Abstract
Study Design
Systematic Review.
Objective
To review the available published literature relating to pedicle lengthening osteotomy (PLO) for lumbar spinal stenosis (LSS), and to compare this technique to existing surgical options.
Methods
PubMed/MEDLINE, Embase, and Scopus were reviewed using the keywords: “pedicle lengthening”, “lumbar”, and “spinal stenosis.” Results were reviewed for relevance and included only if they met inclusion criteria. The SYRCLE checklist was used to assess for sources of bias.
Results
Inclusion criteria were biomechanical or clinical studies evaluating PLO for LSS. A total of 56 studies were identified in the Pubmed/MEDLINE database using the above-mentioned search criteria. Additionally, a total of 83 abstracts were reviewed in Embase, and 76 abstracts were reviewed in Scopus. After screening, 7 articles met inclusion criteria.
Conclusions
PLO is a percutaneous, minimally invasive technique that has been described for the treatment of LSS. Biomechanical studies demonstrate significant enlargements in spinal canal dimensions and improvements in patient reported outcome measures while maintaining kinematic properties similar to the intact spine. Although overall sample size remains small, available data regarding PLO performed for LSS have shown encouraging early results.
Introduction
Lumbar spinal stenosis (LSS) is a common degenerative condition of the lumbar spine projected to increase in prevalence over the next decade as the United States population continues to age.1,2 This condition is most often caused by age-associated osteoarthritic changes of the facet joints, hypertrophy and buckling of the ligamentum flavum, and degenerative spondylolisthesis. 3 Ultimately, patients present with a range of symptoms such as neurogenic claudication, radicular pain, back pain, and sensory changes which may restrict their ability to stand and ambulate.
Although several treatment modalities have been utilized for LSS, lumbar laminectomy remains as the gold standard following failure of nonoperative management. 4 Despite being an effective method to decompress neural elements, laminectomy has several major drawbacks including peridural scarring, iatrogenic instability, and greater morbidity. For this reason, a variety of alternative techniques have been developed to mitigate these disadvantages. Minimally invasive methods using tubular retractors, lateral access, and most recently endoscopic decompression and/or fusion have gained popularity in the last decade. Prior studies have listed many benefits with minimally invasive approaches such as lower blood loss, shorter hospital stays, earlier mobilization, and less muscle damage.5-9 However, these modalities have their own set of limitations including technical difficulty, high learning curves, associated complication risks, and the possibility of inadequate decompression.10,11
Recently, a new procedure has been described for treating LSS. This procedure, referred to as a pedicle lengthening osteotomy (PLO), involves a percutaneous bone cut at the base of the pedicle followed by the placement of an implant that expands the gap between the vertebral body and posterior structures, enlarging the spinal canal to decompress the dural sac and neural foramen. The bone at the osteotomy site heals following the procedure, leading to a permanent enlargement of the spinal canal.
