Abstract
Study Design
Prospective Study.
Objective
The objective of this investigation was to explore the abnormal expression of migration inhibitory factor antisense RNA 1 (MIF-AS1) in lumbar disc herniation (LDH) patients and its relationship to the degree of pain and inflammatory response in LDH, as well as the molecular mechanism of its involvement in LDH.
Methods
This study included 50 patients with LDH. The expression levels of MIF-AS1 were detected by RT-qPCR. The LDH model was constructed in SD rats, and the paw withdrawal mechanical threshold (PWMT) and paw withdrawal thermal latency (PWTL) of LDH rats were detected by behavioral experiments. Enzyme-linked immunosorbent assay (ELISA) was used to detect the concentrations of TNF-α, IL-6, and IL-1β. The targeted regulatory relationships between MIF-AS1 and miR-185-5p, miR-185-5p and VEGFA were verified by a dual-luciferase reporter gene assay.
Result
The expression of MIF-AS1 was up-regulated in LDH patients and correlated with the degree of pain in patients. Low expression of MIF-AS1 reduced the degree of pain and inflammation in LDH rats. In addition, MIF-AS1 may regulate pain and inflammation induced by LDH by modulating the miR-185-5p/VEGFA axis.
Conclusion
MIF-AS1/miR-185-5p/VEGFA axis may be a therapeutic target for LDH.
Introduction
Lumbar disc herniation (LDH) constitutes a prevalent condition within the realm of spinal pathologies, arising from the deterioration and migration of nucleus pulposus. It has garnered global recognition as a significant health concern and is frequently diagnosed as the primary indicator of lumbar degenerative disorders. 1 LDH frequently serves as a primary cause for lower back discomfort, neural impairment, and pain in the gluteal and lower limb regions. Herniation is characterized by the protrusion of the gel-like nucleus pulposus within an intervertebral disc through its fibrocartilaginous ring, which results in pressure being exerted on the neural structures. The effects of LDH can vary widely in intensity, impacting patients from experiencing minor low back and gluteal pain to more severe symptoms that may include an inability to walk and the development of cauda equina syndrome. 2 To date, nonsurgical interventions have remained the primary option for managing LDH. Nonetheless, the approaches primarily alleviate the discomfort associated with LDH rather than providing a curative solution. 3 Surgery is deemed appropriate only for patients with a protracted history of LDH, those experiencing severe pain that markedly compromises their quality of life, or those who have not responded to conservative therapies. 4 Given that LDH significantly affects the work and life of patients, accompanied by substantial medical expenses and considerable physical and psychological burden, there is an urgent need to discover more potent therapeutic strategies.
In the past few years, the involvement of long non-coding RNA (lncRNA) in orthopedic diseases has been a subject of increasing investigation and documentation. For instance, certain lncRNAs are involved in the progression of osteoporosis, including SNHG14, 5 PCBP1-AS1, 6 and SNHG1. 7 Additionally, a subset of lncRNAs exerts significant regulatory functions in the process of fracture healing, such as MAGI2-AS3, 8 HAGLR, 9 and HOTAIR. 10 In addition, some studies have also focused on the abnormal expression of lncRNAs in lumbar disc degeneration (LDD), for example, the expression of lncRNA ZFAS1 is significantly elevated in patients with LDD, making it a prospective marker for predicting the prognosis of LDD. 11 Simultaneously, an increasing number of investigations into lncRNA involvement in LDH have been carried out.1,12
As a novel lncRNA, migration inhibitory factor antisense RNA 1 (MIF-AS1) has been documented to enhance cell growth and attenuate apoptosis in gastric cancer cells. 13 Additional researches have highlighted the significance of MIF-AS1 in the progression of ovarian 14 and breast cancers. 15 It is worth noting that a study conducted to detect lncRNAs linked to the intervertebral disc degeneration (IVDD) and validated eight lncRNAs that were increased in the nucleus pulposus (NP) cells of IVDD, with MIF-AS1 being one of them. 16 However, the role of MIF-AS1 in LDH and its regulatory mechanism have not been elucidated.
Studies have shown that miR-185-5p modulates inflammatory responses and autophagy. 17 Moreover, miR-185-5p also plays a very important regulatory role in cancer. 18 The research also found that miR-185-5p is involved in the process of osteogenic differentiation. 19 It is worth noting that miR-185-5p is downregulated in the chronic constrictive injury induced rat model and is involved in the inflammatory response. 20 By predicting the miRNAs regulated by MIF-AS1 through an online website, we found that miR-185-5p was one of them. However, the mutual regulatory effect between MIF-AS1 and miR-185-5p has not yet been confirmed.
Therefore, this study explored the abnormal expression of MIF-AS1 in LDH patients and its relationship to the degree of pain and inflammation in LDH patients, as well as the molecular mechanism of its involvement in LDH.
Materials and Methods
Study Patients
We included 50 patients who underwent discectomy in author’s institution in this study. All these patients had been diagnosed with lumbar disc herniation (LDH) and presented with unilateral sciatica, as confirmed by magnetic resonance imaging (MRI). Patients suffering from LDH presented with the following symptoms: (1) suffering from lower back pain; (2) experiencing pain in the lower lumbar region accompanied by localized typical sciatica; (3) displaying a reduced amplitude of lumbar flexion; and (4) encountering greater difficulty in performing the straight leg elevation test and intensive test. 21 Individuals with intricate hematological conditions, autoimmune disorders, malignancy, injuries, rheumatoid arthritis, and associated lumbar conditions were excluded from participation in this study. An additional 45 patients who had surgical treatment for lumbar fractures (except for LDH) were selected to serve as a control group.
NP tissue was obtained from all study subjects during surgery and placed in tissue containing 10% fetal bovine serum (FBS; Gibco). This study protocol was approved by the Ethics Committee of author’s institution, and all participants provided informed consent.
