Abstract
Study design
Prospective Clinical Study.
Objective
Allogeneic blood transfusion (ABT) is the current standard of blood replenishment for metastatic spine tumour surgery (MSTS) despite known complications. Salvaged blood transfusion (SBT) addresses majority of complications related to ABT. We aim to conduct a prospective clinical study to ascertain the long-term clinical outcomes of intraoperative cell salvage (IOCS) in MSTS.
Methods
Patients were divided into three groups based on their BT type: no blood transfusion (NBT), ABT and SBT. Primary outcomes assessed were overall survival (OS) and tumour progression (TP), evaluated using RECIST (v1.1) employing follow-up radiological investigations at 6, 12, 24, 36 and 48 months.
Results
We included 98 patients [53:45 (M/F)] with mean age of 60 years old. 33 (33.7%) patients received SBT, 39 (39.8%) received ABT and 26 (26.5%) had NBT. All BT groups were comparable for demographics and tumour characteristics (P = 0.215). Median blood loss was 400 mL and median BT was 620 mL. There were no significant differences between OS of patients who underwent SBT, as compared to ABT or NBT (P = 0.136). On multivariate analysis, SBT did not show increase in 4-year tumour progression (P = 0.423). Total blood loss was not associated with tumour progression (P = 0.260).
Conclusions
MSTS patients who had SBT showed comparable OS and TP to ABT and NBT even on long term follow-up. This is the first long term prospective study to report on the clinical outcomes of SBT in comparison with control groups in MSTS and affirms the clinical role of SBT in MSTS.
Keywords
Introduction
Metastatic Spine Tumour Surgery (MSTS) is an important modality in the treatment of Metastatic Spinal Disease (MSD). With advancement in surgical techniques, it has found its place in the treatment of cord compression, pathological fractures, spinal instability and intractable pain.1,2 Improvements in overall oncological treatments have also allowed patients to have overall increased survival and henceforth patients with more extensive disease are being operated on more commonly.3-5 Recent studies4,5 have also shown the increased longevity in patients with MSD. Coupled with the lowering in our surgical threshold, this has allowed patients with poorer comorbidities to also be considered for surgery. Despite these technological advancements, substantial blood loss is still a significant concern in MSTS.6,7 Average blood loss in open MSTS is 1418 mL (714ml-3120 mL), while it is 745 mL (184-1320 mL) in minimally invasive surgery (MIS). 8 In a meta-analysis of MSTS, the pooled mean blood loss in MSTS patients was found to be 2180 mL, 6 while in a review of cases done in our institution, mean blood loss was 911 mls. 7
Allogeneic blood transfusion (ABT) is the current gold standard of treatment in patients with significant intra-operative blood loss, placing a significant burden on limited blood bank resources.9-11 ABT is associated with post-operative complications predisposing patients to surgical site infections (SSI), immune-mediated complications commonly affecting the lungs (eg, transfusion-related acute lung injury), wound healing, and survival duration, a poorer prognosis, and promotion of tumour growth.12-14 Recent emphasis has also been placed in the development of patient blood management (PBM) measures in MSTS, 15 which is an evidence based and patient tailored approach to reduce the need for ABT and its associated risks. One of the arms of PBM involves the use of intraoperative salvaged blood transfusion (SBT). 16
SBT has an established role in deformity correction and degenerative conditions in the field of spinal surgery. 17 Previous randomized18,19 and non-randomized20,21 studies on the use of SBT in non-oncological surgical procedures indicate that SBT significantly reduced the need for ABT in elective non-oncological spinal surgeries. There has also been ample evidence for the clinical safety of SBT use in gynaecological, urological, hepatobiliary, gastrointestinal, orthopedic, trauma, and cardiac surgical procedures.22-24 The safety profile of SBT have also been previous established in both basic and clinical science studies.25-27 Previously, our prospective paper 28 with 2 year outcomes showed that patients who underwent SBT have lower or similar rates of recurrence compared with the control cohort. The safety profile of SBT has also been shown in a prior retrospective study by Elmslky et al. 29
In the present study, we highlight the 4-year long-term outcomes of a prospective clinical study evaluating the clinical use of salvaged blood in MSTS patients. We discuss the long term evaluation of clinical outcomes such as tumour progression, as well as overall survival of patients who received SBT during MSTS, with a view to compare it with similar outcome measures to patients who have had NBT and ABT.
Materials and Methods
This was a single institution prospective study done on patients who underwent MSTS between January 2014 and December 2017. All of the patients’ records were retrieved through our hospital’s electronic health care records database. Spinal metastases were diagnosed in these patients by one or more of the following investigational modalities: Magnetic Resonance Imaging (MRI), Computed Tomography (CT) and/or nuclear bone scans. Indications for surgery included symptomatic metastatic spinal cord compression and/or instability.
Our study cohort was divided into three categories based on the type of blood transfusion (BT) they received, namely: No blood transfusion (NBT), ABT or SBT (with or without ABT). Primary outcomes were assessed post-operatively through available medical records at 6,12, 24, 36 and 48 months or death, whichever occurred earlier. These postoperative follow-up time points were commensurate with local standard of care for patients undergoing MSTS.