The purpose of this study was to systematically review the biomechanical, computational, and early clinical outcomes of PLO in the treatment of LSS. This systematic review was conducted to answer the following research questions: (1) What are the biomechanical properties of the spine and extent of canal/neuroforaminal enlargement following PLO? (2) What are the clinical outcomes, including pain scores, clinical success, and patient satisfaction in patients who undergo PLO for LSS? (3) What is the rate of complications or re-operation in patients who receive PLO?
Summary of Technique
At our institution, patients selected for PLO have at least 1 symptomatic level of LSS and failed a complete course of non-surgical care, which typically involves physical therapy and epidural steroid injections. Contraindications to the procedure include a pedicle width that cannot accommodate an implant or severe osteoporosis. The procedure can be conducted under local, regional, or general anesthesia. The procedure can be conducted with the assistance of C-arm fluoroscopy, image guidance technology, or robotic assistance.
After localizing the skin incisions, a 10 mm percutaneous incision is made in line with the trajectory of each pedicle to be lengthened. A targeting reamer is used to enter the pedicle and ream a passage through the pedicle to the junction of the pedicle and vertebral body. A special bone saw instrument is used to cut the pedicle from inside out using fine, incremental adjustments of the blade (Figure 1). Because the pedicle is not round in cross section, the initial bone cut is performed with a circumferential technique making a groove inside the bone for the blade to follow. The surgeon then transitions to zonal cutting where the remaining bone is cut in quadrants, avoiding any “pass pointing” of the saw blade to prevent injury to the adjacent nerve root. During the cutting procedure, intermittent fluoroscopy, tactile feedback, and EMG monitoring are utilized to guide the surgeon in determining when the bone cut is complete. After both pedicles have been cut, a specialized bone screw is placed, creating a gap at the site of the osteotomy and leads to expansion of the spinal canal and neural foramen (Figure 2). Bone saw used to cut the pedicle. The instrument has a knob (A, arrow) that can be turned to deploy or retract the cutting blade. The saw blade (B, arrow) extends from the tip of the bone saw. The pedicle-lengthening implant, shown in the shortened (A) and expanded (B) states. The implant is inserted into the pedicle as a typical bone screw and expanded in length after being properly positioned across the osteotomy site by the action of an inner screw. Lateral fluoroscopic (C) and 1-year axial postoperative CT (D) views show the pedicle-lengthening implants placed bilaterally.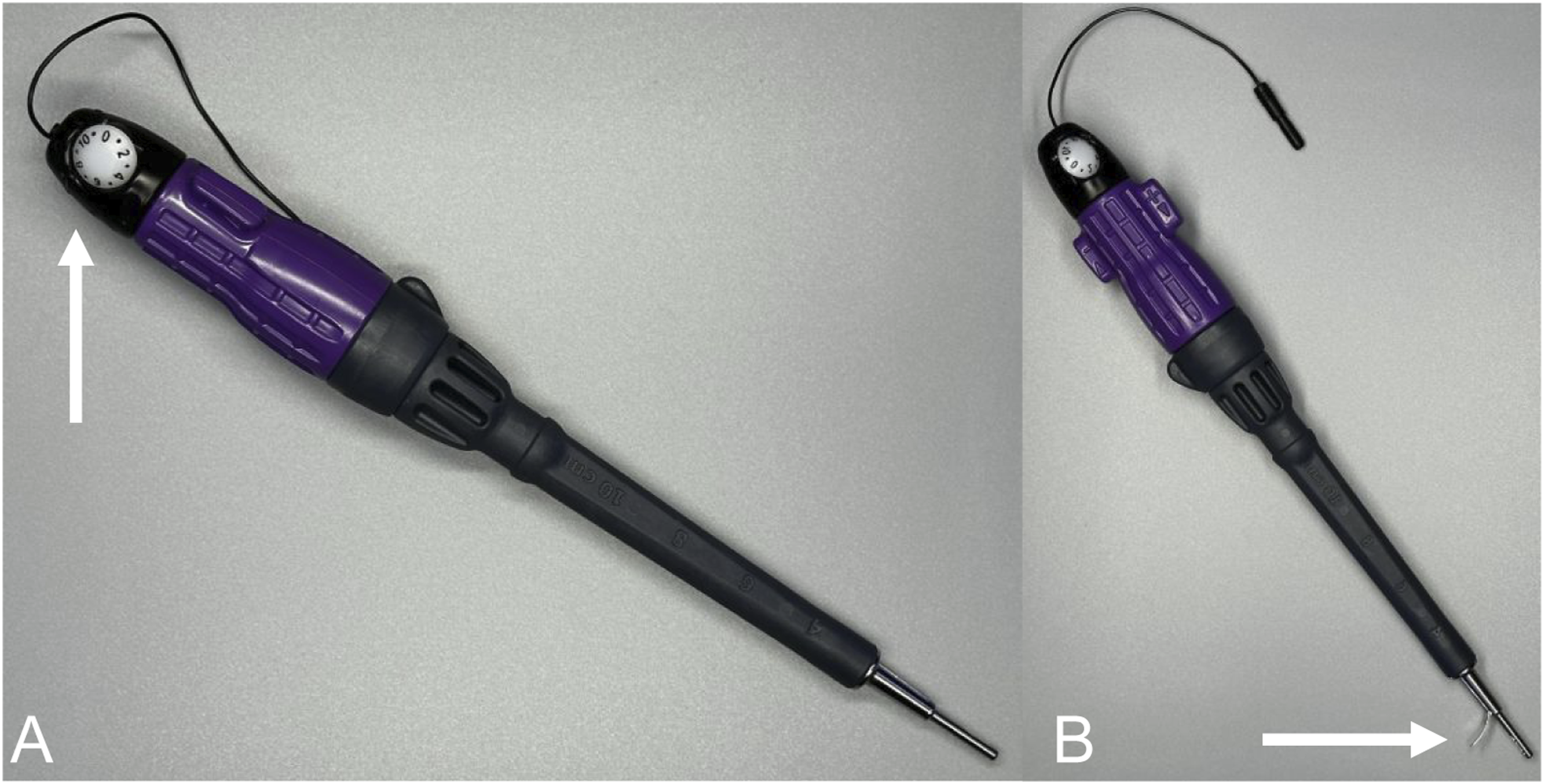
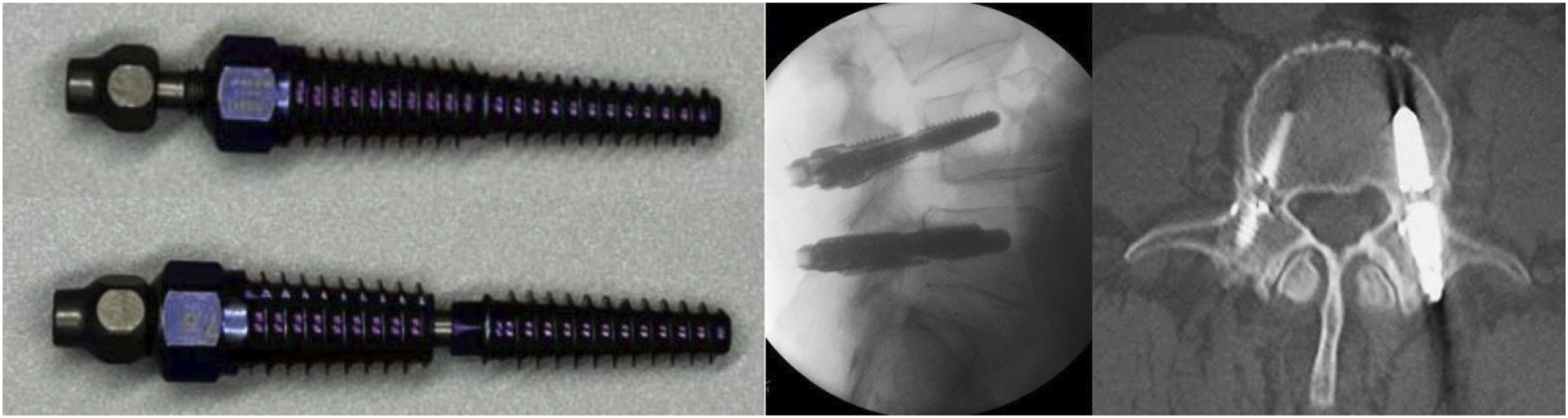
The incisions are closed in a routine fashion and the patient is allowed early mobilization. Early symptom relief is the expected outcome of the procedure. Bone healing takes 6-12 weeks and during this timeframe. The patient is asked to refrain from repetitive bending and twisting activities but encouraged to walk and pursue core muscle strengthening and spinal rehabilitation exercises thereafter.
