Abstract
Study Design
Meta-Analysis.
Objectives
Intraoperative image-guided spinal navigation provides real-time imaging during surgery and can enhance pathology localization, optimize working channel placement, and facilitate the learning process of Endoscopic lumbar spine surgery (ELSS). This meta-analysis examined whether intraoperative image-guided spinal navigation offers perioperative and clinical advantages in ELSS compared with conventional C-arm fluoroscopy.
Methods
We systematically searched PubMed, Europe PMC, Scopus, Cochrane Library, and ClinicalTrials.gov for studies comparing the perioperative and clinical outcome of intraoperative image-guided spinal navigation with conventional C-arm fluoroscopy in ELSS. Results were summarized using the mean difference (MD) or standardized mean difference (SMD) with accompanying 95% confidence intervals.
Results
A total of 19 studies involving 1390 patients were included for meta-analysis. Intraoperative image-guided spinal navigation in ELSS was significantly associated with a shorter total operation time (MD = −11.18 min, P < 0.01), fewer puncture attempts (MD = −2.94 times, P < .01), shorter cannulation time (MD = −12.59 min, P < 0.01), lower fluoroscopy use frequency (MD = −14.75 times, P < 0.01), lower radiation exposure (SMD = −4.18, P < 0.01), and shorter hospital stay (MD = −0.44 days, P = 0.03) compared with C-arm fluoroscopy. No significant differences in back Visual Analog Scale (VAS) scores, leg VAS scores, or Oswestry Disability Index scores were observed at up to 1 year of follow-up.
Conclusion
Intraoperative image-guided spinal navigation in ELSS significantly affects perioperative outcomes. However, clinical outcomes are not affected by the type of navigation. Further research is required to evaluate its cost-effectiveness across diverse health-care systems.
Keywords
Introduction
Endoscopic lumbar spine surgery (ELSS) has emerged as a minimally invasive approach to spinal surgery,1,2 offering advantages such as reduced soft tissue dissection, minimal muscle damage, 3 reduced blood loss, 4 and reduced lamina or facet resection. These advantages may mitigate the risks of epidural fibrosis, iatrogenic instability, and epidural blood supply blockage.4-8 Conventional C-arm navigation in ELSS is limited by its reliance on 2-dimensional imaging, complicating the interpretation of complex anatomical structures in a 3-dimensional surgical environment, increasing the technical difficulty of procedures, prolonging operation time, and increasing the risk of postoperative complications.2,9-11 These limitations hinder the use of endoscopic techniques in complex cases such as scoliosis,12-14 high iliac crests, or other anatomical variations. 15 In addition, repeated fluoroscopic imaging to achieve optimal working channel placement substantially increases radiation exposure, posing risks to both patients and surgical staff.16,17
Various intraoperative navigation systems, including electromagnetic systems, optical systems, and real-time imaging systems, have been developed to assist surgeons in navigating the limited surgical field of ELSS more efficiently and safely.11,18,19 O-arm navigation provides 3-dimensional imaging, 20 whereas electromagnetic navigation (EMN) offers real-time, radiation-free guidance by employing magnetic fields to achieve target positioning. 18 Ultrasound volume navigation (UVN) combines preoperative computed tomography (CT) or magnetic resonance imaging (MRI) data with real-time ultrasound to improve accuracy during surgery. Emerging technologies such as robotic-assisted navigation and augmented reality (AR) further hold promise for enhancing ELSS by reducing surgeon fatigue and improving surgeon ergonomics in the operating room. 21 Studies indicate that these advanced navigation methods may facilitate the learning process of ELSS, reduce the duration of surgery, and minimize radiation exposure for both patients and surgical staff.22-25 Despite these potential benefits, no consensus has yet been reached regarding the advantages of integrating navigation with ELSS and its impact on intraoperative and postoperative outcomes.
In this study, we conducted a systematic review and meta-analysis of matched trials to compare the effectiveness of intraoperative image-guided spinal navigation with that of conventional C-arm fluoroscopy in ELSS.
Materials and Methods
Eligibility Criteria
This systematic review and meta-analysis was conducted in accordance with the 2020 guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (Figure 1). Additionally, the study protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the registration number [CRD42024613611]. The eligibility criteria, structured within the PICO framework, were as follows. In terms of population (P), we included adult patients (18 years of age or older) with lumbar degenerative spinal disease who underwent ELSS. In terms of intervention (I), we included studies involving ELSS performed with any intraoperative image-guided spinal navigation system other than conventional C-arm fluoroscopy. In terms of comparison (C), we included studies involving ELSS performed using conventional C-arm navigation. In terms of outcomes (O), we included studies involving perioperative outcomes such as operation time, number of puncture attempts, cannulation time, fluoroscopy use frequency, and radiation dose and clinical outcomes such as length of stay, Visual Analog Scale (VAS) scores, and Oswestry Disability Index (ODI) scores. “Number of puncture attempts” refers to the number of times the surgeon inserted the puncture needle in an attempt to successfully access the target anatomical space (e.g., interlaminar or transforaminal space) during ELSS. Only studies published in English with full-text availability were included. PRISMA Flowchart. ELSS, Endoscopic Lumbar Spine Surgery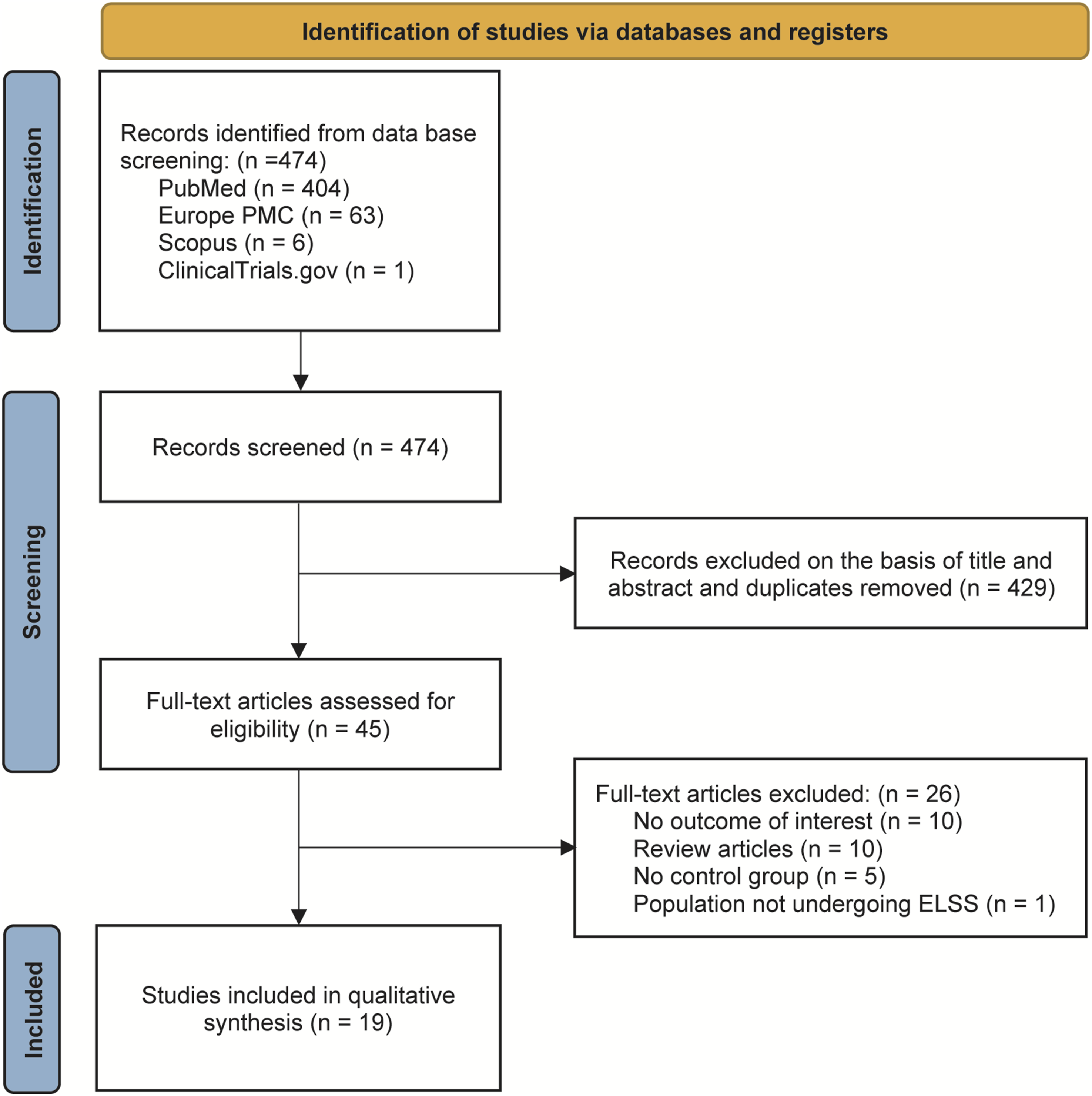
Literature Search and Study Selection
Two authors conducted a systematic search across PubMed, Europe PMC, Scopus, Cochrane Library, and ClinicalTrials.gov for articles published on or before June 3, 2024. To refine the search process, the following terms were employed: (endoscopic spine surgery OR biportal endoscopy OR ‘biportal endoscopic spine surgery’ OR ‘BESS’ OR ‘uniportal endoscopic surgery’ OR ‘UBE’ OR ‘unilateral biportal endoscopy’) AND (electromagnetic navigation OR ‘O-Arm’ OR robotic navigation OR ‘robot-assisted navigation’ OR ‘3D fluoroscopy’ OR ‘three-dimensional fluoroscopy’ OR ‘intraoperative CT’ OR ‘image-guided surgery’). The same authors independently reviewed the articles’ titles and abstracts to identify potentially relevant studies, with duplicate entries removed. After this preliminary screening process, the same authors comprehensively examined the full texts of the selected articles to confirm their eligibility for inclusion. Any disagreements during the review process were resolved through discussion.
