Abstract
Study Design
Retrospective cohort study.
Objectives
To evaluate utilization trends and outcomes of same-day vs staged circumferential lumbar fusion (cLF) in lumbar degenerative disease.
Methods
The PearlDiver national database was queried for patients with lumbar degenerative disease who underwent same-day or staged cLF, defined as anterior lumbar interbody fusion followed by posterior fusion within 1 to 14 days after. Annual utilization trends (2010-2021) were analyzed. Multivariate regression adjusted for age, gender, comorbidity index, obesity, smoking, and multi-level PSF was used to evaluate 30-day complications, readmissions, and revision rates at 1 and 2 years.
Results
Of 45,204 patients undergoing cLF, 40,052 (88.60%) underwent same-day cLF, and 5152 (11.40%) underwent staged procedures. Patients who underwent staged procedures had higher comorbidity rates and longer fusion constructs. Annual utilization of staged cLF increased on average by 3.76% from 2010. Staged procedures had higher risks of 30-day readmissions (aOR: 1.14), infection (aOR: 1.38), DVT (aOR: 2.62), epidural hematoma (aOR: 1.73), seroma (aOR:1.77), AKI (aOR: 1.34), cardiac arrest (aOR: 2.45), blood loss anemia (aOR: 1.56), urinary retention (aOR: 1.50), UTI (aOR: 1.36), ileus (aOR: 1.64), durotomy (aOR: 4.67), mechanical complications (aOR: 3.39), and blood transfusions (aOR: 2.57) (all P < 0.05). No significant differences in revision rates were found.
Conclusion
Staged cLF, utilized in 11.40% of cases, particularly for longer fusions and patients with higher comorbidity risk, is associated with higher risks of hospital readmissions and complications, but is not an independent risk factor. Further research is needed to identify patients who may benefit most from staging.
Introduction
Lumbar degenerative disease and spondylosis are among the most common spinal conditions affecting older adults. 1 For patients failing conservative management with neurological symptoms, surgical decompression and fusion may be indicated. Various surgical techniques have been described including circumferential lumbar fusion (cLF), which combines anterior or lateral interbody fusion with posterior fusion with or without decompression. cLF is associated with higher rates of fusion, lower rates of interbody subsidence, improved restoration of lordosis, and greater improvements in patient-reported outcomes compared to posterior-only techniques.2-4 However, cLF requires 2 approaches (anterior and posterior) and may require patient repositioning during the procedure, which adds logistical complexity and increases operative time. As such, some surgeons perform the procedure in a staged fashion, while others complete it on the same day. The most appropriate strategy is debated and the associated risks and benefits are not well defined in the setting of lumbar degenerative disease.
Advocates for a staged approach suggest that it reduces the well-documented risks associated with lengthy operations, particularly in older adults with multiple comorbidities.5-7 Proponents of the same-day approach emphasize its potential to reduce overall exposure to anesthesia, limit total operative time, and decrease health care resource utilization. 8 Current comparative evidence exists in the context of adult spinal deformity surgery, finding that staged procedures are associated with higher complication rates and longer hospital stays.5,9 To date, only 1 single-center matched cohort analysis of 95 patients has compared these approaches in patients with lumbar degenerative disease. The authors found that patients undergoing staged surgery had longer anesthesia times and higher rates of intraoperative complications, but noted no differences in postoperative complications. 10 However, this study’s relatively small sample size limits its ability to identify rare complications, underscoring the need for larger studies to better understand complications of cLF in this patient population.
Given the increasing prevalence of lumbar degenerative disease and the need for effective surgical strategies, identifying an optimal surgical strategy for cLF in degenerative spine disease is of great interest. To our knowledge, no large-scale studies have compared the outcomes of staged vs same-day circumferential lumbar fusion in this patient population. Therefore, this study aims to leverage a national database to assess the utilization trends and postoperative complications associated with these 2 approaches in patients with lumbar degenerative disease.
Methods
Patient Selection
Patients were identified utilizing the PearlDiver Database (PearlDiver Technologies, Fort Wayne, IN), which provides over 45 billion HIPAA-compliant inpatient and outpatient Medicare records, procedures, demographics, and diagnoses across all payer types. As all patient data is de-identified, informed consent and institutional review board (IRB) approval were not required.