Materials and methods
Literature Search Review
The data used in this systematic review were from published manuscripts that did not contain personal identifiers. Therefore, this study was deemed exempt from the need for informed consent and approval by an Institutional Review Board (IRB). This systematic review was written in accordance with the Primary Reporting Items for Systematic Review and Meta-Analyses (PRISMA) guidelines. 12 Literature search was performed in 3 electronic databases: PubMed/MEDLINE, Embase, and Scopus. Searches were conducted in May 2024. Only full text articles written in the English language were included. Searches were not re-rerun prior to analyses. The following Boolean search string was utilized: pedicle lengthening [title] AND (lumbar [title] OR stenosis [title]). The titles and abstracts of potentially relevant articles were reviewed, and full texts of studies included in this systematic review were obtained. The reference lists of included studies were examined to identify any additional relevant studies. All titles were reviewed by the primary author for relevance.
Studies included met the following criteria: biomechanical studies, computational analyses, clinical trials, and retrospective studies published between 2010 and present; reported data including range of motion, load-bearing, change in central canal/neuroforaminal dimensions; patients >18 years of age diagnosed with LSS treated with PLO with a minimum mean of 1-year post-operative follow-up; containing clinical outcomes with at least 1 of the following: Oswestry Disability Index (ODI), Zurich Claudication Questionnaire (ZCQ) Physical Function and Symptom Severity domains, San Francisco-12 (SF-12) physical and mental component scores, and visual analog scale (VAS), complication or reoperation rates. Exclusion criteria included the following: non-English manuscripts; case reports and abstract only articles; articles that assessed this technique for trauma or oncologic diagnoses.
Quality Assessment
Assessment of the level of evidence for included studies was carried out using the Oxford Centre for Evidence-Based Medicine (OCEBM). 13 Meanwhile, risk of bias for clinical studies was evaluated using the Methodological Index for Non-Randomized Studies (MINORS). 14 Although no standardized scale currently exists which would be suitable to assess the bias of eligible cross-sectional biomechanical studies, methodology was adapted from Hindle et al 15 based on systematic reviews of similar study design. 