Data Extraction
After completing the search process, the same authors independently extracted and organized data on Microsoft Excel 2019. These data included (1) author names; (2) year of publication; (3) geographical location; (4) baseline characteristics, including the number of participants and their ages and sex distribution; (5) intervention details for each group; (6) type of surgery performed; and (7) outcomes of interest.
Risk-of-Bias Assessment
Risk of bias was independently evaluated using the following tools. For nonrandomized studies, the ROBINS-I tool was used to evaluate 7 domains 26 : (1) confounding, (2) selection of participants, (3) classification of interventions, (4) deviations from intended interventions, (5) missing data, (6) measurement of outcomes, and (7) selection of the reported result. Each study was categorized as having a low, moderate, serious, or critical risk of bias. For randomized trials, the Risk of Bias 2 (RoB 2) tool was used to evaluate 5 domains 27 : (1) randomization process, (2) deviations from intended interventions, (3) missing outcome data, (4) measurement of outcomes, and (5) selection of the reported result. Each domain was rated as low risk, some concerns, or high risk, enabling an overall assessment of the risk of bias for each study.
Statistical Analysis
All statistical analyses were conducted using R software for macOS (version 4.4.0; R Foundation for Statistical Computing, Vienna, Austria). In cases of missing or zero standard deviations, we imputed a value based on the lowest value observed in other studies, as described elsewhere. 28 To compare the continuous outcomes of the intervention and control groups, we calculated mean differences with 95% confidence intervals (CIs) by using the inverse-variance method. In studies involving different scales for measuring outcomes, standardized mean differences (SMDs) were employed. For dichotomous outcomes, odds ratios (ORs) with 95% CIs were calculated using the Mantel–Haenszel approach. Given the expected clinical and methodological heterogeneity across studies, random-effects models were applied. Heterogeneity was evaluated using the I2 statistic, with values of ≤25%, 26%–50%, and >50% indicating low, moderate, and high heterogeneity, respectively. 29 Statistical significance was defined at P < 0.05. Meta-regression was conducted to examine the effects of factors such as age, sex, sample size, navigation technique, and surgery type on the outcomes. 30 Additionally, subgroup analyses were conducted using random-effects models to identify potential sources of heterogeneity.
Results
Study Selection and Characteristics
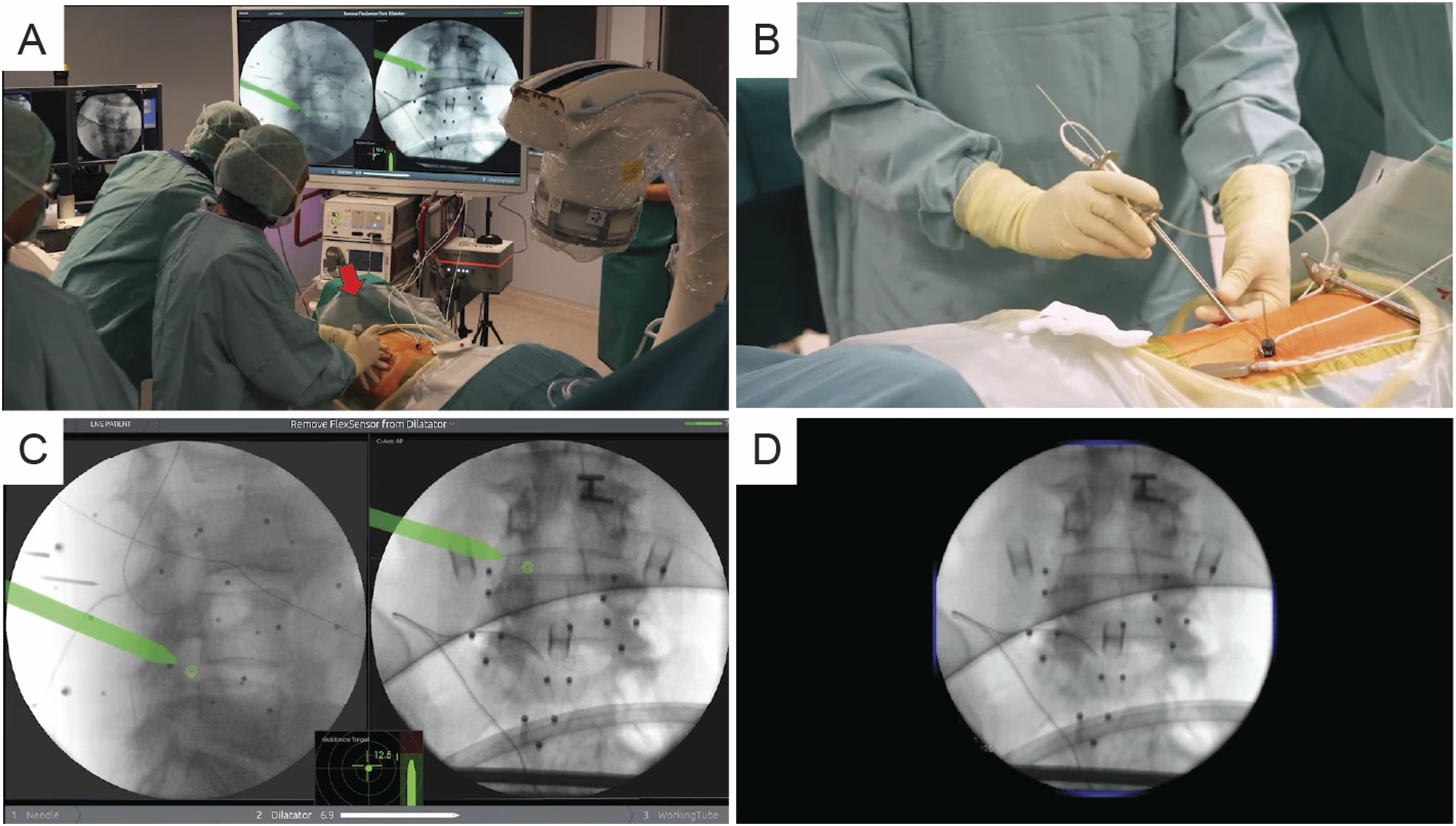
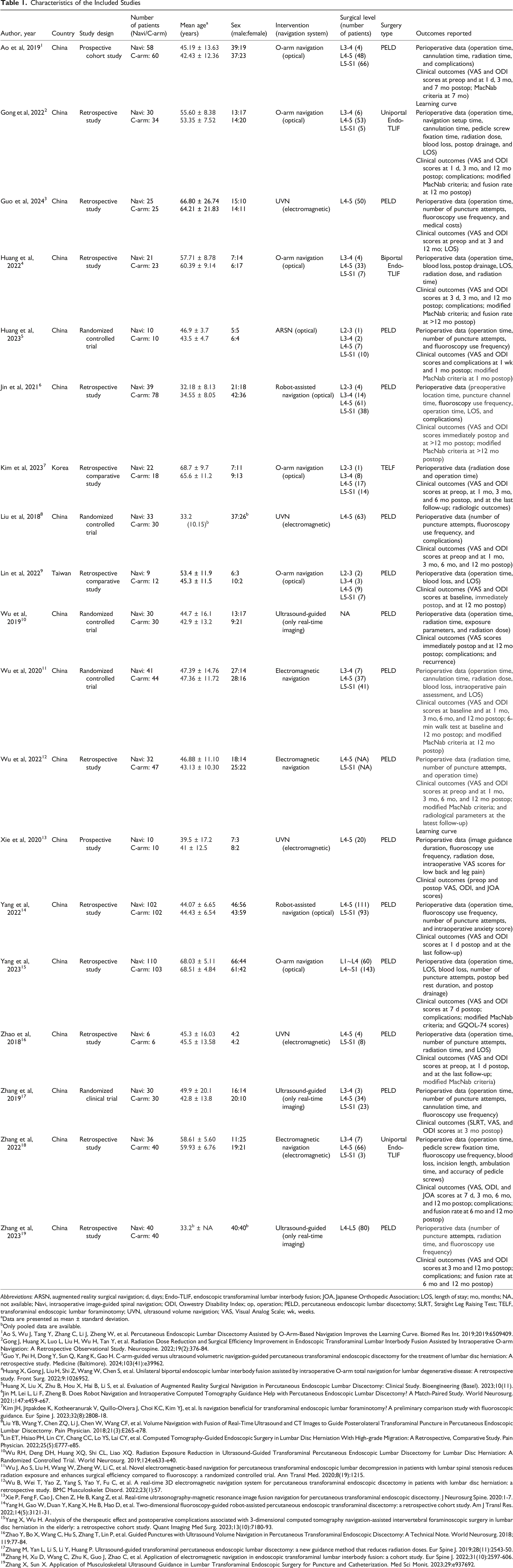
