Abstract
Study Design
Retrospective cohort study.
Objectives
Spinal surgeons face a dilemma regarding the continuation or discontinuation of antiplatelet agents during the perioperative period. Guidelines recommend considering the balance between thrombotic and bleeding risks. However, no consensus exists for the use of these agents for patients who undergo minimally invasive lumbar decompression. This study aimed to assess the effect of continued antiplatelet medication on minimally invasive posterior lumbar decompression surgery outcomes, focusing on perioperative outcomes and 1-year postoperative clinical results.
Methods
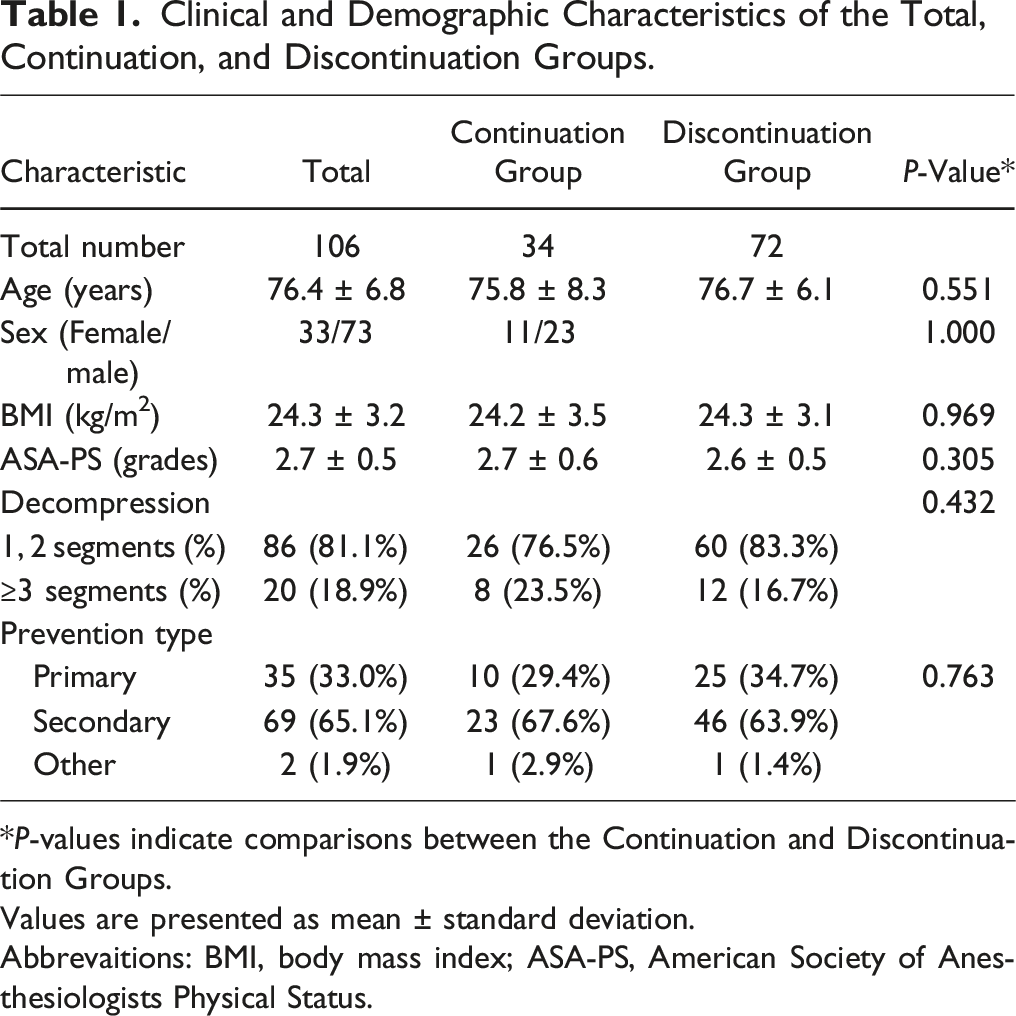
This study included 106 patients who underwent minimally invasive posterior lumbar decompression between 2017 and 2022 and were taking antiplatelet medications before spinal surgery. Patient characteristics, antiplatelet medication type, and perioperative data were analyzed. Patients were divided into “continuation” and “discontinuation” groups based on preoperative antiplatelet medication status. Univariate and multivariate linear regression analyses were performed.
Results
No significant differences were observed between groups in terms of surgical time, intraoperative blood loss, postoperative drain volume, complication rates, and Japanese Orthopedic Association scores and EuroQoL-5 dimensions 5-level at 1 year postoperatively. Similar results were noted in groups focusing exclusively on patients treated with aspirin. Multivariate linear regression revealed that surgical time per level was significantly associated with total blood loss, whereas antiplatelet medications did not show a significant relationship (operative time per level, P < 0.01; antiplatelet drugs, P = 0.459).
Conclusions
This study suggests that minimally invasive posterior lumbar spine decompression can be performed safely and effectively under continuous antiplatelet medication. Further studies with more evidence are required to validate our findings.