Data collected included patient demographics, tumour histology and clinical findings related to both the primary tumour and skeletal metastases. Clinical and investigational findings recorded included neurological assessment, Frankel score, ambulatory status (ECOG score), 30 number of extra-spinal skeletal, vertebral and visceral metastases and Karnofsky Performance Scale (KPS). These were used to calculate the modified Tokuhashi score. 31 Operative variables collected included surgical approach and number of spinal levels decompressed and instrumented. Intraoperative blood loss was recorded, together with blood transfusion (BT) details. Post-operative complications, if any, were recorded. Primary tumour types of MSTS patients were also subgrouped based on their vascularity for analysis. 32 Primary patient outcomes measured were OS and disease progression in terms of tumour progression (TP) [increase in size of existing metastatic lesions and/or the appearance of new metastasis]. TP was evaluated based on RECIST criteria (v1.1). 33 TP was defined as loco-regional or distant increase in size of lesions of ≥20% increment in the sum of diameters of measurable target lesions (eg, lymph nodes, bone metastases), unequivocal progression of non-target lesions (eg, malignant ascites/ pleural effusions) or the appearance of ≥1 new metastatic lesions. 33 Follow-up CT chest and thorax–abdomen–pelvis was used to assess lymph node and visceral metastases. MRI spine and bone scans were studied to detect new spinal/skeletal metastases.
Derived RECIST and modified Tokuhashi scores were independently calculated by the first and senior authors, who were blinded to the patients’ identity. Any discrepancy was re-evaluated and a consensus score was assigned.
Patient Recruitment and Transfusion Protocol
All patients were given the option of not taking part in the study, taking part either as part of the SBT group, or as controls who would receive ABT if needed. Information about theoretical risk of tumour dissemination and the nonstandard and experimental nature of SBT was disclosed. Recent studies26,27,34 disproving this risk were also given to the patients. Patients were given a day to make their decision where clinically possible. Those who agreed to receive SBT, after written informed consent, were transfused with salvaged blood passed through a leukocyte depletion filter (LDF) and when clinically indicated, with additional ABT if the amount of SBT was insufficient. Those who declined to receive SBT but required transfusion were managed with ABT. Patients requiring no transfusion formed the NBT group. Transfusion criteria were standardised at a standard trigger of haemoglobin ≤8 g/dL and/or hemodynamic instability. Patients with large amounts of blood loss, who were at risk of developing dilutional coagulopathy, were managed by the anaesthetic team according to rotational thromboelastometry (ROTEM) findings and given fresh frozen plasma and platelets as indicated. Randomisation was not performed as we did not receive ethical board approval for randomisation and due to the difficulty in recruiting sufficient number of patients if randomisation was performed.
The study was performed in accordance with the ethical standards of the institutional research committee. The National Health care Group Domain Specific Review Board (Singapore) has approved the study, with the study reference being 2022/00866.
Statistical Methods
All statistical analyses were generated using STATA/SE 14.0 (StataCorp LP, College Station, TX USA). Statistical tests were assumed to be two-sided with conventional 5% significance level. Patients’ demographic and clinical characteristics were summarised by mean and standard deviation for continuous variables with approximately normal distribution and median (inter-quartile range) for skewed distribution. Categorical variables were summarised using frequency and percentages. Characteristics related to BT type (NBT/ABT/SBT) were analysed by: (i) one-way ANOVA to compare the means of a normally distributed variable across the groups, (ii) Kruskal–Wallis rank test to compare median, and (iii) Fisher’s exact test for categorical variables. Pairwise comparison of total blood loss and transfusion between different BT types was performed by Wilcoxon rank-sum test with Bonferroni’s correction for multiple testing. For OS, Kaplan–Meier survival curve and log-rank test were adopted for categorical variables. Univariate Cox proportional hazards regression model was used to study individual continuous variables. Multivariable Cox proportional hazards regression was subsequently conducted to adjust for potential confounders for relationship between BT type and OS. Proportional hazards assumption was tested after the final model was obtained. Association between BT type and tumour progression was investigated by competing risks analysis, taking death without tumour progression as the competing event. We plotted cumulative incidence curves of three BT groups nonparametrically. Individual characteristics related to tumour progression were studied by univariate sub-distribution proportional hazards regression. The multivariable sub-distribution proportional hazards regression model was implemented afterwards to adjust for all potential confounders. Finally, Schoenfeld residual of each variable was plotted against survival time and suggested that proportional hazards assumption was adequate.
Results
Demographic and Clinical Characteristics of 98 Patients.
