Abstract
Study Design
Systematic Review and Meta-Analysis.
Objectives
To compare clinical outcomes in degenerative cervical myelopathy (DCM) patients treated with laminectomy alone (LA) vs laminectomy with fusion (LF) and determine post-laminectomy kyphosis incidence.
Methods
We searched PubMed, Scopus, Embase, Web of Science, and MEDLINE from inception-September 2024 for studies comparing LA and LF for DCM. Outcomes assessed included post-laminectomy kyphosis, neurological recovery outcomes, patient-reported outcomes (PROs), and complication rates. Meta-analyses were performed using random-effects models.
Results
Twenty-seven studies including 3286 patients (2272 LA and 1014 LF), met the inclusion criteria. The pooled incidence of post-laminectomy kyphosis in the LA patients was 2.02 events per 100 person-years (95% CI: 1.26-2.78). Post-laminectomy kyphosis declined from 3.67 cases per 100 person-years in pre-2004 studies, to 0.88 cases per 100 person-years in post-2014 studies. No significant differences were observed between LA and LF in neurologic recovery (SMD 0.29, 95% CI 0.02-0.59), pain score improvement (SMD 0.13, 95% CI: −0.38 to 0.64) and complications (OR 0.77, 95 % CI 0.29-2.08), although subgroup analysis demonstrated that the risk of complications in LA may be lower in patients with less than four operative levels.
Conclusions
Although the annual rate of kyphosis after LA is approximately 2 events per 100 person-years, PROs and complication rates may be similar between LA and LF. These findings should be interpreted with the caveat of considerable heterogeneity between studies and further randomized trials are needed to better delineate the benefits of each approach and to optimize patient selection.
Keywords
Introduction
Degenerative cervical myelopathy (DCM) is the leading cause of non-traumatic spinal cord dysfunction in adults worldwide, arising from congenital and age-related degenerative changes that result in compression of the cervical spinal cord. 1 Surgical intervention is the mainstay of treatment for moderate to severe DCM, aiming to decompress the spinal cord and prevent further neurological deterioration. 2 Surgical intervention can be broadly categorized as anterior or posterior approaches, each of which is tailored to address specific patterns of pathology and patient anatomy. While surgical approach decision making can be nuanced, patients with multilevel disease, ossification of the posterior longitudinal ligament or predominant dorsal compression may be good candidates for a posterior approach.3-5
Among posterior surgical techniques, laminectomy alone (LA) and laminectomy with fusion (LF) are widely utilized. LA involves the removal of the lamina to decompress the spinal cord without additional stabilization. Posterior approaches without instrumented fusion offer several advantages, including shorter operative time, reduced blood loss, and preservation of cervical motion segments, which may be beneficial for postoperative range of motion and reduced post-operative pain.6-9 Due to the lack of instrumentation, LA tends to have a reduced cost while also mitigating the potential complications associated with spinal hardware, such as infection, malposition or hardware failure. However, laminectomy in the absence of fusion has been associated with postoperative complications including progressive kyphotic deformity and spinal instability due to the disruption of the posterior tension band.10,11
Despite multiple studies examining the outcomes of these surgical techniques in isolation, there are fewer studies directly comparing LA vs LF in DCM and most of them are observational in nature. 12 Nevertheless, both techniques remain prevalent around the world with limited consensus regarding superiority in well-selected patients. 13 This meta-analysis aims to compare LA and LF for the treatment of DCM. Our specific aims include assessing neurological recovery, patient-reported outcomes (PROs) and complication rates between both procedures and to establish the incidence of kyphosis after LA. Understanding the relative benefits and drawbacks of LA and LF is crucial for optimizing patient outcomes, minimizing complications, and improving surgical decision-making for DCM.
Methods
Study Design
This systematic review and meta-analysis was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 14 The primary study objective was to compare clinical and radiographic outcomes between patients treated with LA compared to those treated with LF for DCM.
Search Strategy
We searched PubMed, Scopus, Embase, Web of Science, and MEDLINE to identify relevant studies published from database inception up to and including September 2024. The search targeted randomized controlled trials, prospective cohort studies, and retrospective cohort studies that compared the outcomes of DCM patients with LA vs LF or studies of LA that reported post-operative kyphosis.
The search strategy utilized a combination of keywords with Boolean operators and Medical Subject Headings (MeSH) terms within article titles, abstracts and keywords related to DCM, surgical interventions, and clinical outcomes. Key terms included “degenerative cervical myelopathy”, “laminectomy,” “posterior decompression,” “with fusion,” and “without fusion” and variations thereof. Articles involving laminoplasty were excluded. Outcome measures searched for included “surgical outcomes,” “functional outcomes,” “quality of life,” “complications,” “kyphosis,” “sagittal balance,” “reoperation,” “revision,” and specific assessment tools such as the Japanese Orthopaedic Association (JOA) score, modified JOA (mJOA) score, Neck Disability Index (NDI), EuroQol- 5 Dimension score (EQ-5D), Short Form Health Survey (SF-36), visual analogue scale (VAS) and numeric rating scale for pain (NRS). Reference lists of retrieved articles were manually screened to identify any further studies that met the inclusion criteria but were not captured in the initial database search. A sample search string is demonstrated in Supplemental Table 1.