Keywords
Introduction
Spine surgeons often face a dilemma regarding whether to discontinue antiplatelet agents during the perioperative period in patients on antiplatelet therapy. Perioperative discontinuation of antiplatelet agents may reduce the risk of intraoperative and postoperative bleeding while increasing the risk of thrombosis, including ischemic heart disease and stroke.1,2 This can lead to serious life-threatening complications. Meanwhile, spinal surgery under continuous antiplatelet medication can reduce the risk of perioperative thrombosis but may lead to transfusion problems due to increased perioperative bleeding and complications such as nerve palsy and asphyxia due to postoperative hematoma.3-5 The relevant treatment guidelines from the United States and Japan suggest that the discontinuation of antiplatelet agents should be decided by considering the balance between thrombotic and bleeding risks.6,7 Discontinuation of antiplatelet drugs is recommended for patients with low perioperative thrombotic risk undergoing high-risk bleeding surgeries, such as spine surgery. 8 However, in a large-scale survey targeting Japanese spine surgeons, 20.4% of respondents who discontinued antiplatelet drugs experienced cerebral infarction and 3.7% experienced myocardial infarction. 9
Although spine surgeries are categorized as surgeries with a high risk of bleeding in the guidelines, some spine surgeries, such as minimally invasive lumbar decompression, cannot be considered as 1 with a high risk of bleeding. For such surgeries, discontinuation of antiplatelet therapy might not be necessary, despite the current guideline recommendations for discontinuation. Meanwhile, minimally invasive decompression surgery has a smaller dead space than traditional open surgery, which could result in postoperative serious palsy due to small hematoma.10,11 Herein, questions arise as to whether the discontinuation of antiplatelet medication is necessary for patients who have undergone minimally invasive lumbar decompression surgery. Therefore, this study aimed to evaluate the effect of continuous antiplatelet medication on minimally invasive lumbar spine decompression surgery by comparing the perioperative and 1-year postoperative outcomes of a group that continued antiplatelet medication with those of a group that discontinued it preoperatively.
Methods
Study Design and Ethical Approval
This study was a retrospective analysis of prospectively collected data of patients who underwent minimally invasive posterior lumbar decompression and were taking antiplatelet medications before surgery. All study participants provided informed consent, and the study protocol was approved by the Institutional Review Board of our institution (No. 3170).
Patient Population
The inclusion criteria were as follows: patients who underwent posterior lumbar decompression surgery with a microendoscope or microscope for lumbar spinal stenosis between 2017 and 2022, were followed up for more than 1 year postoperatively, and were taking antiplatelet medications before spine surgery. Patients were excluded if any of their preoperative or 1-year follow-up data were missing; if they were diagnosed with a bleeding disorder, tumor, trauma, infection, depression, or schizophrenia before the surgery; or if they had routinely taken medications for mental disorders.
Surgical Procedure and Perioperative Protocol
Patients underwent posterior lumbar decompression surgery with a microendoscope or microscope for lumbar spinal stenosis under general anesthesia, as previously reported.12,13 On the day after surgery, all patients were encouraged to stand and walk. Drains were placed intraoperatively in all cases. The drain was removed on the second postoperative day or when the drain volume was <100 mL/day. Patients were discharged when wound healing was confirmed and their activities of daily living were better than those before surgery, as achieved through rehabilitation.
Outcome Assessment
Data regarding the type of antiplatelet medication used and the purpose of antiplatelet therapy were collected as patient information. The total surgical time, surgical time per level, total intraoperative blood loss, intraoperative blood loss per level, total postoperative drain volume, postoperative drain volume per level, length of hospital stay, and the rates of thrombosis-related complications requiring treatment, hematoma requiring unexpected surgery, and those of dural injury and blood transfusion, were investigated as perioperative information. The preoperative and 1-year postoperative Japanese Orthopedic Association (JOA) scores and patient-reported outcomes, including the EuroQoL-5 dimension 5-level (EQ-5D-5 L), were investigated.14,15 The domain scores of the EQ-5D-5 L were converted to index values using the methodology proposed in a previous report. 16
Grouping
The patient cohort was divided into the “continuation” and “discontinuation” groups, which included patients who continued antiplatelet therapy before surgery and those that discontinued it before surgery, respectively. In patients who discontinued their medication, antiplatelet drugs were discontinued 1 week before surgery and restarted the day after drain removal. In accordance with relevant treatment guidelines in Japan, 6 the decision to discontinue antiplatelet drugs was based on balancing the risks of thrombosis and bleeding. Specifically, patients in primary prevention with stabilized conditions were encouraged to discontinue their medication. For those in secondary prevention, the risk of thrombosis was assessed in consultation with the relevant department. Patients at high risk of thrombosis were advised to continue their medication throughout the perioperative period. For other cases, decisions were made on a case-by-case basis.
Statistical Analysis
Univariate analysis was performed to compare the perioperative information between the groups. The Mann–Whitney U test and Studentt test were used to compare non-normally and normally distributed variables, respectively. When comparing the clinical outcomes, the analysis of covariance was used to adjust for covariates that included each preoperative outcome. Finally, multivariate linear regression analysis was performed to evaluate the relationship between the use of antiplatelet medications and total blood loss. In this model, total blood loss was set as the objective variable, and patient characteristics (age and American Society of Anesthesiologists Physical Status [ASA-PS]), body mass index (BMI), operation time/level, and continuation or discontinuation of antiplatelet drugs were included as explanatory variables. BMI and operation time/level were included because of their relationship with blood loss volume.17-19 Subgroup analysis focusing on patients treated exclusively with aspirin, as they constituted the majority of our subjects. To ensure comparability, a 1:1 propensity score matching was performed based on age, sex, BMI, and ASA-PS. The bleeding volume and other perioperative outcomes were then compared between the continuation and discontinuation groups. All statistical analyses were performed using the EZR software (Saitama Medical Center, Jichi Medical University, Saitama, Japan).