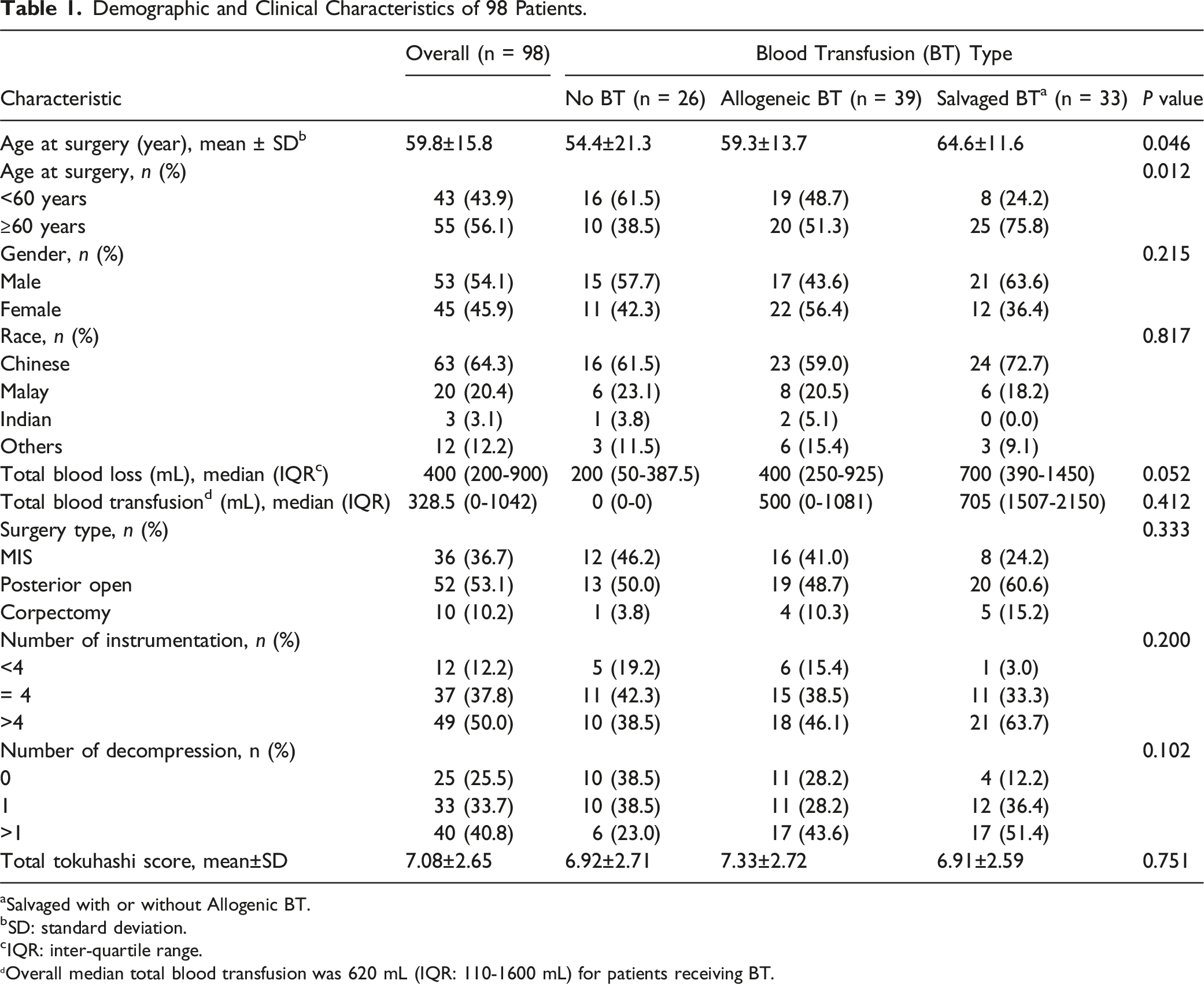
aSalvaged with or without Allogenic BT.
bSD: standard deviation.
cIQR: inter-quartile range.
Overall median total blood transfusion was 620 mL (IQR: 110-1600 mL) for patients receiving BT.
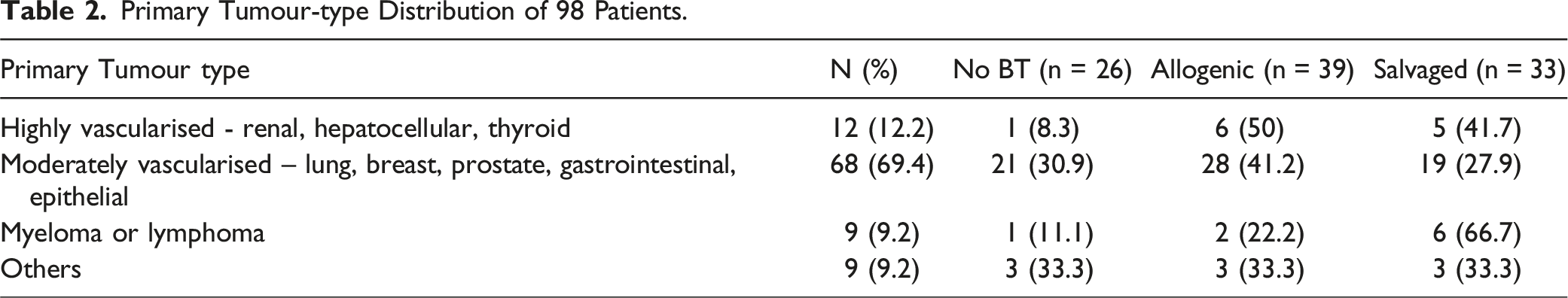
Primary Tumour-type Distribution of 98 Patients.
Characteristics Associated With 4-Year Overall Survival (OS).