International Classification of Diseases ninth and 10th edition (ICD-9 and ICD-10) and Current Procedural Terminology codes were utilized to identify patients who underwent one- to two-level ALIF or OLIF/LLIF for degenerative disease from 2010 to Q3 of 2022 (Supplemental Table 1). We then identified patients who underwent one- to five-level posterior spinal fusion with or without decompression, to create the posterior fusion group. These groups were then combined to identify patients who underwent ALIF/OLIF/LLIF and posterior fusion on the same day, and those who underwent ALIF/OLIF/LLIF and posterior fusion within 1 to 14 days of each other (staged). Patients with surgical indications for rheumatoid arthritis, trauma, malignancy, infection, age <18 years, and a deformity diagnosis were excluded. To further ensure that deformity patients were not captured, patients who underwent long posterior fusion (7+ segments) were excluded. Demographic and procedural data were recorded as mean values and percentages for continuous and categorical variables, respectively.
Primary Endpoints
Outcomes measured included the incidence of same-day vs staged circumferential lumbar fusion in patients with degenerative disease from 2010 to 2022 Q3, as well as the annual utilization trends of staged cLF from 2010 to 2021. Annual trends were analyzed using 2010 as a reference point, as this marks the marks beginning of the study period. The study also compared differences in demographics, comorbidities, number of anterior and posterior levels fused, 30-day postoperative complications, hospital readmissions, and revision rates at 1 and 2 years postoperatively between the 2 groups. Complications included infection, pulmonary embolism (PE), deep vein thrombosis (DVT), epidural hematoma, seroma, acute kidney injury (AKI), pneumonia, surgical site infection (SSI), wound complications, cardiac arrest, anemia, urinary retention, urinary tract infection (UTI), ileus, durotomy, mechanical complications, stroke, and transfusions.
Statistical Analysis
Continuous and categorical variables for patient demographics, comorbidities, and number of levels fused were compared between groups using student’s t tests and Pearson chi-squared tests of independence. Multivariate logistic regression analysis was utilized to compare risks of 30-day postoperative complications, hospital readmissions, and revision rates. Covariates in the model included age, gender, ECI, obesity, tobacco use, and multi-level PSF. All statistical analyses were performed using RStudio (Version 4.4.2) within the PearlDiver Mariner Database program. Statistical significance was set at P < 0.05.
