Abstract
Study design
Prospective within-subjects study.
Objectives
Although motor-evoked potential (MEP) amplitude can reportedly be increased by tetanic stimulation of the peripheral nerves before transcranial electrical stimulation (TES), no reports have described on whether tetanic transcranial stimulation augments the wave amplitudes of spinal cord-evoked potentials (Tc-SCEP). The primary purpose of this study was to investigate whether tetanic stimulation induces waveform amplification of Tc-SCEP. The secondary purpose was to elucidate the mechanism of the amplification effect of tetanic stimulation.
Methods
We recruited 20 patients who underwent cervical or thoracic spine surgeries. We compared the compound muscle active potentials (CMAPs) of Tc-MEP and Tc-SCEP before and after tetanic stimulation of the median or tibial nerve.
Results
Although the CMAP wave amplitudes of the abductor pollicis brevis (APB) and abductor hallucis (AH) showed significant enlargement on Tc-MEP following tetanic stimulation of the median and tibial nerves, an augmentation effect regarding Tc-SCEP (203 μV [without tetanic stimulation], 212 μV [tetanic stimulation of the median nerve], and 208 μV [tetanic stimulation of tibial nerve]) could not be demonstrated.
Conclusion
Although MEP amplitudes can be enlarged by tetanic stimulation of the peripheral nerves, the amplification mechanism may not involve the lateral corticospinal tract from the brain stem to the anterior horn.
Introduction
Recently, intraoperative spinal neuromonitoring (IONM) for spinal cord diseases has become essential for safe spine surgery. Numerous neurophysiological monitoring methods have been used in clinical and surgical settings, including somatosensory-evoked potentials, 1 spinal cord-evoked potentials after transcranial brain stimulation (Tc–SCEP [D-wave]), 2 muscle-evoked potentials after transcranial electrical stimulation of the brain (Tc-MEP), 3 and continuous free-running electromyography. 4 Of those IONM modalities, Tc-MEP is the most frequently used in Japan. 1 Although compound muscle action potentials (CMAPs) recorded from the upper- and lower-limb muscles via Tc-MEP are useful for the intraoperative monitoring of motor function, CMAPs are sometimes unstable owing to anaesthetics, blood pressure, body temperature, operation time, and severe motor paralysis, making it difficult to derive a waveform.2-6 In these cases, several methods of waveform amplification have been reported, particularly for Tc-MEP.7,8 Alternatively, other monitoring modalities can be used.
A novel post-tetanic Tc-MEP (p-Tc-MEP) augmentation technique for CMAP recordings has previously been described. 9 This technique can augment CMAPs in muscles that are innervated or not innervated by stimulated peripheral nerve.8-12 In a clinical setting, this technique improved the success rate of baseline CMAP measurements and led to a decrease in the number of false positives and negatives during p-Tc-MEP monitoring compared with conventional Tc-MEP. 12
However, the precise mechanisms underlying the remote augmentation of MEPs following peripheral nerve tetanic stimulation remain unclear. Prior to this study, we hypothesised that peripheral nerve tetanic stimulation modulates corticomotoneuronal excitability. To clarify our hypothesis, we focused on the D-wave after tetanic stimulation of peripheral nerves. The D-waves were recorded directly from the epidural space and can be used to monitor corticospinal tract integrity. 13 If our hypothesis is accurate, the D-wave amplitude should be augmented after tetanic stimulation of the peripheral nerve. In clinical settings, the possibility of augmented D-waves is crucial because D-waves sometimes reduce the derivation rate in cases of severe paralysis.14,15
Therefore, this study aimed to clarify the effect of D-wave amplitude after tetanic peripheral nerve stimulation (hereafter referred to as post-tetanic Tc–SCEP).
Material and Methods
We prospectively recruited patients who required surgery for spinal cord-level disorders. All patients underwent open surgery. First, the most caudal part of the planned surgical area was decompressed, and a catheter electrode was inserted into the dorsal side of the dura. Subsequently, surgery was performed, and Tc-MEP and Tc-SEP data during surgery and data after tetanic stimulation were also measured. This prospective, within-subjects study was approved by the local institutional review board. All the patients provided written informed consent in accordance with the ethical standards of the 1964 Declaration of Helsinki.
Patients
Patients underwent decompression surgery at a spinal cord level higher than T12 at the hospital between August 2018 and June 2020. The exclusion criteria were patients with lumbar disease, infection, a history of brain disease, or who underwent reoperation.