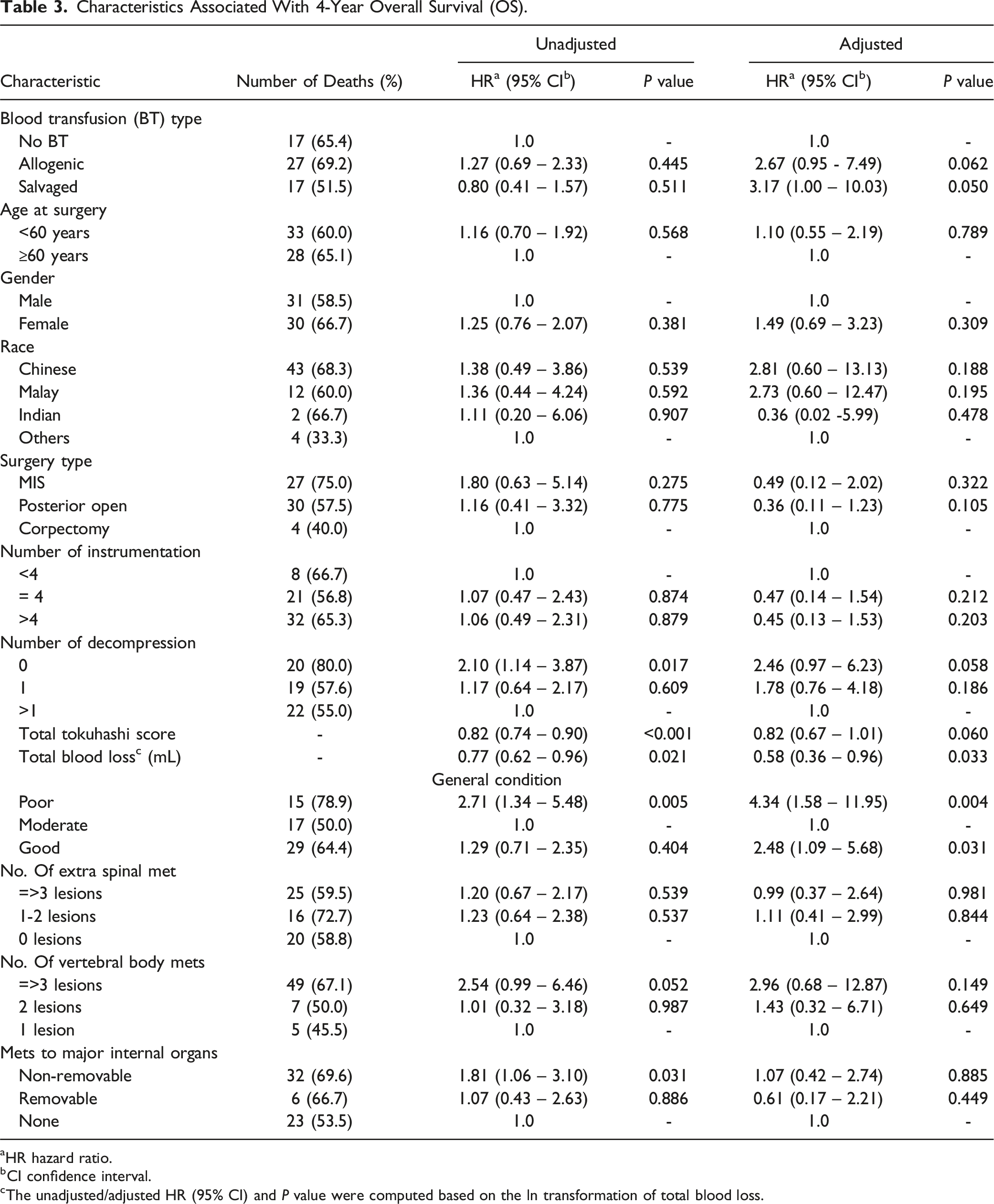
aHR hazard ratio.
bCI confidence interval.
cThe unadjusted/adjusted HR (95% CI) and P value were computed based on the ln transformation of total blood loss.
