Abstract
Objectives
Stand-alone (SA) and anterior cage-and-plate (ACP) have been studied in anterior cervical discectomy and fusion (ACDF). However, fusion assessment methods vary and existing studies are not propensitymatched and often lack patient-reported outcomes (PROMs). We compare fusion rates between propensity-matched single- and multi-level SA versus ACP using a method validated by intraoperative motion testing during revision surgery. We also compare sagittal alignment, perioperative outcomes, and PROMs.
Methods
Patients >18 years who underwent primary ACDF were included. 2:1 propensity score matching was performed. Fusions were assessed using 1 year computer tomography and flexion/extension radiographs via validated fusion assessment methods. ALOD was assessed at least 6-months postoperatively. Sagittal alignment was assessed at preoperative, short-term postoperative (2-6 weeks), and long-term postoperative (6 months or more) time points. PROMs, operative time, blood loss, dysphagia, and complications were analyzed.
Results
153 patients (51 SA) were included after matching. There were no differences in fusion rates overall (
Conclusions
Fusion rates, PROMs, radiographic outcomes, complications, and dysphagia rates were all comparable following single and multi-level ACDF between SA and ACP.
Introduction
Anterior cervical discectomy and fusion (ACDF) can treat cervical radiculopathy and myelopathy by decompressing the spinal cord anteriorly and restoring height of the intervertebral disc.1,2 ACDFs were originally performed using structural autograft or allograft, but more recently performed utilizing synthetic cages. 3 These cages can often be combined with anterior plate and screw fixation to form a cage and plate construct (ACP) that acts as a theoretic tension band. 4 However, applying an anterior cervical plate may increase operative time, rates of dysphagia, adjacent level ossification development (ALOD), and instrument-related complications.5-7
Zero-profile standalone interbody constructs (SA) were introduced to avoid complications related to plate placement. SA cages are equipped with internal vertebral anchoring, eliminating the need for anterior plate fixation. Previous studies and meta-analyses have shown that the use of a SA implant can reduce postoperative dysphagia, operative time, and the incidence of adjacent segment changes while maintaining surgical safety and providing similar radiologic alignment outcomes when compared to ACP constructs.6,8,9 However, such studies have not assessed fusion using previously validated criteria. In fact, assessing successful cervical fusion has been a topic of debate for many years, and numerous methods have been tested10-13 with varying results in sensitivity, specificity, and agreement between assessors. 14
Therefore, the purpose of the present study was to compare the fusion efficacy, perioperative outcomes, radiographic parameters, and patient-reported outcome measures (PROMs) of SA vs ACP constructs in single and multi-level ACDF. To our knowledge, this is the first study to compare these variables in one-, two-, and three-level propensity-matched SA and ACP ACDFs. We hypothesized that fusion rates between implant types would be equivalent for single-level ACDFs, but that ACP would have superior fusion rates in multi-level ACDFs. We additionally hypothesized that perioperative outcomes, radiographic parameters, and PROMs would be comparable for SA vs ACP.
Materials & Methods
Study Population and Data Collection