Stimulators
The intensity of TES was determined at the start of the MEP recording and was set to deliver a constant-voltage stimulus (500 V) using a constant-voltage stimulator (SEN-4100; Nihon Kohden, Tokyo, Japan). The stimulating electrodes consisted of a pair of 14.5 mm silver-plated disc electrodes at the C3 (cathode) and C4 (anode) locations of the international “10-20” system for the scalp and were affixed using conductive paste. A train consisting of 5 monophasic stimulation pulses was delivered with a 2 ms interstimulus interval (500 Hz), and the ground electrode was placed on the body trunk or buttocks.
Post-tetanic Tc–SCEP (D-Wave)
Regarding Tc–SCEP (D-wave), a bipolar catheter electrode was placed into the epidural space caudal to the surgical lesion as the recording electrode. Monophasic stimulation was delivered via the right anode and left cathode-stimulating electrodes. A single transcranial electrical stimulation (single voltage stimulus of 500V; duration: 0.5 ms, frequency: 3 Hz) was applied to elicit a D-wave.
After conventional Tc-SCEP, tetanic peripheral nerve stimulation (50 Hz; stimulus intensity, 50 mA; duration, 5 s) was applied. One second later, transcranial stimulation was applied, and the D-wave was recorded (post-tetanic Tc-SCEP).
The peripheral nerve stimulation site was set in 2 locations: the tibial and median nerve (Figure 1). The stimulation interval was 2 minutes. The CMAPs were recorded simultaneously with the D-wave. The recording sites were the abductor pollicis brevis (APB) and abductor hallucis (AH). CMAPs were also recorded in the APB and AH after post-tetanic stimulation. Schematic diagram of post-tetanic stimulation. Pattern 1: Perform transcranial stimulation after bilateral median nerve stimulation and measure transcranial brain stimulation (Tc-SCEP). Pattern 2: Perform transcranial stimulation after bilateral tibial nerve stimulation and measure Tc-SCEP.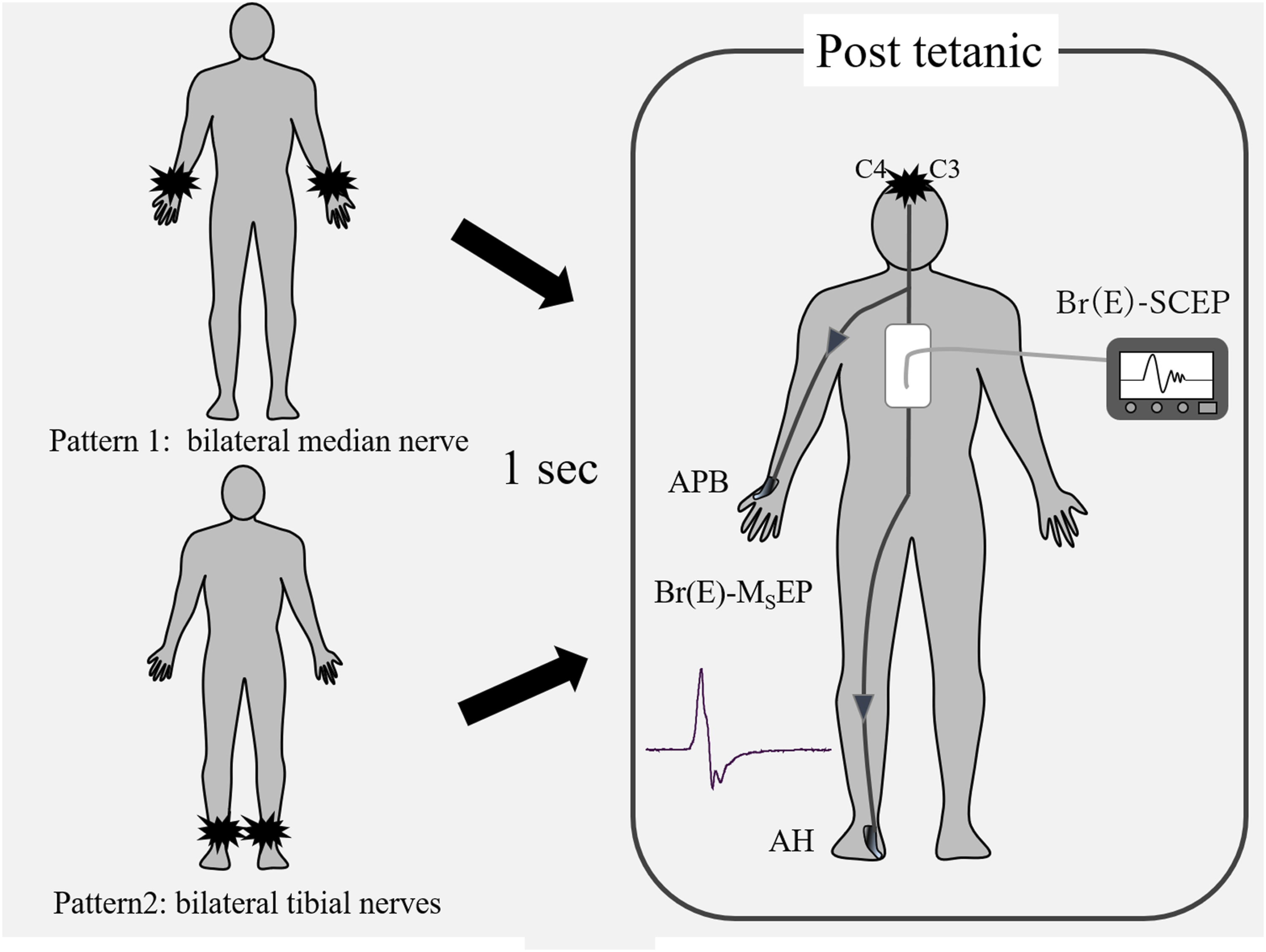
General Anaesthesia
To minimise the suppressive effects of anaesthetic agents and neuromuscular blockade on the CMAP waveforms, anaesthesia was standardised for all patients as follows: No medications were administered before anaesthesia. Anaesthesia was induced using 2-4 μg/kg of fentanyl, 0.25-0.5 μg/kg/min of remifentanil, and 3.0-5.0 μg/mL of propofol, administered via a target-controlled infusion pump (TE-371; Terumo, Tokyo, Japan). After induction, tracheal intubation was facilitated using 0.6 mg/kg of rocuronium. Anaesthesia was maintained with a regimen of propofol (2.0-3.0 μg/mL), fentanyl, and remifentanil (0.20-0.5 g/kg/min), administered via a target-controlled infusion pump. The depth of anaesthesia was adjusted to maintain a bispectral index within the range of 40-60. No additional neuromuscular blockade was administered after tracheal intubation to avoid pharmacological reduction or disappearance of the CMAP waveforms. Sugammadex was only administered to reverse the profound residual neuromuscular blockade induced by rocuronium if the ratio of the fourth response to the first response in the train-of-four monitoring at the time of control CMAP recordings did not return to at least 0.80. After tracheal intubation, the lungs were mechanically ventilated to maintain the partial pressure of end-tidal carbon dioxide within 30-40 mmHg. Subsequently, a mixture of air and oxygen was administered. Rectal temperature was maintained between 35.5°C and 37.0°C.
Statistical Analysis
All statistical analyses were performed using SPSS software (version 17.0; IBM, Chicago, IL, USA). Differences in the rate of decrease between Tc–SCEP (D-wave) and post-tetanic Tc–SCEP (D-wave) were evaluated using the Wilcoxon signed-rank test. The threshold for statistical significance was set at P < 0.05.
Results
Demographic Data
This study recruited 20 patients (12 male and 8 female) ranging from 33 to 82 years old (mean, 64.8 years). The patients were diagnosed with cervical spinal stenosis (n = 14), cervical ossification of the posterior longitudinal ligament (n = 2), cervical intramedullary extraspinal cord tumour (n = 2), thoracic spinal ossification of the ligamentum flavum (n = 1), and thoracic hypertrophic meningitis (n = 1). Preoperative mild to severe motor weakness in any muscle (manual muscle test: 0-3) was present in 2 of the 20 patients (10%). Postoperatively, there were no cases of additional paralysis owing to intraoperative nerve injury.
CMAP Comparison Between Conventional Tc-MEP and Post-tetanic Tc-MEP (Stimulating the Bilateral Median Nerve or Tibial Nerve)
Amplitudes of Conventional Transcranial Electrical Stimulation of Motor Evoked Potentials (C-Tc- MEPs) and Post-tetanic Transcranial Electrical Stimulation of Motor Evoked Potentials (p-Tc -MEPs).