Assessment of Clinical Severity
The level of pain was assessed via the visual analog scale (VAS) score, the functional ability was evaluated using the Japanese Orthopedic Association (JOA) score. In the case of VAS scores, a rating of 0 signifies the absence of pain, while a rating of 10 indicates extremely severe pain. The score increased in direct proportion to the intensity of the pain experienced. 22 The JOA score spans a range from 0 to 29, with higher values indicating reduced pain levels and improved functional ability. 23
LDH Models
The skin and muscle were incised longitudinally along the L4-L6 spinous process with a scalpel, and the paraspinal muscle tissue was bluntly separated. The left inferior articular process of L5, the superior articular process of L6, and the half-laminae of L5 were removed, exposing the L5 and L6 nerve roots. After the rat tail was disinfected with iodine and ethanol, the skin was cut lengthwise along the tail, the knife tip was inserted into the fibrous ring laterally to remove the nucleus pulposus of the tail vertebrae (C7 or C8), and the autogenous nucleus pulposus was placed at the exposed nerve roots to cause mechanical compression. The L5 and L6 nerve roots were filled with an appropriate amount of gelatin sponge and sutured layer by layer. After the wound was sutured, the rat wound was irradiated under an infrared lamp for 1 h to promote wound healing. Behavioral tests were performed 1 day before molding and 1, 3, 5, and 7 days after molding. Finally, the dorsal root ganglion (DRG) tissue of rats was taken for relevant detection.
Animals and Experimental Design
All experiments were carried out in adherence to the NIH Guide for the Care and Use of Laboratory Animals. Furthermore, the study complied with the pertinent regulations set forth by the International Association for the Study of Pain (IASP) to minimize discomfort in rats. A total of 58 female Sprague-Dawley (SD) rats were obtained from Shanghai Laboratory Animal Centre, with a body weight range of 290-380 g and accommodated in a laboratory environment maintained at a temperature of 20°C–25°C. They were kept on a 12-hour light and dark cycle and had unrestricted access to food and water.
SD rats were randomly divided into Sham group and LDH group. Subsequently, to explore the effect of MIF-AS1 in LDH rats, we further divided the model group into LDH+ sh-NC group (a negative control lentiviral vector) and LDH+ sh-MIF-AS1 group (a lentivirus that inhibits MIF-AS1 expression). In an alternative set of rats, antagomiR-185-5p (lentivirus that inhibits miR-185-5p expression) and antagomiR-NC (corresponding control miRNA lentivirus) were injected intrathecally into LDH + sh-MIF-AS1 rats. In addition, to study the regulatory role of miR-185-5p and VEGFA, agomiR-185-5p (miR-185-5p overexpressing lentivirus), agomiR NC, overexpressing plasmid pcDNA3.1-VEGFA (oe-VEGFA) and its negative control pcDNA3.1-NC (oe-NC) were injected intrathecally into LDH rats.
Intrathecal Administration
The animals were induced into anesthesia through the inhalation of 1.5% isoflurane and were fixed in a sitting position. The L5-L6 intervertebral spaces were identified as the insertion points for the needle. Starting at the site of hip joint that was probed by the left hand, the needle was positioned at a point more than 3 cm vertically distant from the highest point on the dorsum, after which the microsyringe needle was carefully inserted in a straight line into the area. When the tail whip occurred, the jugular vein was constricted, and the injection was initiated upon the appearance of cerebrospinal fluid as the plunger was pulled back, delivering a drug solution with a volume of 20 μL (5 μg).
Determination of Paw Withdrawal Mechanical Threshold (PWMT)
The experiment was carried out in a quiet environment. Rats were accommodated in a custom-made glass box featuring a metallic mesh floor. Following a 30-minute acclimation period, the rats’ paws were stimulated using a Von Frey filament, and as the foot withdrew, the Von Frey value was noted. The test was conducted in triplicate, with a 5-minute interval separating each application of the stimulus. Subsequently, the mean value from these three trials was calculated to determine the mechanical pain threshold for the rat.
Determination of Paw Withdrawal Thermal Latency (PWTL)
A heat pain tester was used to detect the PWTL of both posterior feet of rats 1 day before and 1, 3, 5 and 7 days after modeling. The light source was directed at the middle of the feet, and the irradiation was stopped when foot retraction or foot licking occurred. The reaction time was recorded as PWTL, and the automatic cutting time of the instrument was 25 s. The test was repeated 3 times per foot, with an interval of 5 min.
Real-Time Quantitative Reverse Transcription Polymerase-Chain-Reaction (RT-qPCR)
Total RNA was extracted from tissues using Trizol regent (Thermo Fisher Scientific, Waltham, USA). The High-capacity cDNA Synthesis Kits (Genstar, Beijing, China) were employed for the synthesis of cDNA. The relative RNA expressions analyze using the SYBR Green PCR kit (Takara, Japan) on a 7500 fast real-time PCR system (Applied Biosystems, USA). GAPDH and U6 were used as internal reference genes. Relative quantification analysis was performed by using the 2−△△CT method.
Enzyme-Linked Immunosorbent Assay (ELISA)
After the tissues were sufficiently cleaved, the supernatant was collected by centrifugation (10,000 g, 5 min). The concentration of tumor necrosis factor α (TNF-α), interleukin (IL)-6, and IL-1β in tissues of rats were measured by enzyme-linked immunosorbent assay (ELISA) kit. The OD values were read at a wavelength of 450 nm by means of a microplate analyzer (BioTek, USA). The ELISA assay was conducted threefold for accuracy.
Dual-Luciferase Reporter Analysis
The binding sites of MIF-AS1 and miR-185-5p, miR-185-5p and VEGFA were predicted using the online lncRNASNP v3 and starBase database. The wild-type (wt) or mutated (mut) sequences of MIF-AS1 and VEGFA containing the binding sites of miR-185-5p or not were designed and inserted into the dual-luciferase reporter vector pmirGLO. Then these reporter vectors were co-transfected with miR-185-5p mimics, miR-185-5p inhibitors, mimic NC or inhibitor NC into HEK-293T cells, respectively. The luciferase activity was determined by Dual-luciferase reporter assay system.