Patients (18-80 years old) who underwent primary ACDF by 7 surgeons at a single institution between January 2016 and September 2022 with minimum 1-year postoperative radiographs and/or computed tomography (CT) were included retrospectively. Patients who underwent any concurrent (i.e. posterior cervical fusion or hybrid cervical disc arthroplasty) or revision procedure, or who had osteoarthritis, metabolic bone disease (i.e. osteoporosis), active inflammatory arthritis with radiographic erosive changes, chronic steroid use, stage 4 or 5 chronic kidney disease (eGFR 30 mL/min), or had bone morphogenic protein-2 used during their fusion procedure were excluded. Research Electronic Data Capture system (REDCap) was used for data collection under National Institutes of Health award number UL1 TR002384. 15
Primary Outcome
Fusion was assessed utilizing both radiographic and CT measurements. Patients were deemed to be successfully fused if they either: (1) had fewer than 1 millimeter of interspinous motion per fused level between flexion and extension on 1-year postoperative flexion/extension radiographs,16,17 or (2) had the presence of 3 CT slices with bridging bone on 1-year postoperative CT scan.
18
The accuracy of these methods has been previously validated via intraoperative motion testing for fusion (or lack thereof) during revision surgery
18
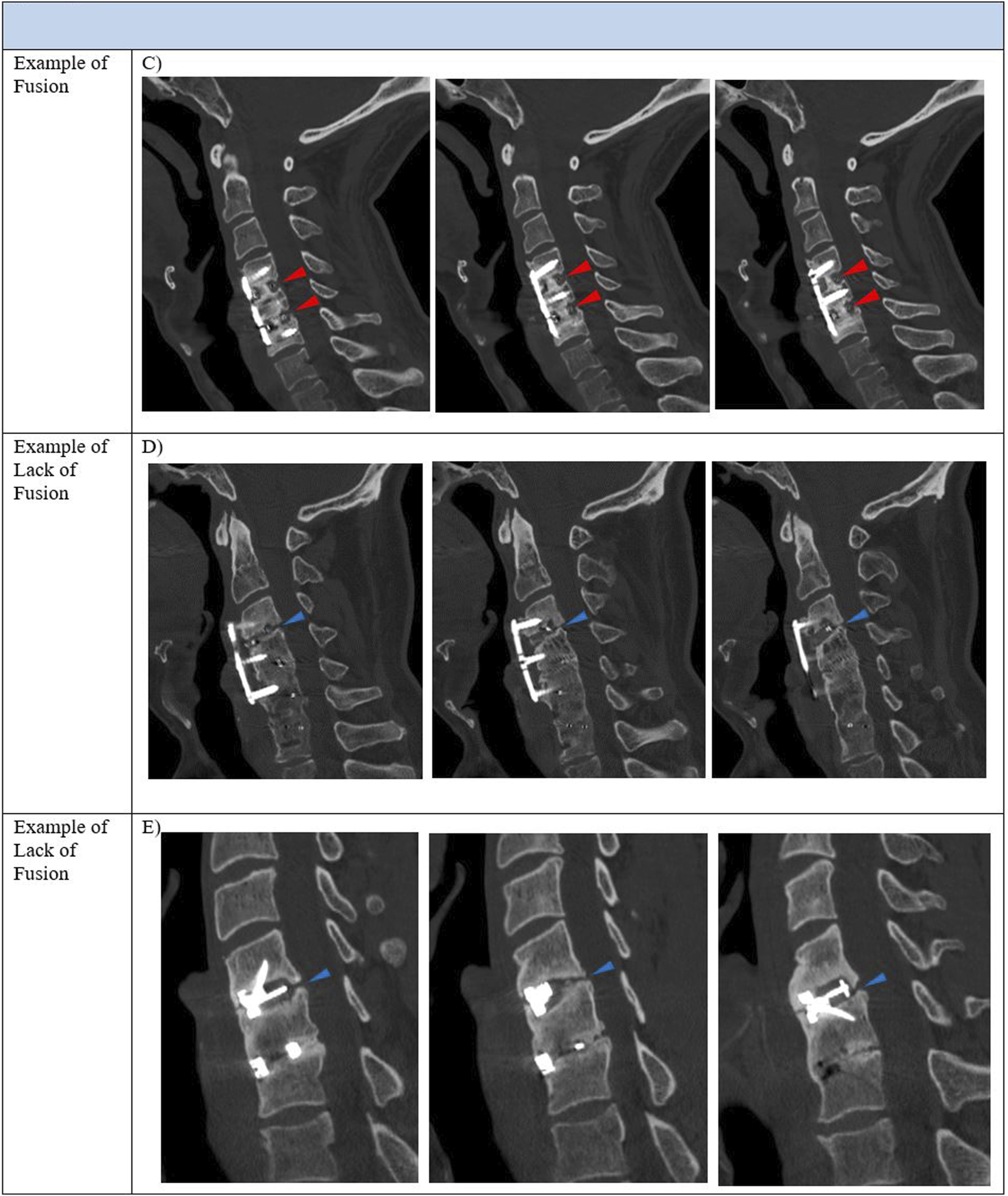
and by rigorous comparison with other available literature.16,17 Fusions were assessed by third- and fourth-year orthopaedic residents. Fusion rates were compared at 1-year postoperative. Examples of fusion and lack of fusion on flexion/extension radiographs and computed tomography are provided (Figures 1 and 2. Example of fusion assessment via flexion/extension sagittal radiographs. Radiographic fusion was determined as < 1 mm of interspinous motion per level across all instrumented levels (panel A). Lack of fusion was determined by Example of fusion assessment via computed tomography. Computed tomographic fusion was determined by 3 or more sagittal cuts demonstrating bridging bones across all instrumented levels (red arrowheads, panel C). Example of one-level lack of fusion at C3-4, with lack of bridging bone, is demonstrated (blue arrowheads, panel D).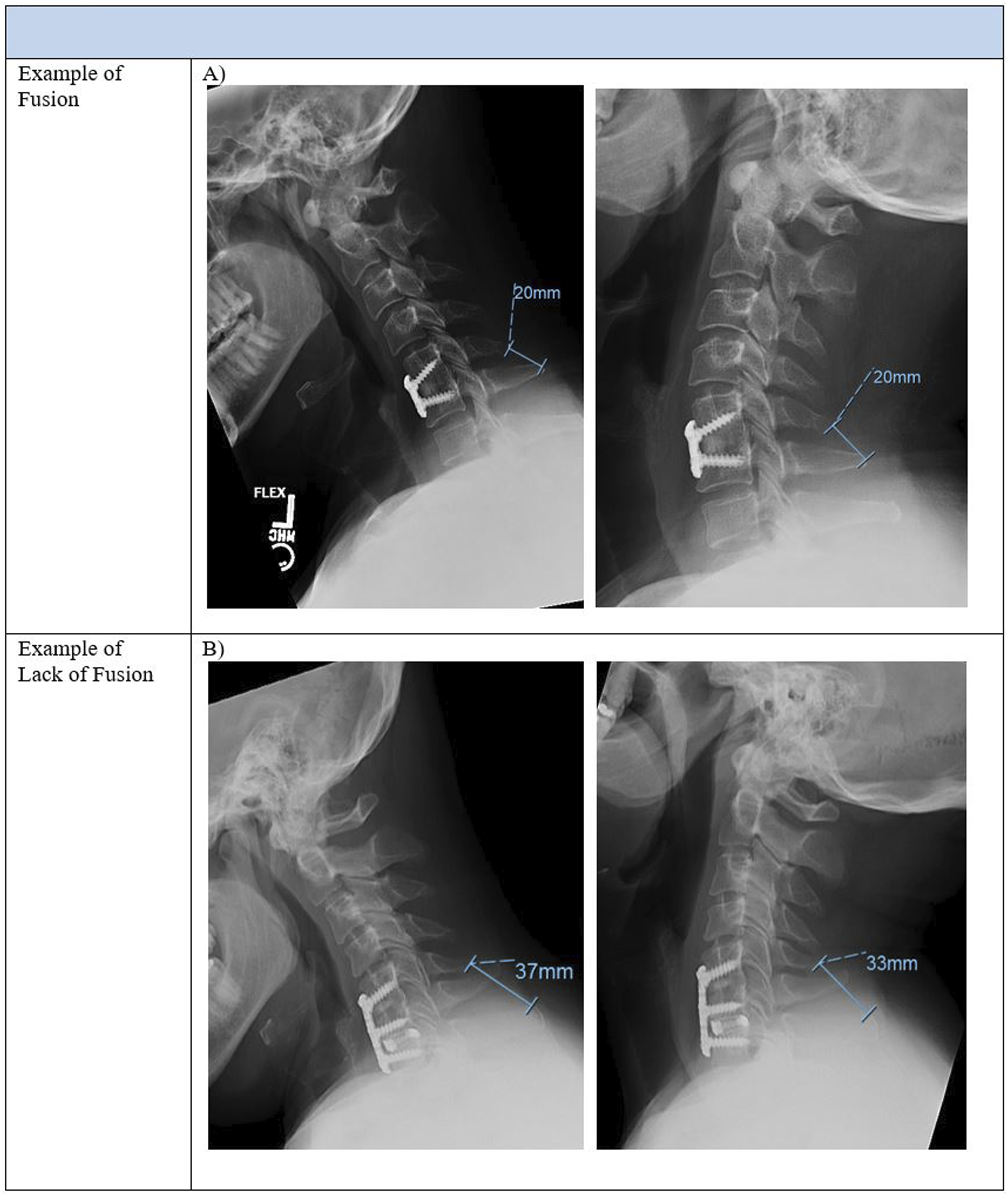
Secondary Outcomes
Data on operative time, blood loss, dysphagia, and surgical complications were collected. Dysphagia was defined as difficulty, discomfort, or choking while swallowing, or explicit mentioning of “dysphagia” in medical records, on the first postoperative visit (2 weeks or 6 weeks postoperatively) or later; immediate postoperative difficulty swallowing prior to the first postoperative visit was not classified as dysphagia. Dysphagia length of symptoms was based on the last clinic visit mentioning dysphagia symptoms. ALOD assessments for upper (cephalad) and lower (caudal) levels adjacent to the fusion were performed on minimum 6-month postoperative sagittal radiographs. ALOD classification based on severity of adjacent osteophyte development was performed according to prior published literature. 19 On preoperative imaging at the adjacent level, if there was: a prior fusion, pre-existing osteophyte grade 2 or more, 19 or difficult visualization, that level was excluded from ALOD subgroup analysis specifically. Plate-to-disc distance, a confounder of ALOD, was accounted for in ACP but not SA constructs. 20 Segmental lordosis, overall lordosis, T1 slope, and T1-slope minus cervical lordosis mismatch (TS-CL) were assessed at preoperative, short-term postoperative (2-week to 6-week), and long-term postoperative (6-month to 2-year) timepoints measured according to available literature.21-23 The most recent available time-point was used for short-term and long-term follow-up windows. Patients with poorly visible cervical levels on sagittal alignment measurements were excluded. In a subset of patients with available PROMs data, we compared Neck Disability Index (NDI), 12-item Short-Form Survey (SF-12), and visual analog scale (VAS) for neck and arm pain at preoperative, short-term postoperative (2-week to 6-week), and long-term postoperative (6-month to 2-year) timepoints. Trends in sagittal alignment and PROMs were plotted on line graphs for clearer visualization.