Results
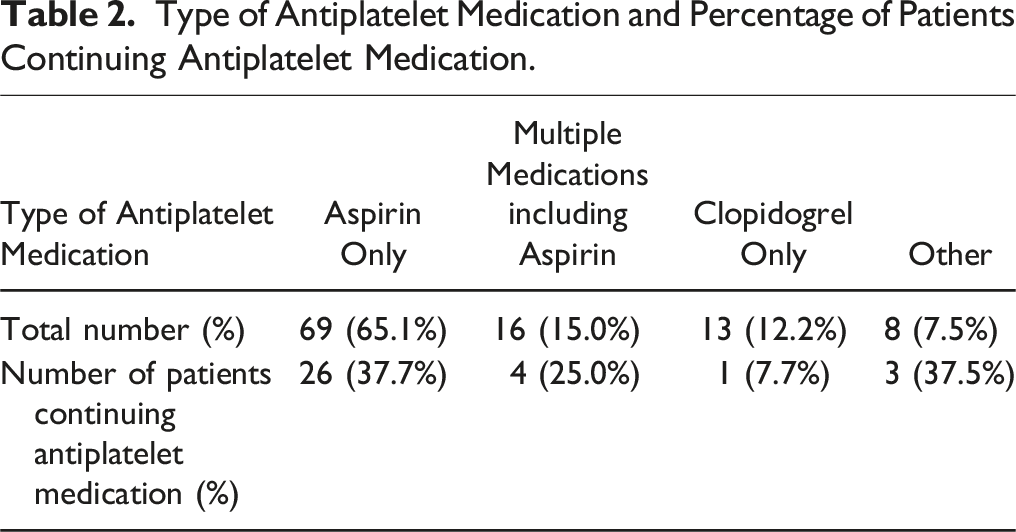
A total of 106 patients were enrolled in this study. The patient selection flow is shown in Figure 1. The clinical and demographic characteristics of the patients are shown in Table 1. In total, 34 and 72 patients were classified into the continuation and discontinuation groups respectively (Table 1). There were no significant differences in age, sex, BMI, severity of comorbidities, the number of decompressions, or prevention type between the 2 groups. Aspirin was the main medication, with about 40% of users continuing it preoperatively (Table 2). Approximately 10% of patients taking clopidogrel continued taking it preoperatively. Flow chart showing the patient selection. Clinical and Demographic Characteristics of the Total, Continuation, and Discontinuation Groups. *P-values indicate comparisons between the Continuation and Discontinuation Groups. Values are presented as mean ± standard deviation. Abbrevaitions: BMI, body mass index; ASA-PS, American Society of Anesthesiologists Physical Status. Type of Antiplatelet Medication and Percentage of Patients Continuing Antiplatelet Medication.
Comparison of Preoperative Information Between the Two Cohorts
Perioperative Characteristics of the Continuation and Discontinuation Groups.
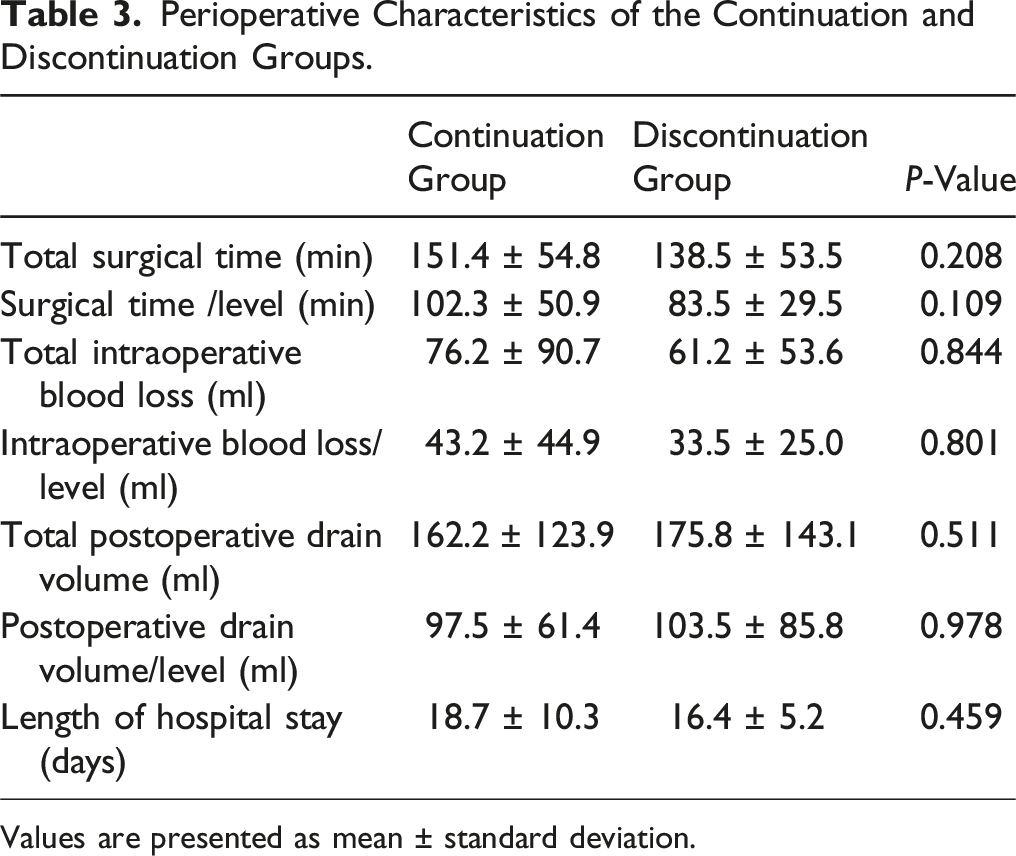
Values are presented as mean ± standard deviation.