15 Regarding this scale, items were scored as 1 where the article provided sufficient evidence in support of criteria, and 0 where criteria was not met. Articles scoring ≥ 67% were considered as having low risk of bias, those scoring between 34-66% having satisfactory risk of bias, and those scoring ≤ 33% having high risk of bias. Only articles scoring low or satisfactory risk of bias were included in this review.
Results
Study Characteristics
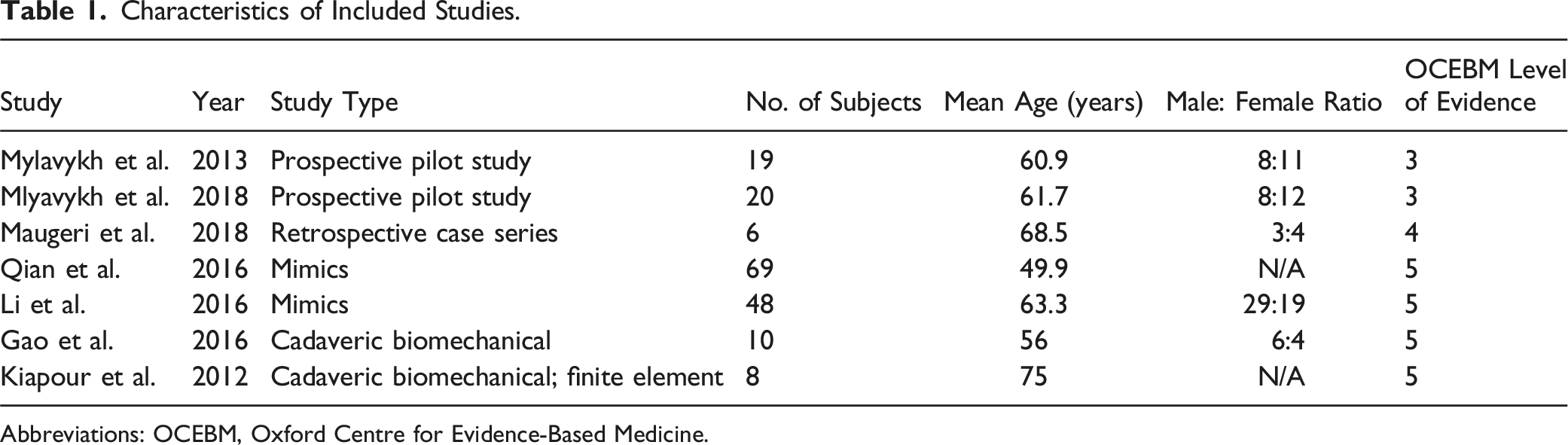
The initial search generated a total of 227 citations from PubMed/MEDLINE, SCOPUS, and Embase. After removal of duplicates, 98 articles were available for title and abstract screening, with 44 articles deemed appropriate for full-text evaluation. A total of 7 studies were included for final analysis (Figure 3). Characteristics and demographics of included studies are included in Table 1. Flowchart depicting strategy for including and screening articles for narrative review. Characteristics of Included Studies. Abbreviations: OCEBM, Oxford Centre for Evidence-Based Medicine.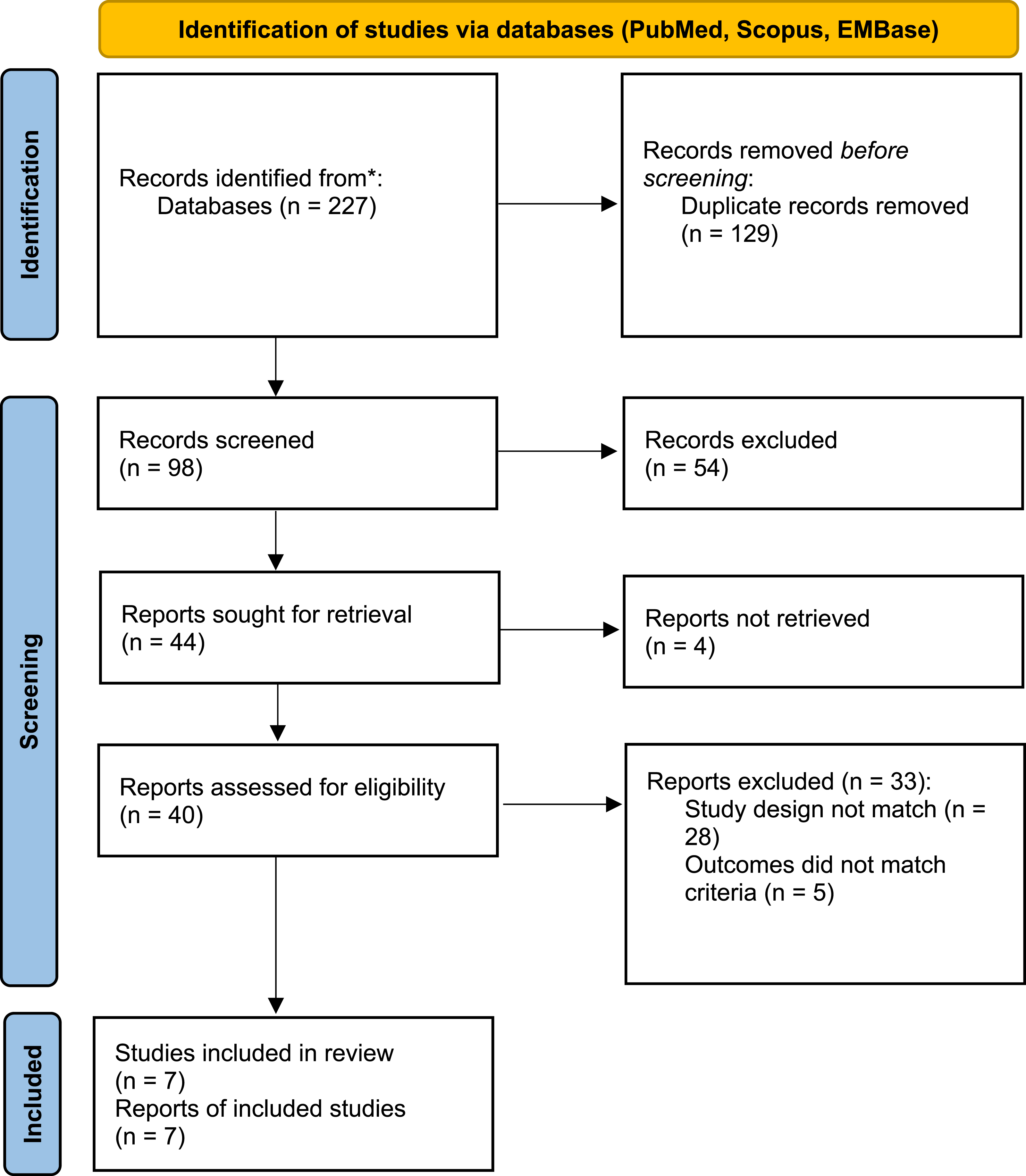
Risk of Bias Assessment.
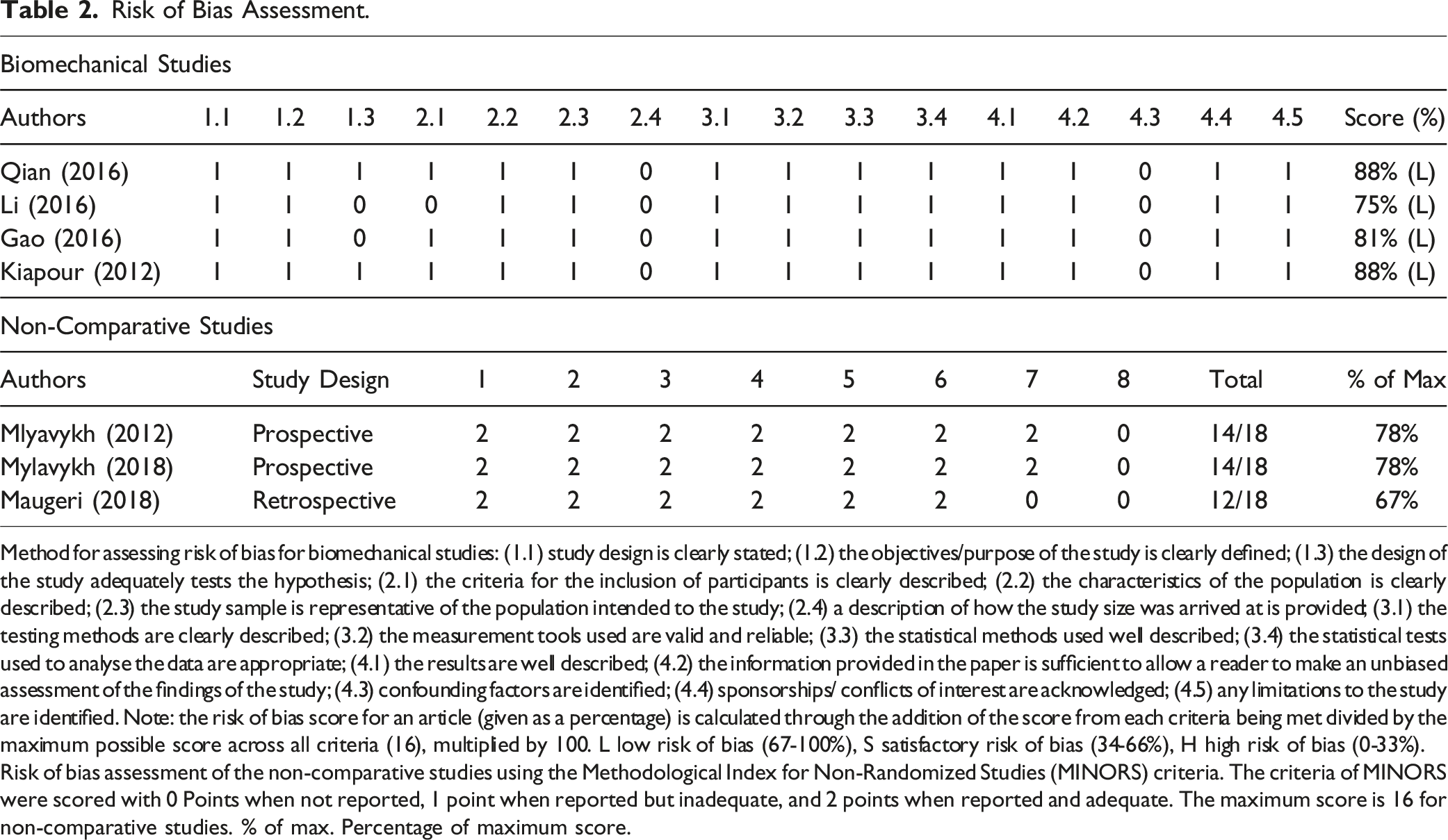
Method for assessing risk of bias for biomechanical studies: (1.1) study design is clearly stated; (1.2) the objectives/purpose of the study is clearly defined; (1.3) the design of the study adequately tests the hypothesis; (2.1) the criteria for the inclusion of participants is clearly described; (2.2) the characteristics of the population is clearly described; (2.3) the study sample is representative of the population intended to the study; (2.4) a description of how the study size was arrived at is provided; (3.1) the testing methods are clearly described; (3.2) the measurement tools used are valid and reliable; (3.3) the statistical methods used well described; (3.4) the statistical tests used to analyse the data are appropriate; (4.1) the results are well described; (4.2) the information provided in the paper is sufficient to allow a reader to make an unbiased assessment of the findings of the study; (4.3) confounding factors are identified; (4.4) sponsorships/ conflicts of interest are acknowledged; (4.5) any limitations to the study are identified. Note: the risk of bias score for an article (given as a percentage) is calculated through the addition of the score from each criteria being met divided by the maximum possible score across all criteria (16), multiplied by 100. L low risk of bias (67-100%), S satisfactory risk of bias (34-66%), H high risk of bias (0-33%).
