Abstract
Study Design
Retrospective Cohort Study.
Objectives
To determine if 3D printed porous titanium (Ti) cages compared to non-porous polyetheretherketone (PEEK) cages predicted improved fusion rate or clinical outcomes after transforaminal lumbar interbody fusion (TLIF).
Methods
All adult patients who underwent one- and two-level TLIF for degenerative conditions by a single surgeon at an academic center between 2017-2021 were retrospectively identified. Patients were dichotomized into porous Ti and non-porous PEEK interbody cage groups. Fusion status was assessed by at 6-months and 1-year postoperatively by CT Bridwell scale and flexion/extension X-Rays. Postoperative complications (CT-based subsidence, adjacent segment disease, reoperation rates) and interval changes in lumbar radiographic alignment at short- and long-term follow-up were determined. Univariate and multivariate analysis compared patient and surgical factors, fusion rates, alignment, and complications across interbody groups.
Results
A total of 136 patients with 169 unique fusion levels (125 Ti cages, 44 PEEK cages) met the inclusion/exclusion criteria. Ti relative to PEEK interbodies had significantly decreased surface area and increased height and lordosis. Ti cage use was associated with greater subsidence (P < 0.001) but did not independently predict maximum subsidence on regression (P = 0.109). In the overall cohort, there were no significant differences in fusion rates or lumbopelvic alignment between interbody groups. Reoperation (Ti: 10.9% vs PEEK: 28.6%, P = 0.026) and reoperation for pseudoarthrosis (Ti: 2.0% vs PEEK: 11.4%, P = 0.038) was significantly more likely in the PEEK relative to the Ti group.
Conclusions
Use of 3D printed porous Ti cages relative to non-porous PEEK cages may mitigate pseudoarthrosis related reoperation after TLIF.
Introduction
Spinal fusion surgery, the fifth most common operation of all surgeries performed in the United States annually, is often performed with an interbody to improve spinal alignment, maximize neuroforaminal height restoration, and increase vertebral stability and fusion rate. 1 In lumbar interbody fusion, transforaminal lumbar interbody fusion (TLIF) may be preferred to enable a single all posterior approach with direct and indirect decompression. However, in TLIF relative to anterior and lateral approaches, the anatomic constraints of the facetectomy window limits cage footprint and surface available for implant to vertebral endplate osseous bridging.2,3 Provided the decreased surface area of TLIF cages, optimization of interbody material and surface properties is crucial for fusion efficacy.
Presently, polyetheretherketone (PEEK) and titanium (Ti)/ Ti-based alloys are the most commonly used materials for interbody cages in surgical practice.4,5 Though PEEK has a close to physiologic range mechanical Young’s modulus, it is associated with hydrophobic surface chemistry, biopassive behavior, and poor surface apatite-formation which limit fibrin clot formation and osteoblast differentiation necessary to achieve osseointegration.6-8 As such, a fibrous interface between the PEEK interbody cages and vertebral endplate may develop creating implant micromotion leading to pseudoarthrosis. 9 Whereas Ti based interbody cages exhibit natural osseoconductive properties developing a surface corrosion- resistant oxide layer that absorbs fibronectin and promotes osteoblastic attachment.10-12 While solid Ti is associated with stress shielding from a supraphysiologic Young’s modulus, the new generation of 3D printed (3DP) Ti cages with bulk and surface porosity optimize device load sharing and maximize the already favorable Ti surface properties for imbibition of blood products that lead to a regenerative microenvironment.13-17 The current study aimed to determine if 3DP porous, Ti cages compared to non-porous PEEK cages predicted improved fusion rate, radiographic alignment, or clinical outcomes after TLIF for degenerative indications.
Methods
Patient Identification
After approval from the Institutional Review Board (IRB: 10-002852), a retrospective cohort study was conducted on all patients over age 18 who underwent one- and two-level TLIF transforaminal lumbar interbody fusion (TLIF) for lumbar spondylosis associated with neurogenic claudication, radiculopathy, and/or spondylolisthesis by a single surgeon at an academic institution between 2017-2021. The study was deemed exempt from needing informed consent by the Institutional Review Board. Additional inclusion criteria required complete patient demographic profiles, surgical characteristics, interbody device and fusion construct records, and preoperative radiographs, and postoperative computed tomography scans obtained greater than 6 months postoperatively. Patients with a traumatic injury, infection, malignancy, previous fusion at the index level, combined anterior/posterior procedures, or surgery with greater than 2 TLIF levels or greater than 4 fusion levels were excluded. Patients were also excluded if their medical records or outcome measures were incomplete. Patient demographic and medical data, surgical factors, perioperative radiographic measures, and duration of follow-up were recorded via chart review.
Operative Treatment
All patients underwent an open midline posterior approach to the lumbar spine. Following fluoroscopic confirmation of the TLIF level, a unilateral complete facetectomy was performed. After disc space distraction was achieved with screw distractors or laminar spreaders, a box annulotomy and disc space preparation were performed. Endplate preparation was achieved with paddle shavers and serrated curettes. The interbody was filled with local autograft and morselized cancellous allograft, with or without bioactive glass for augmentation, and inserted as anteriorly as possible in the disc space under fluoroscopic guidance. Bone morphogenic protein (BMP), if used, was only placed into the lateral gutters. The interbody cage systems utilized were the Conduit Titanium cage (Depuy Synthes, Raynham, MA, USA) and OPAL and T-PAL PEEK cages (Depuy Synthes, Raynham, MA, USA) (Figure 1). All patients had posterior supplemental fixation with Expedium® or Viper® pedicle screw systems (DePuy Synthes, Raynham, MA, USA) and attempted posterolateral fusion. Depiction of (A) 3DP porous, cellular Ti and (B) non-porous PEEK interbody cages. Copyright Depuy Synthes.