Inclusion and Exclusion Criteria
Studies were included if they involved adult patients aged 18 years or older diagnosed with DCM. Studies involving patients with oncologic or traumatic pathologies were excluded. To maintain focus on posterior surgical procedures, only studies examining LA and LF as the primary interventions were considered; studies involving anterior or combined anterior-posterior surgical approaches or those involving laminoplasty were excluded. Eligible studies either directly compared LA with LF or reported on postoperative kyphosis outcomes in the LA group. Inclusion required that studies reported at least one of the following outcomes: postoperative kyphosis, clinical outcomes assessed by the mJOA score or JOA score, the NDI, VAS, NRS, complication rates, or quality of life measures such as the SF-36 or EQ-5D questionnaire. There were no myelopathy severity inclusion criteria applied to study selection. We excluded studies reporting less than 5 patients.
Data Extraction
Initial abstract screening was carried out by a single reviewer (CSL). Full texts were reviewed independently by two reviewers (CSL and AKM), with discrepancies resolved through consensus. Among included articles, information was extracted on the study design, sample size, interventions, baseline demographics, follow-up duration, and outcomes.
Risk of Bias Assessment
The methodological quality of the included studies was assessed by two authors (CSL and NB) using the Newcastle-Ottawa Scale (NOS) for observational studies 15 and RCTs were evaluated using the Cochrane Risk of Bias (RoB 2) tool. 16
Outcome Measures
In order to establish the incidence of post laminectomy kyphosis, we pooled data from both dedicated LA series and the laminectomy arms of comparative LA-versus-LF studies, provided that each study reported the number of patients who developed kyphosis in the LA group. Kyphosis definitions varied across studies; some used a C2–C7 Cobb angle less than 0°, others relied on loss of lordosis measured with the cervical curvature index or other quantitative and qualitative methodologies. Rather than exclude studies based on these methodological differences, we accepted each author’s binary classification of kyphosis (“present” vs “absent”) and pooled the number of counts, adjusting for average follow up time and number of subjects in each study, to generate an incidence expressed as number of kyphosis events per person-year. In order to better assess heterogeneity between studies, we performed subgroup analysis in which we stratified studies by average number of laminectomy levels, age of patients and study publication year.
Neurological recovery was reported with four different instruments: the mJOA, the JOA, the Nurick grade, and the European Myelopathy Scale (EMS). Both JOA instruments quantify motor, sensory, and sphincter function with scores that are summed (to a total of 17 or 18 points). 17 Higher values indicate less neurological impairment. The Nurick grade is a six-point scale (0-5) that focuses on ambulatory function, with higher grades reflecting greater disability. The EMS rates gait, hand function, proprioception, paresthesias and bladder function, with scores ranging from 5-18 with higher scores denoting greater disability. Previous validation studies have shown that recovery rates derived from these scales correlate closely, permitting meaningful cross-scale comparisons of neurologic recovery in DCM. 18
PROs encompassed both neck‐specific and general health measures. Neck‐specific disability was most commonly assessed with the NDI, a 10‐item questionnaire scored from 0-50 (or sometimes reported on a 0-100 scale), where higher values indicate greater disability. Pain intensity was recorded using a VAS, typically ranging from 0 (no pain) to 10 or 100 (worst pain). General health‐related quality of life was evaluated with the SF-36 Physical Component Summary (PCS) and Mental Component Summary (MCS), as well as the EuroQol-5D (EQ-5D). The SF-36 PCS measures physical health domains such as physical functioning, bodily pain, and role limitations, while the SF-36 MCS addresses mental health domains, including emotional well-being, social functioning, energy, and general mental health; higher scores on both indicate better health status. The EQ-5D rates quality of life across five domains (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) on a 0-1 scale, with values closer to 1 reflecting greater overall health.
We also examined overall complication rates across studies. Because reporting varied with some papers listing each complication type (e.g. C5 palsy, wound infection, hematoma), while others provided only a single overall count, we extracted the total number of patients who experienced at least one complication in each treatment arm. When individual complication categories were available, we summed them to obtain a single overall count and when only a summary figure was provided, we used that directly. We generated a pooled odds ratio (OR) of complications in LA vs LF. To explore heterogeneity, we sub grouped studies by average number of laminectomy levels (≤ 4 vs > 4) and by follow‐up duration (≤ 12 months vs > 12 months).