Risk of bias assessment of the non-comparative studies using the Methodological Index for Non-Randomized Studies (MINORS) criteria. The criteria of MINORS were scored with 0 Points when not reported, 1 point when reported but inadequate, and 2 points when reported and adequate. The maximum score is 16 for non-comparative studies. % of max. Percentage of maximum score.
Grading using the OCEBM scale demonstrated that both prospective studies by Mylavykh et al. scored a level of evidence rating of 4, while Maugeri et al. also scored a rating of 4. All biomechanical and/or computation studies, which include Qian, Li, Gao, and Kaipour et al., were given a level of evidence rating of 5.
The mean MINORS score for applicable studies (Mlyavykh 2013, Mylavykh 2018, Kiapour et al.) was 13/16 with a range of 12 to 14. Following risk of bias assessment for all biomechanical/computational studies, all 4 articles were assessed as having a low risk of bias (≥67%).
How Does Pedicle Lengthening Influence Spine Biomechanics?
Kiapour et al 16 examined the effects of pedicle lengthening at the L-4 and/or L-5 levels on lumbosacral cadaveric specimens (L1-S1). 16 Range of motion (ROM) was compared between the intact state and 3 pedicle-lengthening scenarios (L-4, L-5, and L4-5) in flexion-extension, lateral bending, and axial rotation. Additionally, a kinematic profiler device was used to expose lumbosacral specimens to a 400-N compressive load in flexion-extension. Results of this testing revealed no significant differences in ROM in all 3 motion planes. Load-displacement curves for the L4-5 level also generated similar patterns between the intact spine and all 3 lengthening scenarios (intact, L-4, L-5, and L-4 and L-5 pedicle osteotomies).
Gao et al 17 investigated the effect of PLO on coronal and sagittal alignment with computed tomography (CT) scans of cadaveric lumbar vertebral specimens with 3 mm lengthening at the L3, L4, and L5 pedicles. 17 No differences in coronal cobb angle, segmental lordosis, and lumbar lordosis derived from both sagittal Cobb and Harrison’s posterior tangent angles were observed at any segments from L2-3 to L5-S1. Additionally, the relative displacements of the L3, L4, and L5 vertebrae postoperatively were not significantly different (P > 0.05 for all measures).
How Does Pedicle Lengthening Enlarge Canal and Foraminal Dimensions?
Compiled Data of Outcomes From Selected Biomechanical and Computational Studies.