Statistical Analysis
Statistical analysis was performed using GraphPad version 9.0 software. For comparisons between two groups, Student’s t test analysis was used. Additionally, one-way or two-way ANOVA was employed to analyze the significant differences among multiple groups of data.
Result
Comparison of Basic Data Between the Two Groups
Comparison of Basic Data Between the Two Groups.

BMI, Body Mass Index; VAS, Visual Analogue Scale; JOA, Japanese Orthopaedic Association. Data were expressed as mean ± standard deviation or n.
The Expression Level of MIF-AS1 and its Relationship with VAS and JOA Scores
The upregulation of MIF-AS1 expression was observed in the NP tissues of those suffering from LDH (Figure 1A). There was a positive association between the expression level of MIF-AS1 and the VAS scores among LDH patients (Figure 1B), while a negative relationship existed between MIF-AS1 expression and the JOA score of the LDH patients (Figure 1C). The expression level of MIF-AS1 and its relationship with VAS and JOA scores. Expression of MIF-AS1 in control group and LDH patients (A). Correlation between the expression level of MIF-AS1 and VAS scores (B). Correlation between the expression level of MIF-AS1 and JOA scores (C).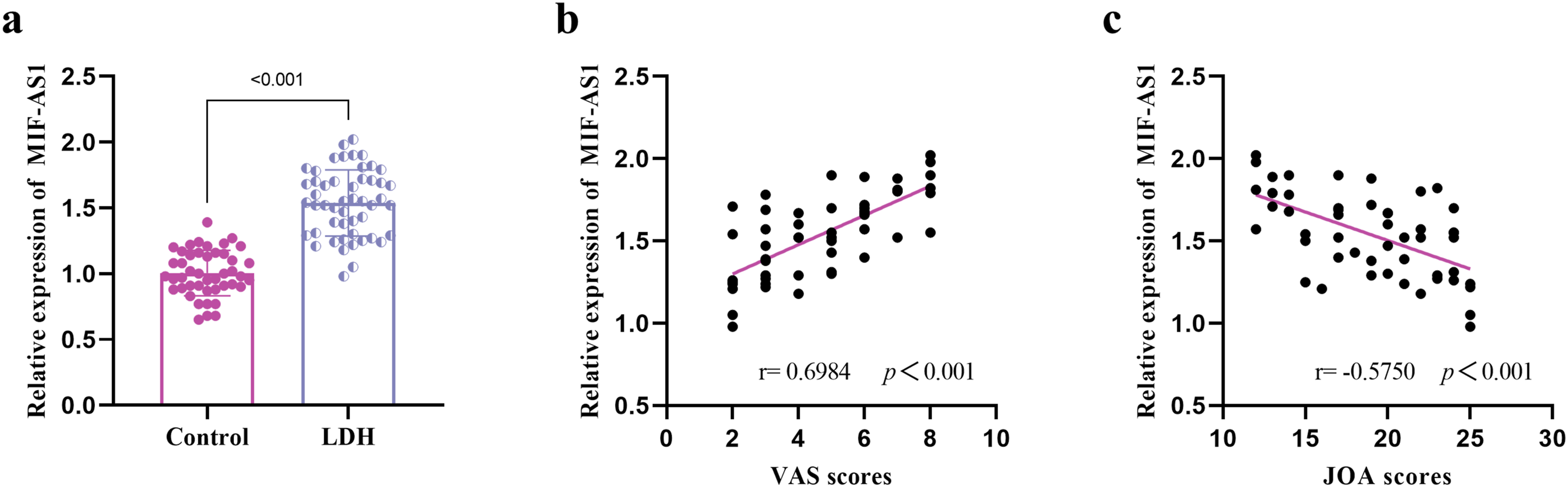
Effect of MIF-AS1 on Pain Degree and Proinflammatory Factors in LDH Rats
In contrast to the Sham group, the levels of MIF-AS1 were increased in the DRG tissues of the LDH rat model. Furthermore, the injection of sh-MIF-AS1 lentivirus resulted in a reduction in the expression of MIF-AS1 (Figure 2A). When compared to Sham rats, the LDH rats exhibited marked reductions in the mechanical pain threshold on the 3rd, 5th, and 7th days (Figure 2B), indicating that the LDH models were effectively established and that LDH rats are more sensitive to mechanical pain than Sham rats. sh-MIF-AS administration via intrathecal injection decreased