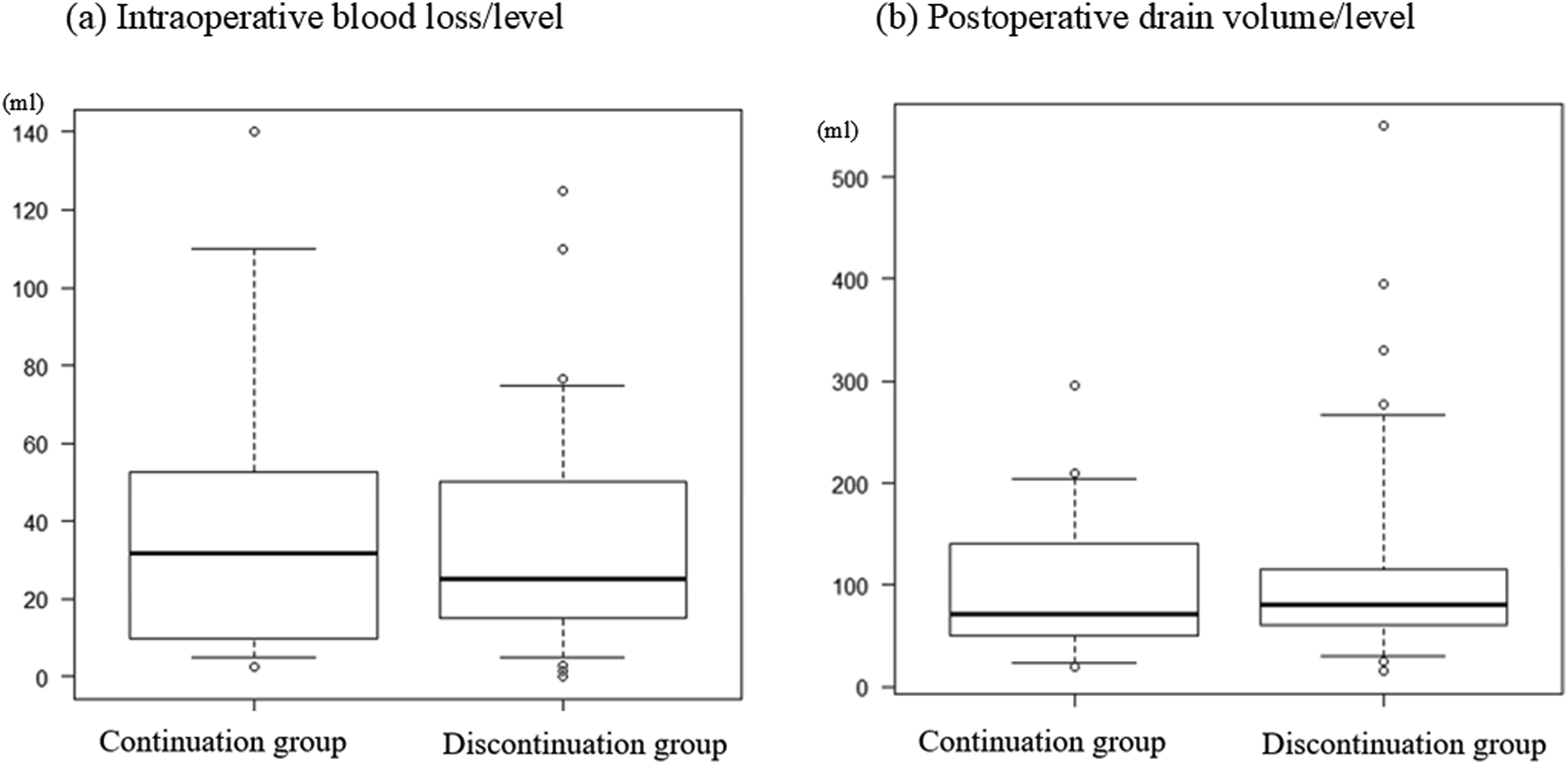
Distribution of blood loss between the groups.
Comparison of Continuation and Discontinuation Groups for Thrombotic and Hemorrhagic Complications.