Radiographic and Construct-Related Variables
Preoperative patient bone quality was determined from the dual energy X-ray absorptiometry (DXA) bone mineral density (BMD) lumbar t-score and the computed tomography (CT) Hounsfield units (HU) of the superior and inferior vertebral levels. Hounsfield units (HUs) of the vertebral body were measured on preoperative axial CT images in the cranial, middle, and caudal aspects of each vertebral body, and an average value per vertebral level was determined. Additionally, preoperative disc space height at the TLIF level (average of anterior, middle, and posterior aspect measurements on mid-sagittal CT view) was measured.
Operative notes and device implantation records were reviewed to determine surgery and instrumentation specific parameters including operative indication, TLIF levels, cage material and surface (3DP porous Ti and non-porous PEEK), cage type (bullet and banana), cage dimensions including length, width, height, and calculation of surface area (assuming rectangular cross-section), cage degree of lordosis, and fusion adjunct use including the use of BMP. Non-porous PEEK interbody use was standard of care from 2017-2019 in this single-surgeon study and transitioned to 3DP porous Ti interbody use from 2019-2021. The normalized anterior-posterior interbody placement location was assessed on postoperative standing lateral radiographs through the calculated center point ratio (distance from center of cage to posterior cortex/ anterior-posterior vertebral dimension) (Figure 2).
18
Center point ratio (CPR) measurement technique.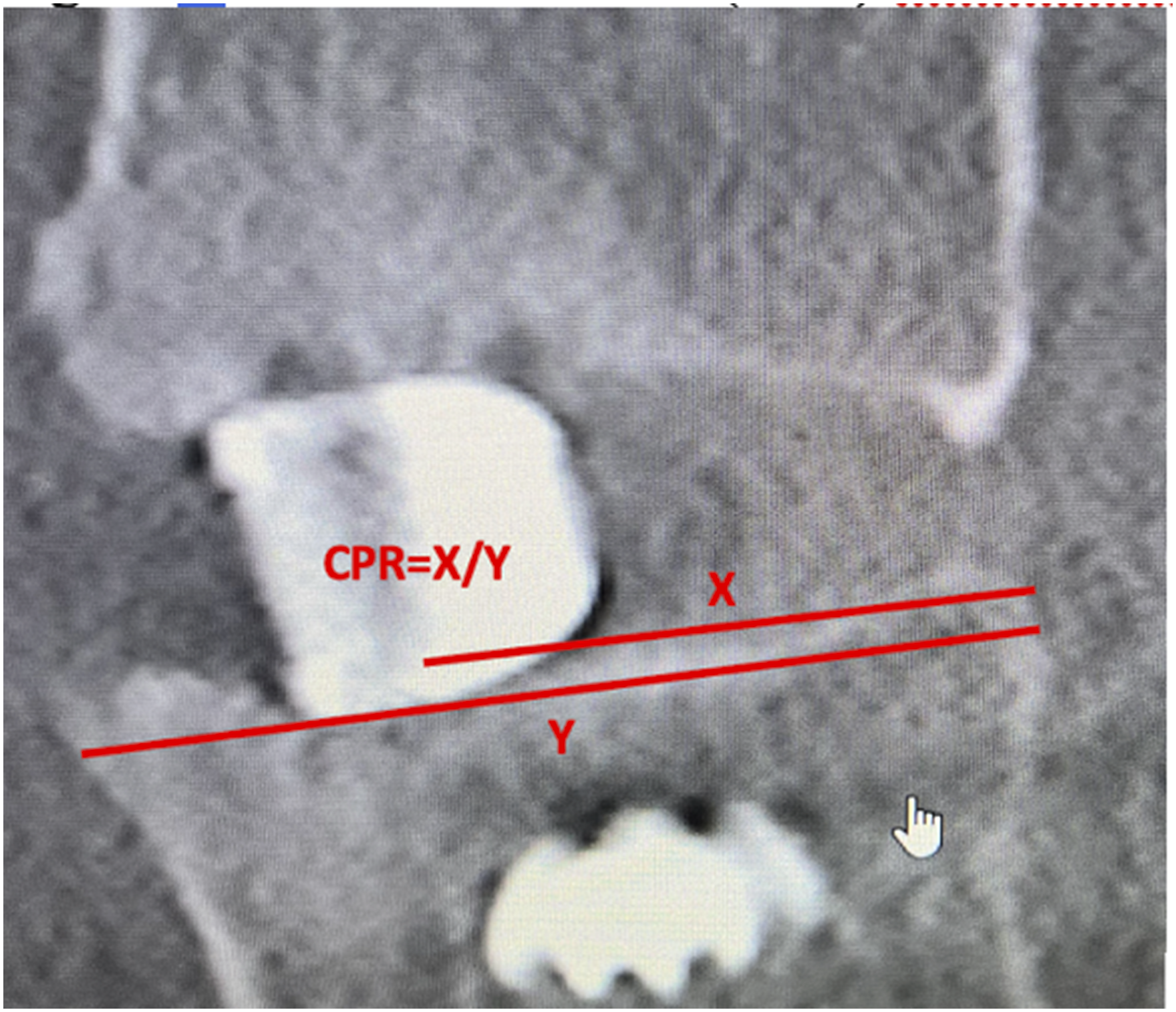
Fusion Assessment
Fusion assessment was performed on X-ray and CT modalities by 2 independent spine fellowship trained reviewers (BG and JB) and discrepancies were resolved via consensus or adjudication by a third reviewer (AS) when necessary. X-Ray fusion assessment was performed separately at 6-months and 1-year postoperatively according to both mobility on flexion/ extension radiographs (<2 mm translation and <3 degrees of angular motion) and the Bridwell scale on intervertebral disc space changes on standard lateral X-ray (I = fused with remodeling and trabeculae present, II = graft intact, not fully remodeled, no lucency present, III = graft intact, potential lucency at the top and bottom of the graft, IV = fusion absent with no visualized bony remodeling with or without graft collapse). 19 Bridwell grade I and II were considered a successful fusion. Patients with successful fusion observed at 6-months were carried through to the 1-year time point if a 1-year X-ray was not available for review. CT fusion status was assessed on the 1-year postoperative CT according to the Bridwell classification (abovementioned scale). Institutional CT properties were as follows: (settings: 140 kV, 280 mAs, and 0.75 mm slice thickness, 0.75 × 0.75 × 0.75 mm resolution). It represented routine institutional practice to obtain CT scans at this interval for fusion monitoring.