the mechanical pain threshold in comparison to the LDH rats, suggesting that sh-MIF-AS has mitigating effects on LDH-induced pain. Consistent results were obtained from the heat pain assay (Figure 2C). Consequently, the LDH models demonstrated heightened sensitivity to pain, which can be mitigated by the intrathecal administration of sh-MIF-AS, partially restoring the mechanical and heat pain thresholds. Effect of MIF-AS1 on pain degree in LDH rats. Expression of MIF-AS1 in LDH model rats (A). Time-dependent changes of thresholds of mechanical pains of rats in different groups (B). Time-dependent changes of thresholds of heat pains of rats in different groups (C).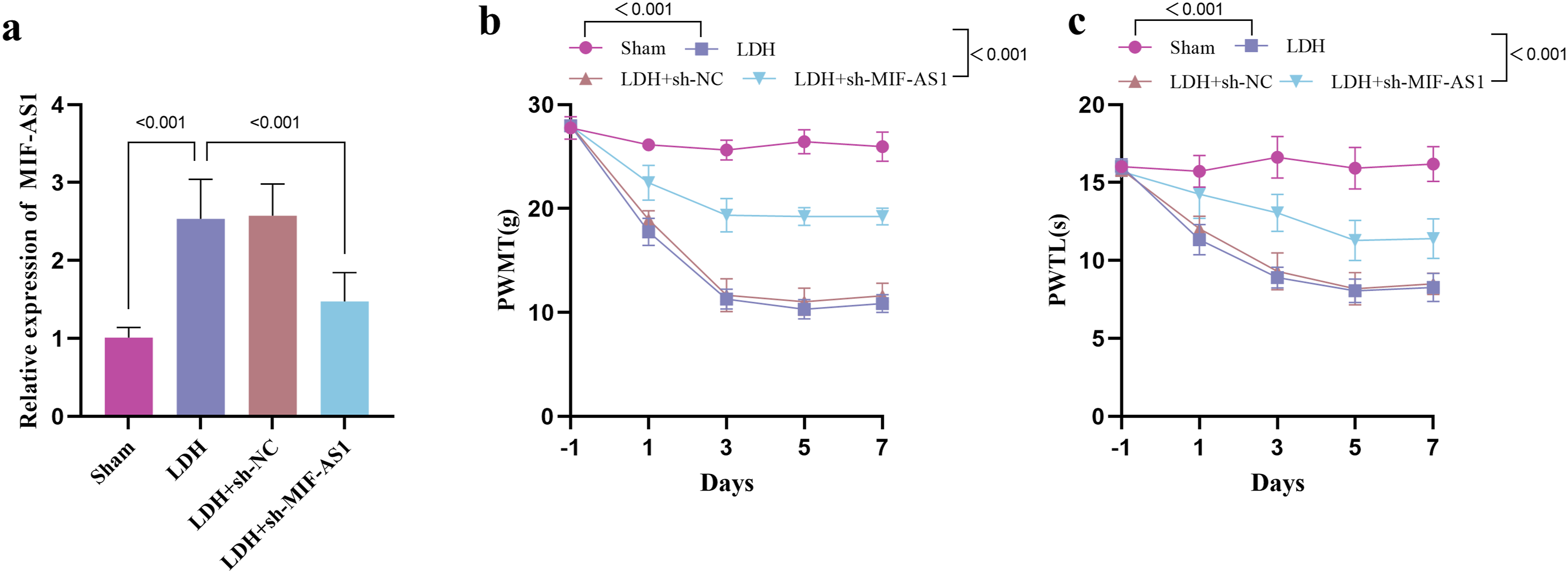
In contrast to the Sham group, the levels of TNF-α (Figure 3A), IL-1β (Figure 3B) and IL-6 (Figure 3C) were elevated in the LDH model rats, and the levels of TNF-α, IL-1β and IL-6 were decreased after the expression of MIF-AS1 was down-regulated. Effect of MIF-AS1 proinflammatory factors in LDH rats. Effects of sh-MIF-AS1 lentivirus injection on levels of TNF-α (A), IL-1β (B), and IL-6 (C) in LDH rats.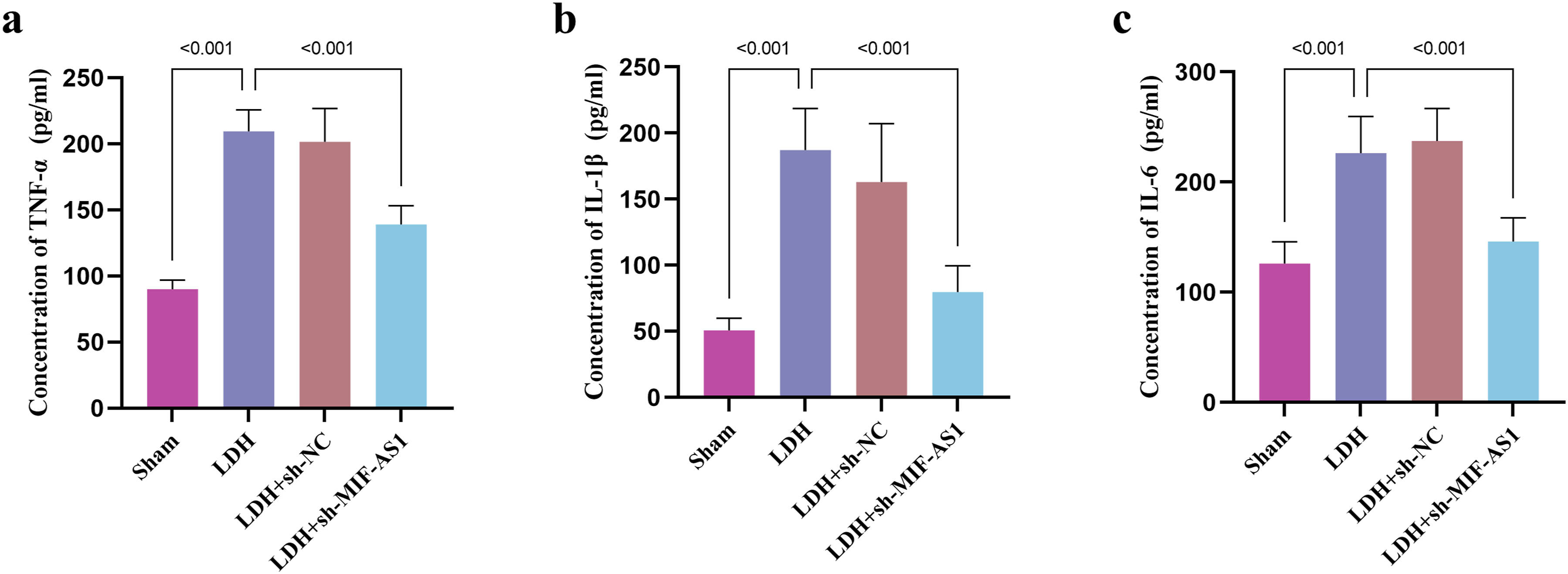
MiR-185-5p Functioned as a Target of MIF-AS1
The level of miR-185-5p was reduced in patients with LDH (Figure 4A), and there was a significant negative correlation between miR-185-5p and MIF-AS1 (Figure 4B). The dual-luciferase assay confirmed the targeting relationship between miR-185-5p and IF-AS1. The miR-185-5p mimic decreased the luciferase activity of wt-MIF-AS1, while the miR-185-5p inhibitor increased the luciferase activity of wt-MIF-AS1 (Figure 4C). Moreover, the expression of miR-185-5p was decreased in LDH rats, and the expression of miR-185-5p was up-regulated after the expression of MIF-AS1 was suppressed, but the injection of antagomiR-185-5p further decreased the expression of miR-185-5p (Figure 4D). miR-185-5p functioned as a target of