Results
The PearlDiver database identified 45,204 patients who underwent circumferential lumbar fusion for degenerative disease from 2010 to Q3 of 2022, of which 40,052 (88.60%) underwent same day cLF fusion, and 5152 (11.40%) underwent a staged procedure.
Patient Demographics
Demographics and Number of Levels Fused.
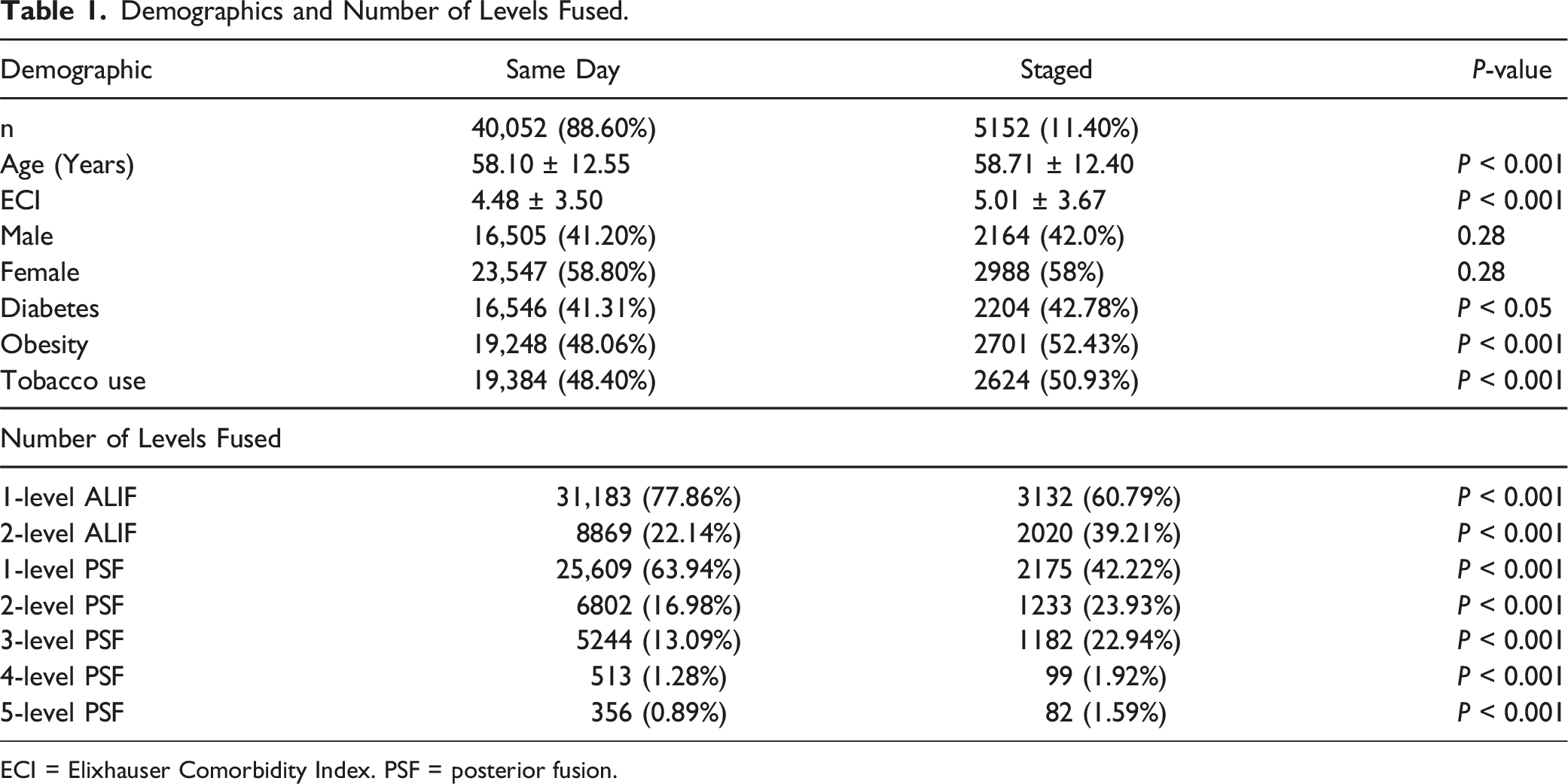
ECI = Elixhauser Comorbidity Index. PSF = posterior fusion.
The staged group had significantly lower rates of one-level ALIFs (60.79% vs 77.86%, P < 0.001) but higher rates of two-level ALIFs (39.21% vs 22.14%, P < 0.001) (Table 1). Regarding posterior fusions, staged cLFs demonstrated lower rates of one-level PSF (42.22% vs 63.94%), but higher rates of multi-level PSFs, including two-level (23.93% vs 16.98%), three-level (22.94% vs 13.09%), four-level (1.92% vs 1.28%), and five-level PSFs (1.59% vs 0.89%) (all P < 0.001).
Utilization Trends
Annual Utilization Rates From 2010 to 2021 for Staged cLF Cases.
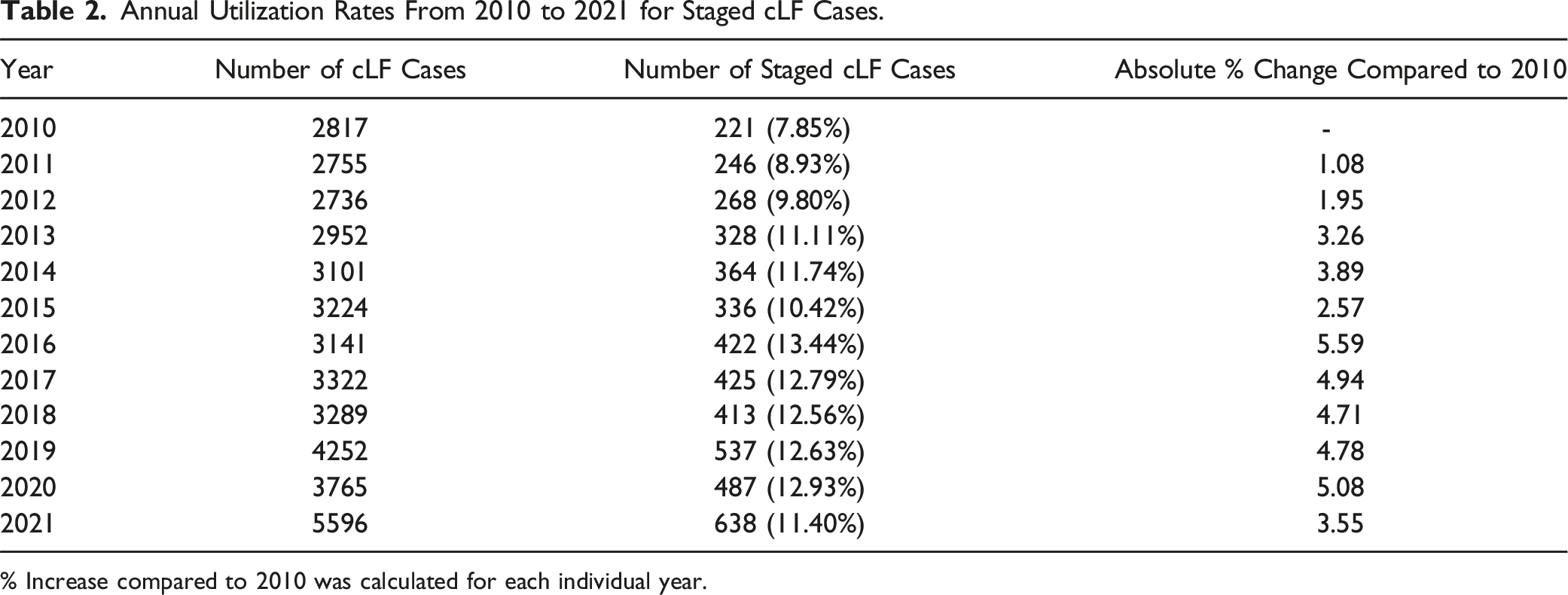
% Increase compared to 2010 was calculated for each individual year.
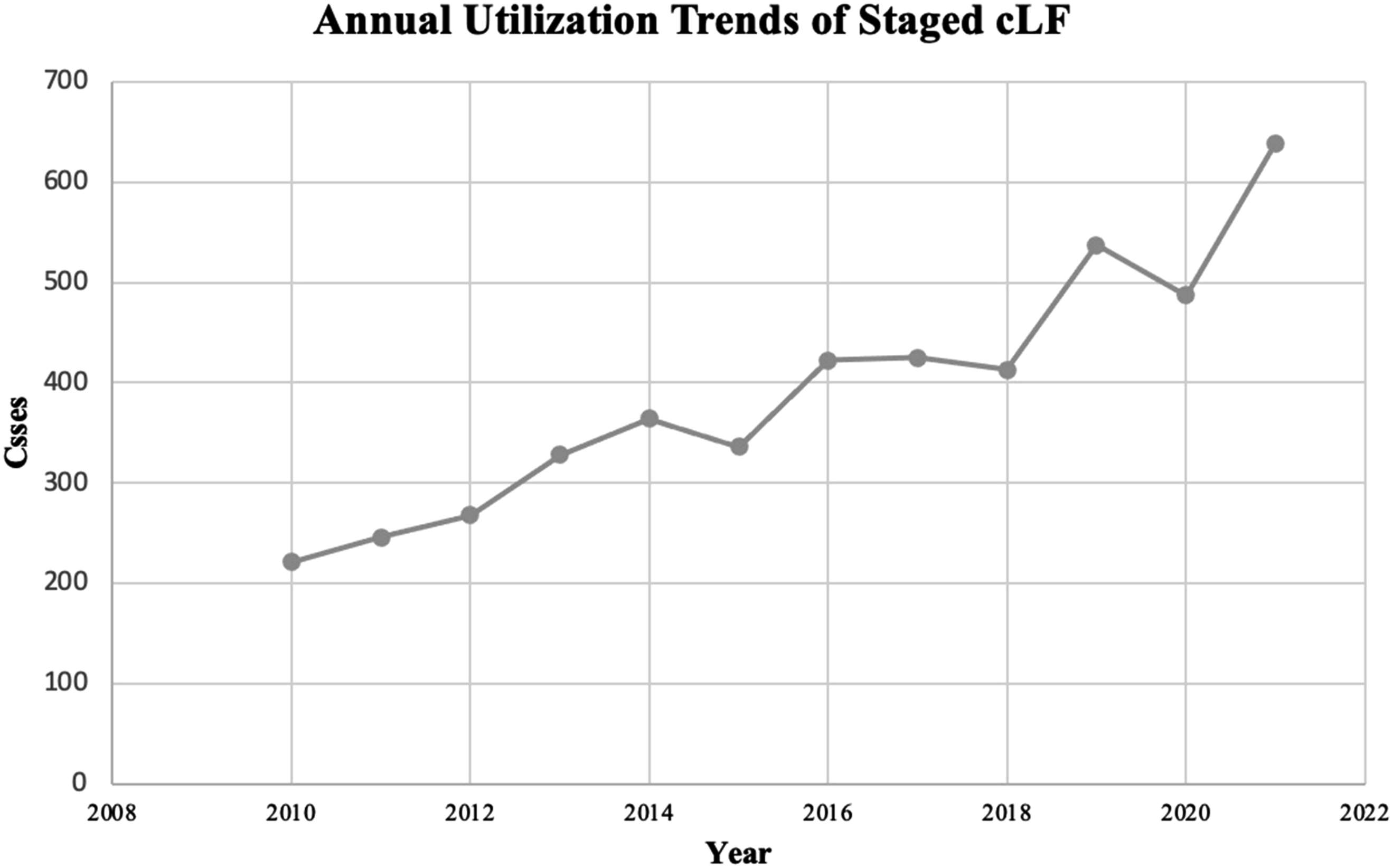
Annual utilization trends of staged cLF.