A total of 19 studies comprising 1390 patients were included for meta-analysis (Figure 1). A total of 6 types of spinal navigation were compared with C-arm fluoroscopy: 6 studies (31.6%) investigated O-arm navigation with an optical system, 2 studies (10.5%) examined robotic-assisted navigation with an optical system, 3 studies (15.8%) evaluated EMN, 3 studies (15.8%) assessed real-time ultrasound-guided navigation, 4 studies (21.1%) focused on UVN, and 1 study (5.3%) used augmented reality surgical navigation (ARSN). Different navigation systems are presented in Figures 2 and 3. Most studies included patients with disc herniations at the L4–S1 levels, with a few also including cases from L1 to L4. None of the included studies reported significant differences in disc level distribution between the comparison groups. These studies explored 2 primary surgical methods: 15 studies (78.9%) investigated percutaneous endoscopic lumbar discectomy (PELD), 1 study (5.3%) examined transforaminal endoscopic lumbar foraminotomy, and 3 studies (15.8%) focused on endoscopic transforaminal lumbar interbody fusion (Endo-TLIF). Table 1 presents the characteristics of the included studies. Optical Navigation System Demonstration. (A and B) Percutaneous Puncture Performed Under Intraoperative O-Arm Navigation. The Red Arrow Indicates the Infrared Tracking Camera of the Optical Navigation System, while the Green Arrow Highlights the O-Arm Device. (C and D) Percutaneous Puncture Performed Under Augmented Reality (AR) Surgical Navigation Utilizing the Optical Navigation System. The Orange Arrow Points to the AR Headset, and the Blue Arrow Identifies the Infrared Tracking Camera of the Optical Navigation System Electromagnetic Navigation (EMN) System Demonstration. (A and B) the Electromagnetic Field Generator was Placed in the Cranial Side of the Surgical Area (Red Arrow), and the Percutaneous Puncture was Performed Under EMN Guidance. (C) Real-Time Inline Navigation Trajectory Help Aim for the Target Point to Superior Articular Process on Lateral (Left) and Anteroposterior (Right) Images. (D) The Mapper Bridge Is Identified by at Least 15 Markers on Anteroposterior Images. Reprinted With Permission From RIWOspine. Available at: https://youtu.be/J3gwEl2q-Ns Characteristics of the Included Studies Abbreviations: ARSN, augmented reality surgical navigation; d, days; Endo-TLIF, endoscopic transforaminal lumbar interbody fusion; JOA, Japanese Orthopedic Association; LOS, length of stay; mo, months; NA, not available; Navi, intraoperative image-guided spinal navigation; ODI, Oswestry Disability Index; op, operation; PELD, percutaneous endoscopic lumbar discectomy; SLRT, Straight Leg Raising Test; TELF, transforaminal endoscopic lumbar foraminotomy; UVN, ultrasound volume navigation; VAS, Visual Analog Scale; wk, weeks. aData are presented as mean ± standard deviation. bOnly pooled data are available. Ao S, Wu J, Tang Y, Zhang C, Li J, Zheng W, et al. Percutaneous Endoscopic Lumbar Discectomy Assisted by O-Arm-Based Navigation Improves the Learning Curve. Biomed Res Int. 2019;2019:6509409. Gong J, Huang X, Luo L, Liu H, Wu H, Tan Y, et al. Radiation Dose Reduction and Surgical Efficiency Improvement in Endoscopic Transforaminal Lumbar Interbody Fusion Assisted by Intraoperative O-arm Navigation: A Retrospective Observational Study. Neurospine. 