MIF-AS1. Expression of miR-185-5p in control group and LDH patients (A). Correlation between the expression level of MIF-AS1 and miR-185-5p (B). Dual-luciferase reporter experiments verified the relationship between miR-185-5p and MIF-AS1 (C). MIF-AS1 regulated the expression of miR-185-5p in LDH rats (D).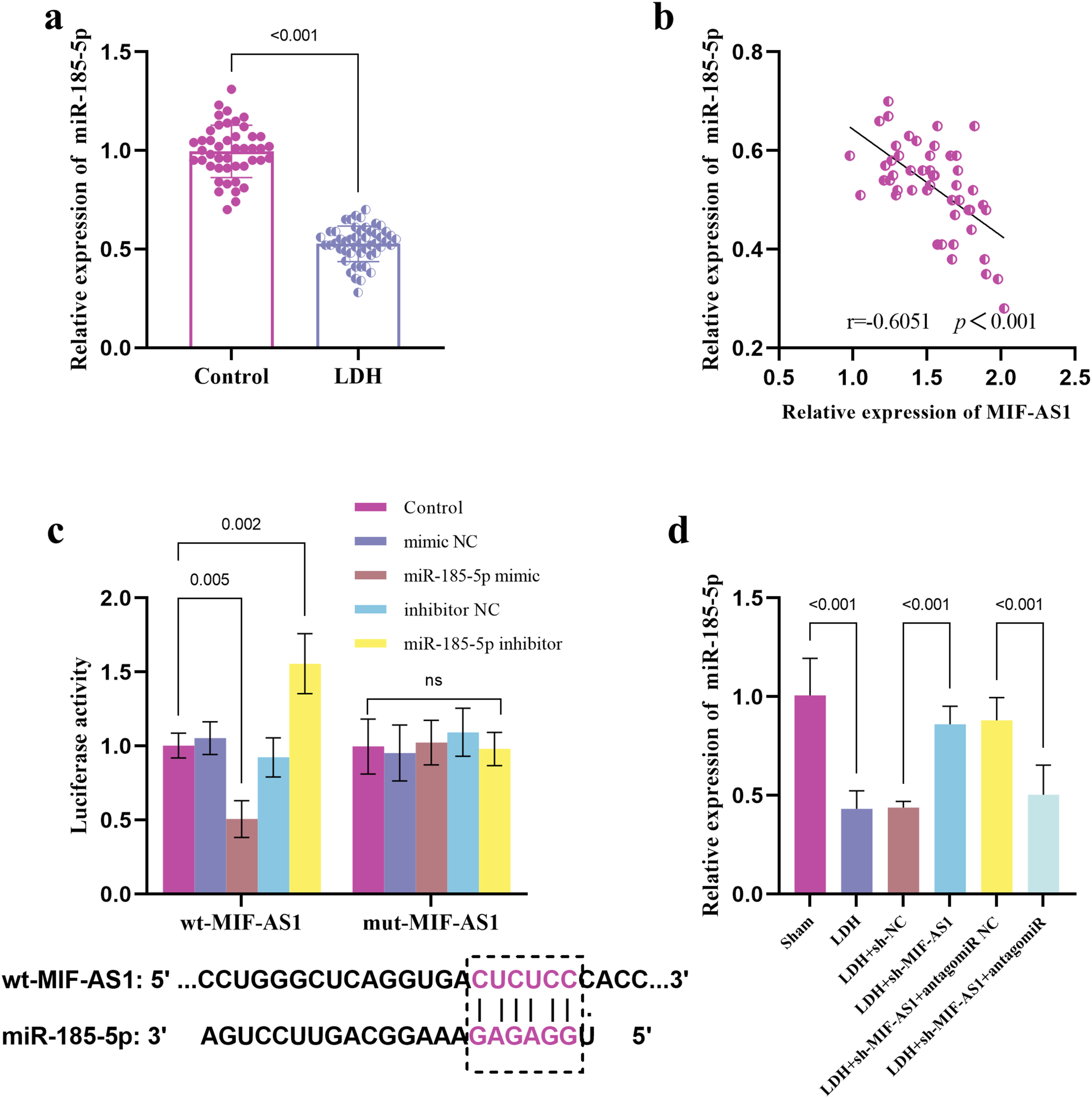
Down-Regulated miR-185-5p Reversed MIF-AS1 Inhibition on Pain and Inflammation Relief in LDH Rats
Behavioral experiments demonstrated that the suppression of miR-185-5p serves to mitigate the augmented PWMT and PWTL observed in LDH rats following the knockdown of sh-MIF-AS1 (Figure 5A and 5B). Moreover, the reduction of miR-185-5p levels countered the downregulation of TNF-α (Figure 5C), IL-1β (Figure 5D) and IL-6 (Figure 5E) induced by MIF-AS1 inhibition, thereby enhancing the inflammatory response in the LDH rat model. Down-regulated miR-185-5p reversed MIF-AS1 inhibition on pain and inflammation relief in LDH rats. Down-regulated miR-185-5p reversed the effect of MIF-AS1 inhibition on thresholds of mechanical pains (A) and thresholds of heat pains (B) in LDH rats. Down-regulated miR-185-5p reversed the effect of MIF-AS1 inhibition on TNF-α (C), IL-1β (D), and IL-6 (E) in LDH rats.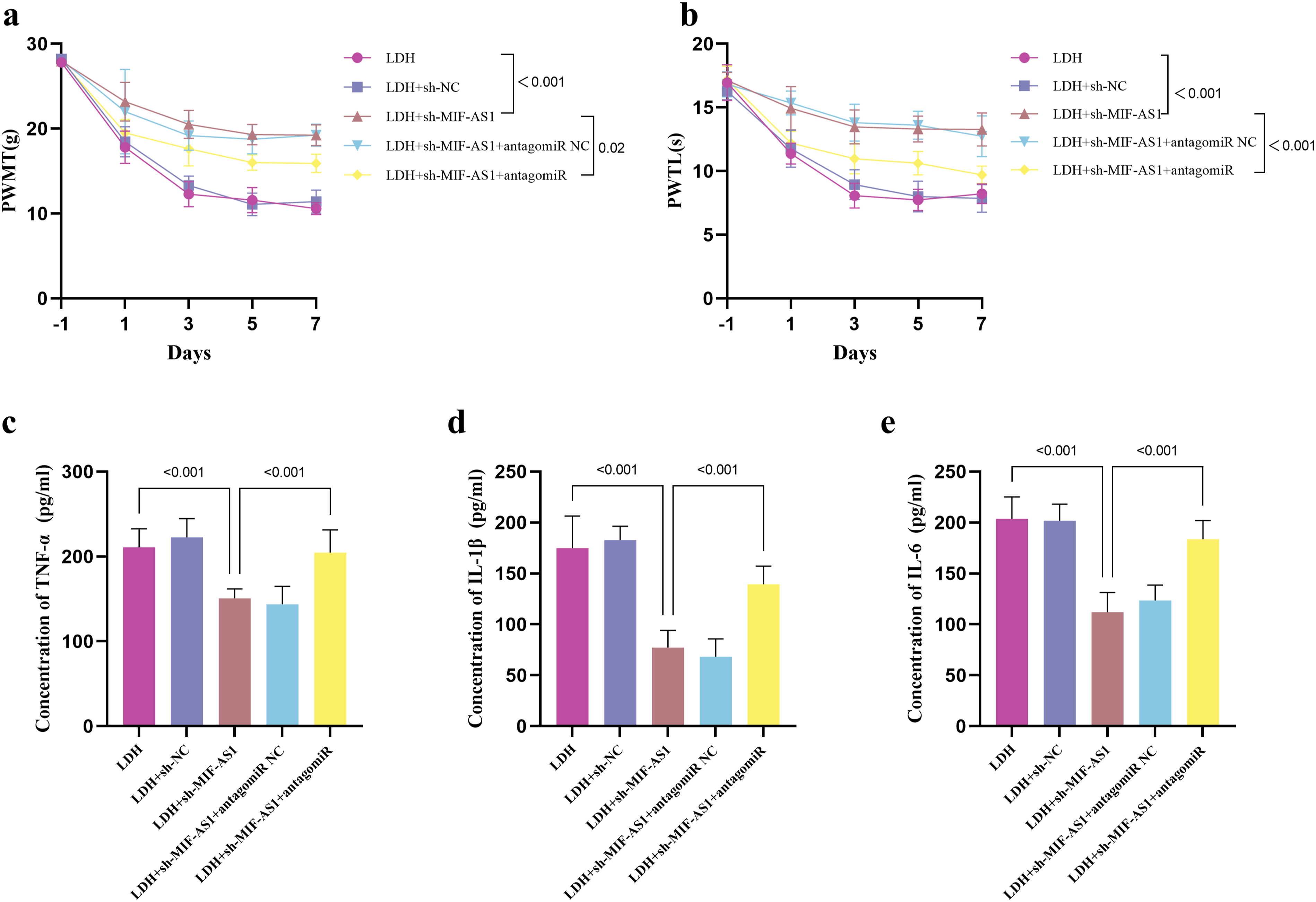
VEGFA Functioned as a Target of miR-185-5p
The level of VEGFA was elevated in those LDH patients (Figure 6A), showing a positive relationship with the level of MIF-AS1 (Figure 6B) yet a negative association with miR-185-5p levels (Figure 6C). Data from dual-luciferase assays revealed that miR-185-5p mimic diminished the luciferase activity of the wt-VEGFA, while miR-185-5p inhibitor augmented the luciferase activity of the wt-VEGFA (Figure 6D). Furthermore, in LDH rats, an upregulation of VEGFA was observed, which was reversed by the overexpression of miR-185-5p. However, this inhibitory effect of miR-185-5p on VEGFA was mitigated by