Statistical Analysis
All statistical analyses were performed using R statistical software (v4.1.2; R Core Team 2021). 2:1 propensity score matching with age, sex, body mass index (BMI), preoperative Charlson Comorbidity Index (CCI), and number of surgical levels (single or multiple level) was utilized to create matched ACP and SA groups. Continuous and categorical variables were compared with Wilcoxon rank-sum tests and Fisher’s exact tests, respectively. Wilcoxon Signed-Rank test was utilized to compare preoperative sagittal alignment and PROMs with short-term and long-term postoperative alignment and PROMs. A
Results
Propensity Matching
Demographics, Preoperative PROMs, Perioperative Data After Propensity Score Matching.
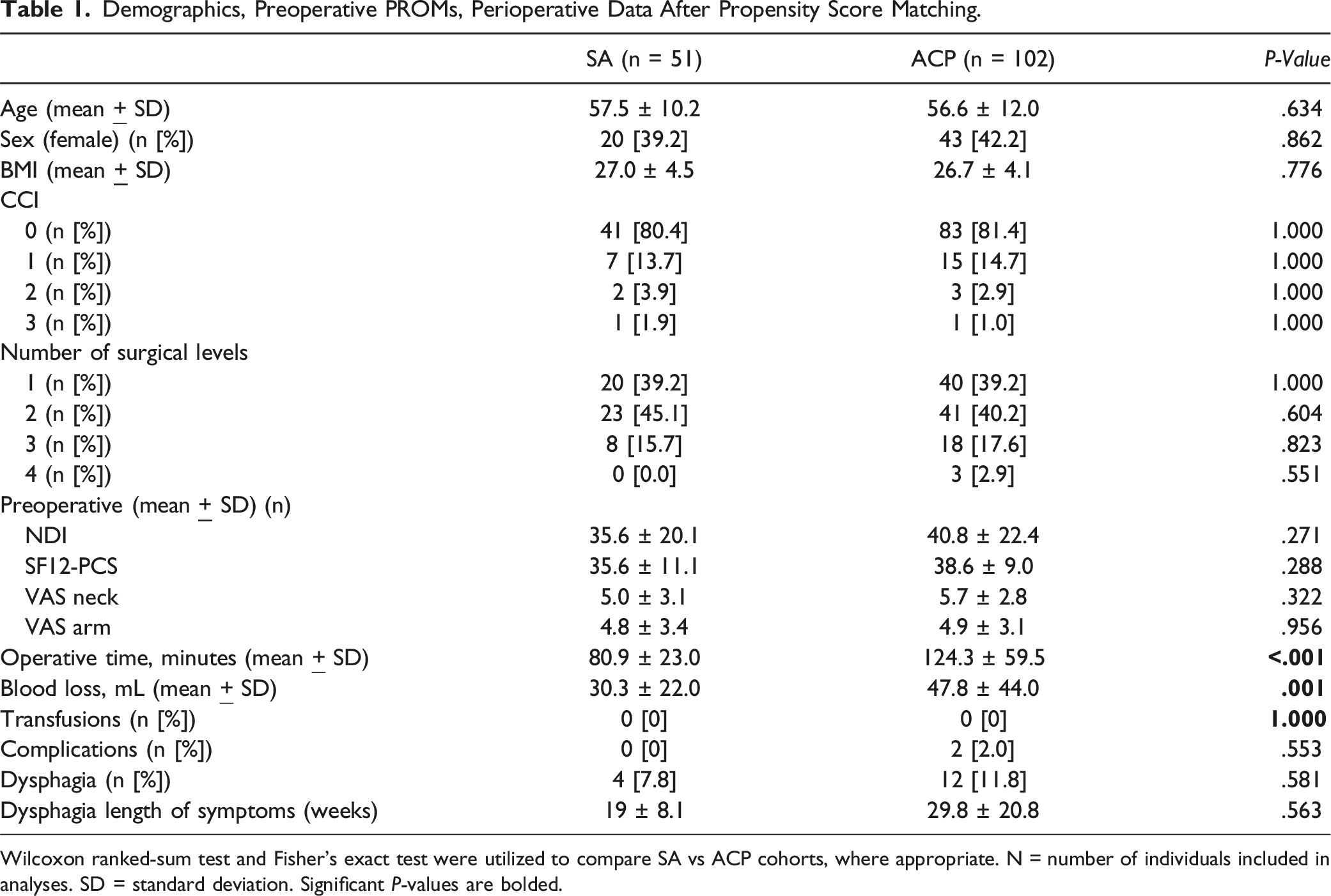
Wilcoxon ranked-sum test and Fisher’s exact test were utilized to compare SA vs ACP cohorts, where appropriate. N = number of individuals included in analyses. SD = standard deviation. Significant
Fusion Rate and ALOD
SA Versus ACP Comparisons of Fusion Rates, ALOD, and Plate-To-Disc Distance.