2022;19(2):376-84. Guo Y, Pei H, Dong Y, Sun Q, Kang K, Gao H. C-arm-guided versus ultrasound volumetric navigation-guided percutaneous transforaminal endoscopic discectomy for the treatment of lumbar disc herniation: A retrospective study. Medicine (Baltimore). 2024;103(41):e39962. Huang X, Gong J, Liu H, Shi Z, Wang W, Chen S, et al. Unilateral biportal endoscopic lumbar interbody fusion assisted by intraoperative O-arm total navigation for lumbar degenerative disease: A retrospective study. Front Surg. 2022;9:1026952. Huang X, Liu X, Zhu B, Hou X, Hai B, Li S, et al. Evaluation of Augmented Reality Surgical Navigation in Percutaneous Endoscopic Lumbar Discectomy: Clinical Study. Bioengineering (Basel). 2023;10(11). Jin M, Lei L, Li F, Zheng B. Does Robot Navigation and Intraoperative Computed Tomography Guidance Help with Percutaneous Endoscopic Lumbar Discectomy? A Match-Paired Study. World Neurosurg. 2021;147:e459-e67. Kim JH, Jitpakdee K, Kotheeranurak V, Quillo-Olvera J, Choi KC, Kim YJ, et al. Is navigation beneficial for transforaminal endoscopic lumbar foraminotomy? A preliminary comparison study with fluoroscopic guidance. Eur Spine J. 2023;32(8):2808-18. Liu YB, Wang Y, Chen ZQ, Li J, Chen W, Wang CF, et al. Volume Navigation with Fusion of Real-Time Ultrasound and CT Images to Guide Posterolateral Transforaminal Puncture in Percutaneous Endoscopic Lumbar Discectomy. Pain Physician. 2018;21(3):E265-e78. Lin ET, Hsiao PH, Lin CY, Chang CC, Lo YS, Lai CY, et al. Computed Tomography-Guided Endoscopic Surgery in Lumbar Disc Herniation With High-grade Migration: A Retrospective, Comparative Study. Pain Physician. 2022;25(5):E777-e85. Wu RH, Deng DH, Huang XQ, Shi CL, Liao XQ. Radiation Exposure Reduction in Ultrasound-Guided Transforaminal Percutaneous Endoscopic Lumbar Discectomy for Lumbar Disc Herniation: A Randomized Controlled Trial. World Neurosurg. 2019;124:e633-e40. Wu J, Ao S, Liu H, Wang W, Zheng W, Li C, et al. Novel electromagnetic-based navigation for percutaneous transforaminal endoscopic lumbar decompression in patients with lumbar spinal stenosis reduces radiation exposure and enhances surgical efficiency compared to fluoroscopy: a randomized controlled trial. Ann Transl Med. 2020;8(19):1215. Wu B, Wei T, Yao Z, Yang S, Yao Y, Fu C, et al. A real-time 3D electromagnetic navigation system for percutaneous transforaminal endoscopic discectomy in patients with lumbar disc herniation: a retrospective study. BMC Musculoskelet Disord. 2022;23(1):57. Xie P, Feng F, Cao J, Chen Z, He B, Kang Z, et al. Real-time ultrasonography-magnetic resonance image fusion navigation for percutaneous transforaminal endoscopic discectomy. J Neurosurg Spine. 2020:1-7. Yang H, Gao W, Duan Y, Kang X, He B, Hao D, et al. Two-dimensional fluoroscopy-guided robot-assisted percutaneous endoscopic transforaminal discectomy: a retrospective cohort study. Am J Transl Res. 2022;14(5):3121-31. Yang X, Wu H. Analysis of the therapeutic effect and postoperative complications associated with 3-dimensional computed tomography navigation-assisted intervertebral foraminoscopic surgery in lumbar disc herniation in the elderly: a retrospective cohort study. Quant Imaging Med Surg. 2023;13(10):7180-93. Zhao Y, Bo X, Wang C, Hu S, Zhang T, Lin P, et al. Guided Punctures with Ultrasound Volume Navigation in Percutaneous Transforaminal Endoscopic Discectomy: A Technical Note. World Neurosurg. 