Postoperative Radiographic Outcomes and Complications
Intraoperative and perioperative outcomes including estimated blood loss (EBL), rates of dural tears, infections, and hematomas were recorded. Lumbar radiographic alignment, specifically pelvic incidence, pelvic tilt, sacral slope, lumbar lordosis, L4-S1 lordosis, and regional lordosis (lordosis of a single operative level) was assessed on standing lateral x-rays at preoperative, immediate postoperative, and long-term postoperative (1-year) time points to determine preoperative to postoperative changes in alignment at immediate and 1-year intervals. Long-term radiographic complications including the occurrence of rod fracture and screw loosening were determined from postoperative CTs and long-term postoperative x-rays. Interbody subsidence, at the superior and inferior endplate of each TLIF level, was directly measured perpendicular to the endplate-facing surface of both mid-coronal and mid-sagittal CT scans obtained at 1-year postoperatively according to previously validated CT methodologies.20-22 Subsidence assessed on postoperative CT has been shown to be more sensitive for the detection of subsidence than lateral x-rays due to improved resolution and lack of dependence on patient positioning.
21
Total subsidence was determined from greatest subsidence measurement with respect to the coronal and sagittal planes. Average subsidence was determined from the average of the superior and inferior measurements in both the coronal and sagittal planes (Figure 3). Interbody subsidence at each operative level was classified as mild, moderate, or severe based on previously documented <2 mm, 2-4 mm, and ≥4 mm thresholds, respectively.21,23 Clinically symptomatic complications secondary to pseudoarthrosis and adjacent segment disease were determined from patient postoperative follow-up encounter notes. The all cause reoperation rate as well as the subset of patients undergoing reoperations for pseudoarthrosis were recorded. Depiction subsidence measurement technique from an example with severe subsidence on (A) mid-sagittal and (B) mid-coronal CT slices.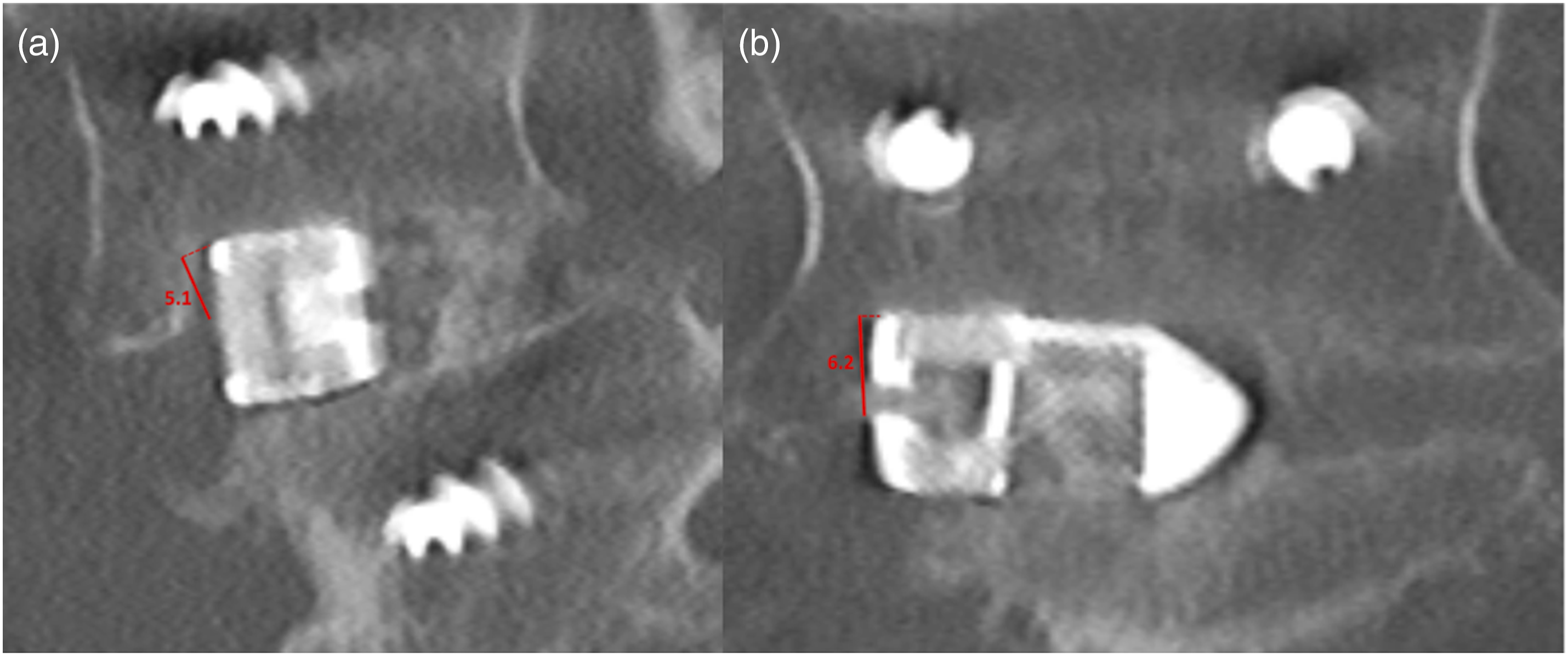