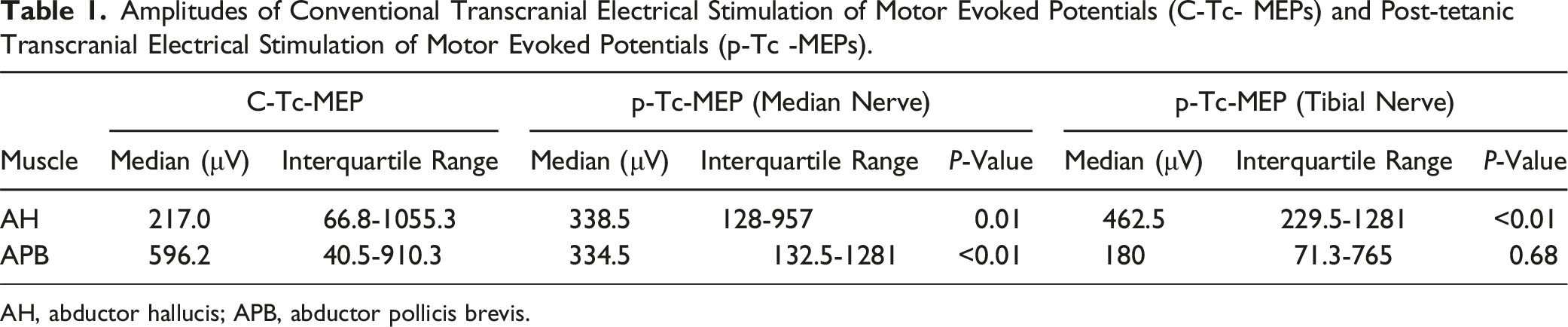
AH, abductor hallucis; APB, abductor pollicis brevis.
D-Wave Amplitude Comparison Between Conventional Tc-SCEP and Post-tetanic Tc-SCEP (Stimulating Bilateral Median Nerves or Tibial Nerves)
The D-wave of conventional Tc-SCEP was 203 (51.5429.1) μV. The D-wave on post-tetanic Tc-SCEP was212 (53,501.6) μV (stimulating the median nerves only). The D-wave on post-tetanic Tc-SCEP was 219 (43.7508.1)μV (stimulating the tibial nerves) (Figure 2). There were no statistically significant differences among the 3 D-wave patterns. Comparison of Tc-SCEP with conventional stimulation method and post-tetanic (median nerve and tibial nerve) Tc-SCEP.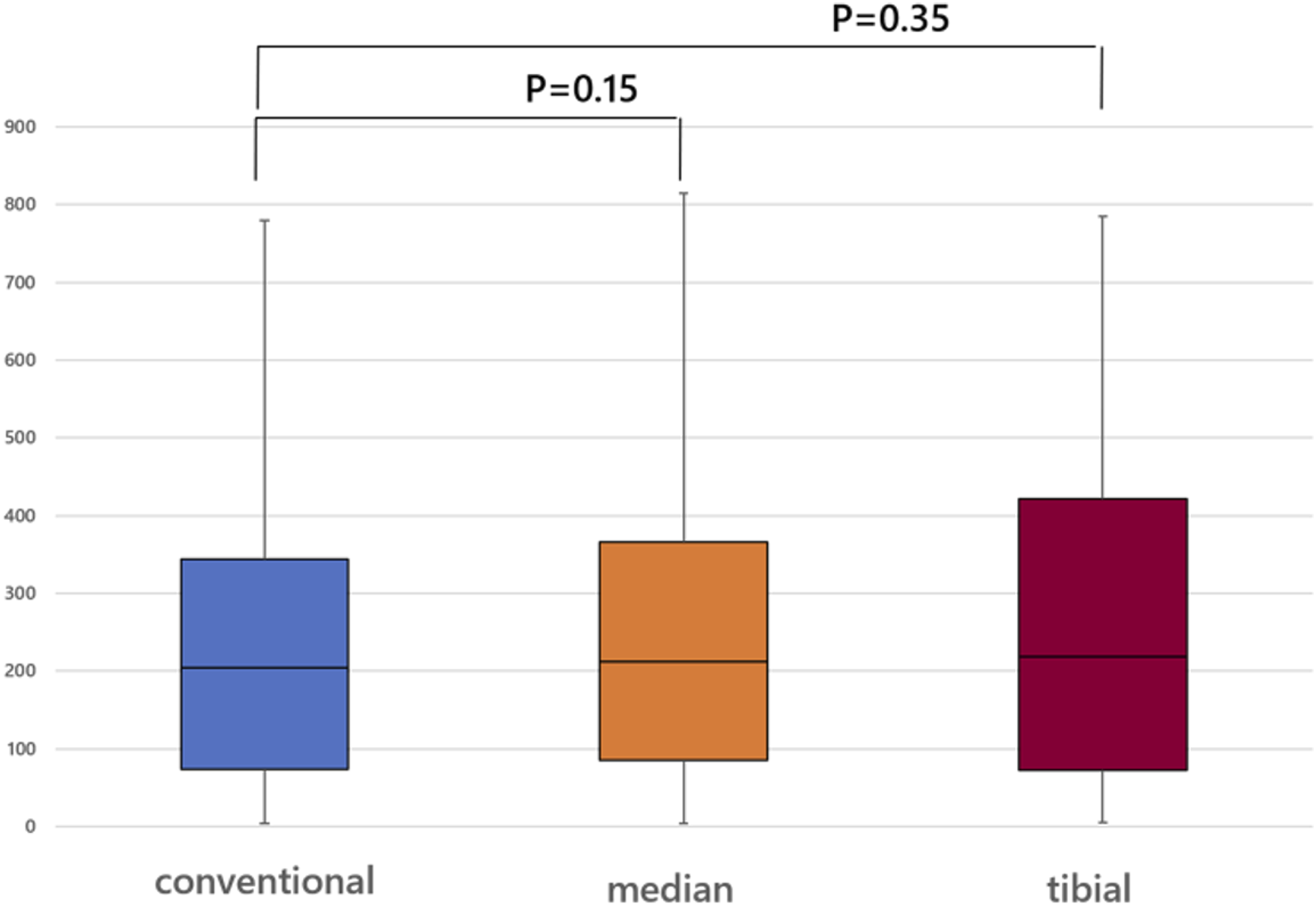
Discussion
In this study, the CMAP of the APB and AH were amplified after median nerve stimulation, while that of the AH was amplified during tibial nerve stimulation. However, no D-wave augmentation was observed after tetanic stimulation of the median or tibial nerves.
Generally, a D-wave is an electric potential recorded from the spinal cord by direct stimulation of the cerebral motor cortex and has been reported as a waveform that is not affected by anaesthetics as it does not pass through synapses. 16 However, because direct stimulation of the brain is invasive in spinal surgery, Tc-SCEPs were used to monitor motor function, similar to D-waves. 17
Kakimoto et al. reported that the CMAPs waveform was amplified by applying tetanic stimulation to peripheral nerves (they used a stimulus intensity of 25-50 mA for a duration of 3-5 s with a post-tetanic interval of 1-5 s) and then performing transcranial stimulation (post-tetanic Tc-MEP). 9 They found that in patients with preoperative muscle weakness, it is also useful because it increases the derivation rate of CMAP waveforms. In addition, Hayashi et al. reported that, as a feature, there is a remote augmentation stimulated by tetanus that has an enhancing effect on muscles other than dominant muscles. 10 Therefore, we can augment the CMAP waveforms using this technique. However, it is still unknown whether the D-wave amplitude can be augmented after tetanic stimulation of a peripheral nerve.