Statistical Analysis
Because our included studies reported outcomes using a variety of scales, it was not feasible to pool raw mean differences since each instrument has a different scoring range. Instead, we used the standardized mean difference (SMD), also known as Hedges’ g, which rescales each study’s treatment‐group difference (LF vs LA) by its own within‐group standard deviation. This approach is recommended whenever outcomes are measured with different instruments but are intended to capture the same underlying construct because it places all studies on a common, unitless scale.19,20 Meta-analysis was performed using random‐effects pooling with Hartung–Knapp–Sidik–Jonkman adjustment, which is recommended when the number of studies is small. 21 We classified heterogeneity as low, moderate, and high to I2 values of 25%, 50%, and 75%, respectively, as suggested by Higgins et al. 22
Data synthesis and meta-analyses were performed using R Statistical Programming Software version 4.2.1 with the meta library package. 23 Forest plots were generated to visualize the effect sizes of individual studies and their pooled results with SMDs for continuous variables and odd ratios for categorical variables.
Results
Search Results
Our search identified 2967 studies. Study selection is depicted in Supplemental Figure 1. After the removal of 1215 duplicates, 1754 studies had abstracts screened, of which 42 studies were identified for full text assessment. Of these, 27 studies met the inclusion criteria for the meta-analysis.24-50 Overall, the risk of bias among the included studies was found to be low to moderate (Supplemental Table 1).
Study Characteristics
Summary of Studies Included in Systematic Review and Meta-Analysis for Laminectomy Alone Cohorts and Comparative Studies of Laminectomy Alone Versus Laminectomy With Fusion
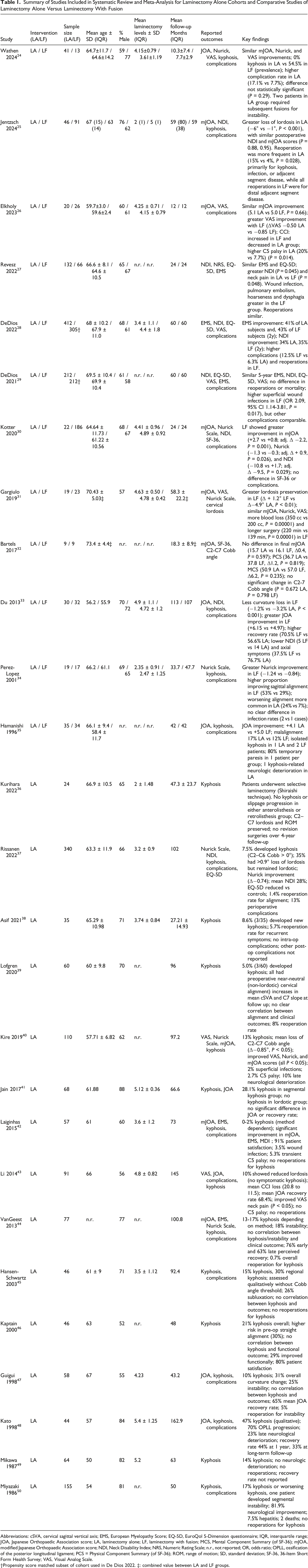
Abbreviations: cSVA, cervical sagittal vertical axis; EMS, European Myelopathy Score; EQ-5D, EuroQol 5-Dimension questionnaire; IQR, interquartile range; JOA, Japanese Orthopaedic Association score; LA, laminectomy alone; LF, laminectomy with fusion; MCS, Mental Component Summary (of SF-36); mJOA, modified Japanese Orthopaedic Association score; NDI, Neck Disability Index; NRS, Numeric Rating Scale; n.r., not reported; OR, odds ratio; OPLL, ossification of the posterior longitudinal ligament; PCS = Physical Component Summary (of SF-36); ROM, range of motion; SD, standard deviation; SF-36, 36-Item Short Form Health Survey; VAS, Visual Analog Scale.
†Propensity score matched subset of cohort used in De Dios 2022. ‡: combined value between LA and LF groups.