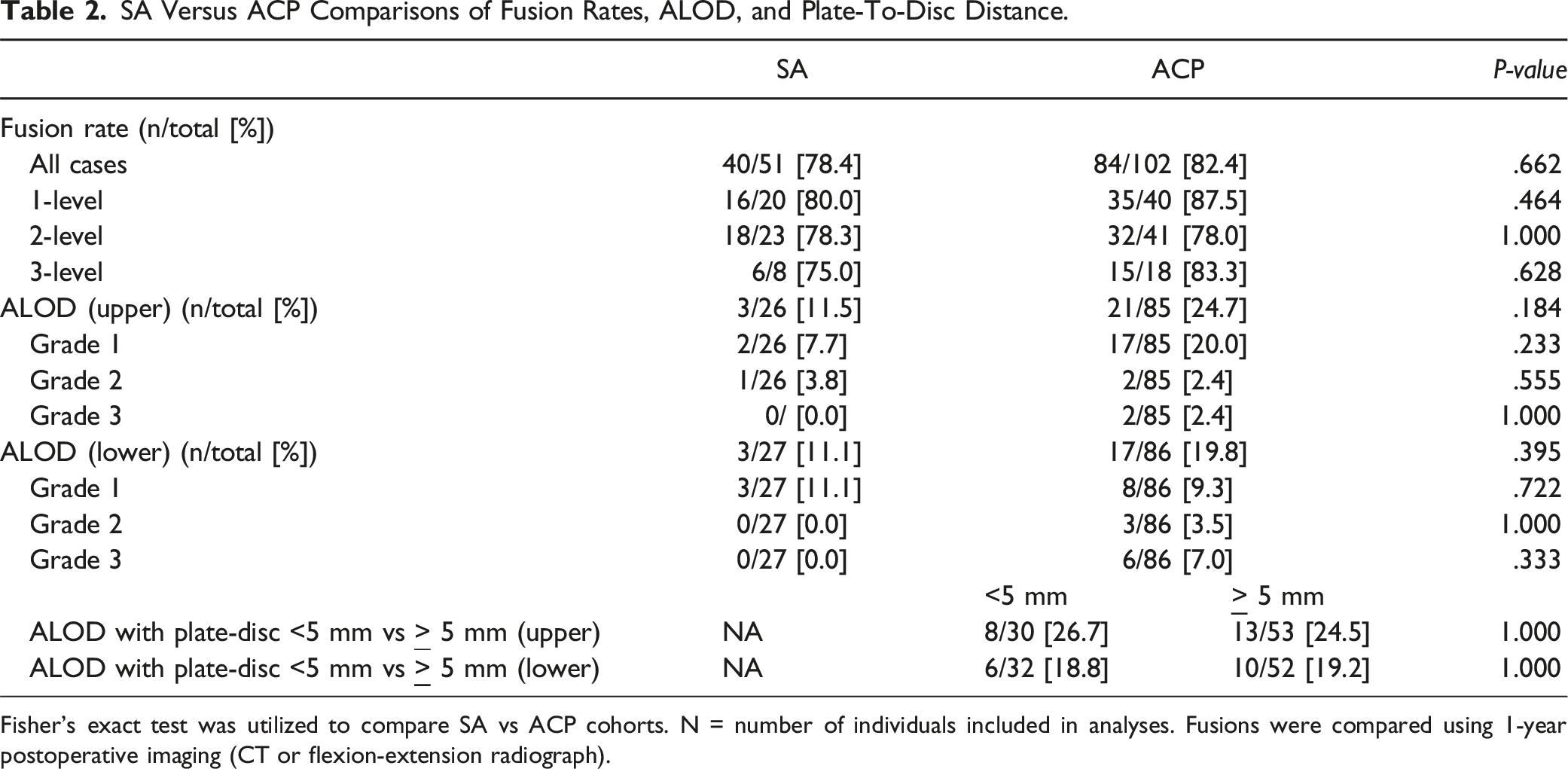
Fisher’s exact test was utilized to compare SA vs ACP cohorts. N = number of individuals included in analyses. Fusions were compared using 1-year postoperative imaging (CT or flexion-extension radiograph).
Changes in Radiographic Alignment
SA
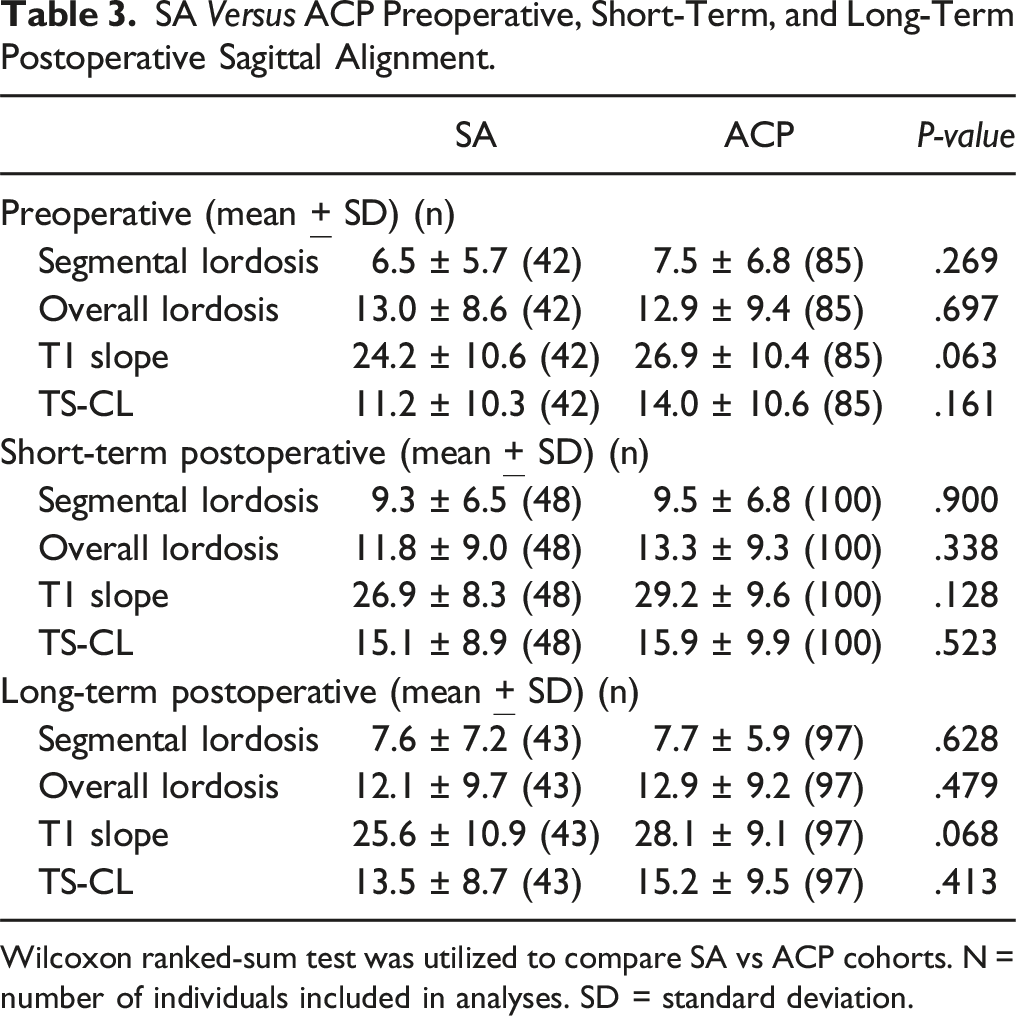
Wilcoxon ranked-sum test was utilized to compare SA vs ACP cohorts. N = number of individuals included in analyses. SD = standard deviation.