2018;119:77-84. Zhang M, Yan L, Li S, Li Y, Huang P. Ultrasound-guided transforaminal percutaneous endoscopic lumbar discectomy: a new guidance method that reduces radiation doses. Eur Spine J. 2019;28(11):2543-50. Zhang H, Xu D, Wang C, Zhu K, Guo J, Zhao C, et al. Application of electromagnetic navigation in endoscopic transforaminal lumbar interbody fusion: a cohort study. Eur Spine J. 2022;31(10):2597-606. Zhang X, Sun X. Application of Musculoskeletal Ultrasound Guidance in Lumbar Transforaminal Endoscopic Surgery for Puncture and Catheterization. Med Sci Monit. 2023;29:e937692.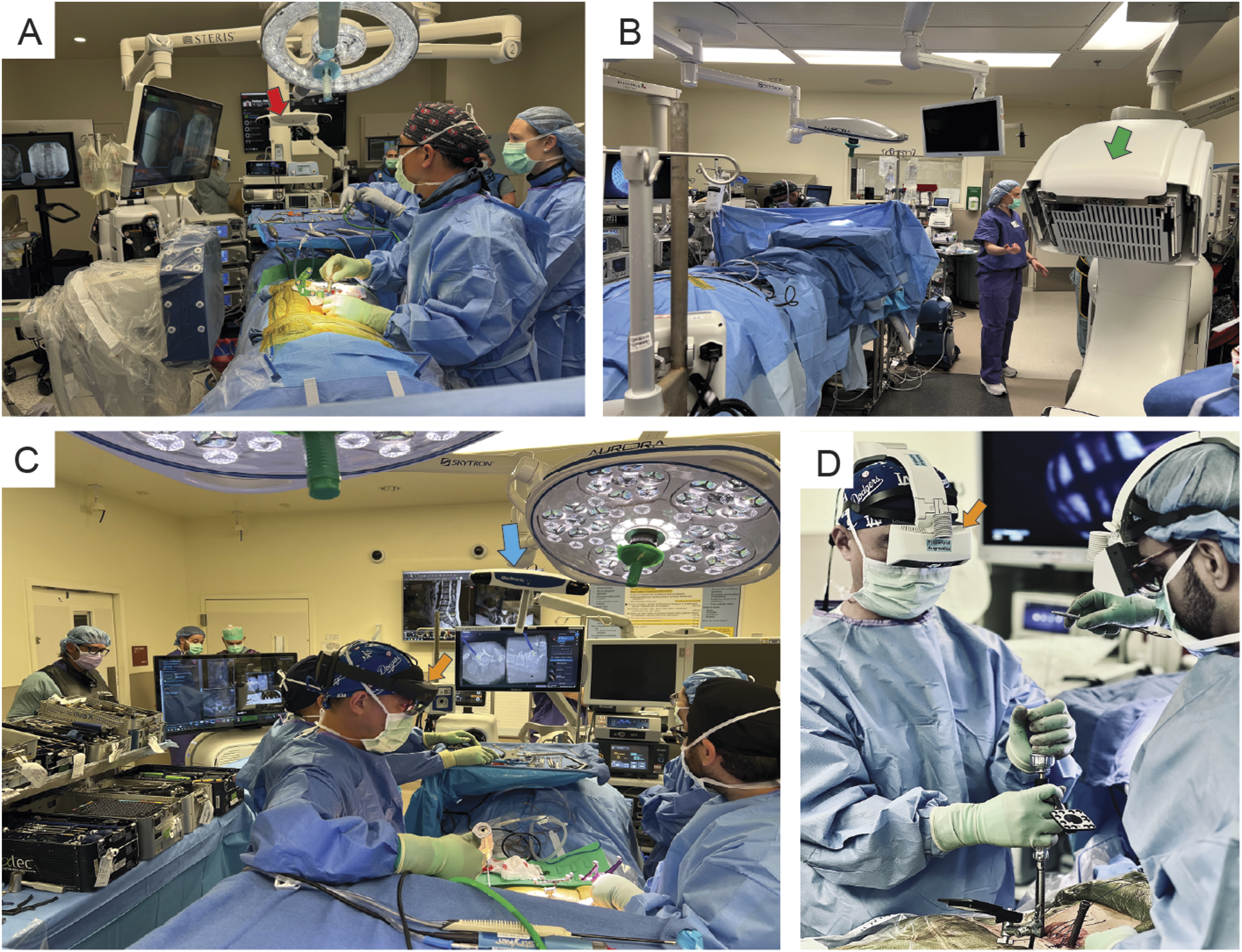
Assessment of Study Quality
A total of 15 nonrandomized studies were evaluated using the ROBINS-I tool, and the results indicated that 7 studies had a low risk of bias, whereas 8 studies had a moderate risk of bias (Figure 4A). A total of 4 randomized controlled trials were evaluated using the RoB 2 tool, and the results indicated that 1 study had a low risk, whereas 3 studies had some concerns (Figure 4B). Risk-of-Bias Assessment of the Included Trials: (A) ROBINS-I; (B) RoB 2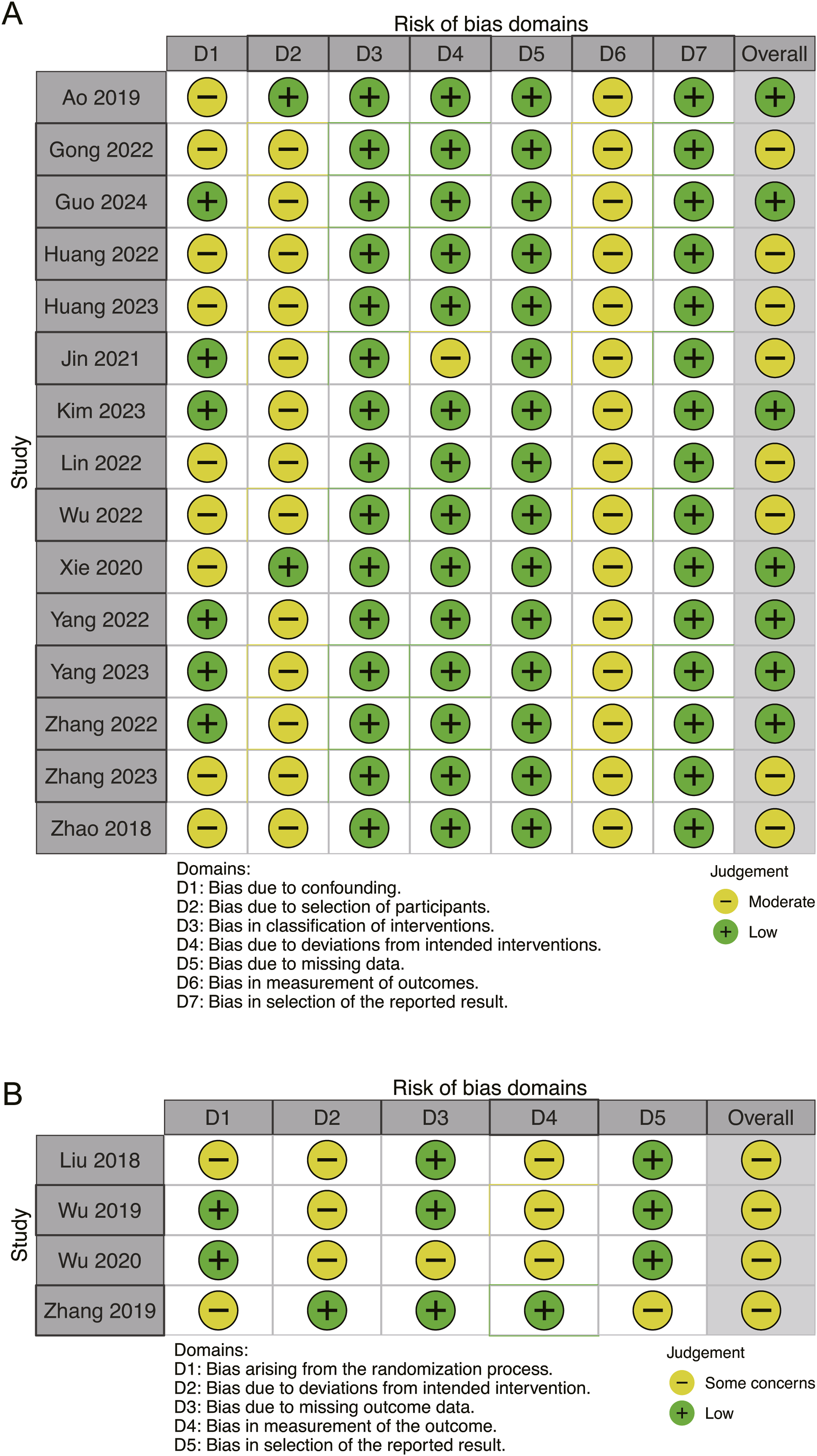
Perioperative Outcomes
Operation Time
Meta-analysis of 16 studies (n = 1263) revealed that intraoperative image-guided spinal navigation was associated with significantly reduced operation time (MD = −11.18 min, 95% CI = −14.67 to −7.70, P < 0.01, I2 = 83%; Figure 5A). Forest Plots Demonstrating the Comparison of Perioperative Outcomes Between Intraoperative Image- Guided Spinal Navigation and C-Arm Navigation in Patients Undergoing ELSS. (A) Operation Time, (B) Number of Puncture Attempts, (C) Cannulation Time, (D) Fluoroscopy Use Frequency, and (E) Radiation Dose. df, Degrees of Freedom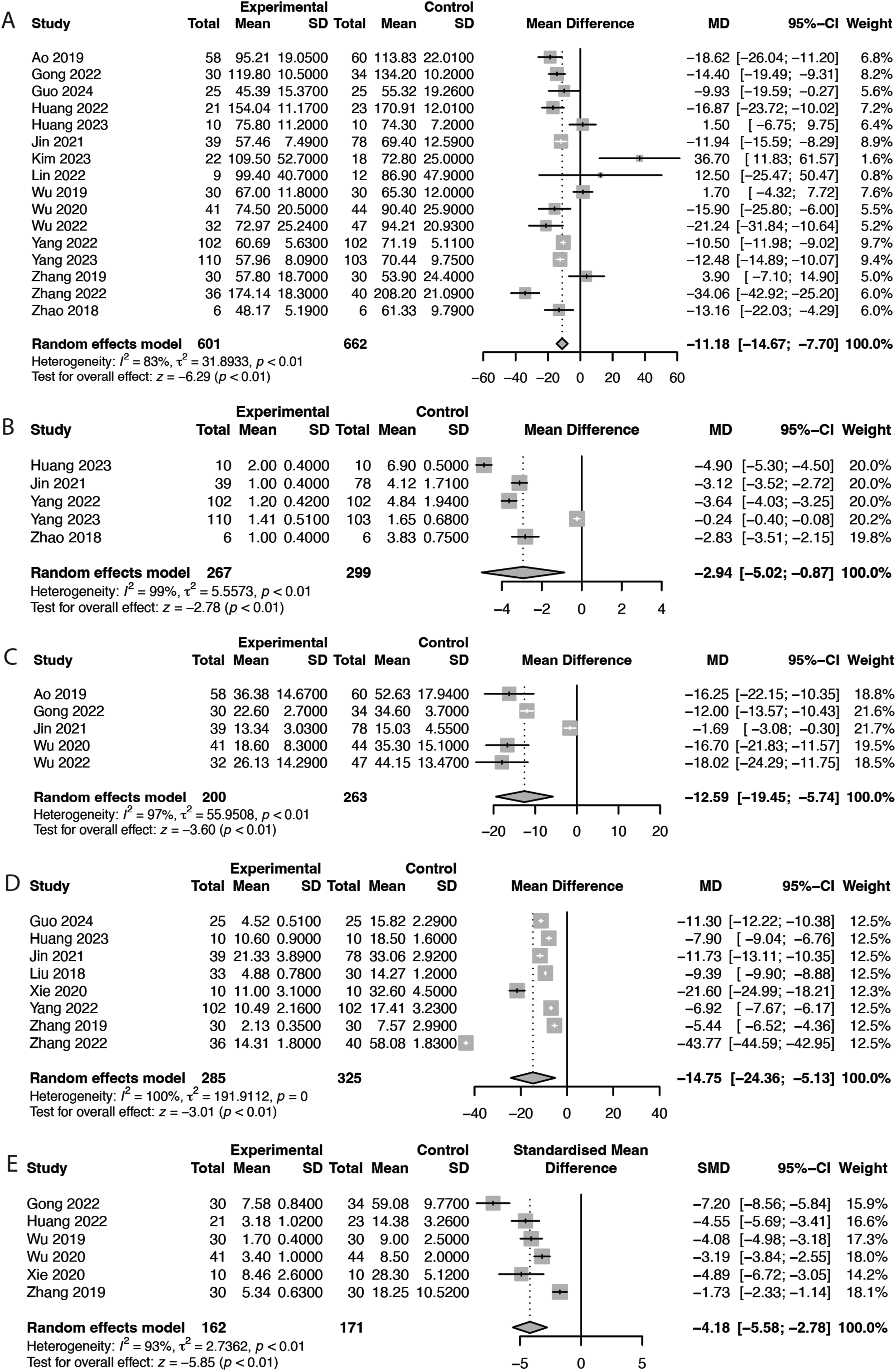
Puncture Attempts and Cannulation Time
Meta-analysis of 5 studies (n = 566) revealed that intraoperative image-guided spinal navigation was associated with a significant reduction in the number of puncture attempts (MD = −2.94 attempts, 95% CI = −5.02 to −0.87, P < 0.01, I2 = 99%; Figure 5B). Similarly, meta-analysis of another 5 studies (n = 463) revealed that intraoperative image-guided spinal navigation was associated with a significant reduction in cannulation time (MD = −12.59 min, 95% CI = −19.45 to −5.74, P < 0.01, I2 = 97%; Figure 5C).
Fluoroscopy Use Frequency and Radiation Dose
Meta-analysis of 8 studies (n = 610) revealed that intraoperative image-guided spinal navigation was associated with a significant reduction in the frequency of fluoroscopy use (MD = −14.75 times, 95% CI = −24.36 to −5.13, P < 0.01, I2 = 100%; Figure 5D). Similarly, meta-analysis of 6 studies (n = 333) revealed that intraoperative image-guided spinal navigation was associated with a significant reduction in radiation exposure (SMD = −4.18, 95% CI = −5.58 to −2.78, P < 0.01, I2 = 93%; Figure 5E).