To date, there have been 3 suggested reasons for the augmentation of CMAP waveforms. First, the mechanism of waveform amplification may be that the amplification effect of the stimulated nerve-dominant muscle is greater than that of the nondominant muscle, which may promote transmission at the neuromuscular junction. 11 Second, based on F-wave studies, the excitability of spinal cord anterior horn cells may be increased by the peripheral nerve tetanus stimulation, regardless of the level of nerve stimulation. 18 Finally, regarding the effect of excitation induction in the corticospinal tract within the brain, Sun et al. did not observe a waveform amplification effect due to tetanic stimulation of the facial nerve, suggesting that it may originate in the subcortex rather than that in the motor cortex. 19 However, Kaelin et al showed that peripheral electrical stimulation increases corticomotoneural excitability. 20
Although the pathway by which the D-wave potential passes through the spinal cord has not been fully clarified, D-waves reportedly reflect corticospinal tract function. 21 Transcranial stimulated-SCEPs stimulate the deep parts of the brain, such as the brain stem, and cortical stimulation stimulates the superficial parts of the brain. 16 Thus, the stimulated parts of the brain may vary between transcranial and direct cortical stimulation.
This study has some limitations. Firstly, the sample size was relatively small, which limited the statistical power of our findings. Secondly, the study included various types of target diseases, and it remains unclear whether the waveform of D waves induced by tetanus stimulation changes depending on the type of disease. In the future, we believe that a unified study of diseases is necessary.
Conclusion
In this study, we did not observe an augmentation effect on the D-wave after tetanic stimulation of the peripheral nerves (median and tibial nerves). These results suggest that the tetanic stimulation of peripheral nerves does not activate corticomotoneuronal excitability in the brain.
Footnotes
Acknowledgments
The authors thank Sayomi Yamamoto, Junko Kato, and Tomoshige Miyabayashi for their technical assistance with TES-MEP.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