Statistical Analysis
Patients were dichotomized into 3DP porous Ti and non-porous PEEK interbody cage groups. Analysis was performed on a per-patient basis for demographics, medical factors, general operative factors, and complications and on a per-level basis for interbody factors, fusion status, and interbody cage subsidence. Descriptive statistics were used to compare patient demographics and delta outcomes in terms of mean and standard deviation, median and interquartile range, or number of occurrences and percent of total. Continuous and categorical variables were analyzed with t-tests and chi-square tests, respectively, or the corresponding non-parametric tests. Univariate analysis compared patient demographics, medical comorbidities, preoperative musculoskeletal parameters, and intraoperative factors, and postoperative outcomes among interbody cage groups. Logistic regression models isolated the effect of cage type on postoperative outcomes of interest. All statistical analysis was performed with R Studio Version 4.0.2 (Boston, MA). A P-value <0.05 was statistically significant.
Results
A total of 136 patients with 169 unique TLIF fusion levels (103 one-level, 33 two-level) met the inclusion/ exclusion criteria. Specifically, 101 patients (125 TLIF levels) underwent TLIF with 3DP porous Ti interbody cages and 35 patients (44 TLIF levels) with underwent TLIF with non-porous PEEK interbody cages. The average radiographic follow-up duration in the overall cohort was 1.04 [0.99, 1.79] years postoperatively.
Demographics and Surgical Factors
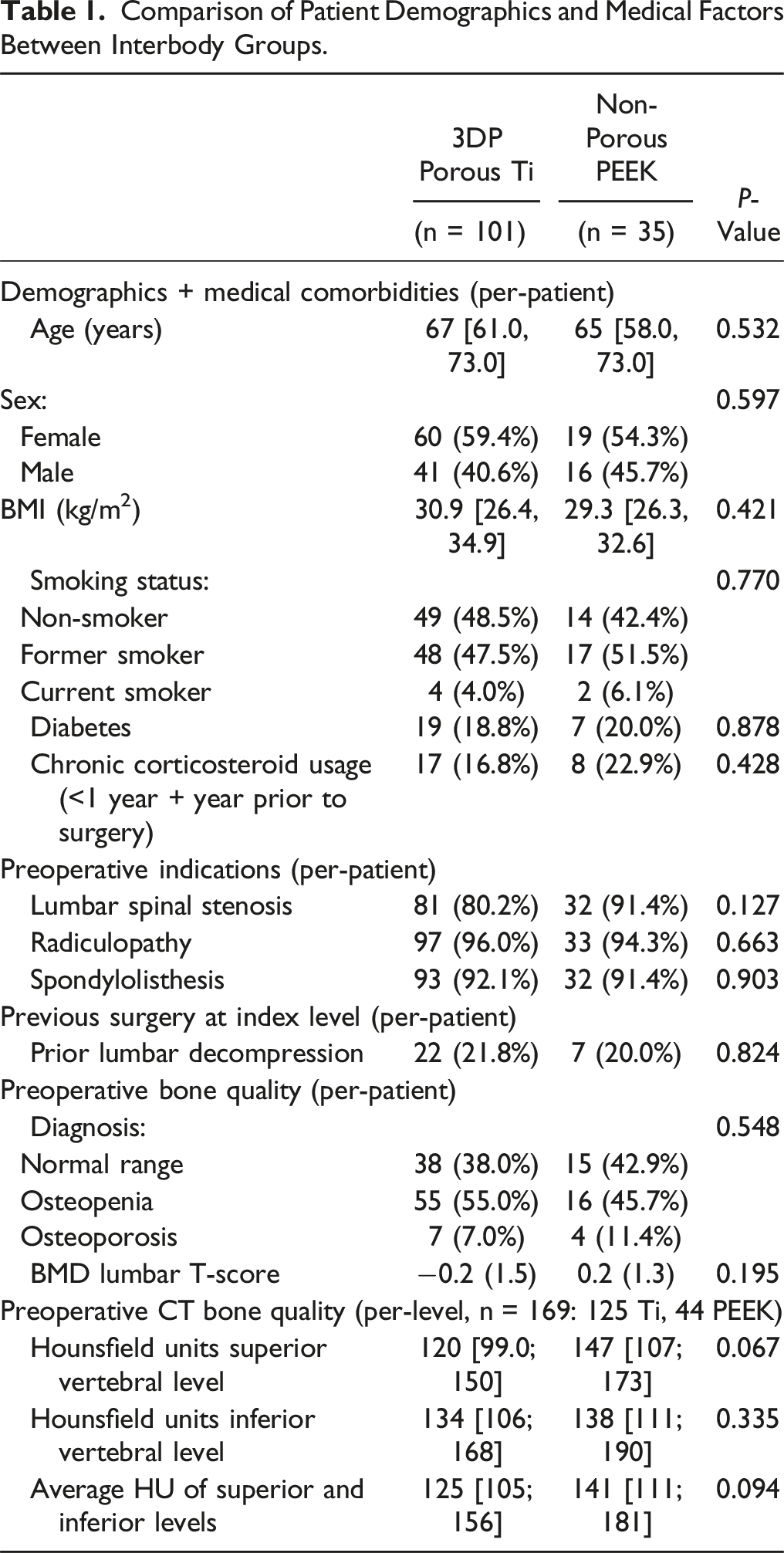
Comparison of Patient Demographics and Medical Factors Between Interbody Groups.