Clinical Outcomes
Length of Stay
Meta-analysis of 9 studies (n = 646) revealed that intraoperative image-guided spinal navigation was associated with a significant reduction in length of stay (MD = −0.44 days, 95% CI = −0.84 to −0.03, P = 0.03, I2 = 83%; Figure 6A). Forest Plots Demonstrating the Comparison of Clinical Outcomes Between Intraoperative Image-Guided Spinal Navigation and C-Arm Navigation in Patients Undergoing ELSS. (A) Length of Stay, (B) Back VAS Score at 1 Month, (C) Back VAS Score at 3 Months, (D) Back VAS Score at 6 Months, and (E) Back VAS Score at 12 Months. df, Degrees of Freedom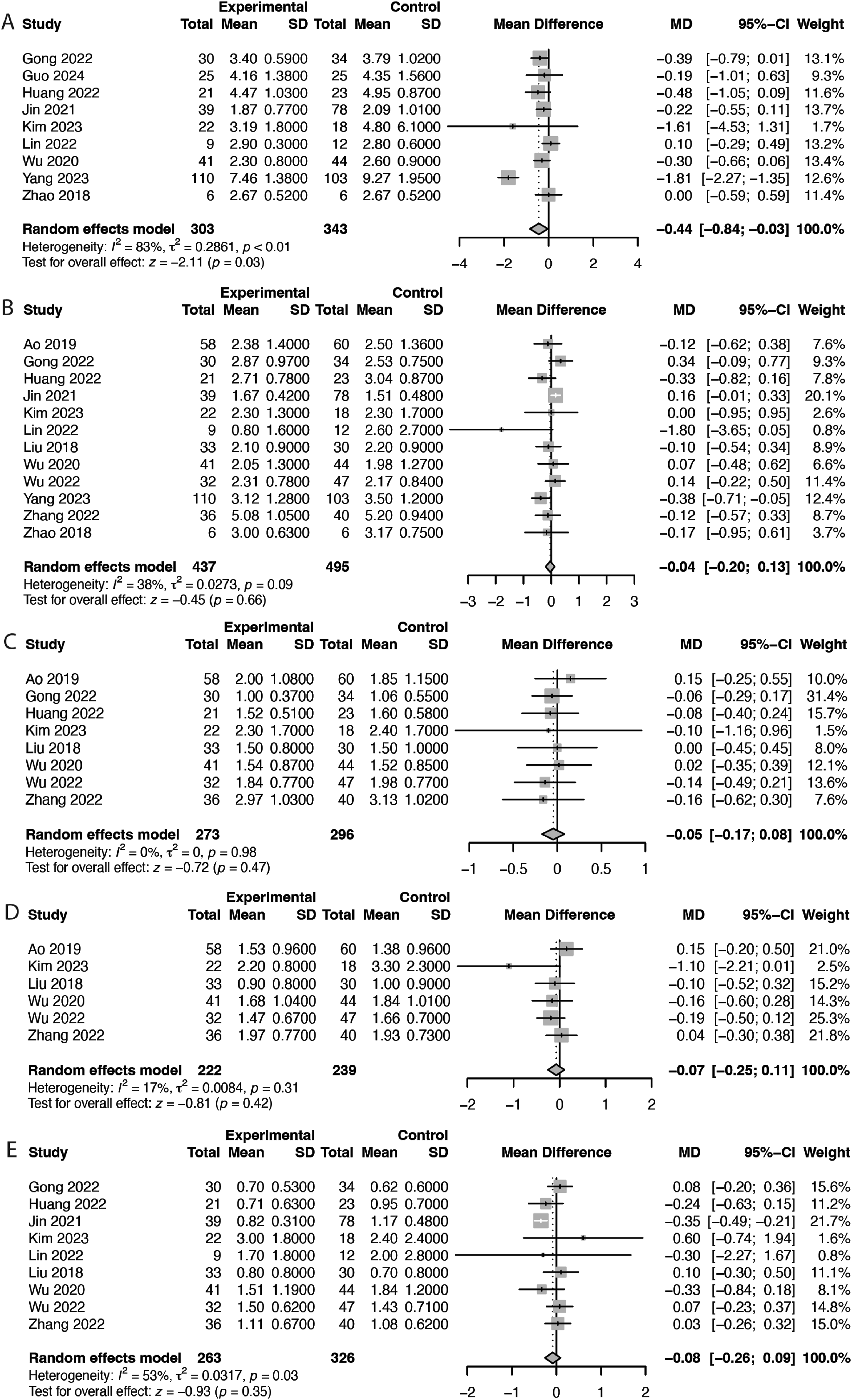
Back and Leg VAS Scores
Pooled analysis revealed no significant differences in back VAS scores at any of the following follow-up intervals: 1 month (MD = −0.04, 95% CI = −0.20 to 0.13, P = 0.66; Figure 6B), 3 months (MD = −0.05, 95% CI = −0.17 to 0.08, P = 0.47; Figure 6C), 6 months (MD = −0.07, 95% CI = −0.25 to 0.11, P = 0.42; Figure 6D), and 12 months (MD = −0.08, 95% CI = −0.26 to 0.09, P = 0.35; Figure 6E). Similarly, no significant differences in leg VAS scores were observed at any of the following follow-up intervals: 1 month (MD = 0.06, 95% CI = −0.05 to 0.17, P = 0.26; Figure 7A), 3 months (MD = 0.03, 95% CI = −0.12 to 0.17, P = 0.73; Figure 7B), 6 months (MD = 0.07, 95% CI = −0.13 to 0.27, P = 0.50; Figure 7C), and 12 months (MD = −0.02, 95% CI = −0.16 to 0.12, P = 0.80; Figure 7D). Forest Plots Demonstrating the Comparison of Clinical Outcomes Between Intraoperative Image-Guided Spinal Navigation and C-Arm Navigation in Patients Undergoing ELSS. (A) Leg VAS Score at 1 Month, (B) Leg VAS Score at 3 Months, (C) Leg VAS Score at 6 Months, and (D) Leg VAS Score at 12 Months. df, Degrees of Freedom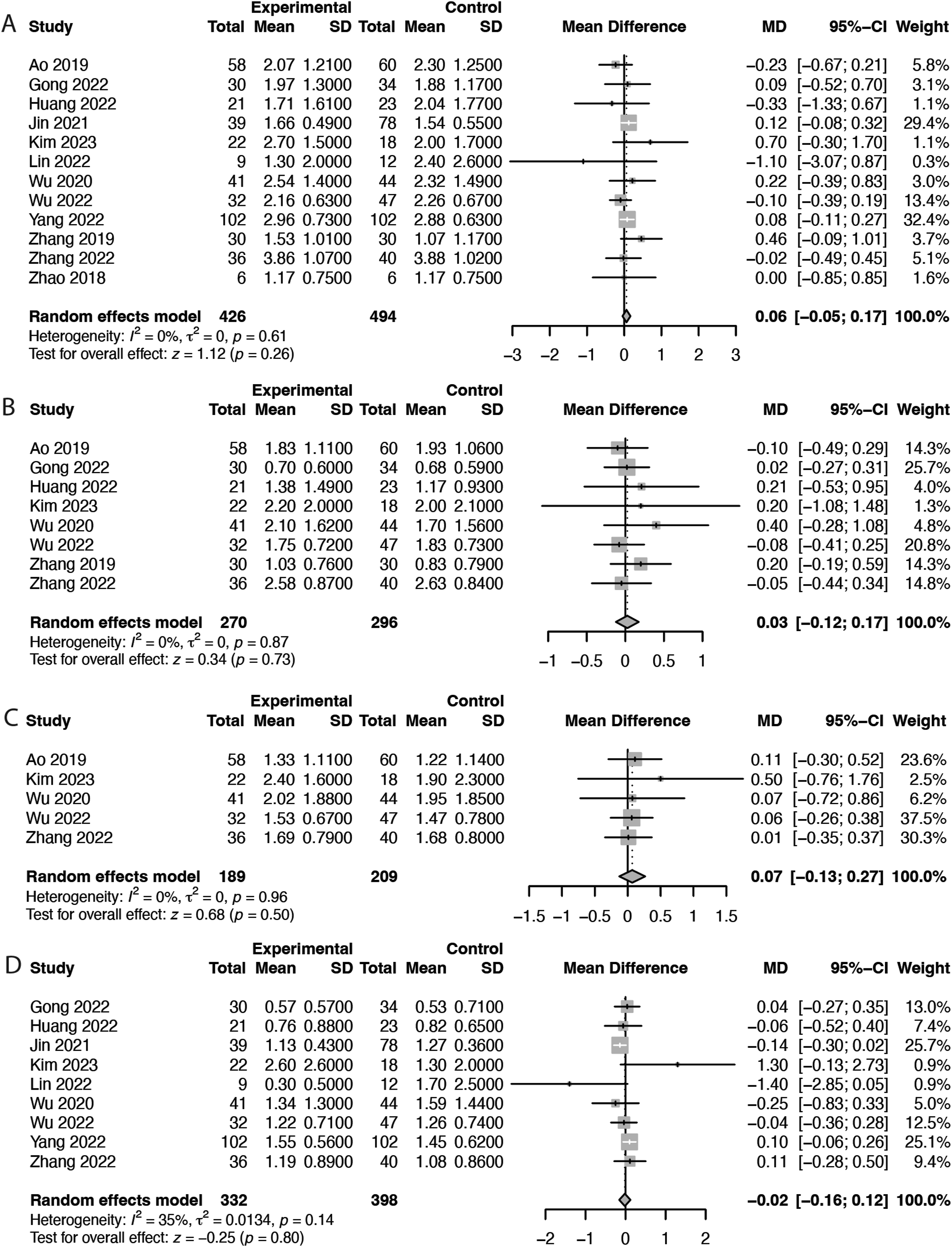
ODI
Pooled analysis revealed no significant differences in ODI scores at any of the following follow-up intervals: 1 month (MD = −0.14, 95% CI = −1.27 to 1.00, P = 0.81; Figure 8A), 3 months (MD = −0.64, 95% CI = −2.00 to 0.72, P = 0.36; Figure 8B), 6 months (MD = −0.25, 95% CI = −0.90 to 0.39, P = 0.44; Figure 8C), and 12 months (MD = −0.03, 95% CI = −0.46 to 0.39, P = 0.88; Figure 8D). Forest Plots Demonstrating the Comparison of Clinical Outcomes Between Intraoperative Image-Guided Spinal Navigation and C-Arm Navigation in Patients Undergoing ELSS. (A) ODI Score at 1 Month, (B) ODI Score at 3 Months, (C) ODI Score at 6 Months, and (D) ODI Score at 12 Months. df, Degrees of Freedom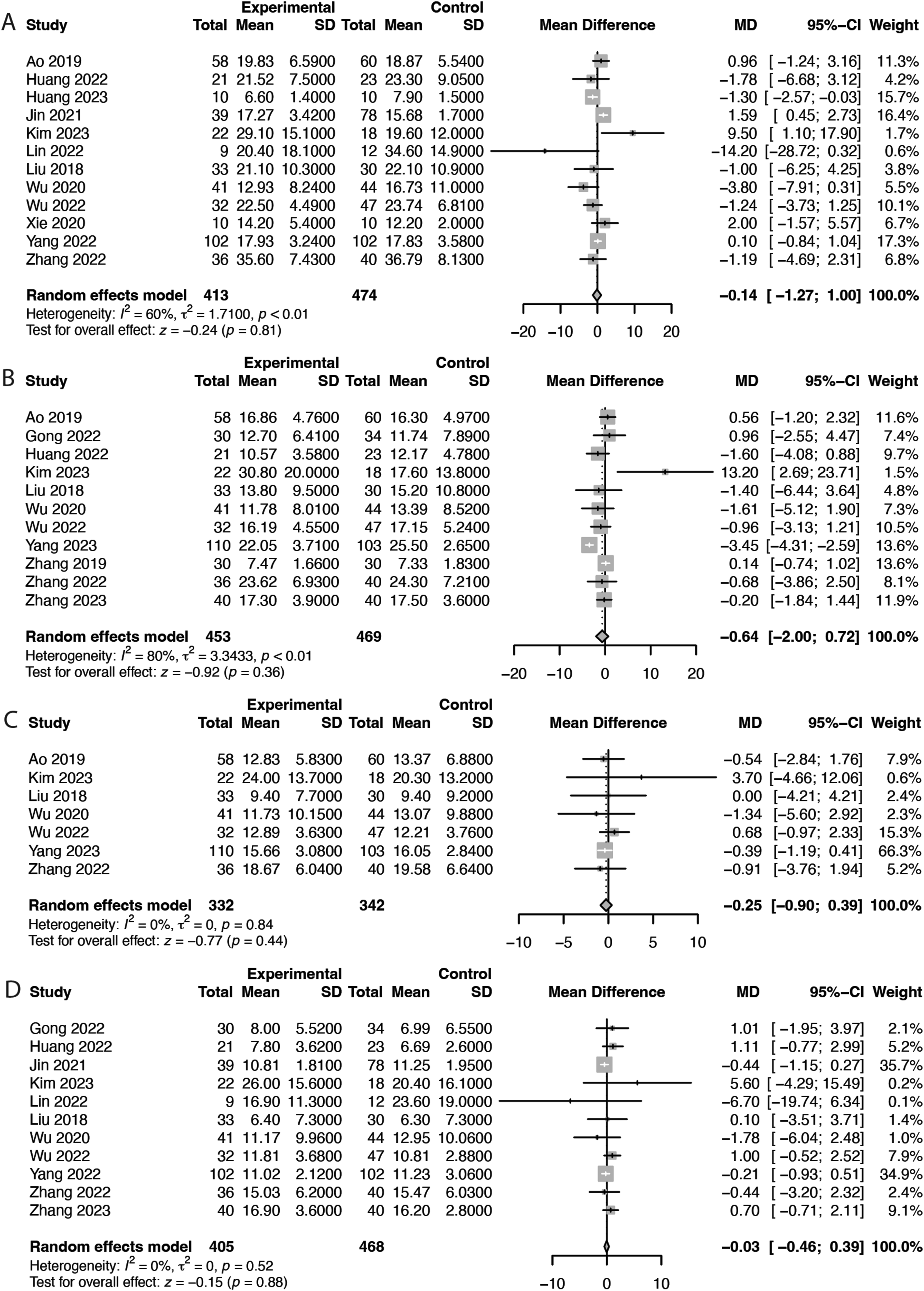
Meta-Regression
Meta-regression was conducted to identify factors that may influence the outcomes. The results indicated that length of stay was significantly influenced by age (P = 0.045) and sample size (P = 0.0137), whereas fluoroscopy use frequency was significantly influenced by the type of surgical method employed (P = 0.0009, Supplemental File 1).