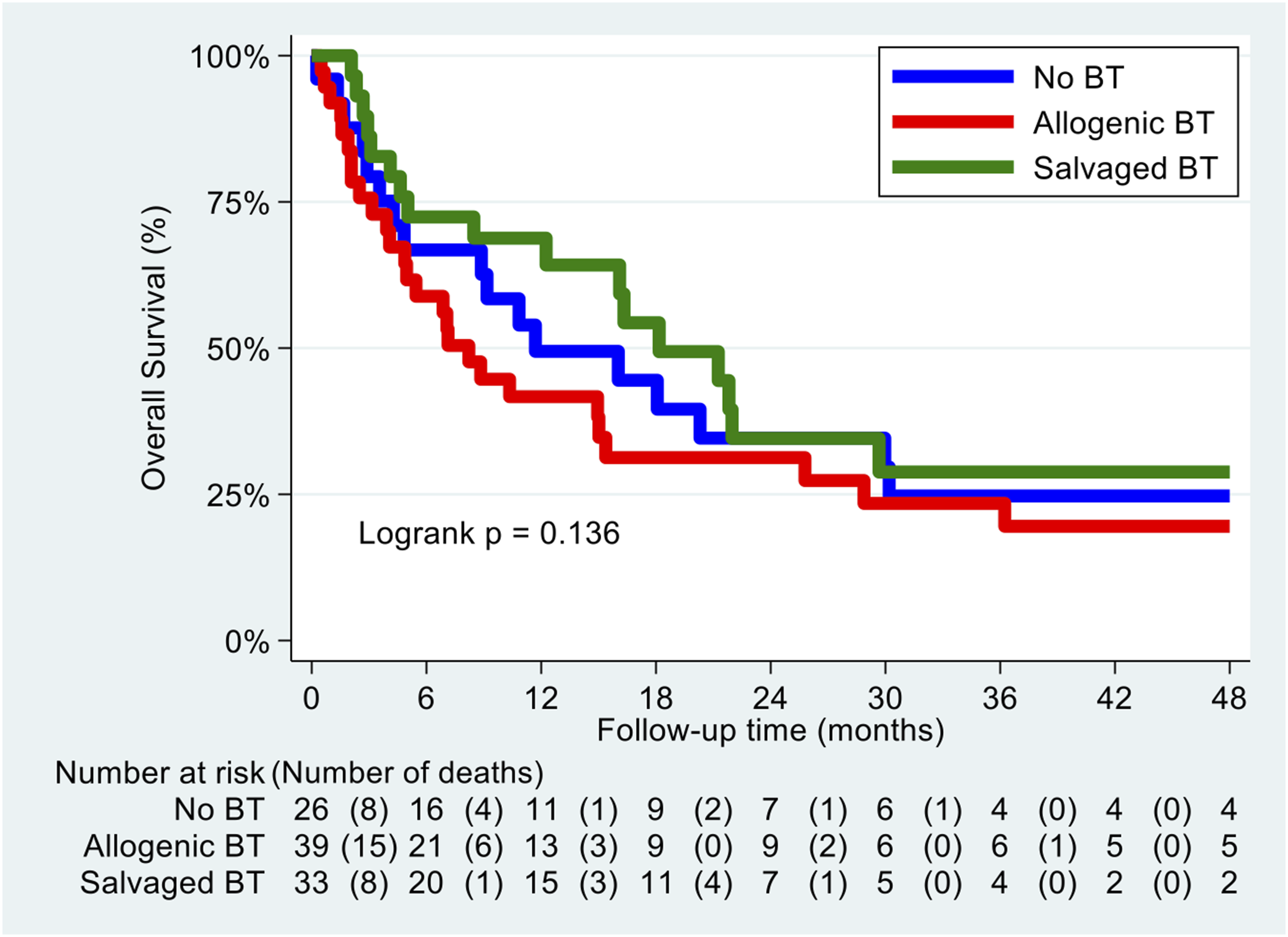
Adjusted survival graph based on blood transfusion (BT) types.
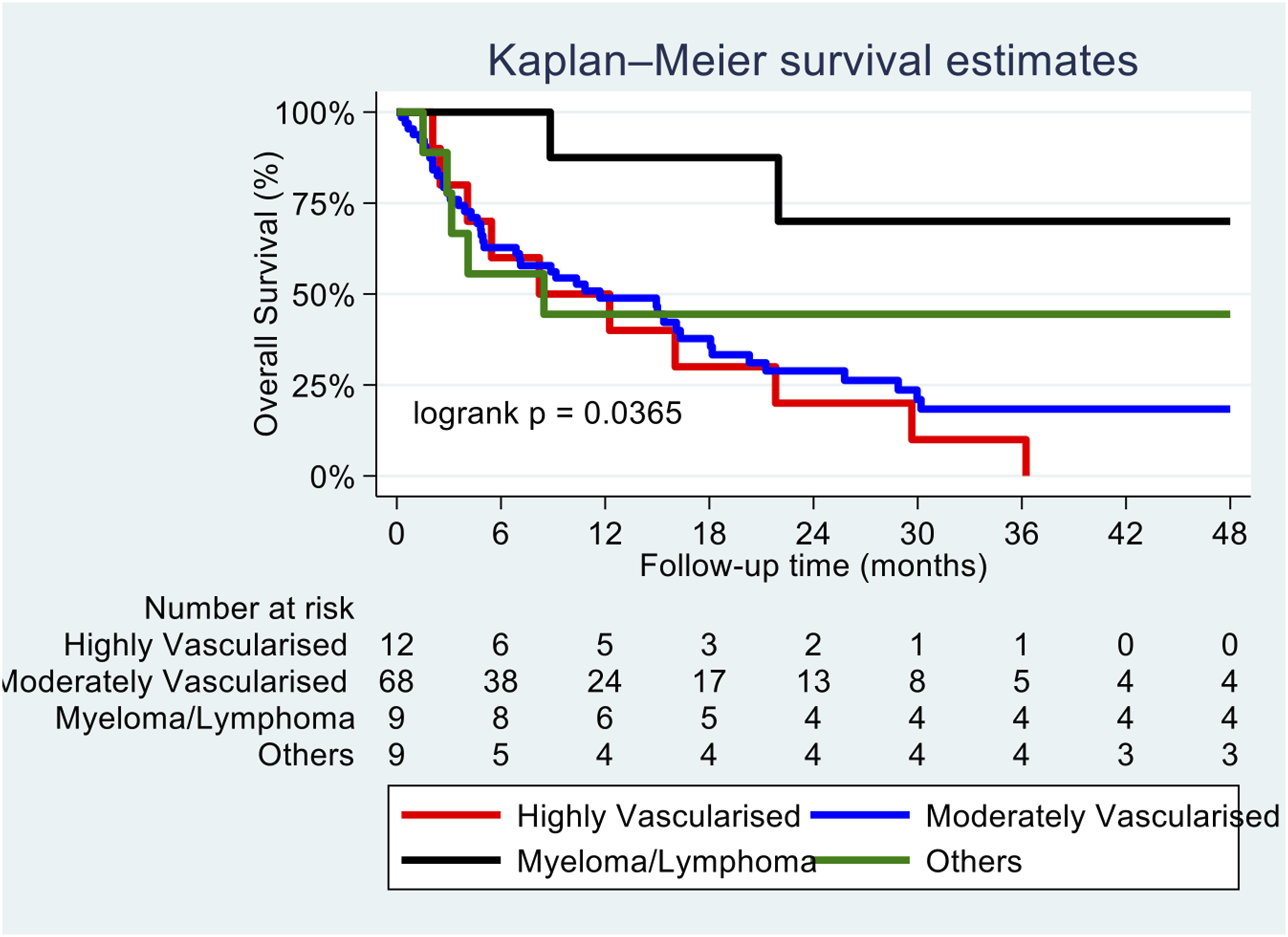
Adjusted survival graph based on vascularity of primary tumour.