30-Day Postoperative Complications and Hospital Readmissions
Multiple Logistic Regression for 30-Day Postoperative Complications for Same Day and Staged Groups.
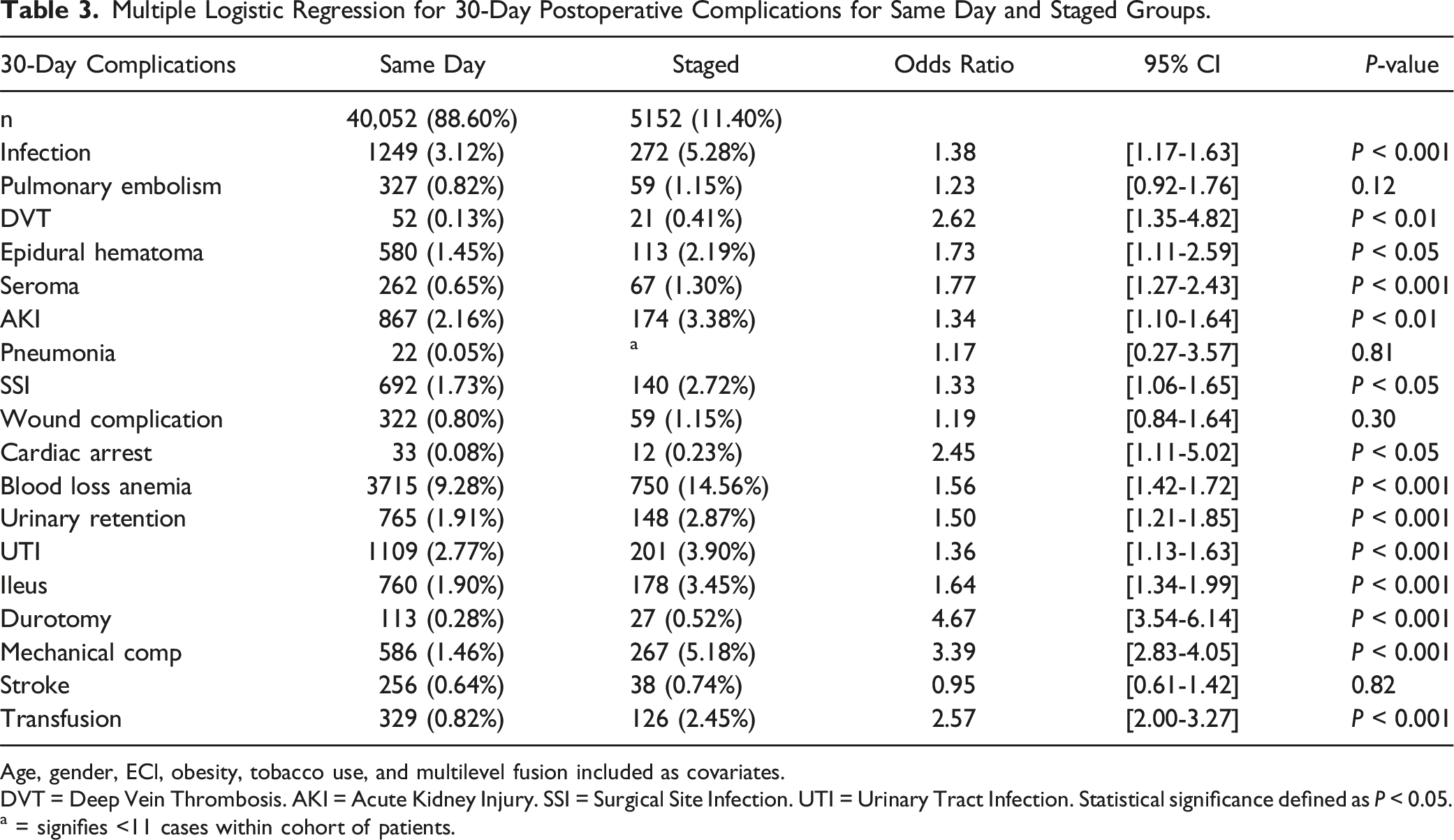
Age, gender, ECl, obesity, tobacco use, and multilevel fusion included as covariates.