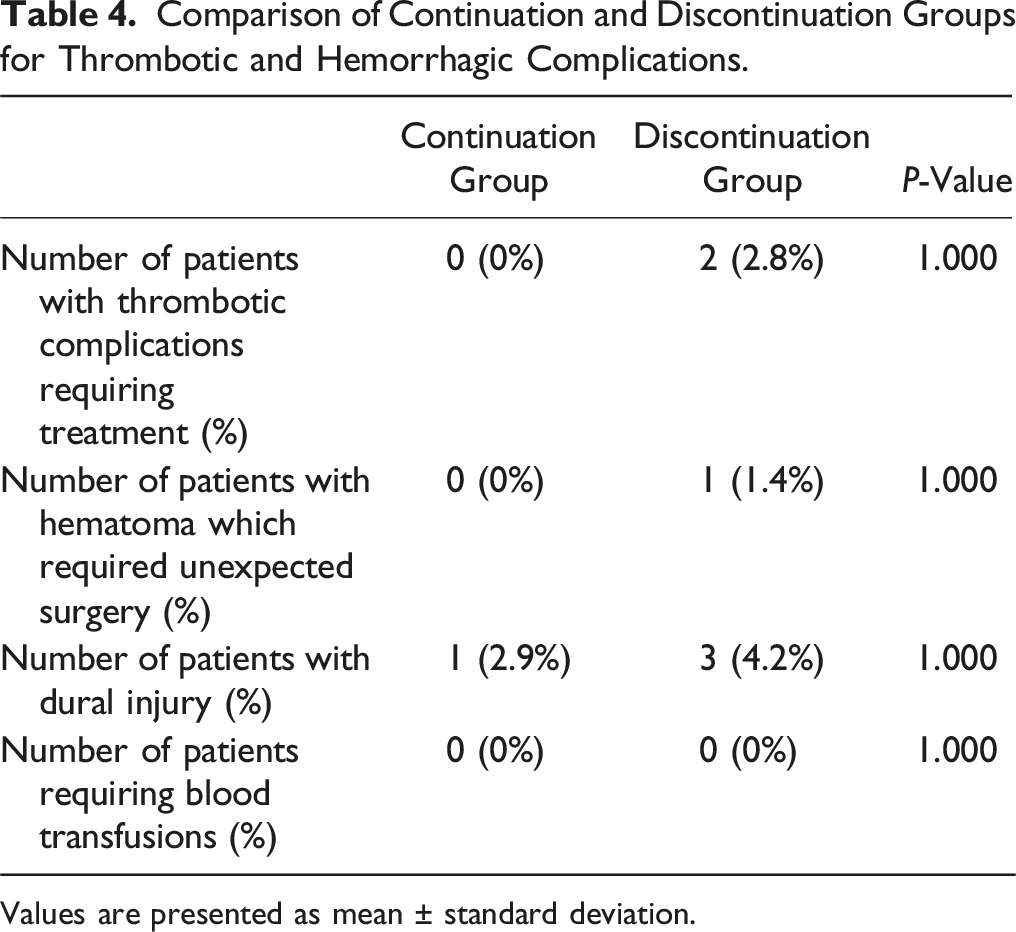
Values are presented as mean ± standard deviation.
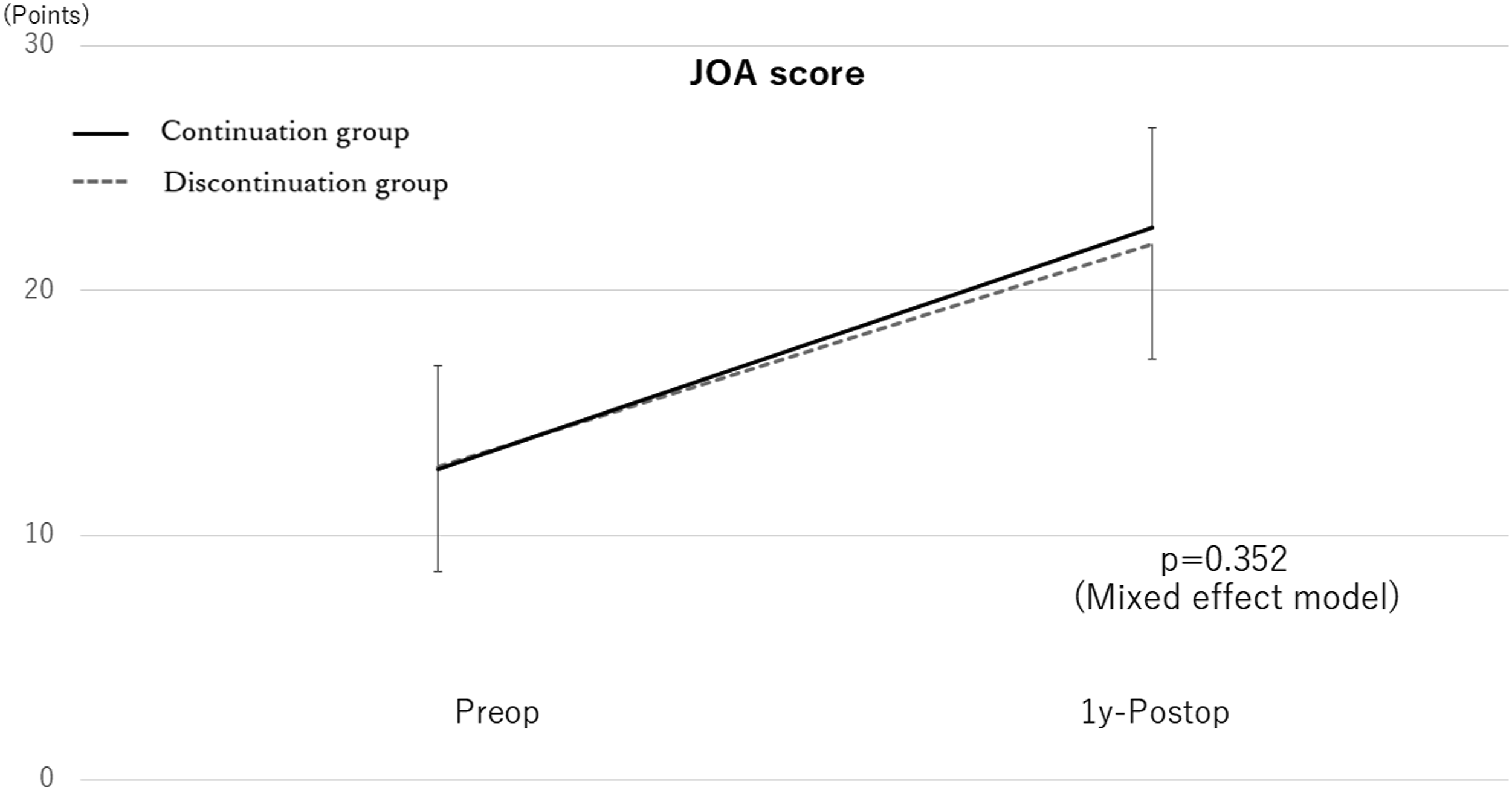
Comparison of the JOA scores between the groups JOA, Japanese Orthopedic Association.