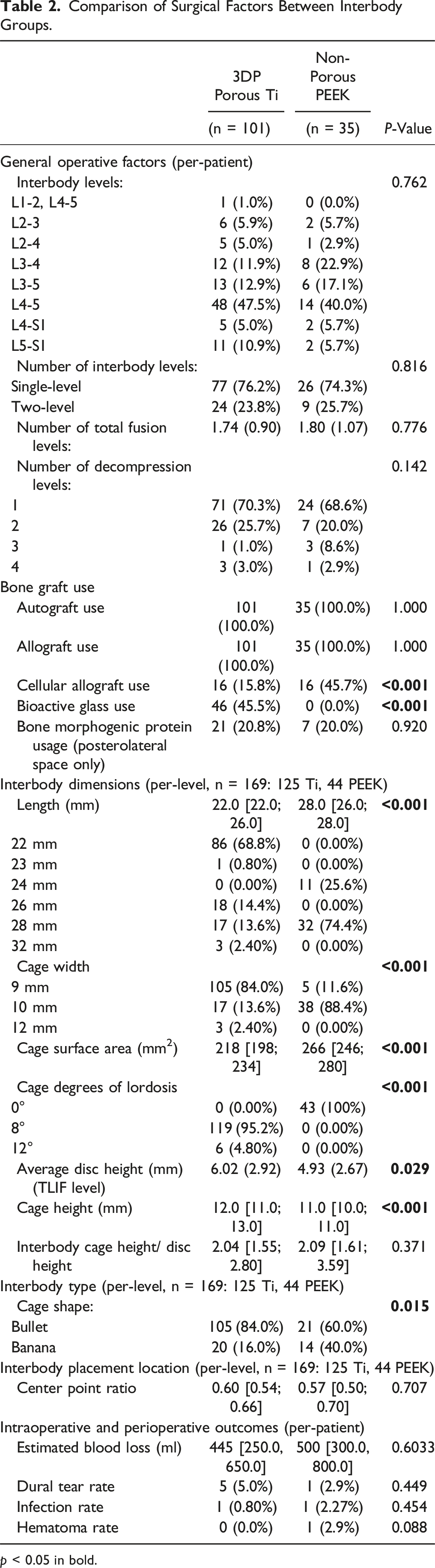
Comparison of Surgical Factors Between Interbody Groups.
p < 0.05 in bold.
In aggregate, the PEEK cage surface area (length x width) was significantly larger than the Ti cage surface area (Ti: 218 mm2 vs PEEK: 266 mm2). Ti interbody cages had either 8° (95.2%) or 12° (4.8%) of lordosis whereas PEEK interbody cages were all 0° designs (P < 0.001). While the mean Ti cage height was greater than the PEEK cage height, this scaled appropriately with the differences in preoperative disc height between groups such that the interbody cage height to disc height ratio were not significantly different between groups (P = 0.371) (Table 2). The majority of interbody cages in both groups were bullet designs (Ti: 84.0% vs PEEK: 60.0%), however there was greater banana shaped cage use in the PEEK group (Ti: 16.0% vs PEEK: 40.0%) (P = 0.015). There was no significant difference in cage anterior-posterior placement, assessed with center point ratio, between groups (P = 0.707). Intraoperative (EBL) or perioperative outcomes (dural tears, infections, and hematomas) did not differ significantly between groups (Table 2).