Post-laminectomy Kyphosis
Across 19 laminectomy-only cohorts (8214 person-years of follow-up), 140 patients developed new postoperative kyphosis, yielding a pooled random effects incidence of 2.02 cases per 100 person-years (95 % CI 1.26-2.78, Figure 1) with moderate to high heterogeneity (I2 = 73.3). Pooled Incidence of Post-laminectomy Kyphosis Events per Person Year, Stratified by Publication Era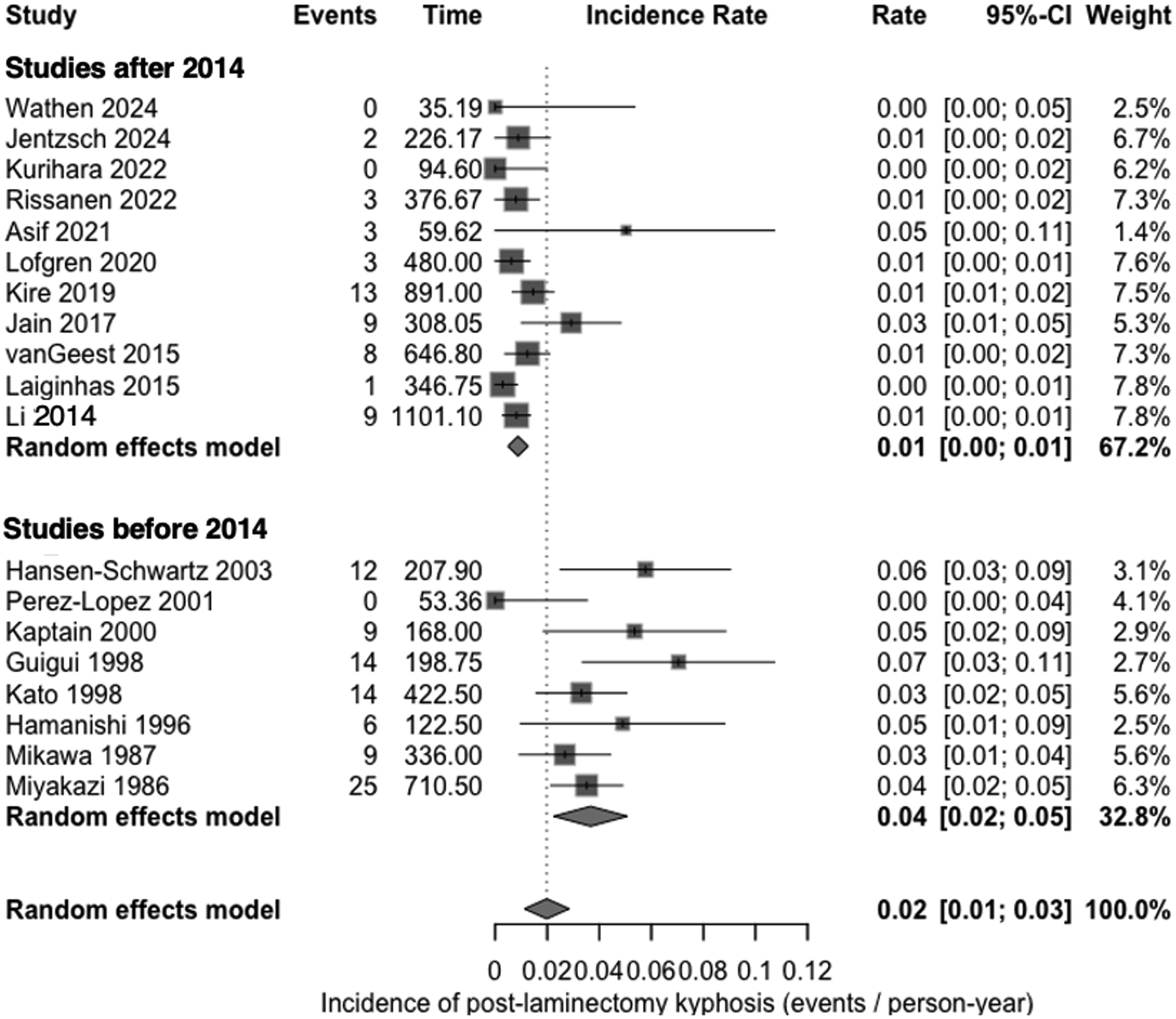
To better explore heterogeneity, we performed subgroup analysis based on average number of operative levels, average patient age and publication era. When studies with an average of four or more levels were compared with those averaging fewer than four, the incidence was increased to 2.40 vs 0.81 cases per 100 patient years (χ21 = 5.77, P = .016). Stratification by cohort mean age suggested lower risk in cohorts aged ≥ 65 years (1.03 vs 2.17 cases per 100 patient years for < 65 years), although this difference did not reach significance (χ21 = 3.60, P = .06). Studies published before 2004 resulted in a pooled incidence of 3.67 cases per 100 person-years (95 % CI 2.26-5.08), whereas those published after 2014 reported only 0.88 cases per 100 person-years (95 % CI 0.49-1.27); the between-era difference was significant (χ21 = 20.1, P < .001).
Neurologic Recovery in LF vs LA
Ten effect estimates from seven studies (n = 983 total patients: 602 in LF, 381 in LA) were included in the meta‐analysis of neurologic recovery. Five studies (Jentzch 2024, Bartels 2017, De Dios 2021, De Dios 2022 and Perez-Lopez) could not be included in the neurologic function meta-analysis because insufficient reporting of pre- or post-operative data precluded calculation of the SMD. Among included studies, four reported changes in mJOA (Wathen 2024, Elkholy 2023, Kotter 2020, Gargiulo 2019); three studies reported Nurick grade changes (Wathen 2024, Kotter 2020, Gargiulo 2019); two studies reported JOA changes (Du 2014, Hamanishi 1996); and one study reported EMS change (Revesz 2022).