the administration of oe-VEGA (Figure 6E). VEGFA functioned as a target of miR-185-5p. Expression of VEGFA in control group and LDH patients (A). Correlation between the expression level of MIF-AS1 and VEGFA (B). Correlation between the expression level of miR-185-5p and VEGFA (C). Dual-luciferase reporter experiments verified the relationship between miR-185-5p and VEGFA (D). miR-185-5p regulated the expression of VEGFA in LDH rats (E).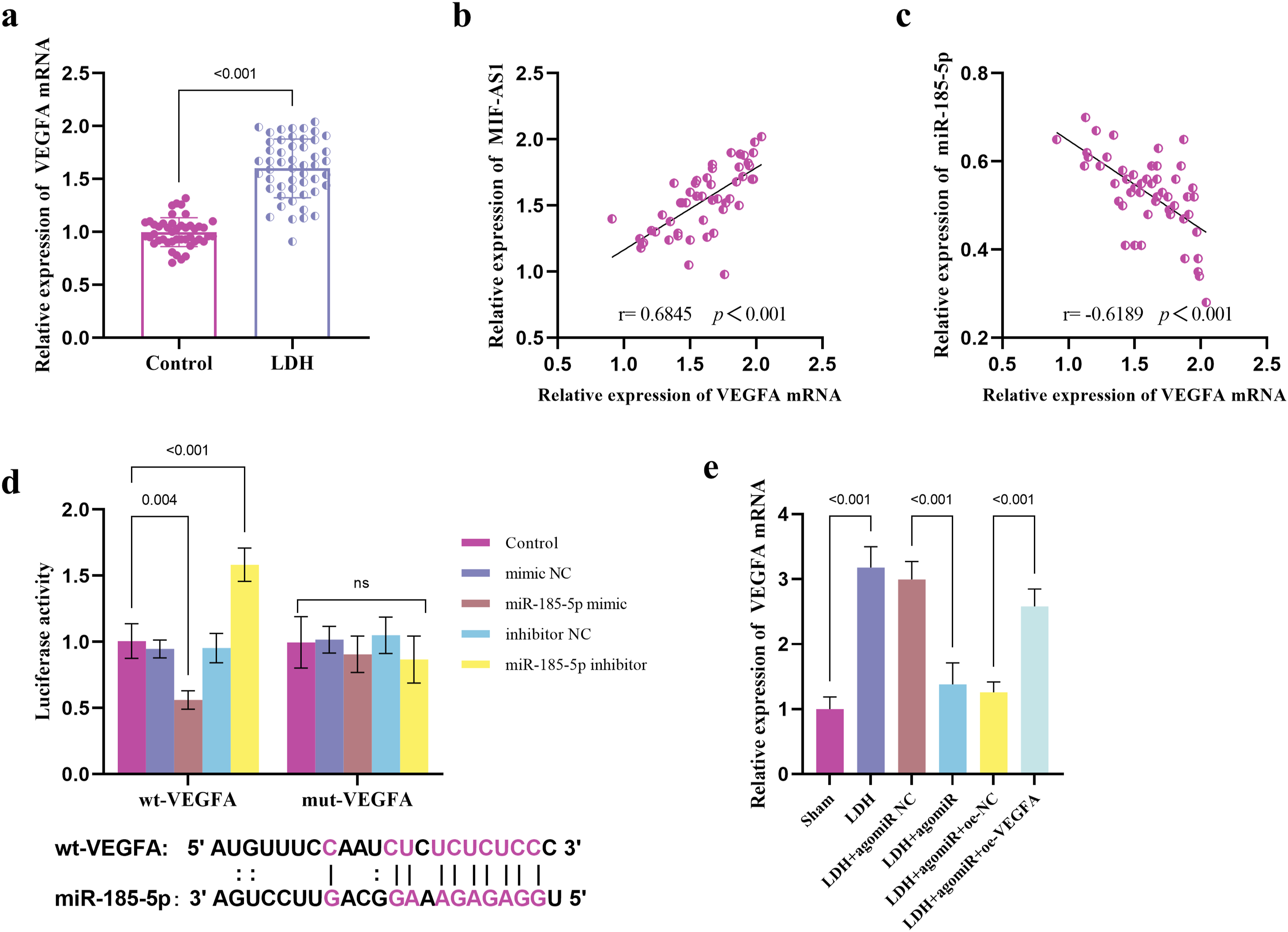
Discussion
Multiple significant discoveries regarding lncRNAs in LDH have been documented. For instance, the expression of lncRNA MT1DP is upregulated in the NP tissues patients with LDH, which contributes to the progression of LDH by reducing mitochondrial function and the capacity to scavenge reactive oxygen species. 12 Simultaneously, research has uncovered a strong association between lncRNAs and pain mechanisms. 24
We observed that the expression of MIF-AS1 was substantially elevated compared to that in the control group, suggesting that MIF-AS1 may be involved in the pathophysiology of sciatica in patients with LDH. Furthermore, our results indicated a positive correlation between the levels of local MIF-AS1 in NP tissues and VAS scores, as well as a negative correlation with JOA scores. Building on this, our study showed that increased expression of MIF-AS1 in LDH patients was associated with heightened radicular pain.
As described in the previous study, 25 the herniated tissue is highly dynamic in its expression of inflammatory molecules. The chemical irritation resulting from the release of bioactive substances by the NP, as well as an autoimmune reaction directed against the tissue itself, can both trigger inflammation This includes the release of TNF-α and other cytokines including IL-1β and IL-6, as well as the stimulation of circulating monocytes and macrophages.26,27 To investigate the impact of MIF-AS1 