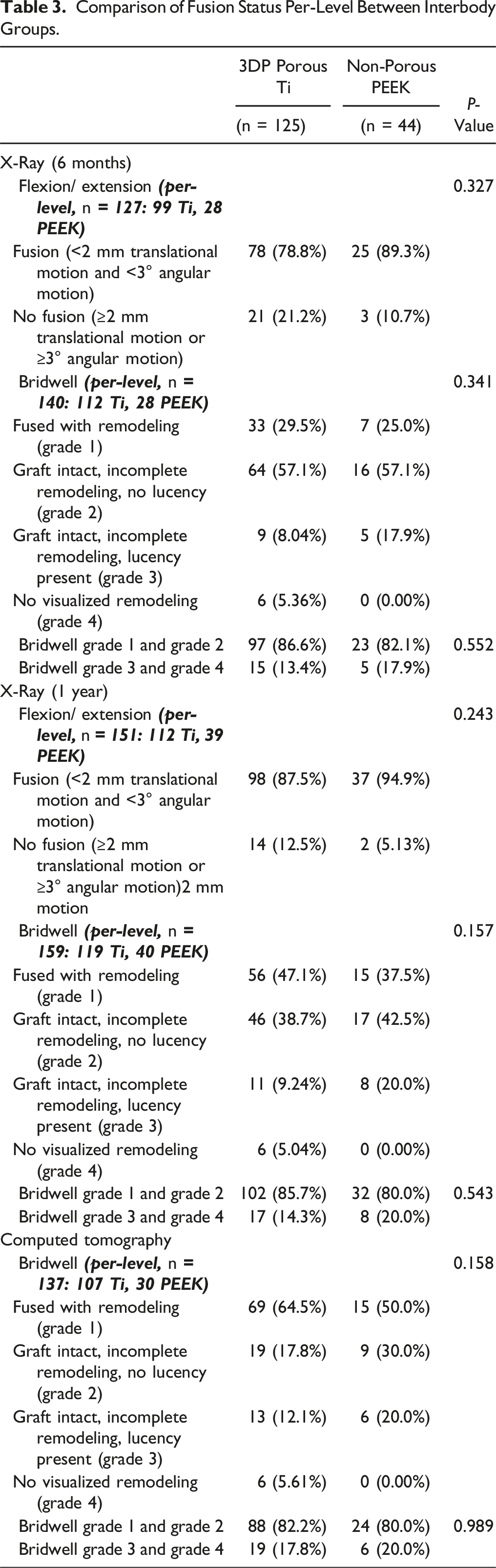
Fusion Rate
Comparison of Fusion Status Per-Level Between Interbody Groups.
Radiographic Outcomes and Complications
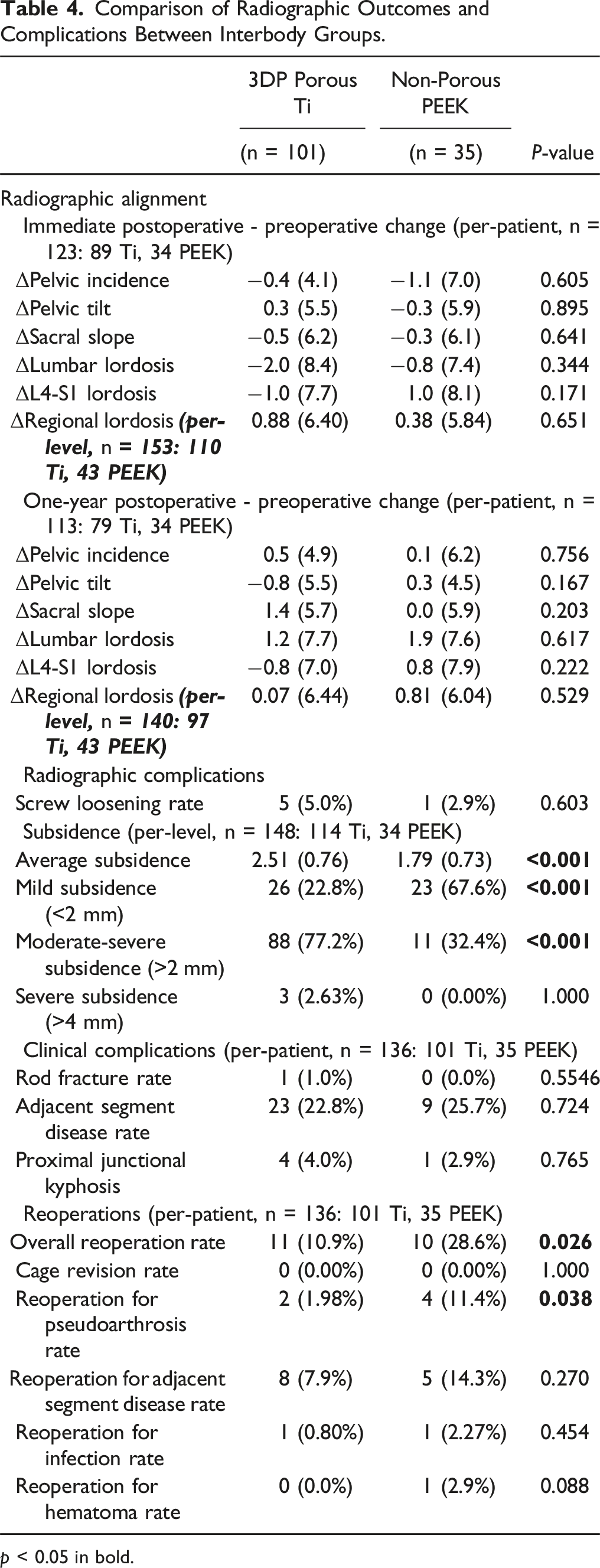
Comparison of Radiographic Outcomes and Complications Between Interbody Groups.
p < 0.05 in bold.
Logistic Regression Analysis for Moderate Subsidence.