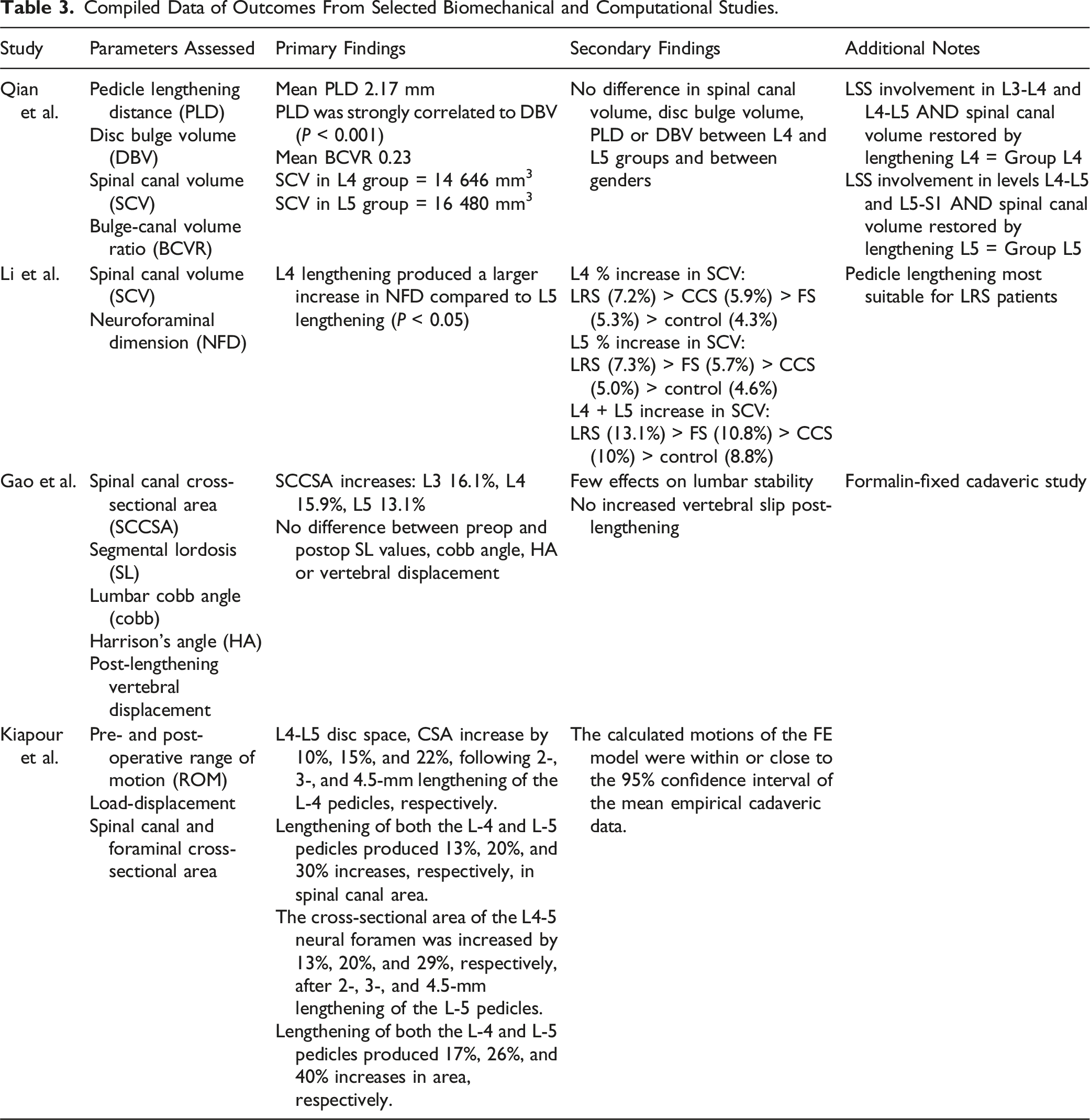
Qian et al. conducted a CT-based study with reconstructions using the Mimics software. The authors simulated pedicle lengthening at a mean distance of 2.17 mm (0.5 – 4.8 mm) and bulge-canal volume ratio (calculated as R = Vb/Vc, where Vb = disc bulge volume and Vc = canal volume) of 0.23 (0.05 – 0.48), with most bulge-canal ratios and lengthening distances falling between 0.10 – 0.30 and 1.0 – 3.0 mm, respectively. 18 They then derived an equation to predict desired pedicle lengthening distance for a given R value as follows: L = 0.06 + 9.06 R, where L equals the pedicle lengthening distance.
Li et al 19 simulated pedicle lengthening at 1, 2-, 3-, 4-, and 5-mm intervals about the L4 and L5 pedicles to assess changes in spinal canal volume (SCV) and neural foramen dimension (NFD) in CT-based cadaveric model in patients with lateral recess (LRS), foraminal stenosis (FS), and central canal stenosis (CCS). 19 The largest changes in SCV were seen in the LRS group after lengthening in all configurations, with 7.2%, 7.3%, and 13.1% per-millimeter increases following lengthening at L4, L5, and L4-L5, respectively. Per-millimeter SCV gains of at least 5% were noted in all other scenarios aside from the control group. Regarding NFD, lengthening at L4 alone at all distances caused the greatest increases in the FS groups at 14.3% per-millimeter. Lengthening of 1 mm at L5 alone caused the greatest increases in NFD in the control group, while lengthening from 2 to 5 mm caused the greatest increase in the CCS group. On average, lengthening at L5 produced per-millimeter gains of 10.4% and 9.7% in the CCS and LRS groups, respectively. Finally, lengthening at L4 and L5 at all distances caused the greatest increase in the FS group at 16.9% per millimeter. Furthermore, the most significant increases in SCV (CCS: 10.0%, LRS 13.1%, FS 10.8%) and NFD (CCS: 15.5%, LRS 14.6%, FS 16.9%) across all models occurred after lengthening of both the L4 and L5 levels.