When all ten observations were combined the overall pooled SMD was 0.29 (95 % CI: −0.02 to 0.59, P = .061), favouring LF, but not reaching statistical significance (Figure 2A). Between‐study heterogeneity was moderate ( I2 = 62.6 %). In subgroup analyses for individual scales, the pooled SMD for mJOA was 0.21 (95 % CI: −0.28 to 0.70; I2 = 22.6 %), 0.78 (95 % CI: −4.77 to 6.32, I2 = 82.1%) for the JOA and 0.21 (95 % CI: −0.86 to 1.28; I2 = 60.2 %) for the Nurick scale. A test for subgroup differences did not reach statistical significance (Q = 3.27, P = 0.352). (A) Meta-Analysis of Neurologic Improvement with Subgrouping by Measurement Instrument and Overall Pooling. (B) Meta-Analysis of Neck Pain Improvement with Subgrouping by Measurement Instrument and Overall Pooling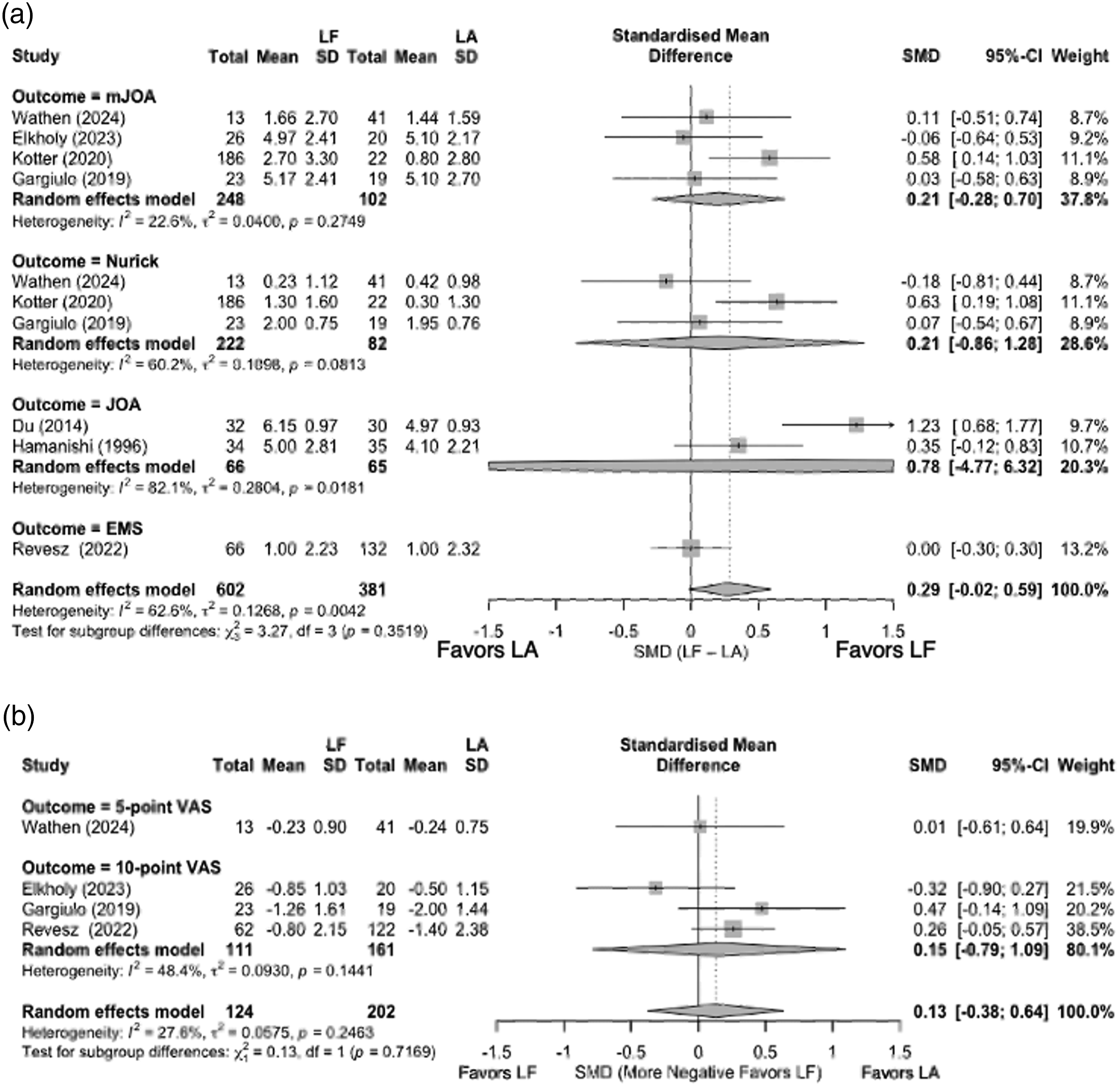
Patient Reported Outcome Measures
Four studies had sufficient reporting of data regarding neck pain score improvement (n = 316 total patients: 111 in LF, 205 in LA) and thus were included in a meta‐analysis (Figure 2B). One study used a 5‐point VAS (Wathen 2024), and three studies used a 10‐point VAS (Elkholy 2023, Gargiulo 2019, Revesz 2022). The overall random-effects pooled SMD was 0.13 (95 % CI: −0.38 to 0.64; P = .61), indicating a small, difference in pain reduction favouring LA, but not reaching statistical significance (Figure 2B). Between‐study heterogeneity was low to moderate (I2 = 27.6 %). The test for subgroup differences between the 5‐point and 10‐point VAS formats was not significant (χ21 = 0.13, P = .72).