DVT = Deep Vein Thrombosis. AKI = Acute Kidney Injury. SSI = Surgical Site Infection. UTI = Urinary Tract Infection. Statistical significance defined as P < 0.05.
a = signifies <11 cases within cohort of patients.
Conversely, no statistically significant differences were found for PE (aOR: 1.23, [0.92-1.76], P = 0.12), pneumonia (aOR: 1.17, [0.27-3.57], P = 0.81), wound complications (aOR: 1.19, [0.84-1.64], P = 0.30), and stroke (aOR: 0.95, [0.61-1.42], P = 0.82) (Table 3).
Multiple Logistic Regression for 30-Day all Cause Hospital Readmissions and 1-, 2-Year Revision Rates for Same Day and Staged Groups.
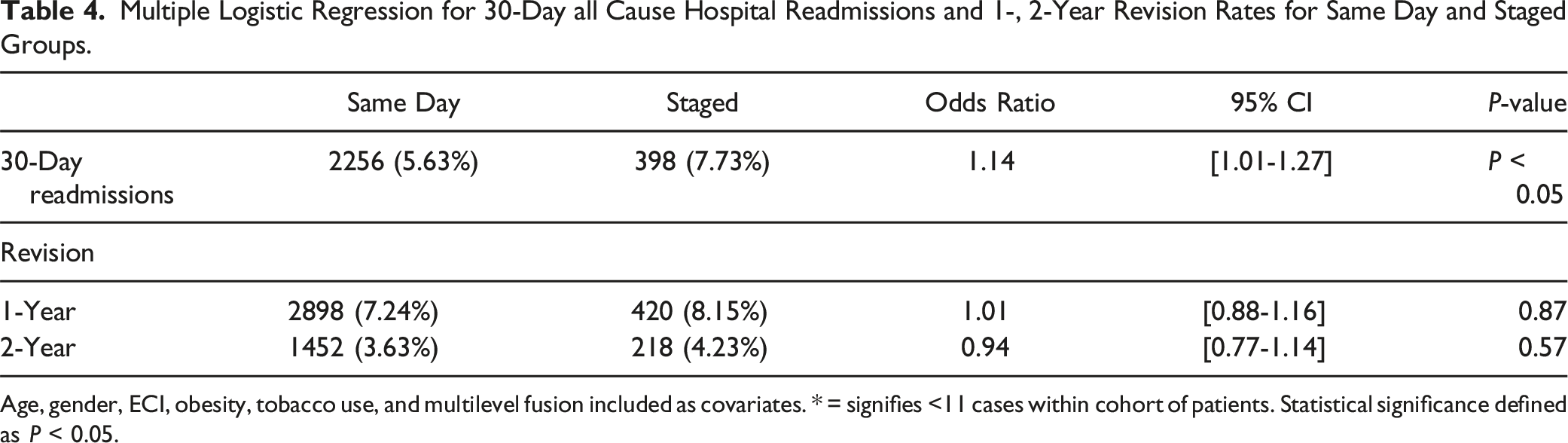
Age, gender, ECI, obesity, tobacco use, and multilevel fusion included as covariates. * = signifies <11 cases within cohort of patients. Statistical significance defined as P < 0.05.
Revision Rates
Multivariate analysis revealed no significant differences in revision rates between staged and same day cLF at 1-year (aOR: 1.01, [0.88-1.16], P = 0.87) or 2-years (aOR: 0.94, [0.77-1.14], P = 0.57) postoperatively (Table 4).