Patient Reported Outcomes following Pedicle Lengthening
Compiled Data of Outcomes From Selected Clinical Studies.
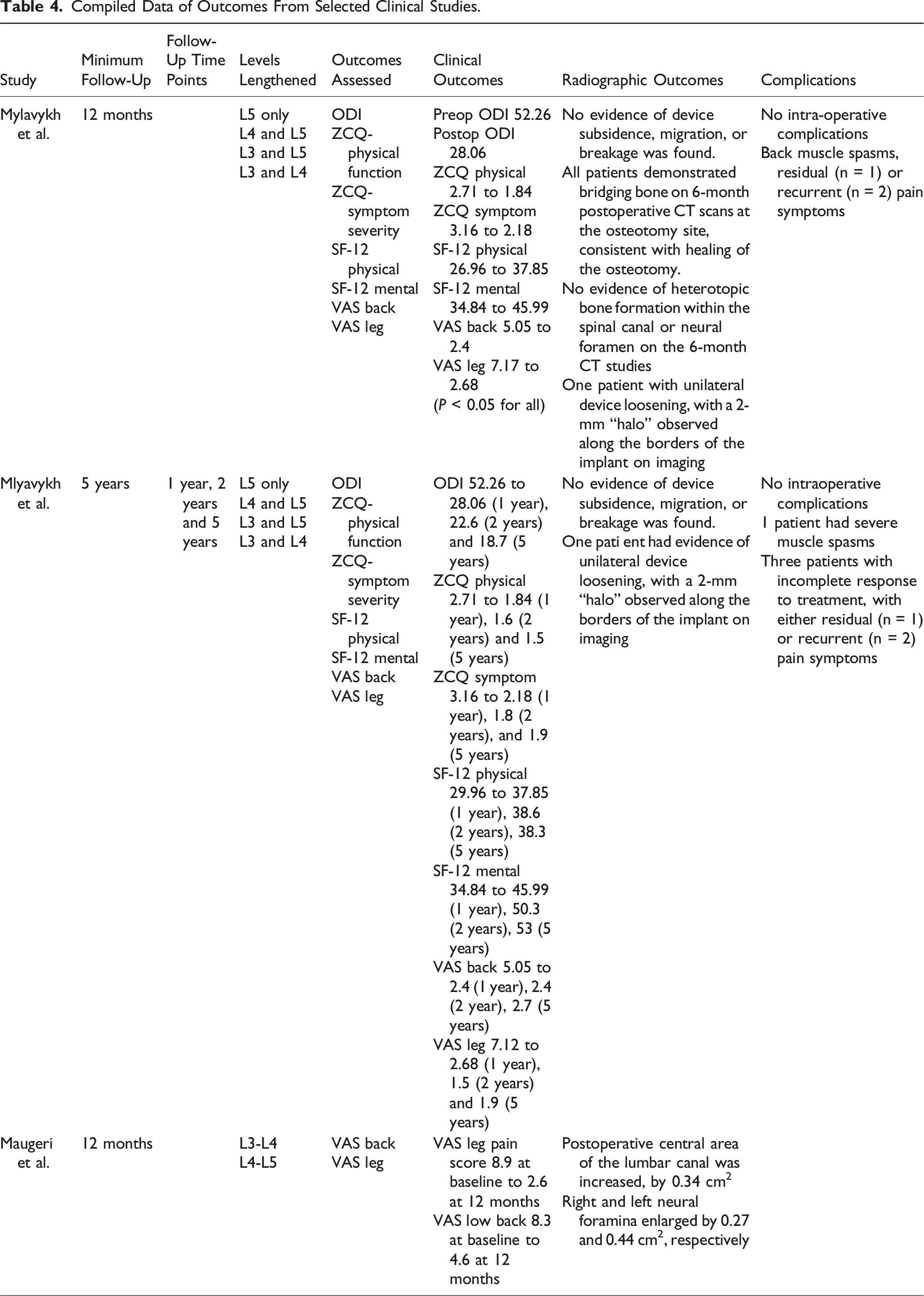
Maugeri et al. conducted a single-center study evaluating VAS back and leg scores in a cohort of 7 patients treated for LSS. 22 VAS back (preoperative: 8.3 vs 12-month: 4.6, P < 0.01) and VAS leg (preoperative: 8.9 vs 12-month: 2.6, P < 0.05) scores both significantly improved compared to baseline. Canal dimensions increased considerably, with mean increases in central canal area of 30.4%, right foramen of 36.5%, and left foramen of 59.5% postoperatively (P < 0.05).
Complication Rates
In their 12-month pilot study involving a cohort of 19 patients, Mlyavykh et al. observed no intraoperative complications including dural tears, nerve root injuries, or new neurologic deficits. 20 4 postoperative adverse events were encountered. One patient experienced severe low-back muscle spasms which resolved within 24 hours. Three others reported incomplete response to treatment with residual symptoms potentially related to residual lumbar stenosis. All 3 patients underwent open lumbar laminectomy between 6-18 months after the index procedure, with 2 receiving posterolateral fusion for concomitant degenerative spondylolisthesis. Complication rates remained low in Mlyavykh’s 5-year trial including 20 patients, except for 1 bone screw loosening event in a patient with severe medical comorbidities (ASA class IV), later found to have severe osteoporosis, an overlooked exclusionary criterion for the study. 21 Maugeri’s study reported no complications, with no dural tears, cerebrospinal fluid leaks, bleeding, or wound-healing complications noted. 22
Regarding intraoperative measures, Mlyavykh et al. described a mean procedure time of 63 minutes for a 1-level (2 pedicles) procedure and 118 minutes for a 2-level (4 pedicles) procedure. 20 Only several lightly bloody sponges used for wound cleaning were noted, and no suction was involved during the procedure.18,19 Radiographically, all patients in Mylavykh’s study had bridging bone at their osteotomy sites on 6-month postoperative spiral CT. Additionally, Maugeri observed no instances of device subsidence, migration, breakage, or heterotopic ossification. 22 Patients may mobilize quickly after surgery, with Maugeri’s cohort mobilized at 24 hours after the procedure, allowed to ambulate on postoperative day 2, and discharged on postoperative day 3 with a nonrigid lumbosacral vest to be worn for 40 days thereafter.