Characteristics Associated With 4-Year Tumour Progression (TP).
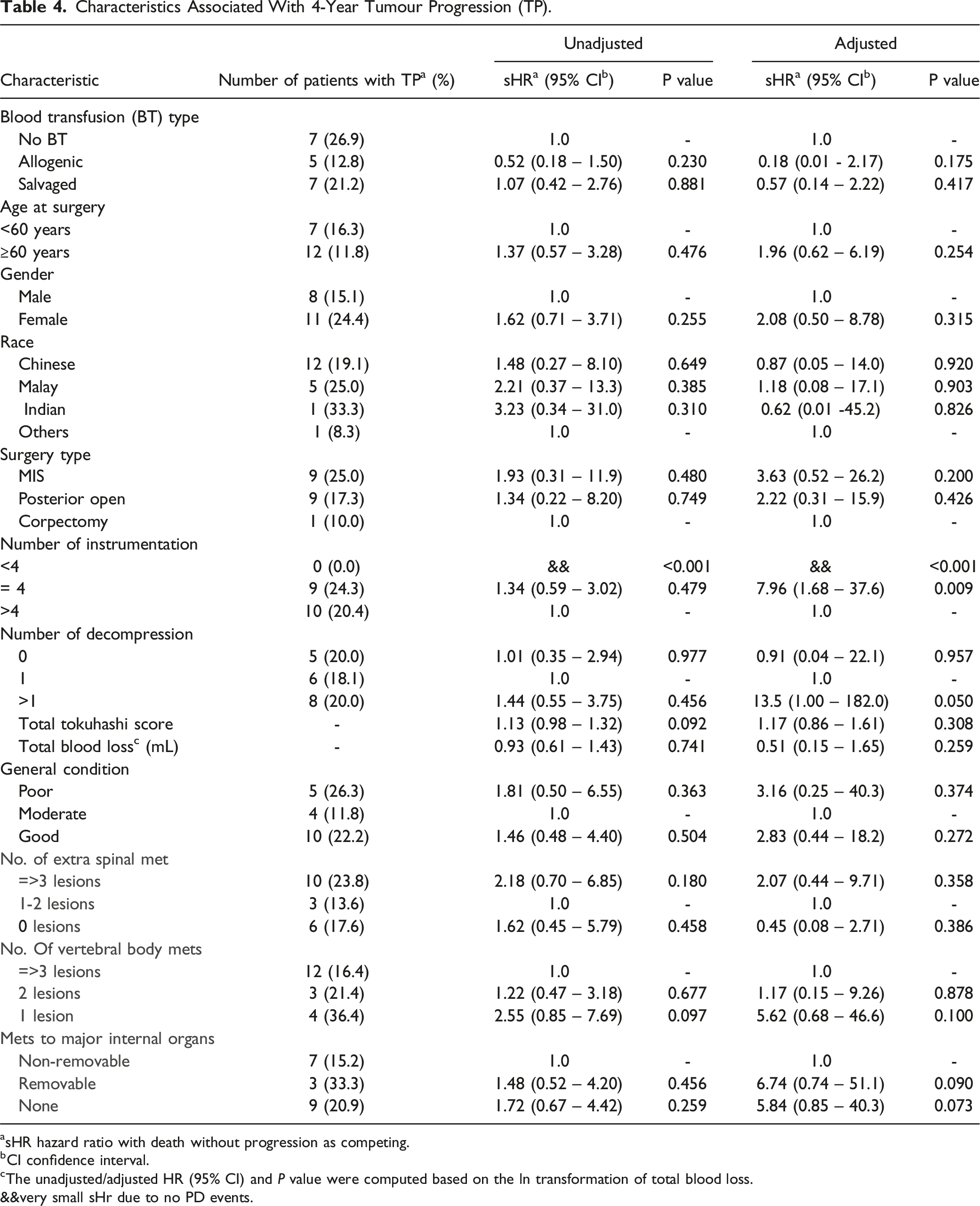
asHR hazard ratio with death without progression as competing.
bCI confidence interval.
cThe unadjusted/adjusted HR (95% CI) and P value were computed based on the ln transformation of total blood loss.
&&very small sHr due to no PD events.