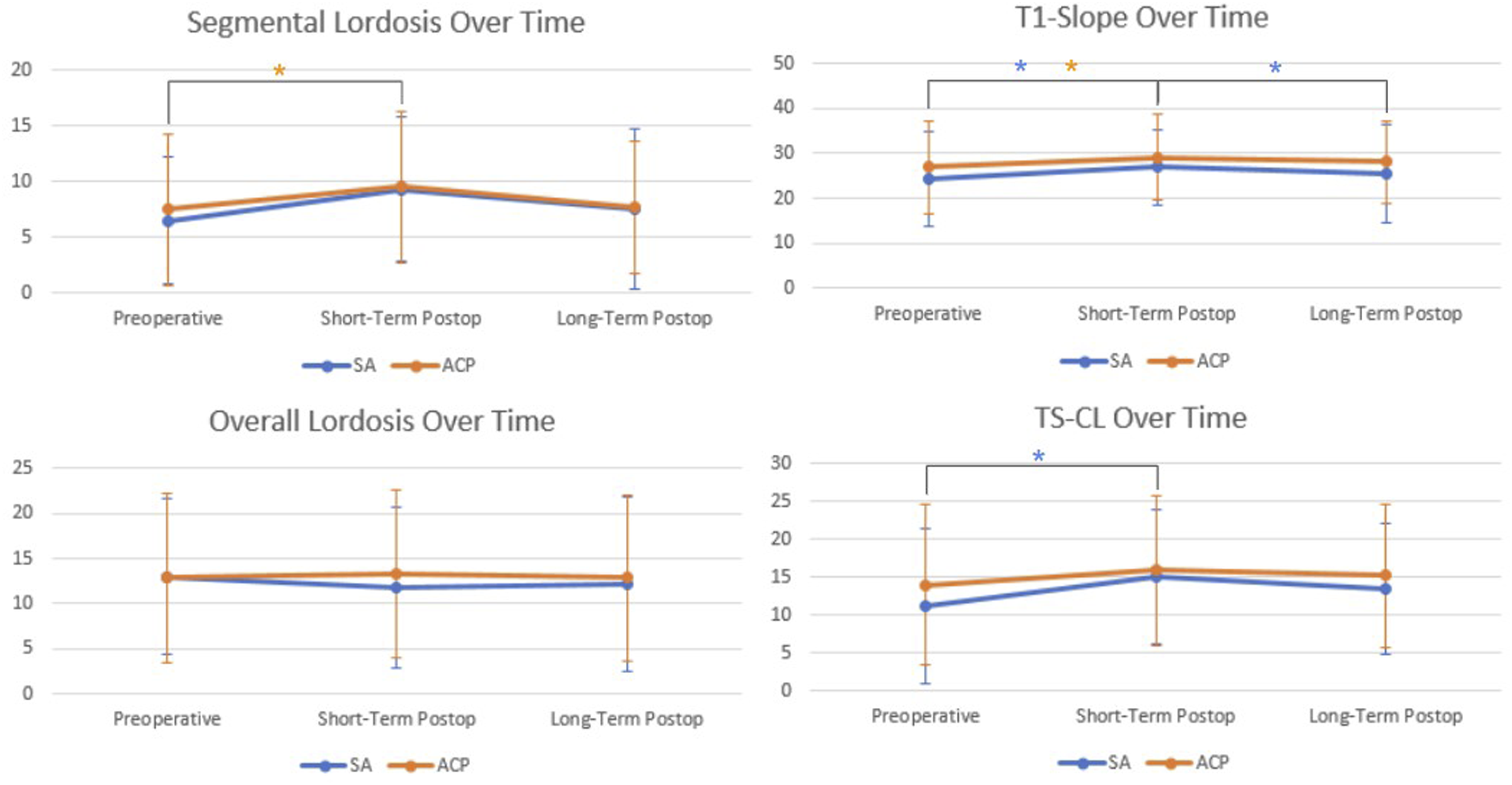
Trends in cervical alignment over time. Preoperative, short-term postoperative, and long-term postoperative time points are shown. Black brackets between time points indicate a significant difference (
PROMs Subset Analyses
SA
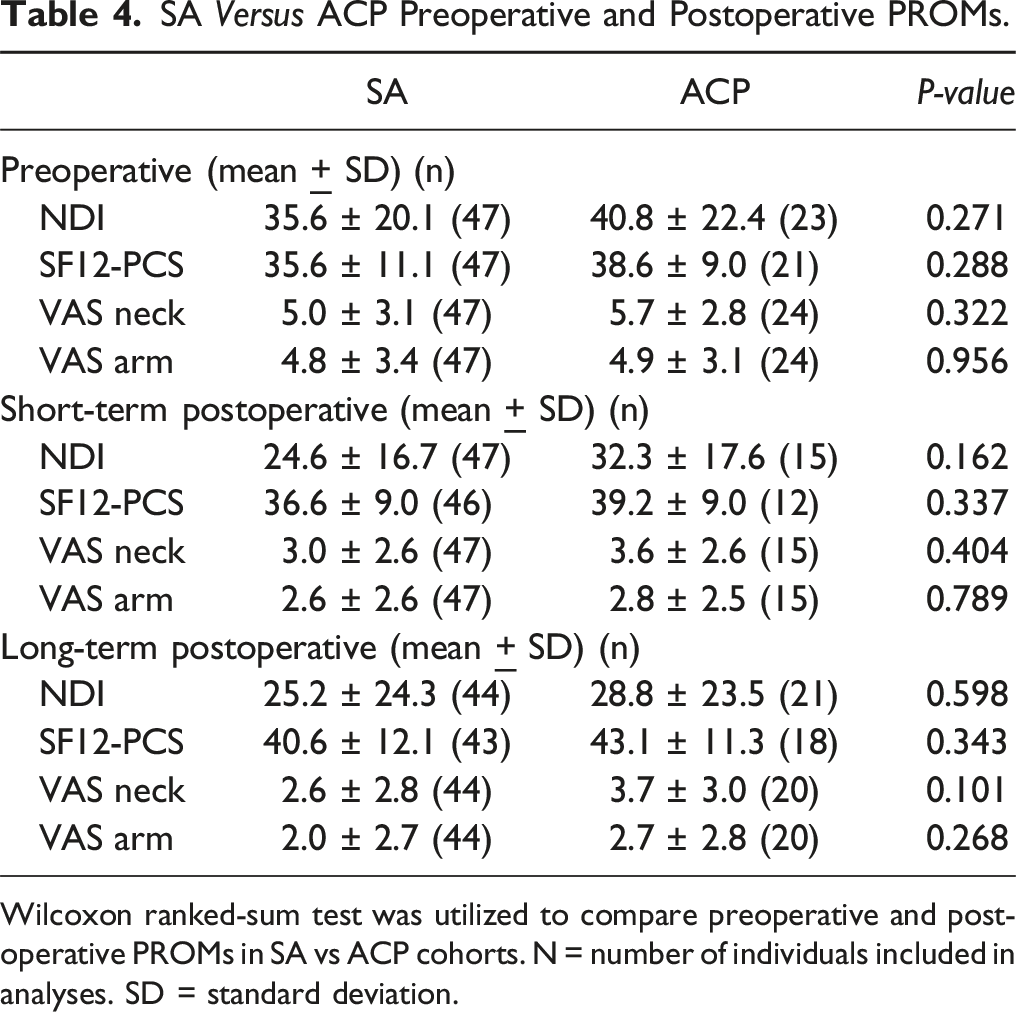
Wilcoxon ranked-sum test was utilized to compare preoperative and postoperative PROMs in SA vs ACP cohorts. N = number of individuals included in analyses. SD = standard deviation.