Discussion
LSS can lead to significant patient disability resulting in limited ability to stand or ambulate for more than a couple minutes. Nonsurgical treatment incorporates a multimodal approach of manual therapy, anti-inflammatory medications, and steroid injections, which may provide some patients significant relief.23-26 Unfortunately, these options may be ineffective compared to surgical decompression for certain patients, especially those with severe LSS, medical comorbidities, and advanced age.27-29 Notably, the Spine Patient Outcomes Research Trial (SPORT) noted that nonoperative management was inferior to surgical decompression at a minimum of 4 years postoperatively for symptomatic LSS. 30
Prior studies have attempted to correlate radiographic findings with the presence of symptomatic LSS. Hamanashi et al. reported that intermittent claudication occurs when the dural tube CSA measures 100 mm2 or less in 2 or 3 segments from L2-L3 and L4-L5. 31 Bolender corroborated these findings, demonstrating that dural CSA in CT myelogram images of patients with LSS measured similarly compared to 180 mm2 in asymptomatic patients. 32 Thus, sufficient expansion of the spinal canal must be performed to achieve clinical improvement, whereas failure to do so may portend poor outcomes. For example, Yamazaki et al. described poor post-operative clinical outcomes in patients with limited dural expansion, grouping of the cauda equina, and decreased dural cross-sectional area. 33
Open laminectomy, the traditional treatment for LSS, may bestow significant improvements in lower extremity pain, intermittent claudication, and quality of life.34-36 Instrumented fusion may also be indicated for LSS secondary to spondylolisthesis and degenerative scoliosis, or for iatrogenic instability.37-39 However, these methods carry risks for significant blood loss, wound infection, perineural scarring, medical complications, and prolonged recovery, with life-threatening complication rates ranging from 2.3% for those having decompression alone to 5.6% among patients undergoing complex fusions.37,39 Wide laminectomies may also harm posterior soft tissue structures and impart iatrogenic instability, decreasing the lumbar spine’s shear force to failure, thus hastening degeneration.16,40-42 Additionally, prior literature suggests that suboptimal clinical outcomes may occur following laminectomy in patients with hypermobility or excessive vertebral slippage in the sagittal plane.43,44
Minimally invasive surgery (MIS) boasts advantages including less muscle damage, blood loss, and recovery time.6-9 Podichetty described successful outcomes with minimally invasive techniques in the treatment of LSS in an elderly population. 11 In an evaluation of over 10 000 patients in the Scoliosis Research Society Database, Fu et al. observed significantly lower rates of death and neurologic complications in patients receiving minimally invasive decompression compared to open surgery for LSS. 8 Over the last 2 decades, numerous MIS techniques have been introduced, including interspinous implants, tubular-retractor based approaches, and endoscopic- or microscopic-assisted “over the top” decompression through a unilateral laminotomy. 45
In the present review, all relevant studies reported significant increases in central canal and neuroforaminal dimensions following PLO which was shown to occur in a length-dependent manner. 18 Agreement was found between studies that pedicle lengthening be performed at the levels cranial and caudal to the stenotic region to maximize gains in spinal dimensions, yielding central canal and neuroforaminal area gains of up to 30% and 40%, respectively.16,18 Through computational and cadaveric means, PLO maintained spinal kinematics akin to the intact spine without increased risk of iatrogenic instability or listhesis, posing mechanical advantages compared to open laminectomy, fusion, and interspinous spacers.16,17 While dural tube measurements were not specifically performed, observed spinal dimension changes likely provided sufficient decompression given the extent of clinical improvement seen across multiple trials. Existing studies report that PLO grants considerable improvements in PROMs immediately postoperatively that persist up to 5 years post-operatively with virtually no major complications and low conversion rates to open decompression with fusion.19-21
Despite the above advantages, pedicle lengthening osteotomy is a relatively new procedure with limited documentation. Clearly, there is much to be learned regarding this technique. Studies comparing the outcomes of pedicle lengthening osteotomy with laminectomy are needed to understand how this procedure compares scientifically in a well-controlled environment. Additionally, as seen in Mylavykh’s 12-month pilot study, residual and recurrent symptoms may persist following pedicle lengthening which could be attributable to hypertrophied ligamentum flavum, epidural lipomatosis, and higher grade listhesis causing persistent central stenosis which may not be fully addressed with pedicle lengthening alone. The use of pedicle osteotomies for spinal deformity surgery and vertebral cancer resection is similarly new and without sufficient outcome data to allow for comparison of these techniques to traditional approaches. Despite these limitations, the availability of the pedicle bone saw in the surgeon’s armamentarium provides a potentially useful method that is worthy of further study.
Conclusion
Pedicle lengthening osteotomy is a novel technique that adds another potential treatment option for patients with LSS. Translation research has suggested this technique does not significantly affect spinal biomechanics. Clinically, the greatest experience has been in the setting of LSS and although there has been early clinical success with minimal complications, early return of LSS should be expected in a subset of patients. Understanding which patients most benefit from this surgery is necessary prior to universal adoption.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