Discussion
The utilization rates and complication profiles of staged cLF in the setting of degenerative spine disease have yet to be examined using data from a large, nationwide database. Although staged cLF is traditionally reserved for long fusions in deformity cases, our analysis revealed that 11.40% of patients undergoing cLF for degenerative indications also underwent staged procedures, and found that its utilization has steadily increased throughout the past decade. Patients who underwent staged procedures generally presented with a higher baseline comorbidity burden, and higher rates of obesity and tobacco use. Furthermore, staged procedures were more commonly utilized for longer anterior and posterior fusions. After adjusting for age, gender, comorbidity index, obesity, tobacco use, and the number of posterior levels fused, multivariate analysis demonstrated that the staged cohort had significantly higher risks of 30-day hospital readmissions and a broad spectrum of postoperative complications. However, no significant differences were observed in revision rates within 2 years of surgery. For the first time we have demonstrated the growing popularity of staged cLF in degenerative spine disease, especially in high-risk patients undergoing multi-level fusions, while highlighting its association with a higher postoperative complication profile.
To our knowledge, only 1 other study has focused on outcomes of cLF in the setting of degenerative disease. 10 In a single-institution study of 120 patients, Thompson et al. reported a higher rate of durotomy in same-day cLF cases (9% vs 0%), attributing this to surgeon fatigue during lengthy operations. 10 In contrast, our analysis identified an almost 5-fold increased risk of incidental durotomy in the staged cohort. Furthermore, Thompson et al found no significant difference in postoperative complications up to 1 year following the procedure. The disparity in findings between our study and theirs could be attributed to differences in sample size, as our large national cohort offers greater statistical power to detect subtle variations in complication rates.
Our findings align more closely with larger studies that have examined outcomes of staged fusions in deformity patients. In a meta-analysis, Daher et al. found longer total operating times for the staged group. 9 While the use of the PearlDiver database in our study precluded direct measurement of operative time, our findings of increased rates of blood loss anemia and transfusions in the staged group align with the longer operating times reported by Daher et al, as these complications are commonly associated with prolonged surgeries.9,11 Several studies have reported longer hospital stays in staged cohorts, which likely reflects the hospitalization required between the 2 stages of the procedure.5,9,12 Prolonged hospitalizations increase the risk of nosocomial infections, which may in part explain the higher rates of infection and UTI observed in our staged group. 13 Moreover, this may also be explained by the elevated rate of transfusions, as prior research has found an association between blood transfusion and infection risk among spine surgery patients. 14
The almost 3-fold increase in thrombosis risk observed in the staged cohort is particularly sobering. This finding aligns with findings by Daher et al, which reported a 4-fold increase in thrombosis risk, and Edwards et al, who identified an 8.17-fold increased risk.9,15 Staged operations may contribute to prolonged perioperative immobility and delayed physical therapy, both of which are well known risk factors for DVT. 9 Given the already high baseline prevalence of DVT following single-level lumbar fusion, this risk warrants attention. 16 While specific guidelines for DVT prophylaxis after elective spine surgery do not exist, the North American Spine Society (NASS) suggests considering postoperative chemoprophylaxis for combined anterior-posterior approaches, further corroborating our findings and highlighting the need for tailored thromboprophylaxis strategies in staged procedures. 17
Furthermore, we found an elevated risk of epidural hematoma in the staged group, which is likely associated with our findings of increased use of staged procedures for longer fusions. These more extensive surgeries often necessitate earlier initiation of VTE prophylaxis due to the heightened risk of VTE with multi-level fusions, potentially contributing to the increased risk of bleeding complications. 18 Our findings also demonstrate that the risk of thrombosis outweighs the risk of epidural hematoma, supporting the NASS guidelines for the administration of DVT prophylaxis in this setting to mitigate the elevated risk of thromboembolic events associated with combined anterior-posterior fusions.
We found no significant differences in revision rates up to 2 years postoperatively between the 2 cohorts. Similarly, 3 studies examining staged vs same-day fusions in deformity patients also reported no differences in reoperation rates.5,19,20 These findings underscore that while staged procedures are associated with an increased complication profile, they do not compromise short-term revision rates, making them a potential viable option in specific clinical scenarios.