Six studies measured NDI, however only three studies had sufficient reporting of data to calculate SMDs (Kotter 2020, Revesz 2022 and De Dios 2021). The remaining studies (Jentzch 2024, De Dios 2022 and Du 2013), did not report unadjusted pre and/or follow up means and standard deviations required for the pooled SMD calculation. Given the small number of studies with sufficient data, a formal meta-analysis was not performed, however in summary, Kotter et al and Du et al both found greater NDI improvements in the LF group, whereas Jentzch et al observed higher (worse) postoperative NDI scores in the LF group (without adjusting for baseline scores). Finally, Reversz et al and De Dios et al reported comparable improvements in the LA and LF groups.
Health-related quality of life outcomes assessed by the SF-36 PCS and MCS and EQ-5D were infrequently reported and varied in the degree of available pre- and post-operative data, precluding formal meta-analysis. In studies where these outcomes were available, both surgical strategies demonstrated improvements from baseline, with no clear advantage observed for either approach.
Complications
Among six comparative studies (646 patients in the laminectomy-alone groups and 613 in the laminectomy-with-fusion groups), 223 complications were recorded. The pooled random-effects model showed no statistically significant difference in overall complication between procedures (OR 0.77, 95 % CI 0.29-2.08; P = .53; I2 = 60.8 %, Figure 3). Meta-Analysis of Complications in LA Versus LF Stratified by Studies with an Average of ≤4 Operative Levels and >4 Operative Levels and Overall Pooling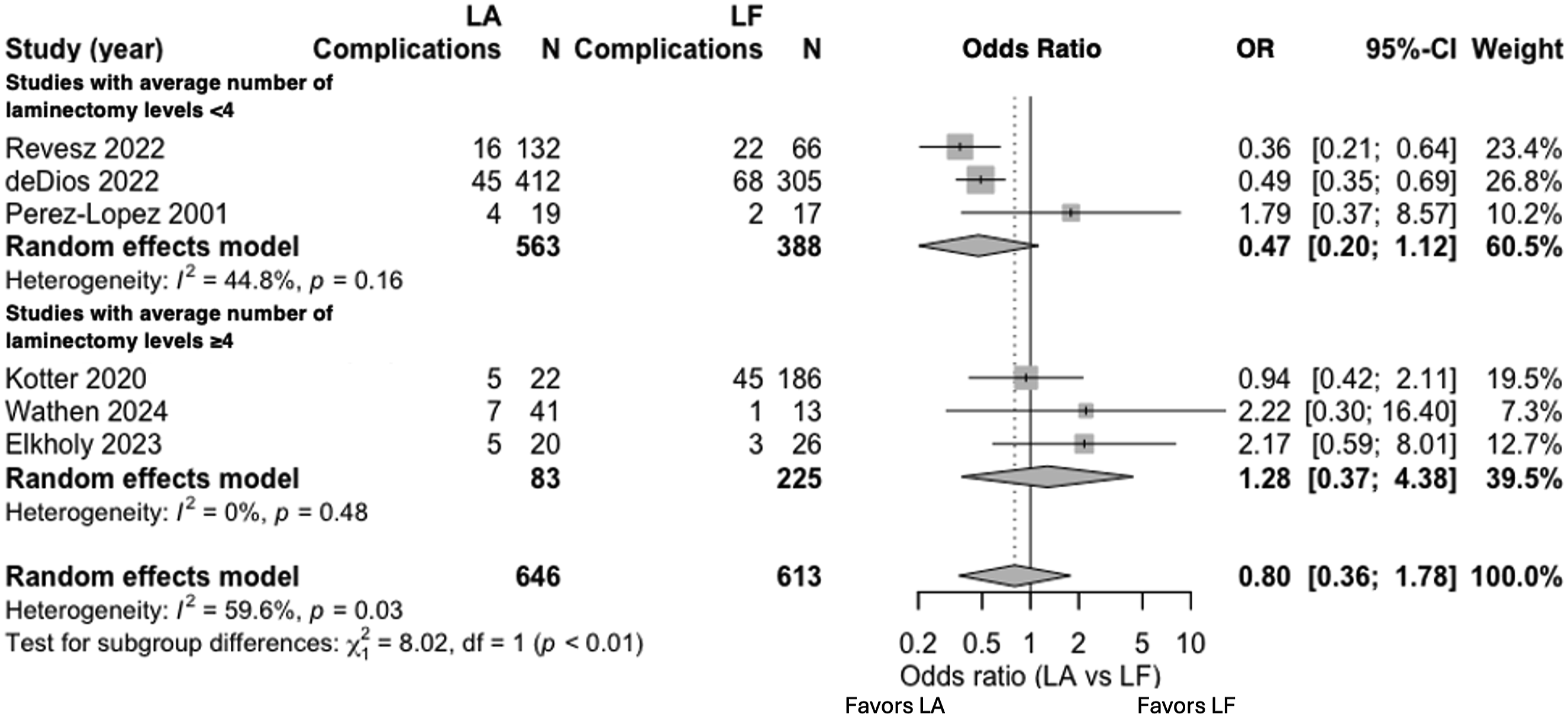
Studies were then grouped by the mean number of decompressed levels in the LA arm. In the three cohorts with ≤ 4 operated levels the pooled OR was 0.41 (95 % CI 0.14-1.22) whereas in the three cohorts with > 4 operated levels the pooled OR was 1.38 (95 % CI 0.31-6.24). A test for subgroup differences indicated that these two pooled estimates differ (χ2 = 7.97, P = .005).