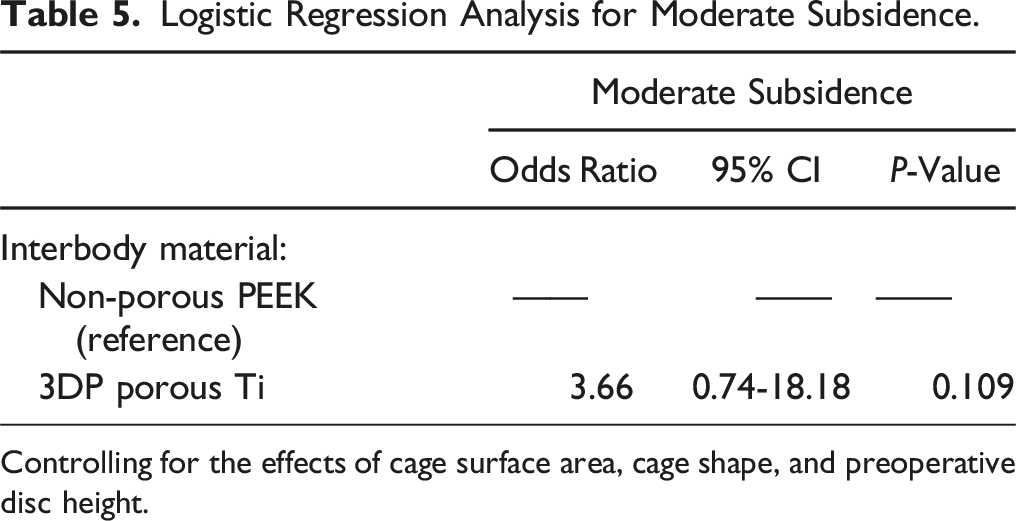
Controlling for the effects of cage surface area, cage shape, and preoperative disc height.
Reoperation (Ti: 10.9% vs PEEK: 28.6%, P = 0.026) and reoperation for pseudoarthrosis (Ti: 2.0% vs PEEK: 11.4%, P = 0.038) was significantly more likely in the PEEK relative to the Ti group. In reoperations for pseudoarthrosis, revision fusion was attempted in the posterolateral space and interbody cages were not removed. There were no significant differences in the reoperation rate for adjacent segment disease between groups (Table 4).
Discussion
Comparing the 2 most common spinal interbody cage materials, Ti/ Ti alloys are associated with naturally osseoconductive surfaces but supraphysiologic moduli, whereas PEEK demonstrates a physiologic range modulus but has a hydrophobic surface with poor apatite-forming ability that is necessary for osteoblast differentiation.10,12,24,25 The introduction of surface and bulk porosity in 3DP Ti-based interbody cages offers potential advantages of decreased modulus and more favorable load sharing than their non-porous cage counterparts.13,17,26,27 Interbody cage porosity may also decrease interbody micromotion limiting fibrotic response, maximize osteoconductivity, and promote osseointegration.9,16,17,28-30 While prior preclinical and lateral and posterior lumbar interbody fusion (LLIF and PLIF) studies have previously demonstrated increased fusion rate and decreased time to fusion of porous Ti cages compared standard of care PEEK cages, there is a relative dearth of high quality evidence in TLIF.31-37 Our results suggest that porous Ti interbody compared to non-porous PEEK interbody usage was associated with decreased rates of reoperation for pseudoarthrosis.
Preclinical models of ovine lateral lumbar interbody fusion comparing porous Ti alloy to PEEK cages have demonstrated significantly increased bone ingrowth and bone volume in the graft interval and significantly decreased flexion-extension range of motion in the Ti alloy group at early and late sacrifice time points.31,35-37 These results have been largely substantiated in clinical LLIF and PLIF investigations comparing 3DP porous Ti interbody cages to their previous standard-of-care non-porous PEEK interbody.33,34 Specifically, the standalone LLIF study demonstrated a significantly increased fusion rate in the group of patients with 3DP porous Ti relative to non-porous PEEK interbody fusion rate based on postoperative CT at approximately 8 months, suggesting a shorter time to achieve segmental stability. 34 However, they failed to detect a significant difference in 3DP porous Ti relative to non-porous PEEK interbody fusion rate in the group of patients with postoperative CTs at a longer time point of approximately 18 months. Additionally, in this study all lateral interbody cages were packed with BMP, a known efficacious osseoinductive agent, which likely cofounds the effects of cage material and surface properties on disc space fusion. 34 The single-surgeon PLIF study, performing bilateral facetectomy and bilateral interbody insertion, found a significantly increased fusion rate (by CT and flexion-extension X-ray) was achieved with 3DP porous Ti relative to PEEK cages at both one- and two- year postoperative time points. They also demonstrated that 3DP Ti was an independent predictor of CT fusion status. 33
In TLIF, relative to LLIF and bilateral PLIF, the interbody cage footprint representing the surface available for disc space fusion is substantially reduced and the potential fusion benefit of 3DP Ti compared to PEEK is less obvious. In a small single-center blinded, randomized controlled trial (RCT) of patients undergoing TLIF with bilateral facetectomy with a 3DP Ti vs PEEK interbody, there were significant differences in intervertebral fusion rates (according to the Brantigan-Steffe-Fraser classification) at the initial 6-month assessment that favored 3DP Ti, which led to the trial being stopped early. 38 Of note, there was no significant difference in fusion rate assessed by flexion-extension imaging at 6-months. Furthermore, because of the early trial conclusion it is unclear whether this PEEK related pseudoarthrosis risk would have been sustained in the long-term. This was also explored in a recent retrospective single-surgeon cohort of patients undergoing minimally invasive surgery (MIS) TLIF with a 3DP Ti compared to PEEK interbody. This study found significantly greater attainment of Bridwell Grade 1 fusion (complete remodeling) at 6-months and 1-year in the 3DP Ti group, however the differences in overall fusion rates (Bridwell Grade 1 and 2) were not significantly different across groups. 39 In the current study, while there was up to a 15% greater achievement of CT Bridwell Grade 1 fusions in the 3DP Ti relative to PEEK groups, the differences in Bridwell fusion status in all time points and imaging modalities were not significant. Contrary to prior literature, the rate of intervertebral fusion, as defined in this study, was not accelerated in the porous Ti group with no significant difference in X-ray Bridwell classification between interbody groups at 6-months and less than a 5% difference in the portion of interbody levels that had achieved complete osseous remodeling at this earlier time interval. Similar to the randomized trial, there were also no significant differences in X-ray flexion-extension assessment of fusion at either the 6-month or 1-year time interval. This may reflect differences in posterolateral gutter or contralateral facet fusion that are independent of interbody properties.