Preoperative
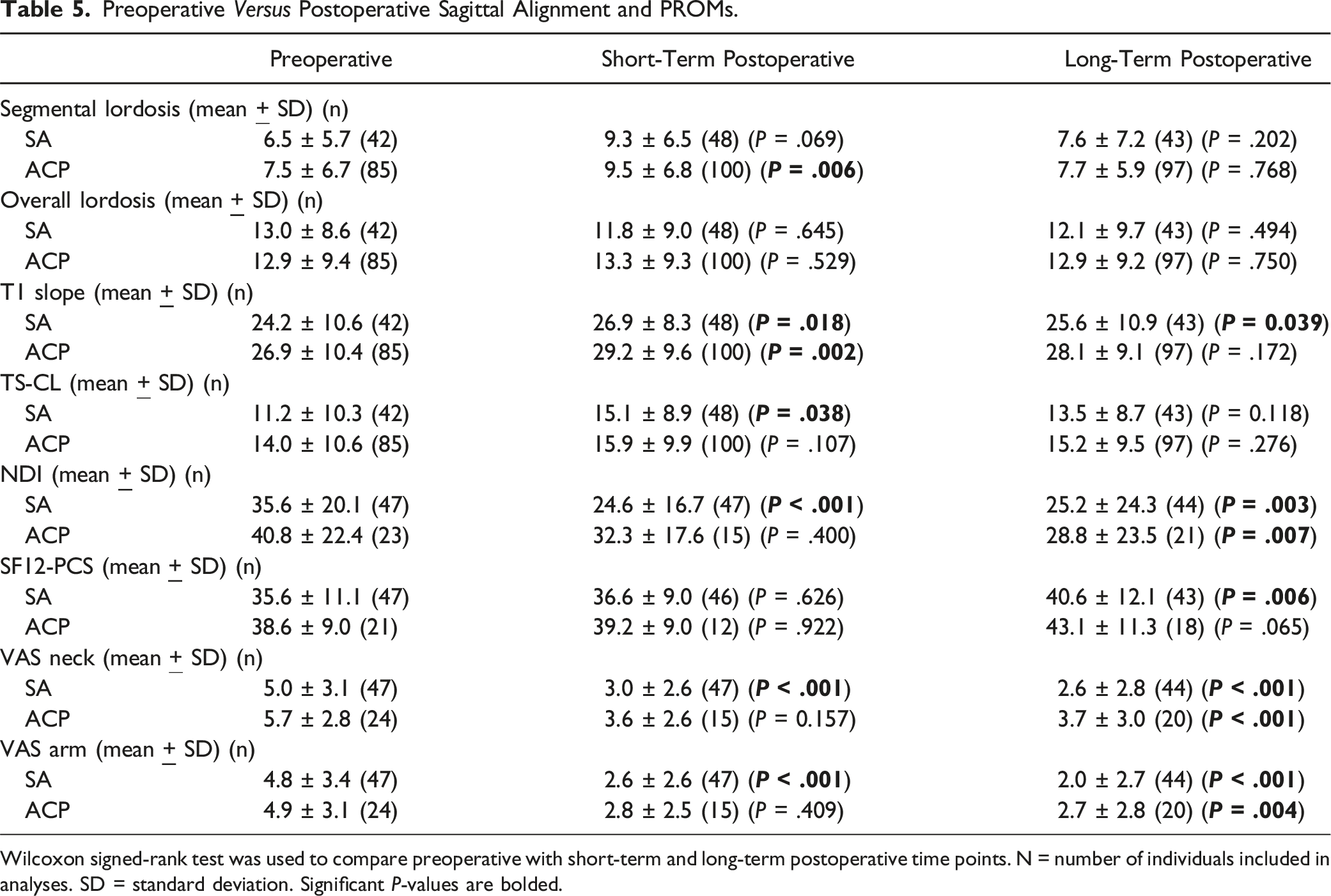
Wilcoxon signed-rank test was used to compare preoperative with short-term and long-term postoperative time points. N = number of individuals included in analyses. SD = standard deviation. Significant
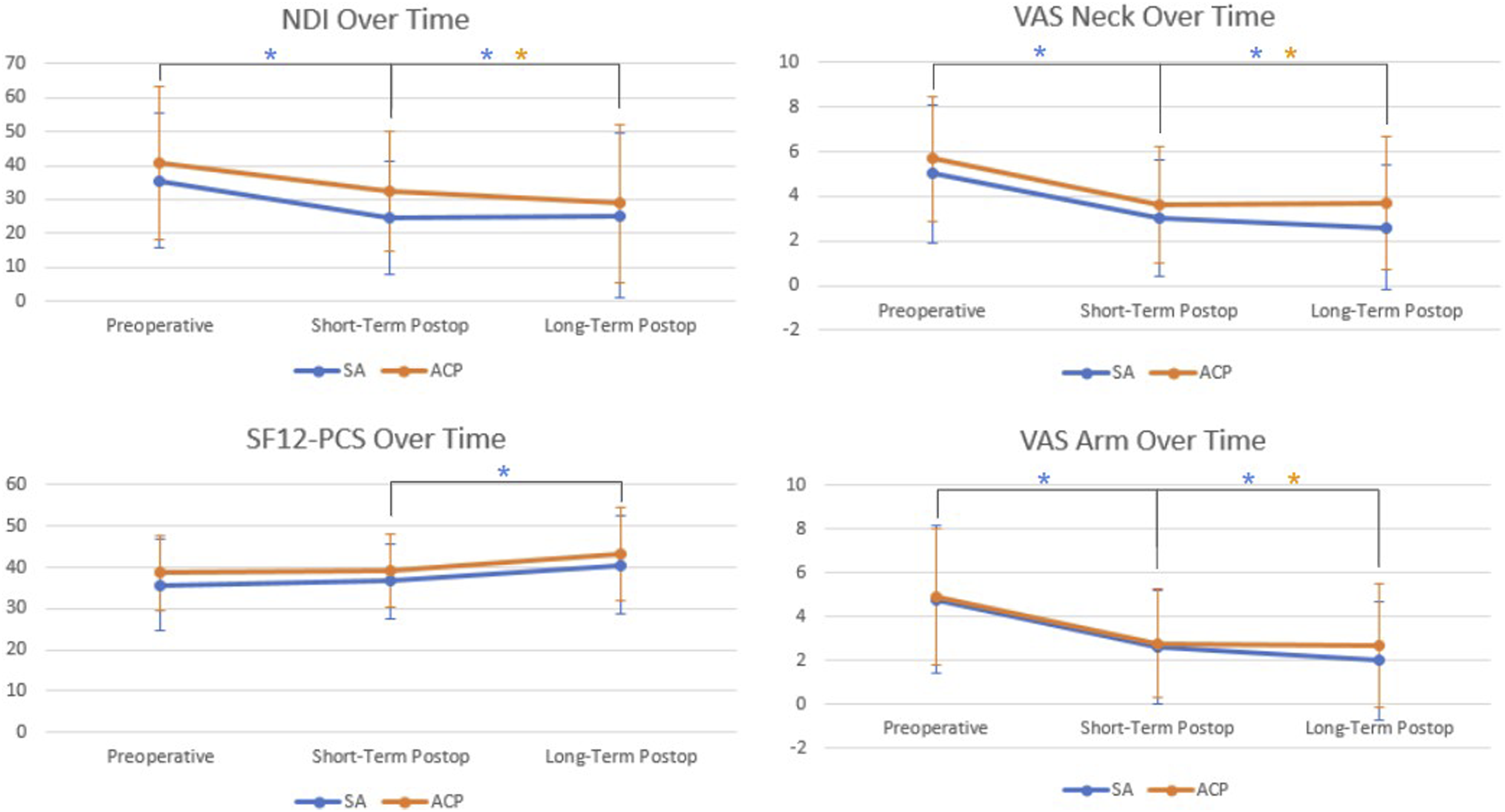
Trends in PROMs over time. Preoperative, short-term postoperative, and long-term postoperative time points are shown. Black brackets between time points indicate a significant difference (
Perioperative Outcomes and Complications
Average operative time was shorter in the SA cohort by over 40 minutes (
Discussion
While ACP and SA constructs are both used in ACDF, these approaches may differ regarding biomechanics. In our understanding, no studies currently exist that compare fusion rates following single and multi-level ACP vs SA ACDFs using validated fusion assessment methods in propensity-matched cohorts. Our study found no statistically significant difference in 1-year fusion rates following single and multi-level ACP vs SA ACDFs. Furthermore, there were no differences in long-term PROMs, ALOD, or sagittal alignment. While operative time and blood loss were greater in the ACP cohort, there were no differences in rates of dysphagia or length of dysphagia symptoms.