expression on the pain and inflammatory responses induced by LDH, we constructed an LDH model in SD rats. Our results showed that the downregulation of MIF-ASI not only relieved pain in LDH rats but also reduced the tendency of LDH-induced elevations of IL-6, IL-1β and TNF-α. Some researchers have developed LDH models and discovered that TNF-α compromises DRG neurons, leading to the occurrence of radiating pain. 28 TNF-α and IL-6 can stimulate the production of multiple pro-inflammatory cytokines, making them key markers of the extent of inflammation and tissue damage. These pro-inflammatory cytokines can initiate action potentials in peripheral nociceptor cells, leading to the influx of sodium and calcium ions, which ultimately results in pain. 29 Given the role of inflammatory cytokines in the generation and progression of pain, investigating the influence of MIF-AS1 on these pro-inflammatory mediators is of critical importance.
MicroRNAs (miRNAs) are a family of small non-coding RNAs, serving crucial functions in modulating gene expression and involving in a variety of vital physiological and pathological processes. 25 LncRNAs are recognized for their ability to bind to miRNAs, which in turn lessens the control that miRNAs exert over mRNA expression. 30 To delve deeper into the molecular mechanism through which MIF-AS1 modulates LDH-induced pain and inflammation, we sought to identify miRNAs that are controlled by MIF-AS1. Among these miRNAs, miR-185-5p stood out as a notable candidate. miR-185-5p was found to be abnormally down-regulated in disc degeneration, 31 and overexpression of miR-185-5p in NP cells significantly reduced the increased extracellular matrix (ECM) catabolism brought on by treatment with IL-1β and TNF-α. 32 The current investigation revealed that MIF-AS1 modulated the pain response and inflammatory reaction in LDH rats through the manipulation of miR-185-5p expression.