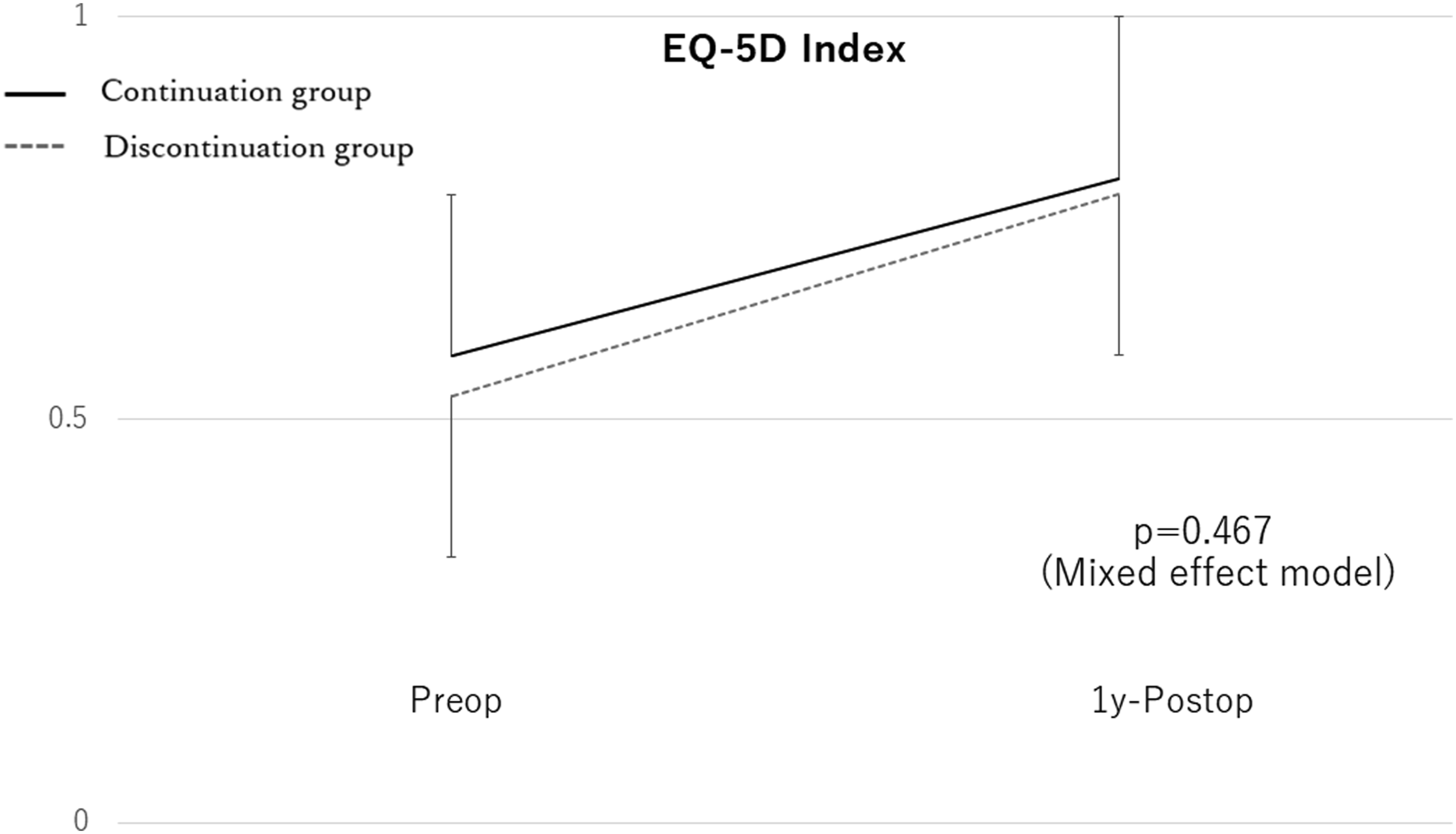
Comparison of the EQ-5D Index between the groups EQ-5D, EuroQoL-5 dimension.
Results of Multivariate Linear Regression Analysis of Factors Influencing Total Blood Loss.