Although other studies have investigated fusion rates between SA vs ACP, these studies involve a wide variety of assessment methods, including plain and dynamic radiography, CT, and magnetic resonance imaging, with varying degrees of sensitivity, specificity, and percent agreement between observers. 14 This study is unique in that it implements a fusion assessment method that has been previously validated by intraoperative motion testing. Importantly, imaging outcomes in this study are also supplemented by PROMs, which provide key clinical insights often lacking in other similar studies.
The current available literature shows similar rates of fusion between SA and ACP cohorts following single-level ACDF. For instance, in a retrospective study of 68 patients, Lan et al reported no significant difference in fusion following single-level SA vs ACP ACDFs, with all patients in both cohorts achieving fusion during follow-up. 24 Our study findings corroborate the findings of Lan et al and support the notion that SA cages are comparable to ACP in achieving fusion following single-level ACDF. Additional studies and meta-analyses have reported similar findings in SA vs ACP fusion rates in single-level ACDF.25,26
Our findings also show no significant difference in fusion rate even after two- and three-level ACDF. This is supported by Chen et al, who reported similar fusion rates between multi-level ACP and SA cohorts even 3-year postoperative. 27 While Chen et al was limited by using only radiographs in determining fusion, the extensive follow-up period provides valuable data in assessing longer-term rates of fusion. Our results also demonstrate comparable fusion rates in multi-level ACP and SA cohorts, which is supported by a growing body of literature.28-31 Importantly, in a separate study by Chen et al of multi-level ACP vs SA ACDFs, both cohorts also experienced improvements in Neck Disability Index (NDI) and Japanese Orthopaedic Association (JOA) scores compared to preoperative, with no differences between cohorts. 31 Our study similarly found that both cohorts improved in all PROMs postoperatively compared to preoperative, with no significant differences in any PROMs at any timepoint, suggesting that in addition to fusion rates, key clinical metrics of pain and functionality are also comparable between multi-level SA and ACP. Indeed, lack of fusion following ACDF may not equate to poor patient outcomes; as seen in Chen et al, even patients with pseudoarthrosis following 3-level ACDF may be asymptomatic and may not require surgical revision. 31 Therefore, clinical and functional outcomes are important supplements to imaging findings, especially since fusion assessment methods vary widely in sensitivity, specificity, and percent agreement between observers, even when gold-standard practices are used. 14
In the literature, various studies report increased ALOD in ACP vs SA constructs following single-level and multi-level ACDF.7,8,32,33 While our study found that ALOD was approximately twice as common in the ACP cohort, differences were not significant at the upper nor the lower level. Additionally, there were no differences in ALOD severity based on prior classifications, 19 and contrary to the literature,20,34 there was no significant impact of plate-to-disc distance on ALOD development. While the above studies suggest that ACP increases ALOD, Chen et al did not demonstrate significant differences in adjacent level degenerative changes in ACP vs SA following 3-level ACDF. 29 Importantly, the development of ALOD is likely multifactorial, possibly involving changes in biomechanics,35,36 mobility,37,38 sagittal alignment,39,40 and surgeon-specific factors like disruption of the anterior longitudinal ligament.27,41,42 Given we excluded many patients due to prior adjacent fusion and pre-existing adjacent osteophytic degeneration, decreased sample size and lack of statistical power in ALOD analysis may have also prevented us from detecting significant differences. Further investigations with larger cohorts are needed to elucidate the impact of anterior plating and fusion constructs on development of ALOD.
Our study found no differences in segmental or overall lordosis at preoperative or any postoperative time point between ACP and SA cohorts. Interestingly, when comparing short-term postoperative to preoperative, the ACP cohort achieved a significant increase in segmental lordosis while the SA cohort did not. Perrini et al also showed that ACP constructs may be advantageous in achieving segmental lordosis at 1-year postoperative vs SA. 43 In our study, however, both cohorts saw a loss of lordosis at long-term postoperative follow-up, with no differences between cohorts. While various investigations have suggested advantages in lordosis with ACP constructs,44,45 it remains unclear whether ACP is truly superior to SA regarding lordosis, as other investigations and meta-analyses have shown no difference in segmental and overall lordosis between ACP and SA.9,46,47 Although loss of lordosis has been associated with postoperative disability and pain, 48 our study demonstrated preservation of PROMs improvements even at long-term follow-up despite loss of lordosis. This finding again emphasizes the importance of supplementing imaging findings with PROMs as imaging findings alone may not capture the entire clinical picture.
Other factors such as operative time, surgical blood loss, complications, and dysphagia also have important clinical consequences and may differ between ACP and SA. Our study found that the ACP cohort experienced longer operative times and increased blood loss, supporting the existing literature.49,50 This may be related to decreased instrumentation and surgical dissection required during SA compared to ACP ACDF, the latter requiring additional anterior plating. Surgical complications are another important consideration. Postoperative plate-specific complications may increase proportionally to number of surgical levels, suggesting that procedure invasiveness is related to risk for complications.4,51 Complications are also dependent on surgeon skill, and in a tertiary care center such as ours with highly-specialized surgeons, complications are quite rare, as was seen in our study. Larger propensity-matched studies are needed to discern differences in complications in single- and multi-level ACP vs SA ACDFs. Another undesirable outcome after ACDF is dysphagia. Various studies have suggested that ACP constructs are associated with higher rates of dysphagia in single and multi-level ACDF.8,52-54 Other studies, however, have not shown any significant difference in dysphagia after single and multi-level ACDF, even with 3-year follow-up.27,55,56 While our study did find increased rates of dysphagia and duration of dysphagia symptoms in the ACP cohort, differences were not significant. Importantly, definitions and approaches to measuring dysphagia vary between studies, which may help to explain the lack of agreement across studies. Additionally, while it is believed that the presence of an anterior plate drives the development of dysphagia, the thickness of the anterior plate may also play a role. 57 Therefore, different ACP models may have varying risks of dysphagia development, which should be considered when comparing ACP vs SA constructs. Larger propensity-matched studies are needed to discern any differences in dysphagia between single- and multi-level ACP vs SA ACDF.