When the six studies were stratified by average follow-up duration (≤ 12 months, n = 4; > 12 months, n = 2), no significant difference was found between the strata (χ2 = 0.15, P = .70).
Discussion
The objective of the present study was to establish the incidence of post-laminectomy kyphosis and to compare outcomes between LA and LF. We found that, although LA carries an approximate 2 events per 100 person-year risk of kyphosis (with lower rates in more recent series), there was no significant difference between LA and LF in terms of neurologic recovery (mJOA, JOA, Nurick, EMS), neck pain improvement (VAS), or overall complication rate. Stratified analysis suggested that when fewer than four levels are decompressed, the incidence of kyphosis is reduced significantly. In addition to multiple decompression levels, other known risk factors for post laminectomy kyphosis include preoperative loss of physiologic lordosis (i.e. a straight or kyphotic cervical alignment), removal of more than 50% of the facet joints (which disrupts the posterior tension band), evidence of preoperative instability on flexion–extension films, and younger patient age (especially in children/adolescents).6,47,51,52 Thus in patients with these clinical features or patients requiring long multi-segment decompressions, LF could mitigate kyphosis risk. Conversely, patients requiring only short segment decompressions or patients with preserved lordosis, minimal instability, and significant comorbidities could be suitable candidates for LA, especially if their life expectancy is less than the many years that it may take for clinically significant kyphosis to develop.
In our review of both comparative LA vs LF studies and standalone LA cohorts, the range of post-LA kyphosis ranged between 0% to 47%. The wide range reported can likely be attributed, at least in part, varying follow up periods across studies, spanning from 10 months to over 13 years. The temporal trend we observed in reduced rates of kyphosis in more recent studies is likely attributable to awareness for improved patient selection and operative technique. Starting in the late 1980 and 1990s, cadaveric studies demonstrated the biomechanical alterations induced by multilevel cervical laminectomies and the destabilizing effects of foraminotomies and partial facetectomies.53-56 The importance of patient selection, in particular, preoperative alignment was also described. For instance, Kaptain et al. in 2000 noted patients with preoperative “straightened” cervical alignment, significantly increased the risk of kyphosis compared to patients with lordotic alignment. 46 Guigui et al. in 1998 advocated for pre-operative flexion–extension radiographs for detecting dynamic instability; in their cohort of 58 patients who underwent multilevel laminectomy, 31% developed postoperative kyphosis and 5 % eventually required revision fusion for stabilization. 47
The clinical impact of post-laminectomy kyphosis is uncertain. For instance, Kato et al. observed 47% occurrence of postoperative kyphosis in their LA series, however, the presence of kyphosis did not appear to be associated with neurological decline. 48 Similarly, Mikawa et al. found that 36% experienced changes in postoperative spinal alignment, with 14% developing kyphosis, but again the development of kyphosis did not appear to correlate with neurologic worsening. 49 In contrast, van Geest et al, reported on 77 undergoing LA with an incidence of kyphosis of 15% and 5 patients requiring reoperation for symptomatic deterioration related to kyphosis. 44 Although our meta-analysis of neurologic recovery did not show a significant difference in LA and LF, the included studies varied significantly in follow up time (e.g. 10 months to over 9 years). The study that showed the largest effect size favouring LF was by Du et al, which also happened to have the longest follow up of the included studies, suggesting the benefit of adjunctive fusion in preserving neurologic function from late symptomatic kyphotic progression may only become apparent with longer‐term follow-up. Further long-term, prospective studies with standardized assessment intervals are needed to clarify the true impact of adjunctive fusion on preventing late deterioration.
Across the included studies, both LA and LF were associated with improvements in patient-reported measures of disability, pain, and quality of life measures. Given the diversity of PROs employed across the literature and the variability in reporting, we could not meaningfully pool most PROs, precluding formal assessment. We were able to pool neck pain scores, showing a trend towards superior pain improvement in LA, however it did not reach statistical significance. LA may result in reduced pain, at least in the initial post operative period due to reduced muscle dissection, operative time and blood loss. However, patients with existing loss or lordosis, instability or significant axial neck pain may stand to gain more from fusion in addition to decompression. 5
Complications following LA vs LF have traditionally been thought to favor the LA approach because fusion typically involves longer operative times, greater blood loss, and the potential for instrumentation‐related issues.12,57 Previous systematic literature review indicates that the overall complication rate among cohorts of LF is between 15-25% and 13-25% among LA cohorts.12,58 Certainly, fusion adds the potential for instrumentation-related complication risks that can be avoided in LA. 58 In our pooled analysis, there was no statistically significant overall difference in the odds of experiencing complications between LA and LF, however this relationship may depend on the number of operative levels. Ideally, we would have also stratified by severity or type of complication however this was precluded by granularity of reporting across studies.