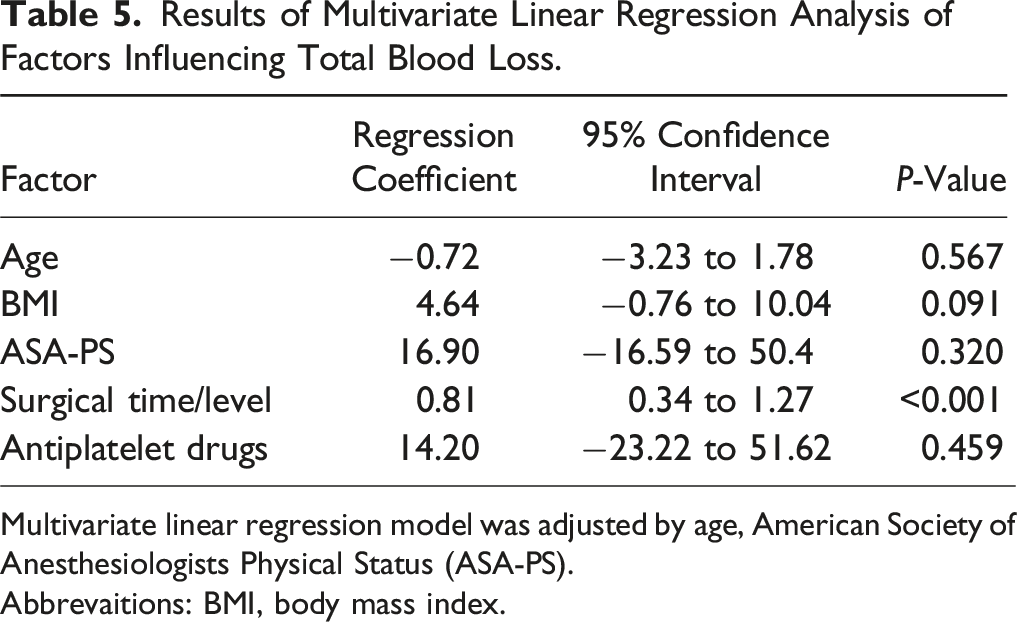
Multivariate linear regression model was adjusted by age, American Society of Anesthesiologists Physical Status (ASA-PS).
Abbrevaitions: BMI, body mass index.
Comparison of Only Those Taking Aspirin
Preoperative and Perioperative Characteristics of the Continuation and Discontinuation Groups in Matched Cohorts.
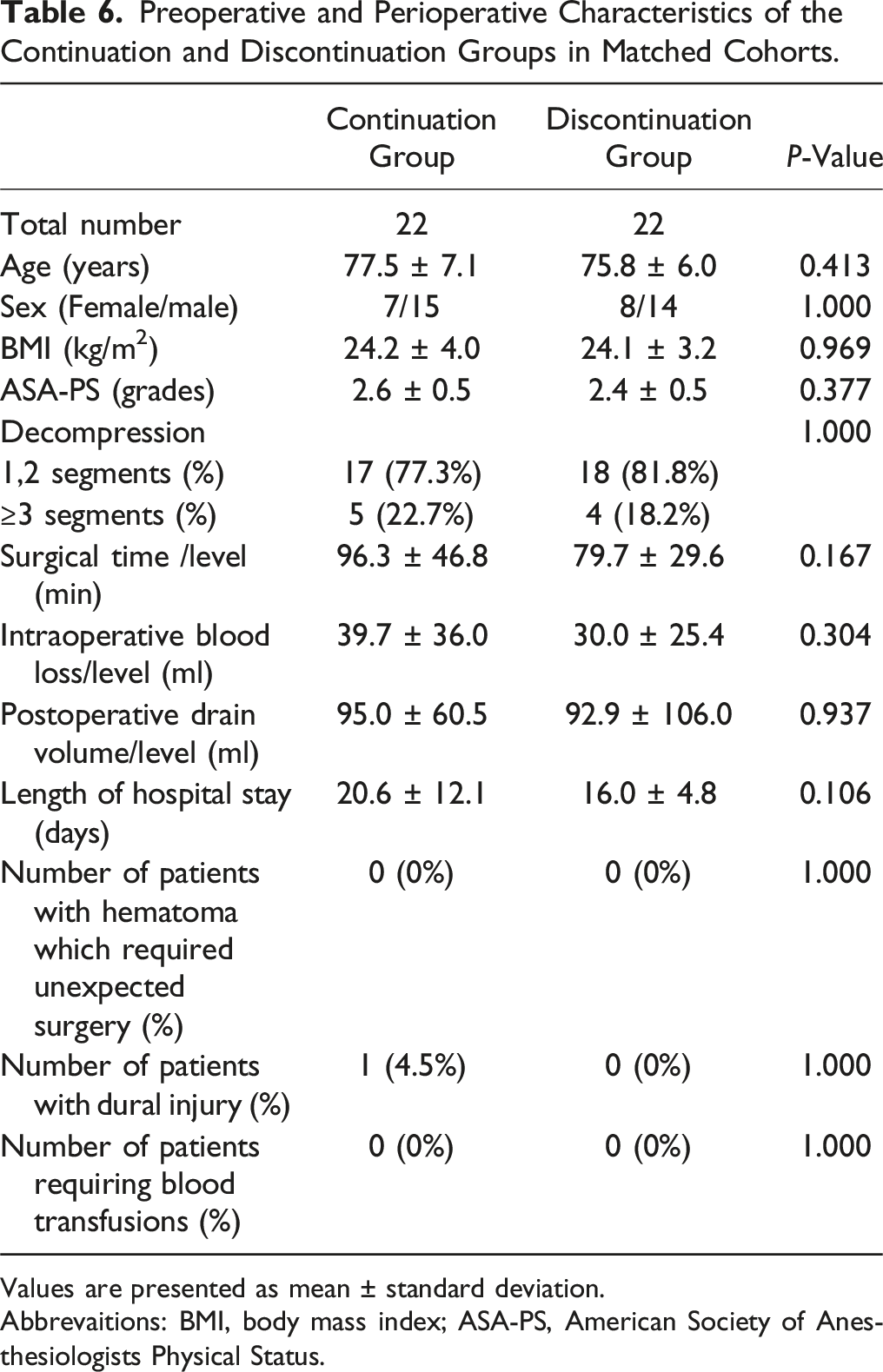
Values are presented as mean ± standard deviation.