For instance, Thompson et al. noted that staging may be necessary when urgent surgery is required but limited by operating room or staff availability. 10 Furthermore, staging may also be required for multi-level fusions from a logistical standpoint, allowing strategic allocation of operating room time to accommodate other surgeons and cases. Staged procedures also offer the advantage of allowing patient re-evaluation after indirect decompression achieved through ALIF, which may help avoid unnecessary posterior decompression and lowers the risk of associated complications. 10 Moreover, our study found that patients undergoing staged cLF had a higher comorbidity index and were more likely to be obese and use tobacco, suggesting that surgeons often select staged procedures for patients presumed to be at higher risk and less likely to tolerate prolonged anesthesia. The increasing utilization of stage procedures, particularly for longer fusion constructs, may reflect a perception that staging offers a safer alternative for these high-risk patients by dividing complex spine fusion cases into 2 shorter, less physiologically demanding operations. However, our findings challenge this assumption. The observed increase in readmissions and postoperative complications in the staged cohort suggests that this approach may not provide the protective benefit it is often believed to confer. These findings underscore that surgeons need a critical reevaluation of the clinical decision making surrounding staged procedures. Future prospective, institutional studies are required to elucidate whether cLF should be staged in the setting of degenerative spine disease, and in which patient population.
Limitations
The PearlDiver database relies on accurate coding, introducing potential for human error. Thus, we may have over- or under-estimated the complication rates between the 2 groups. Additionally, intraoperative variables such as operative time, blood loss, and patient reported outcomes were unavailable. While we accounted for age, gender, ECI, obesity, and tobacco use in the multivariate model to address differences in comorbidities, statistical adjustment cannot fully eliminate confounding variables when key clinical decision-making variables are absent. The 10-fold difference in sample size between the groups further amplifies this concern, as even small differences in staging indications could disproportionately affect outcomes.
Furthermore, we were also unable to determine whether staging was preplanned or prompted by intraoperative factors such as excessive blood loss, neuromonitoring signal changes, prolonged operative time, or anesthesia-related concerns. In such cases, staging may have been intended to reduce complications rather than serving as an independent variable. We were also unable to control for surgeon experience, surgical technique as well as graft and instrumentation type which could potentially influence the outcomes of cLF. Additionally, while surgical techniques likely evolved over the study period, we could not account for these practice shifts within the database.
Staging decisions may also be influenced by factors beyond patient characteristics, such as case complexity, prior surgeries, surgeon preference, OR scheduling, and institutional policies, none of which were captured in this analysis. These limitations introduce the potential for selection bias. As a result, our findings demonstrate an association rather than a direct causal relationship between staged cLF and increased complications.
However, the study’s greatest strength is its large sample size, which allowed us to find statistically significant differences between the 2 groups even after controlling for several factors. Due to this, we were also able to identify a wide range of complications, affecting various organ systems, that smaller institutional studies would not have been able to.
Conclusion
Staged cLF for degenerative spine disease is increasingly utilized in more complex cases and among patients with a higher comorbidity burden. While this study identifies an association between staging and increased postoperative complications and hospital readmissions, it does not establish staging as an independent risk factor. These findings underscore the need for careful patient selection and tailored perioperative management strategies to optimize outcomes. Future research should employ prospective institutional studies that account for surgical complexity and decision-making factors to better assess whether staging itself contributes to adverse outcomes.
Supplemental Material
Supplemental Material - Staged Versus Same-Day Circumferential Lumbar Fusion for Degenerative Disease: Which is the Safer Approach?
Supplemental Material for Staged Versus Same-Day Circumferential Lumbar Fusion for Degenerative Disease: Which is the Safer Approach? by Henry Avetisian, MS, Mirbahador Athari, MD, William Karakash, BS, Jordan O. Gasho, BS, Matthew C. Gallo, MD, Jeffrey C. Wang, MD, Raymond J. Hah, MD, and Ram K. Alluri, MD in Global Spine Journal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Henry Avetisian, Mirbahador Athari, William Karakash, Jordan O. Gasho, and Matthew C. Gallo have nothing to disclose. Jeffrey C. Wang has received intellectual property royalties from Zimmer Biomet, NovApproach, SeaSpine, and DePuy Synthes. Raymond J. Hah has received grant funding from SI bone, consulting fees from NuVasive, and support from the North American Spine Society to attend meetings. Ram K. Alluri has received grant funding from NIH, consulting fees from HIA Technologies, and payment from Eccential Robotics for lectures and presentations.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
Data is not publicly available but can be available upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
