to home to a native IVD, yet these MSCs enhanced GAG production in the IVD. Other studies have shown MSC homing to the disc in the setting of IVD degeneration, and that the damaged nucleus and annulus attract cellularity more than healthy discs.46,47 In our study, a targeted recruitment of MSCs was sustained to the IVD in a preclinical rat model with several chemotactic factors, including RANTES and MCP-1.
Chemokines may affect MSC recruitment to injured tissues. In our study, of the three chemokines assessed, RANTES produced the highest MSC recruitment to the IVD. Other studies have shown that, in humans, RANTES is not expressed in normal tissues, but is upregulated and highly expressed in degenerative tissues. 48 Therefore, degenerative IVDs are subject to and susceptible to RANTES upregulation. 49 Additionally, increased levels of RANTES have been isolated in painful IVDs in humans undergoing surgery. 50 Pattapa et al conducted an in vitro analysis of the chemokines in degenerative discs that may be responsible for the ability to attract MSCs and found that RANTES/CCL5 was the key chemoattractant in drawing MSCs to the disc space. 51 Our study results agree with this existing literature, and provide a possible explanation for the association between RANTES-based therapy and successful recruitment of MSCs to the disc.
AMD3100 is an FDA-approved, systemic CXCR4 antagonist that has a safe and effective profile in humans. 52 AMD3100 mobilizes endogenous stem cells and has been explored in several pathologies.53,54 AMD3100 acts by generating a CXCL12 gradient across the bone marrow sinusoidal endothelium to promote migration of MSCs into the peripheral blood stream. 55 Increases in circulating MSCs has been shown to be advantageous in several musculoskeletal models, including degenerative IVD, although the exact mechanisms of these endogenous MSC-based effects remain unknown. 56 In our study, AMD3100 treatment groups showed evidence of larger NPs and lower Rutges scores compared to other groups. Despite these protective effects, AMD3100 had little effect on IVD height, suggesting indirect effects on the IVD.
In our study, the combination of AMD3100 and RANTES provided a synergistic improvement in some measures of IVD degeneration, particularly in terms of NP size and histologic vertebral endplate damage. The mechanism by which these two interventions act in a synergist manner is unknown, but we believe that RANTES homes MSCs to the IVD space to allow them to exert their effects more efficiently. Additionally, RANTES has been shown to be angiogenic, perhaps allowing MSCs more hematogenous access to the IVD. 57 Regardless, in our study, although both RANTES and AMD3100 improved measures of IVD degeneration individually, they exerted a greater effect together.
This study included several limitations. First, the chemokine (RANTES) selection was based on the ability of MSCs to migrate to a healthy IVD immediately after chemokine injection. Although all chemokines were examined using the same methods, a different chemokine may have been more effective in the setting of a degenerative IVD, as the damaged IVD nucleus and annulus are more receptive to MSC migration than a healthy disc. 47 Additionally, although we utilized a chronic degenerative model which most closely resembles the pathophysiologic state in humans, this model does lead to some inconsistency in the grade of degeneration, with some rats sustaining nucleotomy despite consistent/controlled injuries. This mild variation may have some effect on our observations and results. Finally, as with most studies examining disc intervention, this study uses an intervention-induced degeneration model, which may affect our results. While this study included Sham and Hydrogel control groups to isolate these intervention-based changes, there may still be persistent and/or global effects.
While this study demonstrates the promise of the combination of intradiscal RANTES and systemic AMD3100 in mitigating intervertebral disc (IVD) degeneration in a preclinical model, important translational considerations remain. First, further study is warranted to determine the optimal delivery method for RANTES in the clinical setting. Intradiscal injection may pose technical challenges and potential risks in humans, including infection, disc injury, and leakage of injectate. 58 Additionally, sustained and targeted chemokine delivery in humans may require harnessing novel biomaterials or release systems to ensure localized effects while minimizing off-target activity. Second, AMD3100 is currently approved for short-term mobilization of hematopoietic stem cells in oncology settings, but the effects of its systemic use for musculoskeletal indications are unclear.59,60 Future studies should therefore investigate these delivery and systemic considerations to enable safe and effective clinical translation of this therapeutic strategy.
In conclusion, we found that intradiscal delivery of RANTES via hydrogel promoted a sustained, targeted recruitment of MSCs to the IVD. Additionally, we showed that the use of systemic AMD3100, in combination of intradiscal delivery of RANTES, mitigated IVD degeneration using a multimodal characterization approach. Future studies should examine the optimum dosing, timing, and concentrations of these interventions, as well as investigate the mechanisms by which MSCs exert their regenerative effects.
Footnotes
Ethical Consideration
The study was performed under Institutional Review Board approval.
Informed Consent
All described procedures were conducted under an Institutional Animal Care and Use Committee-approved protocol.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work was provided by a 2016 Resident/Fellow Research Grant from the Cervical Spine Research Society.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