Subgroup Analysis
Navigation Methods
Subgroup analysis revealed a statistically significant effect of navigation method on operation time (P < 0.01), number of puncture attempts (P < 0.01), cannulation time (P = 0.00), fluoroscopy use frequency (P < 0.01), and radiation dose (P < 0.01). Spinal navigation methods, including robotic-assisted, electromagnetic, and ultrasound volume methods, outperformed conventional C-arm navigation methods in reducing the operation time, with the greatest reduction observed with EMN (MD = −23.96 min, Figure 9A). In terms of the number of puncture attempts, ARSN, robotic-assisted navigation, O-arm navigation, and UVN outperformed conventional C-arm navigation, with greatest reduction observed with ARSN (MD = −4.9 times) and the smallest reduction observed with O-arm navigation (MD = −0.24 times, Figure 9B). Cannulation time was shorter with O-arm navigation, robotic-assisted navigation, and EMN than with C-arm navigation, with the greatest reduction observed with EMN (MD = −17.23 min) and the smallest reduction observed with robotic-assisted navigation (MD = −1.69 min, Figure 9C). The frequency of fluoroscopy use was significantly reduced across all types of spinal navigation, except with O-arm navigation, because no studies reported this outcome for O-arm navigation. The greatest reduction was observed with EMN (MD = −43.77 times, Figure 9D). Radiation exposure was significantly lower with ultrasound-guided navigation, EMN, and UVN than with conventional C-arm navigation, with the greatest reduction observed with UVN (SMD = −19.84). No significant reduction was observed with O-arm navigation (Figure 9E). In addition, no significant subgroup effect was observed for length of stay (P = 0.62, Figure 9F). Forest Plots of Perioperative and Clinical Outcomes From the Subgroup Analysis of different Navigation Types Between Intraoperative Image-Guided Spinal Navigation and C-Arm Fluoroscopy in Patients Undergoing ELSS. (A) Operation Time, (B) Number of Puncture Attempts, (C) Cannulation Time, (D) Fluoroscopy Use Frequency, (E) Radiation Dose, and (F) Length of Stay. df, Degrees of Freedom
Surgery Type
Subgroup analysis revealed significant effects on fluoroscopy use frequency (P < 0.01), with Endo-TLIF exhibiting a greater reduction (MD = −43.77 times) than that of PELD (MD = −10.29 times) compared with C-arm navigation (Figure 10D). Similarly, a significant subgroup effect was observed on radiation dose (P < 0.01), with uniportal Endo-TLIF achieving the greatest reduction (SMD = −51.50), followed by biportal Endo-TLIF (SMD = −11.20) and PELD (SMD = −11.10, Figure 10E). No significant subgroup differences were observed in operation time (P = 0.06, Figure 10A), cannulation time (P = 0.85, Figure 10C), or length of stay (P = 0.92; Figure 10F). Because only studies involving PELD reported on the number of puncture attempts, no subgroup analysis for this outcome was conducted (Figure 10B). Forest Plots of Perioperative and Clinical Outcomes From the Subgroup Analysis of different Surgery Types Between Intraoperative Image-Guided Spinal Navigation and C-Arm Fluoroscopy in Patients Undergoing ELSS. (A) Operation Time, (B) Number of Puncture Attempts, (C) Cannulation Time, (D) Fluoroscopy Use Frequency, (E) Radiation Dose, and (F) Length of Stay. df, Degrees of Freedom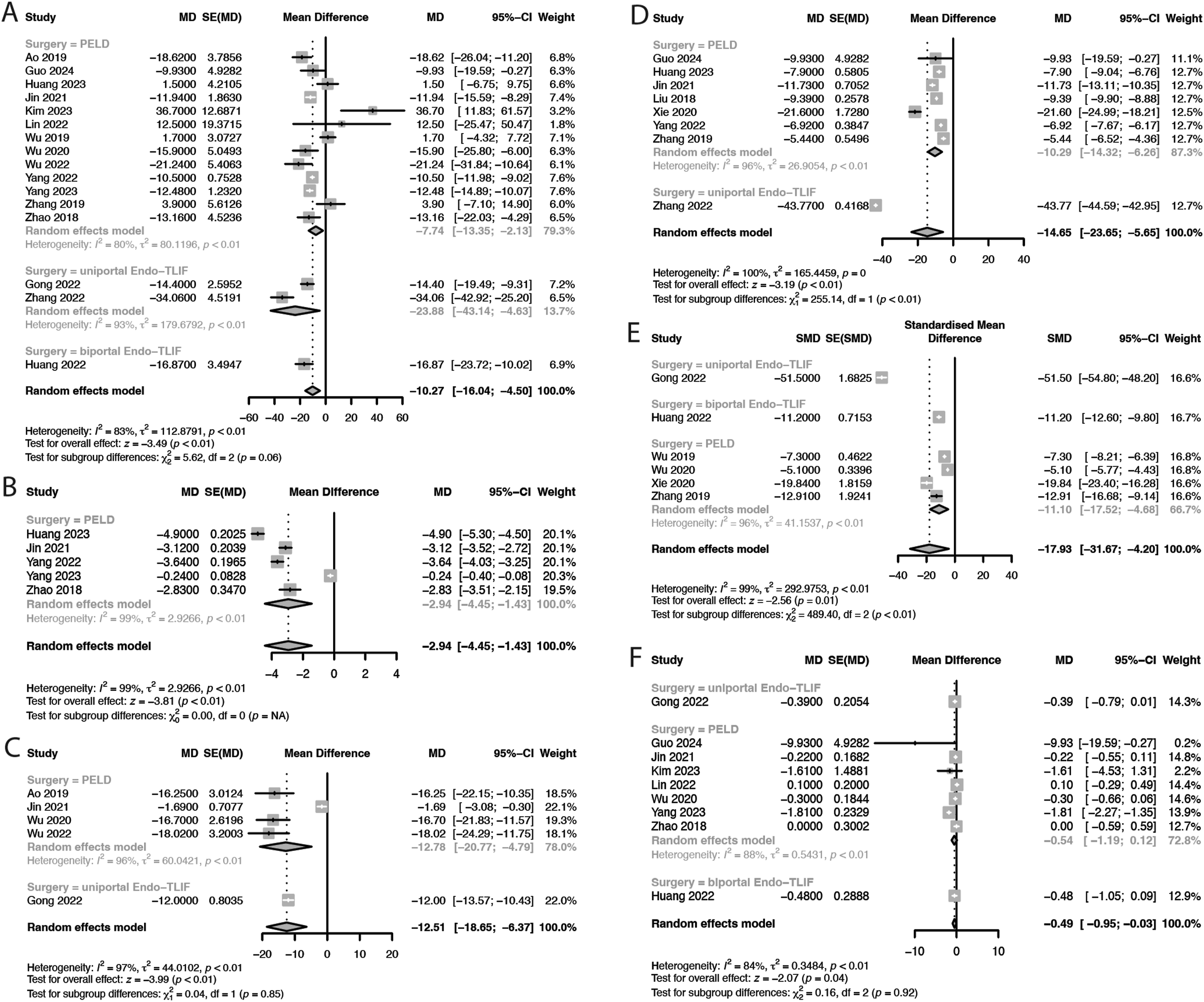
Discussion
To the best of our knowledge, this is the first meta-analysis to evaluate the effect of intraoperative image-guided spinal navigation on ELSS. Our findings indicate that image-guided navigation significantly reduces operation time, the number of puncture attempts, cannulation time, fluoroscopy use frequency, radiation exposure, and length of stay. Our postoperative clinical outcomes, including VAS and ODI scores, were comparable between the 2 groups. Meta-regression analysis revealed that length of stay was significantly influenced by patient age and sample size, whereas fluoroscopy use frequency was influenced by the surgical method employed. Subgroup analysis by navigation type revealed EMN and UVN as providing the greatest perioperative benefits. By contrast, subgroup analysis by surgery type revealed a greater reduction in fluoroscopy use frequency and radiation dose with Endo-TLIF than with PELD.