Quality Assessment of Studies Comparing LA and LF
We conducted quality assessments of the studies comparing LA vs LF for both observational trials and RCTs (Supplement Table 2). The study by Bartels et al was the only RCT and was assessed using the Cochrane Risk of Bias (RoB 2) tool. 16 It was found to have an overall low risk of bias. Strengths included adequate randomization and handling of missing data, though some concerns were noted due to the lack of blinding for participants and outcome assessors, which may have introduced some performance and detection bias. Additionally, this study had a very small sample size (18 patients, 9 in the LA group and 9 in the LF group), which limits its statistical power and the generalizability of its findings.
The observational studies comparing LA to LF, evaluated using the Newcastle-Ottawa Scale (NOS), 15 demonstrated varying, but generally moderate to high, levels of methodological rigor. Generally, studies performed well in areas such as comparability of the cohort and follow-up adequacy. Some articles were found to have limited representativeness and varying levels of comparability, which reduced their overall scores. Despite these limitations, the observational studies still provide valuable insights, particularly in long-term outcomes which are often harder to capture in RCTs.
Limitations
Several limitations of this systematic review and meta‐analysis warrant consideration. First, the included studies were highly heterogeneous in design, patient selection, and outcome measurement. Such variability diminishes comparability between LA and LF cohorts and limits the generalizability of our pooled estimates. Second, the low number of studies included in meta-analysis may make it difficult to detect true differences when they exist and raises concerns about the representativeness of the pooled estimates. Third, the majority of data originated from retrospective observational cohorts, which are subject to selection bias and unmeasured confounding, particularly related patient selection, surgical technique and follow up protocols. Important covariates that might influence both treatment selection and outcomes, such as baseline cervical alignment, bone quality (osteoporosis/osteopenia), and relevant comorbidities were not consistently included or adjusted for. Finally, cost considerations and variability in access to instrumentation, factors not assessed here, may also influence decision making across different geographic regions and healthcare settings. Taken together, these limitations demonstrate the need for prospective, randomized studies with standardized outcome definition with an emphasis on PROs to more definitively compare LA and LF in DCM.
Conclusion
While LA carries a clear risk of postoperative kyphosis, it remains uncertain whether this significantly translates into worse clinical outcomes. In our pooled analysis, neurologic recovery, pain improvement, and overall complication rates did not differ significantly between LA and LF. However, subgroup analysis suggested that patients undergoing LA across more than four levels may face higher kyphosis and complication rates, indicating that extensive multilevel decompression without fusion could confer additional risk. Substantial heterogeneity in patient selection, surgical technique, and outcome measurement and the potential for unmeasured confounding likely obscures true differences between approaches. Prospective randomized comparisons of LA and LF using standardized outcome measures, such as the ongoing POLYFIX-DCM trial, 59 represent the definitive way to clarify the benefits of both surgical approaches. Future studies should employ standardized criteria for selecting candidates for fusion vs decompression alone and utilize uniform, validated outcome measures.
Supplemental Material
Supplemental Material - Cervical Laminectomy for Cervical Degenerative Cervical Myelopathy: Is There a Role in the Modern Day? A Systematic Review and Meta-Analysis
Supplemental Material for Cervical Laminectomy for Cervical Degenerative Cervical Myelopathy: Is There a Role in the Modern Day? A Systematic Review and Meta-Analysis by Christopher S. Lozano, Armaan K. Malhotra, Husain Shakil, Vishwathsen Karthikeyan, Neevya Balasubramaniam, Uzondu Agochukwu, Ankit Mehta, Aditya Vedantam, Rory Goodwin, Nathan Evaniew, Aminul I. Ahmed, Ricardo Rodrigues Pinto, James Harrop, Shekar Kurpad, and Jefferson R. Wilson in Global Spine Journal
Footnotes
ORCID iDs
Author Contributions
CSL, AKM, and JRW contributed to the conception and design of the study. Data collection and analysis were performed by CSL, AKM, NB. CSL drafted the manuscript. All authors provided critical revisions. Study supervision was provided by JRW.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Goodwin has received other unrelated research funding disclosed on the ICMJE disclosure form. Dr. Vedeantam has received other unrelated research funding disclosed on the ICMJE disclosure form.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Spinal Cord Injury, a focused group of international spine experts, and AO Spine North America. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization based in Davos, Switzerland
Supplemental Material
Supplemental material for this article is available online.