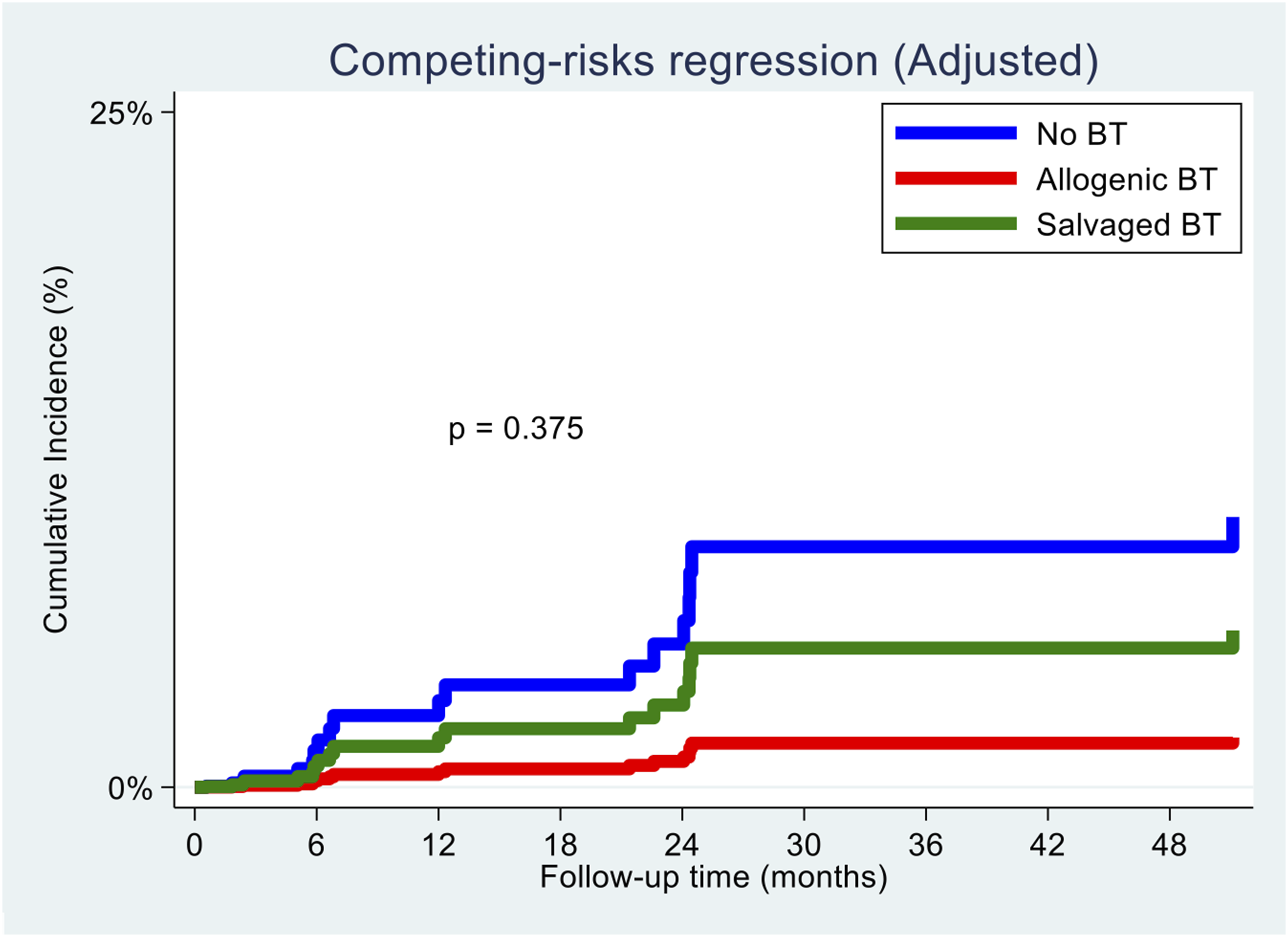
Cumulative incidence of tumour progression for all blood transfusion (BT) types.
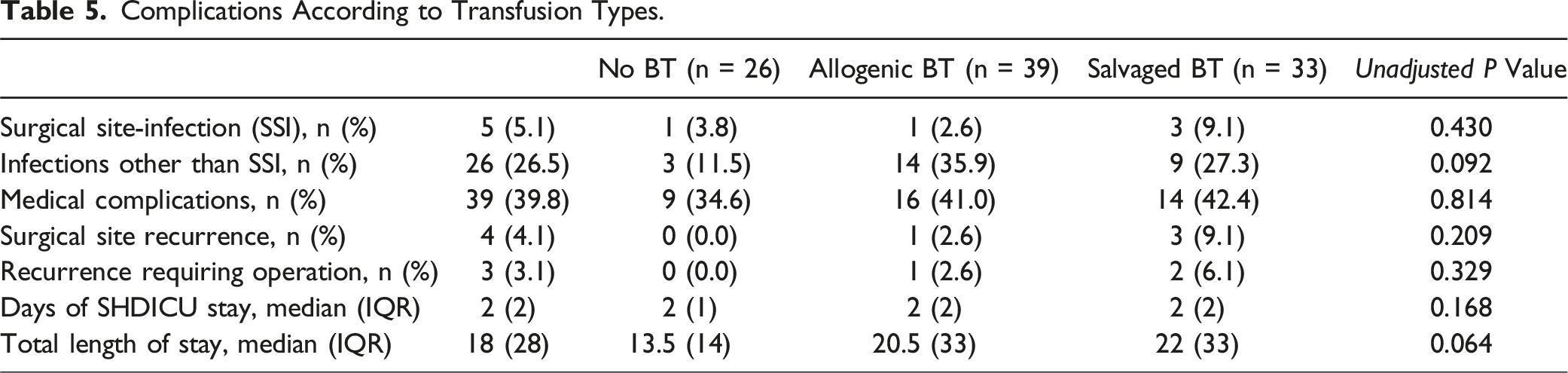
Complications According to Transfusion Types.