Abbrevaitions: BMI, body mass index; ASA-PS, American Society of Anesthesiologists Physical Status.
Discussion
The current study showed no significant differences in surgical time, intraoperative and postoperative blood loss, complication rate, and improvement of the symptoms and QOL at 1 year postoperatively according to univariate analysis between patients who continued antiplatelet therapy and those who discontinued antiplatelet therapy. There was no significant difference in the incidence of hematoma requiring unexpected surgery, which is a critical issue particularly in minimally invasive lumbar spine decompression surgery. Even multivariate linear regression analysis revealed that total blood loss was significantly associated with surgical time/level, while antiplatelet medications were not. Similarly, among the patients treated exclusively with aspirin, who constituted 70% of our subjects, no significant differences were observed in intraoperative or postoperative blood loss, thrombotic rates, transfusion rates, or hematoma rates.
A single-center retrospective study involving 5866 orthopedic surgeries indicated a significant increase in perioperative aspirin continuation rates among patients with coronary artery disease in recent cases, accompanied by a significant decrease in the incidence of postoperative myocardial infarction. 20 In addition, Okamoto et al. conducted a multicenter study on the impact of perioperative antithrombotic medications, including aspirin, on 9853 posterior spine surgeries. 21 Using propensity score matching, they found no significant differences in intraoperative blood loss and epidural hematoma rates between the anticoagulant-discontinued and non-anticoagulant groups. Postoperative cardiovascular events occurred in 3 patients (0.27%) in the discontinuation group, whereas no such event occurred in the non-anticoagulant group. In addition, some studies reported that aspirin continuation in non-cardiac surgeries increased the risk of bleeding by 1.5 times, but did not lead to serious events.22,23 Although the current study did not include enough patients to identify the effect of discontinuation of antiplatelet therapy on serious postoperative complications, such study supports the continuation of antiplatelet therapy when surgery can be performed safely.
However, several studies do not support the continuation of aspirin during the perioperative period. Kang et al. reported significant differences in postoperative blood loss and transfusion rates in 76 lumbar fusion cases in the withdrawal group compared with the non-use group. 24 Park et al. compared discontinuation and non-use of antiplatelets in 182 cases of lumbar fusion and demonstrated a significant difference in postoperative blood loss in the 2-level fusion group only. 25 In the current study, which focused on minimally invasive lumbar decompression surgeries and compared aspirin discontinuation and continuation groups, there were no significant differences in intraoperative and postoperative blood loss, transfusion rates, hematoma rates, or dural injury rates. The results of these studies suggest that particular attention should be paid to intraoperative hemostasis during lumbar fusion surgery.
Consistent with our results, several studies have recommended continued perioperative aspirin therapy. Cuellar et al. compared a withdrawal group with a continuation group in 200 cervical or lumbar cases, including fusion cases, and found no differences in intraoperative blood loss, transfusion rates, complication rates, or readmission rates. 2 Soleman et al. compared a withdrawal group with a continuation group in 102 cases of posterior lumbar decompression and found no differences in intraoperative or postoperative blood loss, operative time, hospital length of stay, or complication rates. 1 Inoue et al. reported similar results in 395 patients who underwent cervical laminoplasty. 26 Based on our results, it is encouraging that minimally invasive decompression surgery can be performed with the continuation of antiplatelet medications.
This study had the following strengths. First, it evaluated the effects of continuation of antiplatelet medication using a control group. Second, the conditions were uniform. The surgical technique was standardized to a minimally invasive lumbar decompression surgery, and the surgical time, intraoperative blood loss, and postoperative drain volume were calculated at each level. Third, it examined the clinical outcomes. 27
This study had several limitations. The biggest limitation was selection bias. The surgeon determined the discontinuation criteria. In cases of aspirin discontinuation, the attending surgeon might have selected patients at low risk of complications due to therapy discontinuation. The small sample size is another limitation of this study. The reported incidence of epidural hematomas ranged from 0.2–0.4%,28,29 suggesting that epidural hematomas may not have been detected. There is also the issue of generalizability. Since most patients received aspirin monotherapy, the applicability to other antiplatelet drugs is limited. To address this limitation, we applied propensity score matching in our subanalysis focusing on patients treated solely with aspirin. However, to validate our results, further evidence from a randomized controlled study with a large sample size should be conducted.
Conclusion
We retrospectively investigated the effects of continued antiplatelet medication in minimally invasive posterior lumbar decompression surgery and found no significant differences in surgical time, intraoperative blood loss, complication rate, or 1-year postoperative clinical outcomes. Similar results were observed in the subgroup analysis focusing on patients treated solely with aspirin. Continued aspirin therapy in the perioperative period may be recommended for minimally invasive lumbar spine decompression. However, further studies with larger samples are required to validate our findings.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