The research revealed that increased expression of vascular endothelial growth factor (VEGFA) in individuals suffering from lumbar spinal stenosis is linked to heightened inflammation and a protracted course of the illness. 33 The expression level of VEGFA was notably elevated in cases of degenerative disc disease. 34 miR-185-5p can target VEGFA to prevent angiogenesis, consequently enhancing the progression of polycystic ovary syndrome. 35 In line with this, we also verified in this study that miR-185-5p targeting regulates VEGFA expression, so we considered that the regulatory effect of MIF-AS1 on pain and inflammation induced by LDH is achieved by regulating the miR-185-5p/VEGFA axis, but this may need more experiments to confirm. The proliferation of venous endothelial cells driven by VEGFA contributes to angiogenesis in the context of neuroinflammation. 36 Research has found that the binding of VEGFA with neuropilin-1 (NRP-1) and the co-receptor tyrosine kinase receptor VEGFR2 enhances the activity of voltage-gated sodium and calcium channels, sensitizing nociceptors and thereby causing pain. 37 Moreover, research has found that VEGFA regulates neuropathic pain through the AKT/TRPV1 pathway. 38 Therefore, VEGFA may cause changes in the microenvironment around the nerve roots by promoting local angiogenesis, thereby triggering neuroinflammation and pain. Moreover, the mechanism by which VEGFA affects the inflammatory response and pain in LDH still requires further experimental exploration. In the future, it is very necessary to use VEGFA inhibition or overexpression models to confirm its functional role in the pathology of LDH.
Although previous studies have confirmed the general role of miR-185-5p in inflammation regulation, such as its regulation of CDK6 expression and participation in the inflammatory response during myocardial ischemia-reperfusion injury. 39 Additionally, although some studies have shown that miR-185-5p targets VEGFA in diseases such as cervical cancer 40 and recurrent miscarriage, 41 its specific function in LDH has not been systematically explored. This study is the first to reveal that miR-185-5p regulates pain and inflammatory responses in LDH by directly targeting VEGFA. As LDH patients often suffer from chronic pain and recurrent inflammation, the MIF-AS1/miR-185-5p/VEGFA axis proposed provides a new direction for the development of targeted gene therapy. For example, by inhibiting MIF-AS1 (such as siRNA or antisense oligonucleotides) or supplementing miR-185-5p mimics, more precise regulation of pain and inflammation may be achieved. This study not only fills the gap in the mechanism of non-coding RNA in LDH but also provides a molecular-level reference framework for the study of other spinal degenerative diseases.
Studies have found that in LDH, the nuclear factor-κB (NF-κB) pathway is significantly activated. Inhibiting the NF-κB pathway alleviates the pain associated with LDH. 42 Additionally, Mitogen - Activated Protein Kinase (MAPK) expression is upregulated in LDH rats and may mediate the immune-inflammatory response in these animals. 43 The MAPK/NF-κB pathway is involved in regulating the pain and inflammation induced by autologous nucleus pulposus transplantation in rats with LDH. 44 LDH significantly increases thermal mechanical allodynia and induces degeneration of the DRG by inactivating the PI3K/Akt pathway. 45 miR-185-5p affects the progression of periodontitis by regulating NF-κB signaling. 46 Additionally, miR-185-5p regulates the PI3K/AKT pathway in prostate cancer. 47 Therefore, the MIF-AS1/miR-185-5p/VEGFA axis may also regulate NF-κB, MAPK, and PI3K/AKT signaling pathways to control the progression of LDH. This hypothesis is worthy of systematic verification in the future through RNA sequencing (RNA-seq) or protein interaction analysis.
Overall, this study explored the effect of MIF-AS1 on LDH pain and inflammation, but there are some limitations to this study. First, the clinical samples included in this study are not enough, which may cause a certain degree of bias in the research results. More sample sizes should be included in the future to verify the conclusions of this experiment. The miRNAs regulated by MIF-AS1 may include miR-185-5p and others. Due to the limitations of experimental conditions, we did not explore multiple molecular mechanisms in this study. Future research is intended to conduct more experiments to investigate the downstream miRNAs of MIF-AS1. In addition, although in vivo experiments verified the function of the MIF-AS1/miR-185-5p/VEGFA axis in the overall tissue system, future in vitro studies using human nucleus pulposus cell cultures and dorsal root ganglion neurons could better help us understand the cellular-level interactions of MIF-AS1, miR-185-5p, and VEGFA in LDH. In future experiments, we plan to use CRISPR/Cas9 technology to knock out MIF-AS1 to confirm its direct impact on cell survival, inflammatory response and the expression of pain-related genes. Moreover, this study did not extensively investigate the role of the MIF-AS1/miR-185-5p/VEGFA axis and its associated pathways in inflammation and neuropathic pain. Future studies plan to verify and explore these interactions through RNA-seq and protein-protein interaction analysis, and it is necessary to use VEGFA inhibition or overexpression models to confirm its role in LDH pathology. Another significant limitation is that this study did not confirm how MIF-AS1 affects mechanical stimulation caused by segmental protrusion, nor did it prove the role of MIF-AS1 in the regression of intervertebral disc protrusion. This will be another focus of our future research. We plan to directly verify the effect of MIF-AS1 on mechanical stimulation by introducing an in vitro mechanical stress model, and in animal experiments, we plan to examine the changes in intervertebral disc structure or imaging, while also studying the role of MIF-AS1 in extracellular matrix remodeling.
Conclusion
The levels of MIF-AS1 were found to be elevated in patients with LDH and associated with the intensity of pain experienced by these patients. Decreased MIF-AS1 alleviated pain and inflammatory responses in LDH rats. Furthermore, MIF-AS1 might influence pain and inflammation caused by LDH through regulation of the miR-185-5p/VEGFA pathway. Therefore, the MIF-AS1/miR-185-5p/VEGFA axis could potentially serve as a therapeutic target for managing LDH.
Footnotes
Authors’ contributions
Conceptualization, G.L., J.LIN, J.LI; Data curation, H.Z., L.L., M.W.; Formal analysis, L.L., M.W., Y.G.; Funding acquisition, J.LI; Investigation, H.Z., L.L.,; Methodology, G.L., J.LIN, H.Z., L.L., J.LI; Project administration, J.LI.; Resources, H.Z., L.L.,; Software, L.L., M.W., Y.G.; Supervision, J.LI; Validation, L.L., M.W., Y.G.; Visualization, G.L., J.LIN; Roles/Writing - original draft, G.L., J.LIN; Writing - review & editing, J.LI.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by Zhongshan Science and Technology Bureau (No. 2023B3020, No. 2024B1131).
Ethical Statement
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