Although the potential efficacy of porous Ti cages has been evaluated in numerous image-based fusion studies, the aforementioned clinical studies either failed to include if the pseudoarthrosis cases were clinically symptomatic or did not identify a difference in revision rates between groups. This is the first investigation to establish a direct clinical benefit from porous Ti relative to non-porous PEEK interbody use, finding a protective effect on revision surgery for pseudoarthrosis. Specifically, there was a 9% greater prevalence of reoperations indicated for pseudoarthrosis in the non-porous PEEK group among patients with noted clinically symptomatic and painful pseudoarthrosis. These pseudoarthrosis related differences were also reflected by the elevated overall reoperation rate in the PEEK group, where adjacent segment pathology did not differ significantly between groups.
However, the role of cage material and porosity on subsidence occurrence as well as the clinical importance of mild subsidence remains an area of ongoing debate.40-42 It is possible that the osseoconductive properties of Ti relative to PEEK may confer earlier stability protecting against subsidence, as preliminary LLIF study results suggest. 10 While stiffness of porous Ti is decreased notably relative to non-porous Ti, the modulus of porous Ti cages still remains higher relative to trabecular bone which could risk subsidence in TLIF where the smaller, single interbody cage footprint already leads to higher pressures. 43 A single-surgeon investigation in bilateral PLIF found that no significant differences in subsidence rates between the 3DP porous Ti and the non-porous PEEK control group, when subsidence was defined by a 3 mm threshold on 1-year CT scans. 33 These same findings have been corroborated by some but not all TLIF studies, where a single-surgeon MIS TLIF study that assessed subsidence by segmental vertebral body height loss >2 mm on final X-Ray found no significant difference in subsidence rates between 3DP Ti and PEEK groups. 39 Whereas a TLIF RCT defining subsidence by ≥ 2 mm endplate depression on 6-month CT found that subsidence rates were over 40% significantly greater in the PEEK relative to 3DP Ti group. 38 However, there was no regression analysis or comparison of HU between groups to clarify this large difference. In a retrospective study that focused on the relative importance of risk factors for TLIF subsidence classified on CT, PEEK relative to Ti (both 3DP and non 3DP) was associated with moderate (≥2 mm) and severe (≥4 mm) on univariate analysis, but after controlling for cage surface area, interbody placement location, and local HU was not a predictive of either moderate or severe subsidence in the predictive logistic regression model. 44 Conversely, in the current study, moderate but not severe subsidence, assessed on CT, occurred more commonly in the 3DP porous Ti relative to the PEEK group, but this relationship was cofounded by differences in cage designs across groups, most notably smaller surface areas in the Ti group. After controlling for these factors, cage material was not predictive of subsidence. Furthermore, despite the potential for subsidence to cause local kyphotic changes, our study did not find any significant differences in long-term radiographic outcomes changes across interbody type groups.
This study is not without limitations including those inherent to retrospective data collection. Although patients were included in a systematic method, potentially relevant cases were identified based on availability of follow-up parameters, introducing a selection bias. Specifically postoperative CT was not available on all patients and X-ray based assessment of fusion may under or over predict fusion rates. Although this study did not find a decreased time to fusion in the porous Ti group, postoperative CT rather than X-ray at the early postoperative time point would be necessary to more precisely characterize early intervertebral osseous consolidation. Given the limited cohort size and retrospect nature of this multi-surgeon study, inherent heterogeneity in bone graft utilization, interbody characteristics, and operative techniques may serve to confound the effect of interbody cage material and porosity on radiographic outcomes and complications in univariate analysis, where sub-stratification by all of these components was not numerically plausible. Logistic regressions for significant subgroup differences in Bridwell fusion status at 1-year were not able to be performed due to the overall small number of patient with non-union (Bridwell grade 3 and 4) at that assessment period. Further prospective randomized studies are needed to better standardize patient and operative factors across interbody cage groups and include multiple surgeons and institutions to increase the study generalizability. Radiographic data were extracted until final postoperative follow-up at one-two years after surgery. While this time period is likely sufficient to detect pseudoarthrosis in the lumbar spine it may be insufficient to detect all adjacent segment complications. Limited completion of patient outcomes questionaries did not facilitate analysis in this study. Finally, existing TLIF subsidence literature that employs X-ray based detection methods may underestimate subsidence due to patient rotation and low resolution in comparison CT-based subsidence measurement utilized in this study, preventing direct comparison of subsidence rates.
Conclusion
PEEK TLIF cages were associated with a greater reoperation for pseudoarthrosis rate than 3DP porous Ti cages, demonstrating that porous Ti cages may mitigate pseudoarthrosis related complications after TLIF. Furthermore, porous Ti relative standard PEEK interbody cages were not independently predictive of interbody subsidence and did not associate with postoperative changes in lumbar alignment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was supported by an educational grant from DePuy Synthes to cover the costs of statistical analysis and administrator work hours. Synthes had no direct access to the data or input in the final study results.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