The transforaminal approach to the L5–S1 level is considered technically challenging due to the high iliac crest, narrow foraminal space, and obstructing anatomy such as a large L5 transverse process or hypertrophic facet joints, which may influence perioperative outcomes.31-33 In our meta-analysis, most included studies involved L5–S1 cases, but none reported significant differences in disc level distribution between the navigation and control groups. Thus, the overall results are unlikely to be biased by surgical level. Nonetheless, future studies should further investigate the impact of different lumbar levels on surgical outcomes.
Only 2 studies have evaluated the learning curve associated with ELSS with intraoperative navigation and conventional C-arm navigation. Ao et al 20 used a regression model with operation time as the dependent variable and case number as the independent variable. Their findings revealed a significantly steeper learning curve for ELSS with conventional C-arm navigation than with O-arm navigation, with surgical proficiency achieved in case 13 for O-arm navigation and case 32 for C-arm navigation. Similarly, Wu et al 34 reported a more favorable learning curve with EMN than with C-arm navigation. However, because of the limited number of available studies on this topic, a comparative analysis of learning curves for different navigation methods was not feasible in this meta-analysis. Nonetheless, the observed reductions in operation time, number of puncture attempts, and cannulation time associated with intraoperative navigation suggest its potential to accelerate the learning process of ELSS.
Intraoperative navigation systems can broadly be classified into 2 primary categories: optical and electromagnetic. 18 Optical systems, which are used in intraoperative O-arm and robot-assisted navigation, rely on infrared tracking cameras for guidance. By contrast, electromagnetic systems involve specialized sensors that can be detectable when moving in an electromagnetic field. 35 Studies indicate that EMN offers substantial advantages over optical systems by eliminating line-of-sight restrictions through its tissue-penetrating field. This capacity enables seamless device registration, thereby eliminating the need for re-registration and reducing operation time.36,37 Our subgroup analyses corroborate these findings, demonstrating that electromagnetic systems generally outperform optical systems in terms of perioperative outcomes, particularly by reducing total operation and cannulation time. UVN, which combines real-time ultrasound with preoperative CT or MRI data and employs electromagnetic guidance, demonstrated comparable results. UVN not only enhances navigation but also provides greater radiation dose reduction compared with standard EMN, which remains a major concern in navigation-assisted endoscopic procedures.
Utilizing robotic mechanical arms to autonomously locate puncture points along the planned surgical path may improve the outcomes of optical navigation. Robotic arms can help reduce surgeon fatigue by maintaining steady positioning during surgery, which may be particularly helpful during the initial steps of the surgery. Integrating AR headsets into optical navigation systems may further enhance these outcomes by enhancing surgeon ergonomics and projecting navigation data directly to the surgeon’s field of view over the patient’s body. 18 These innovations are expansion of computer-assisted navigation, with navigation as the fundamental building block that both robotics and AR builds upon. These innovations expand the capabilities of computer-assisted navigation. In contrast, real-time ultrasound guidance without computer-assisted navigation can only reduce radiation exposure.
In this study, pooled analysis revealed a significant reduction in hospital stay duration for patients in the navigation group, potentially indicating improved clinical outcomes or fewer complications associated with navigation-assisted surgery. However, direct clinical outcomes were not available for analysis. While all included studies applied consistent protocols across comparison groups, this finding may still be influenced by confounding factors such as differences in surgical practices, healthcare systems, and cultural norms regarding postoperative care. Further high-quality studies are needed to strengthen the evidence supporting our findings.
Intraoperative navigation serves as a key tool for enhancing surgical precision and accuracy, enabling surgeons to more effectively target anatomical structures. This precision may assist surgeons in achieving the goals of surgery more effectively by addressing symptomatic pathology and mitigating the risks of incomplete decompression. Despite these theoretical advantages, our pooled analysis revealed no significant differences in ODI scores at any of the 4 follow-up intervals until 12 months after treatment. This finding suggests that the theoretical advantages of navigation in improving perioperative metrics may not yet translate into meaningful improvements in long-term patient-reported clinical outcomes.
This meta-analysis has several limitations that should be acknowledged. First, our subgroup analyses revealed notable differences between various navigation systems and surgical procedures. However, the lack of comprehensive perioperative data across the included studies resulted in smaller sample sizes for each subgroup analysis. This limitation may have reduced the statistical power of our findings and limited our ability to draw robust conclusions regarding the comparative effectiveness of specific navigation technologies or surgical techniques.
Second, although intraoperative image-guided navigation demonstrated clear advantages in terms of perioperative outcomes, these benefits did not consistently translate into statistically significant improvements in the majority of clinical outcomes when compared with conventional C-arm navigation. Therefore, adopting advanced technologies such as navigation, robotics, and AR on a wider scale requires careful consideration of the cost–benefit ratio and may necessitate case-specific evaluation.
Third, the financial burden associated with these technologies is substantial, particularly given the limited reimbursement models available in many regions of the world. Many studies included in this meta-analysis were conducted in academic centers with access to advanced navigation technologies, which may not be available in hospitals with limited resources. Additionally, certain surgeons may have additional expertise with navigation and may be adept at leveraging its advantages, potentially influencing their operation time.
Fourth, most of the included studies were retrospective in nature, introducing an inherent bias influenced by the authors’ preferences.
Lastly, although intraoperative navigation involves substantial equipment costs, none of the included studies reported on the cost–benefit aspects of these technologies, thereby precluding a formal analysis of the financial implications. To address this gap, future research should evaluate the cost-effectiveness of various intraoperative navigation methods.
Conclusion
Intraoperative image-guided spinal navigation in ELSS significantly affects perioperative outcomes by reducing total operation time and radiation exposure for both patients and surgical staff. It also facilitates the learning process of ELSS by reducing the number of puncture attempts and cannulation time. Overall, postoperative clinical outcomes are not affected by the type of navigation. However, to comprehensively evaluate the cost-effectiveness of intraoperative navigation, additional data are necessary to compare its impact across various health-care systems.
Supplemental Material
Supplemental Material - Effect of Intraoperative Image-Guided Spinal Navigation Technologies on Endoscopic Lumbar Spine Surgery: A Systematic Review and Meta-Analysis
Supplemental Material for Effect of Intraoperative Image-Guided Spinal Navigation Technologies on Endoscopic Lumbar Spine Surgery: A Systematic Review and Meta-Analysis by Yu-Che Wang, Hsu-I Chou, Ying-Fong Su, Rafael Garcia de Oliveira, Abhinav K. Sharma, Yang-Ching Chen, Anh Tuan Bui, Ching-Yu Lee, Jowy Tani, Cheng-Chun Chang, Takaki Yoshimizu, Wongthawat Liawrungrueang, Tsung-Jen Huang, Daisuke Sakai, Klaus John Schnake, Jason Pui Yin Cheung, MBBS, Don Young Park, and Meng-Huang Wu Global Spine Journal
Footnotes
Acknowledgments
The authors acknowledge RIWOspine’s provision of video and illustration for the Electromagnetic Navigation System Demonstration.
ORCID iDs
Ethical Considerations
As this article does not contain studies involving human participants or animals conducted by any of the authors, ethical approval and informed consent were not required.
Author Contributions
Conceptualization: Yu-Che Wang, Hsu-I Chou, Ying-Fong Su, Rafael Garcia de Oliveira, Abhinav K. Sharma, Ching-Yu Lee, Don Young Park, Meng-Huang Wu. Data Curation: Yu-Che Wang, Hsu-I Chou, Formal Analysis: Yu-Che Wang, Hsu-I Chou, Anh Tuan Bui, Cheng-Chun Chang. Methodology: Anh Tuan Bui, Jowy Tani, Don Young Park, Meng-Huang Wu. Software: Yang-Ching Chen, Cheng-Chun Chang. Visualization: Takaki Yoshimizu, Wongthawat Liawrungrueang, Tsung-Jen Huang, Daisuke Sakai, Klaus John Schnake, Jason Pui Yin Cheung, Don Young Park, Meng-Huang Wu. Project Administration: Don Young Park, Meng-Huang Wu. Writing – Original Draft: Yu-Che Wang, Hsu-I Chou, Ying-Fong Su, Rafael Garcia de Oliveira, Abhinav K. Sharma. Writing – Review & Editing: Yang-Ching Chen, Anh Tuan Bui, Ching-Yu Lee, Jowy Tani, Cheng-Chun Chang, Takaki Yoshimizu, Wongthawat Liawrungrueang, Tsung-Jen Huang, Daisuke Sakai, Klaus John Schnake, Jason Pui Yin Cheung, Don Young Park, Meng-Huang Wu.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information. Data may be available upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