Discussion
Patient blood management strategies in recent years have emphasised the use of salvaged blood transfusion for MSTS.22,23 SBT has shown to be safe, with minimal to no tumour cells present in the salvaged blood after passage through a leukocyte depletion filter. 25 Previously, retrospective studies29,35 have also highlighted the advantages of SBT to reduce ABT without an increase in overall costs for the patients. SBT has been used extensively in other oncological procedures such as GI or gynaecological conditions.18,19
Despite the above advantages, there still remains a major reluctance in the uptake of SBT in clinical practice, which can be due to the current lack of long term prospective clinical studies establishing the clinical safety profile of SBT for MSTS patients. In the present study, we manage to establish how SBT is an effective tool for use in MSTS, without increase in risk of death or tumour progression in MSTS patients, on long term clinical follow-up. This is especially important since blood loss is a significant concern in MSTS, with studies showing pooled mean blood loss in MSTS patients to be up to 2180 mLs. 6 Previously, the main method of blood transfusion has been the use of ABT, but studies have shown an increase in complications in patients with ABT. In a multicentre prospective review of 1601 patients by the American College of Surgeons National Quality Improvement Program database, 36 623 patients (38.9%) received packed cell transfusion, and those who had a transfusion had significantly higher complication rate when compared to non-transfused patients, (22.3% vs 15.0%, P = 0.00). Vinas-Rios et al13,37 also found ABT to be an independent risk factor for higher risks of complications while Purvis et al 37 found that a liberal transfusion policy was associated with an independently higher risk of perioperative morbidity and increasing perioperative costs.
To date, this is the first long-term study to compare prospectively the survival and tumour progression results in patients undergoing MSTS who received SBT. There was no significant difference in the oncological characteristics of the three groups [Tables 1 and 2]. Overall, SBT was associated with a decreased risk of death compared to ABT and NBT in univariate analysis, and comparable OS outcomes on long term follow-up in multivariate analysis. As mentioned in previous studies,38,39 the improved OS for SBT compared to ABT can be theoretically attributed to ABT-related immunosuppression which increase the likelihood of postoperative complications in such as infection reduces overall survival. Immunosuppression is unlikely to happen with SBT, resulting in better survival.
Our study also used the recently established tumour vascularity model 32 which divides group of tumours based on their vascularity to show the correlation between tumour vascularity and OS as well as TP. As established in previous studies,32,40 tumour vascularity was shown to be a significant risk factor in OS, with highly vascular tumours showing significant increase in risk of death. Tumour vascularity did not show significant influence on TP on competing risks analysis. Our study did not find any factor that had significant association with TP. This is supported by studies41,42 which showed that TP is usually due to inherent primary tumour characteristics and also affected by ongoing genetic mutations that leads to local and distant progression. As established by our previous basic science publications,25,26 salvaged blood has no viable tumour cells after passing through a filter, and hence is unlikely to affect TP.
One interesting finding in our study is that every unit increase in the natural log scale of blood loss was associated with a decreased risk of death [Adjusted HR 0.58; 95% CI 0.36-0.96; P = 0.03]. This is also supported by a decreased risk of death with greater number of instrumentation in our study, though not significant [Adjusted HR 0.45; 95% CI 0.13-1.53; P = 0.203]. These findings can possibly be explained by the undertaking of larger debulking surgeries such as partial or near total corpectomy for certain tumours subgroups, 43 with greater number of instrumentation, which can aid in more extensive removal of tumour material and hence leading to improved OS. In such cases, the surgery itself may be curative.
The absence of any complications from SBT, and overall comparable trend of OS and TP to ABT during the 4 year follow-up period in our study highlights the long term safety profile of SBT for use in MSTS. From our knowledge, the usage of SBT in MSTS has yet to be established in clinical practices around the world due to the theoretical concerns of tumour seeding. In MSTS, patient blood management (PBM) is an important concept to improve outcomes and readmission free survival. Hence, the utilisation of SBT as a form of intra-operative PBM will only help in overall management of blood loss, improvement in outcomes and reduction in length of hospital stay.
Limitations
This prospective study is the first to highlight the long term safety profile of SBT in MSTS patients but it also faces some limitations. Firstly, this study was not randomised as it is not ethically reasonable to control the treatment which each group of patients received since each patient was managed according to their clinical presentation based on department treatment protocols. Secondly, the relatively small number of patients in each BT subgroup makes it difficult to generalise our results to all MSTS patients. However, further propensity matched studies from large tumour databases can be done to validate the findings of our present prospective study.
Conclusion
SBT has shown to be promising and useful in the intra-operative blood management of MSTS patients. It has comparable overall survival, as well as tumour progression outcomes to those who received ABT. Complication rates of patients who received SBT are also comparable to those who received ABT or NBT.
The utilisation of SBT in MSTS can help to reduce ABT requirement and its related risks, improving overall outcomes of patients with MSTS. SBT should be incorporated to the practice of MSTS surgeons around the world, as part of PBM protocols, which have shown to benefit morbidity and mortality of MSTS patients.
Supplemental Material
Supplemental Material - Does Intraoperative Cell-Salvaged Autologous Blood Transfusion in Metastatic Spine Tumour Surgery Impact Clinical Outcomes: A Prospective Clinical Study With 4-year Follow-Up
Supplemental Material for Does Intraoperative Cell-Salvaged Autologous Blood Transfusion in Metastatic Spine Tumour Surgery Impact Clinical Outcomes: A Prospective Clinical Study With 4-year Follow-Up by Naresh Kumar, Si Jian Hui, Yiong Huak Chan, Yong Hao Tan, Praveen Jeyachandran, Laranya Kumar, Laranya Kumar, Jiaqian Cui, Rohan Parihar, James Thomas Patrick Decourcy Hallinan james, Rohit Vijay Agrawal, and Jiong Hao Tan in Global Spine Journal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by NMRC/CNIG/1161/2016(App I/D: CNIG 15nov018).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