There are several study limitations. One limitation is small sample size due to strict inclusion and exclusion criteria and propensity matching. While our criteria reduced the effect of potential confounders to fusion such as osteoarthritis, osteoporosis, and inflammatory bone diseases like rheumatoid arthritis, these are common conditions in the general population and many potential candidates were excluded based on these criteria. Additionally, propensity matching helped mitigate potential confounders but decreased sample size. We believe future studies should maintain strict study criteria and propensity matching in order to limit the effect of confounding variables. Furthermore, while we investigated fusion rates at 1 year and secondary outcomes between 6-month and 2 years, longer follow-ups would be beneficial. In a study comparing SA and ACP 3-level ACDFs over a 5-year follow-up period, Sun et al found that both cohorts lost disc height and cervical lordosis by 5 years post-operation. 58 This suggests that cervical changes following ACDF may occur well past the 1- and 2-year follow-ups, necessitating studies with longer follow-up periods. We recognize that our grouping of 6-month follow-up through 2-year follow-up under “long-term follow-up” is also a limitation, since fusion rates may change during this critical time; the same limitation also applies to ALOD assessments, as bony osteophyte remodeling has been shown to continue even at 2-year postoperative and beyond. 59 Lack of longer-term follow-up may similarly limit analyses in cervical alignment and PROMs measurements; future studies should aim for longer term analyses when possible. Additionally, it is important to note that cage material, the use of biologics, and the type of biologic may all impact fusion rates.60-62 Our study was limited in that this data was not available for analyses; furthermore, attending surgeons in our study varied in their choice of cage material and use of biologics. Not only could these decisions affect fusion rates, but also these choices may vary greatly in their economic impact, both to the hospital as well as to the patient. Given that studies have already suggested cost differences between SA vs ACP following ACDF, 63 the use of biologics may further complicate the issue of cost-effectiveness versus clinical outcomes. Therefore, in addition to the above recommendations, we believe future studies should incorporate data on construct type, use of biologics, and associated costs into analyses, to better understand these nuances that affect fusion rates as well as healthcare spending. Finally, this study was conducted at a single-institution urban academic center. This may limit the generalizability of study results to a larger and global patient population because patients in this study were more likely to be complex cases requiring an urban tertiary referral center.
Conclusion
This study investigated fusion rates, PROMs, ALOD, sagittal alignment, and perioperative outcomes after single- and multi-level SA vs ACP ACDF using validated fusion assessment criteria in propensity-matched cohorts. We found that both constructs are effective in achieving cervical fusion following single and multi-level ACDF, with no significant differences in dysphagia rates, sagittal alignment, and PROMs with short-term and long-term follow-up. These findings may allow surgeons more flexibility in implant and technique selection based on individual patient characteristics.
Footnotes
Author Contributions
Image measurements and data collection: Eric Zhao, Robert Kamil, Austin Kaidi, Tomoyuki Asada, Arsen Omurzakov, Tim Xu, Andrew Kim, Anthony Pajak, Gregory Kazarian, Troy Amen, Jung Mok, Yousi Oquendo, Olatunde Badejo, Nathan Varady, Mitchell Johnson, Bo Zhang. Manuscript writing, revising, and editing: Eric Zhao, Robert Kamil, Austin Kaidi, Tomoyuki Asada, Sravisht Iyer. Statistical analysis: Eric Zhao, Tomoyuki Asada. General project oversight: Mitchell S. Fourman, Sravisht Iyer, James Dowdell, Han Jo Kim, Sheeraz Qureshi.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sheeraz Qureshi has the following disclosures: AMOpportunities: Other financial or material support; Annals of Translational Medicine: Editorial or governing board; Association of Bone and Joint Surgeons: Board or committee member; Cervical Spine Research Society: Board or committee member; Contemporary Spine Surgery: Editorial or governing board; Globus Medical: IP royalties; Paid consultant; Paid presenter or speaker; Hospital Special Surgery Journal: Editorial or governing board; HSS and ASC Development Network: Ownership/Equity/Investment; HS2, LLC: Stock or stock Options; International Society for the Advancement of Spine Surgery (ISASS) - Program Committee member: Board or committee member; Lifelink.com: Other financial or material support; Lumbar Spine Research Society: Board or committee member; Minimally Invasive Spine Study Group: Board or committee member; North American Spine Society: Board or committee member; See All AI: Other financial or material support; Board or committee member; Sustain Surgical, Inc.: Board of Directors, Ownership/Equity/Investment; Simplify Medical, Inc.: Other financial or material support; Society of Minimally Invasive Spine Surgery (SMISS) - Program Committee member: Board or committee member; Spinal Simplicity: Other financial or material support; SpineGuard, Inc.: Paid consultant; Stryker: IP royalties; Paid consultant; Surgalign: Paid consultant; Tissue Differentiation Intelligence: Stock or stock Options; Viseon, Inc.: Paid consultant; Research support; Clinical Spine Surgery: Editorial or governing board. Sravisht Iyer has the following disclosures: Bioventus: Paid consultant; Globus Medical: Paid presenter or speaker; HSS and ASC Development Network: Other financial or material support; JEASO: Other financial or material support; Intrinsic Therapeutics Inc.: Other financial or material support; Stryker: Paid presenter or speaker; Vertebral Columns/International Society for the Advancement of Spine Surgery (ISASS): Editorial or governing board; HS2, LLC: Ownership/Equity/Investment; Innovasis: Research Support (either personally or through institution). Han Jo Kim has the following disclosures: Alphatec: Consultant; Acuity Surgical: Royalties from Intellectual Property; Aspen Medical Products: Royalties from Intellectual Property; Consultant; Blackstone: Consultant; FOCOS Hospital: Board of Directors; Highridge: Royalties from Intellectual Property; Consultant; HS2, LLC: Ownership/Equity/Investment; International Spine Society Group (ISSG): Research Support (either personally or through HSS); K2 Medical, LLC: Royalties from Intellectual Property; Speakers' Bureau; Manhattan School of Music: Board of Directors; Mirus: Consultant; SI Bone: Consultant; SpineSTUD: Ownership/Equity/Investment. Francis Lovecchio has the following disclosures: Seaspine: Consultant; SI Bone: Consultant. James Dowdell has the following disclosures: Bioventus: Consultant; Cerpedics: Speaker; Globus: Providence Medical: Consultant; Speaker; Stryker/VB spine: Consultant. Mitchell Fourman has the following disclosures: Johnson and Johnson: Consultant.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No direct funding was received for this study. However, the study used REDCap (Research Electronic Data Capture) hosted at Weill Cornell Medicine Clinical and Translational Science Center supported by the National Center For Advancing Translational Science of the National Institute of Health (NIH) under award number: UL1 TR002384.
Drug(s)/Device(s) Statement
This manuscript submitted does not contain information about medical device(s)/drug(s).
